Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is the less strong acid from H_2S and H_2O ? Why? |
| Answer» Solution :`H_2O` is less STRONG and `H_2S` is more strong O and S both are in same group. and the atomic SIZE of S is more than O because `H_2S` is more and `H_2O` is less acidic. | |
| 2. |
When NH_(4)NO_(2)(s) decomposes at373K , it forms N_(2)(g) and H_(2)O(g). The DeltaH . For the reactionat one atmosphericpressureand 373 K is -223.6 kJ mol^(-1)of NH_(4)NO_(2)(s) decomposed. What is the value of DeltaU for the reaction under the same conditions ?( Given R = 8.31 J K^(-1) mol^(-1)) |
|
Answer» `DELTAU = DeltaH - Deltan_(g) RT = - 223.6 kJ mol^(-1) - ( 3 mol) xx ( 8.314 xx 10^(-3) k J K^(-1) mol^(-1)) xx ( 373K)` `= - 232 kJ mol^(-1)` |
|
| 4. |
What is tetraethyl lead ? How is it prepared ? |
| Answer» SOLUTION :Tetraethyl lead is `(CH_(3) CH_(2))_(4)Pb`. It is prepared as FOLLOW : <BR> `4CH_(3) - CH_(2) Br + 4 NA -Pb overset("Dry ether") (to) underset("Tetraethyl lead (TEL)") ((CH_(3) - CH_(2))_(4) Pb) + 4 NaBr + 3 Pb` | |
| 5. |
What are Troposphere and Stratosphere ? |
|
Answer» 10 - 20 KM and 1 - 10 km |
|
| 6. |
Two moles of N_(2) and two moles of H_(2) are taken in a closed vessel of 5 litre capacity and suitable conditions are provided for the reaction. When the equilibrium is reached, it is found that a half mole of N_(2) is used up. The equilibrium concentration of NH_(3) is |
|
Answer» 0.2 `=("No. of moles at equilibrium")/("Volume")=(1)/(5)=0.2` |
|
| 7. |
What is the structure of BeCl_(2)molecule in gaseous and solid state ? |
Answer» Solution :In gaseous state above 1200K, `BeCl_(2)` exists as a monomer having linear structure and with ZERO dipole MOMENT.`Cl-Be-Cl`. In solid state, it exists in polymeric chain structure in which each Be atom is surrounded by 4Cl ATOMS. 2Cl atoms through covalent bonds and other 2Cl atoms through COORDINATE bonds and give bridge structure. 
|
|
| 8. |
Which of the following combines with blood to form oxy-haemoglobin |
|
Answer» `CO` |
|
| 9. |
What type of crystalline solid is graphite ? |
| Answer» SOLUTION :GRAPHITE is COVALENT CRYSTALLINE SOLID. | |
| 10. |
Using the VSEPR theory , identify the type of hydridisation and draw the structure of OF_(2) . What are oxidation states of O and F ? |
Answer» Solution :Electron dot structures of `OF_(2)` is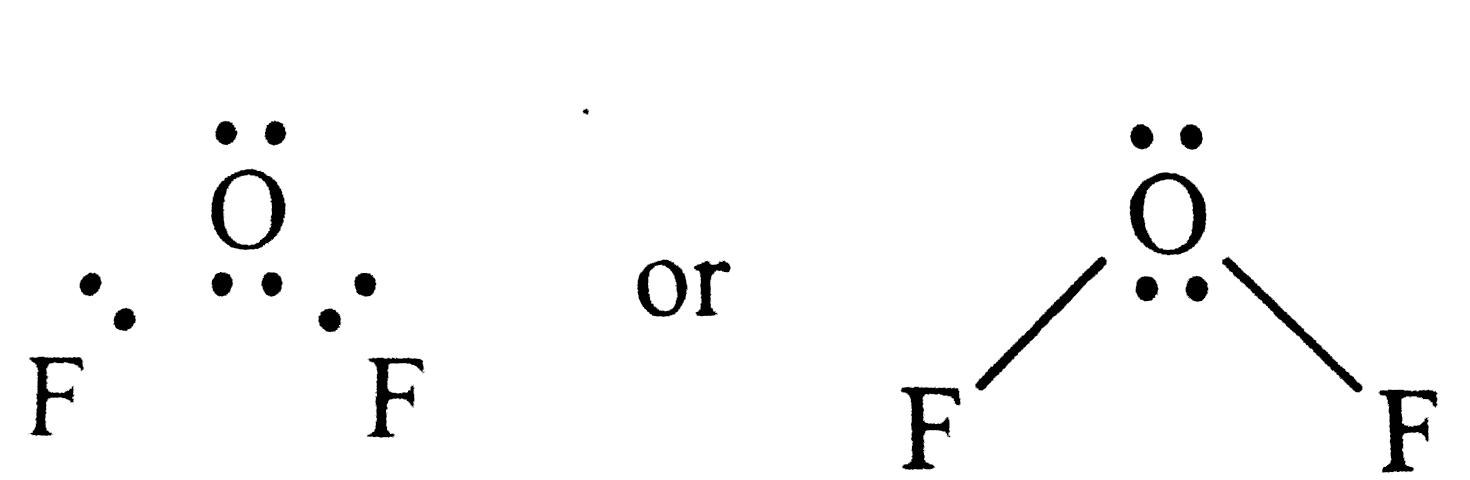 Thus , the central ATOM (O-atom) has 4 pairs of electrons (2bond paris ). HENCE oxygen in `OF_(2) is SP^(2)`hybridised and the molecular is V-shaped . Oxidation state of F = - 1 Oxidation state of O = + 2 . |
|
| 11. |
Which of the following reagent(s) will be most suitable reagent to remove completely NO_(2)^(-) from the mixture of NO_(2)^(-) and NO_(3)^(-)? |
| Answer» Answer :D | |
| 12. |
Which type of isomerisam is seen in 1-propanemine and 2-propanemine? |
|
Answer» Functional group isomerism The position of `-NH_(2)` in obth structure is DIFFERENT so, it is position isomerism |
|
| 13. |
Which of the following reactions is an example of use of water gas in the synthesis of other compounds ? |
|
Answer» `CH_(4(g)) +H_2O_((g)) underset"Ni" overset"1270 K" to CO_((g)) + H_(2(g))` `CO_((g)) + 2H_(2(g)) overset"[CO]"to CH_3OH_((l))` This EQUATION indicate the use of water gas is in prep. of methanol. |
|
| 14. |
When1g liquidnaphthalene ( C_(10) H_(8)) solidifies.149 jouls of heat is evolved. Calculate the enthalpy of fusion of naphthalene. |
|
Answer» Solution :Molar mass of NAPHTHALENE `(C_(10)H_(8))= 128 g mol^(-1)` When 1 g of liquidnaphthalene solidified, HEAT evolved `= 149 xx 128 ` joules `= 19072` Joules Since fuson is reverse of solidification, therefore, heat absorbed for FUSION of one mole of naphthalene `=` 19072 joules, i.e., Enthalpy of fusion `(Delta _(FUS) H )= + 19072` joules `//` mole |
|
| 15. |
Which of the following compounds is a peroxide ? |
|
Answer» `KO_(2)` |
|
| 16. |
When BCl_(3) is treated with water, it hydrolysesand forms [B(OH)_(4)]^(-) only whereasAlCl_(3)in acidifiedaqueous solutionforms [Al(H_(2)O_(6))]^(3+) ion. Explain what isthe hybridisationof boron and aluminium in these species ? |
|
Answer» Solution :When `BCl_(3)` reacts with water, it first undergoeshydrolysis to form boric acid, `B(OH)_(3)`.Due to small size and high electronegativity of B, `B(OH)_(3)`polarizes `H_(2)O` moleculeacceptingan `OH^(-)` ion to form `[B(OH)_(4)]^(-)`speciesand releasinga proton : `BCl_(3) + 3H_(2)O rarr B(OH)_(3) + 3HCl : B(OH)_(3) + H_(2)O rarr [B(OH)_(4)]^(-) + H^(+)` Since B lies in the 2ndperiod,it has onlyibe s-and three p-orbitalsbut no d-orbitals .In other words, at the maximum, it canhave fourpairs ofelectrons in the valenceshell, ie., its maximumcoordinationnumberis In contrast, `AlCl_(3)` undergoes hydrolysis in acidfied aqueous solution to form `[Al(H_(2)O)_(6)]^(3+)` `AlCl_(3) + "water" overset("HCL")rarr[Al(H_(2)O)_(6)]^(3+) + 3Cl^(-) (aq)` This may be explainedas follows : (i) In acidicmedium, the conc.of `OH^(-)`IONS is much lower than that of`H^(+)` atomstherefore,`Al^(3+)` C ions coordinatewith `H_(2)O` moleculesand not with `OH^(-)` ions. (ii) Due to the presence of vacant d-orbital in `Al^(3+)`ions,ITCAN expand its coordinationnumber from 4 to 6and hence forms `[Al(H_(2)O)_(6)]^(3+)` in whichhybridizationof `Al` is `sp^(3)d^(2)`. 
|
|
| 17. |
What is the resonance energy of Benzene in the same units? |
Answer»  `Delta H_(th) = -30` units But practical value `= -24` units `:.` RESONANCE ENERGY `= -24 - (-30) = 6` units |
|
| 18. |
Which scientists gave gas laws from the basis of physical properties of gas ? Give their Laws. |
|
Answer» Solution :(i) Boyle : Robert Boyle in 1662 did trustable measurement of gaseous properties. Boyle.s Low : At constant temperature, volume of fixed amountof any gas is inversely proportional to its pressure... `p prop (1)/(V )""` (at constant T and n) `therefore p=k_(1)(1)/(V)` (II) JACKS Charle.s Law : Jacks Charle.s performed several experiments on gases independently to improve upon hot air BALLOON technology. Their inverstigations showed that for a fixed mass of gas at constnat pressure, volume of gas increases on increasing temperature and decreases on cooling. `V prop T` (at constnat n and p) (ii) Joseph Gay LUSSAC : Pressure in well inflated types of automobilesis almost constant, but on a hot summer day this increases considerably and tyre may burst if pressure is not asdjucted properly. During winters, on a cold morning one may find the pressure in the types of vehicle decreased considerably. Law : ..At constant volume, pressure of a fixed amount of a gas varies directly with the temperature. `p prop T` (at constant n and V) `therefore (p)/(T)=k_(3)=` constant (iv) Avogadro Law : In 1811 Italian Scientist Amedeo Avigardro tried to combined conclusions of Dalton.s atomic theory and Gay Lussac.s law of COMBINING volumes which is now known as Avogadro Law. Law : It states that equal volumes of all gases under the same conditions of temperature and pressure contain equal number of molecules. `V prop n` (at constant T and p) where, n is the number of moles of the gas. `therefore V=k_(4)n` |
|
| 19. |
Which of the following is hyperconjugation of overset(+)(C )_(2)H_(5) ? |
|
Answer» `H- UNDERSET(underset(H)(|))OVERSET(overset(H)(|))(C )-underset(underset(H)(|))overset(overset(H)(|))(C ^(+))` |
|
| 20. |
Which of the following has least nucleophilicity? |
|
Answer» `F^(-)` |
|
| 21. |
Two salts A and B both give white ppt. with BaCI_(2), (insoluble in conc. HCI and conc. HNO_(3)) and brown ppt. with alkaline Nessler's reagent. Salt A gives blue colour with reagent X. Reagent X on oxidation gives reagent Y. The reagent Y gives blue colour with salt B. (X and Y are potassium contaning reagents.) What will be the oxidation state of the red-brown coloured residue obtain by thermal decomposition of salt B? |
|
Answer» `+2` (b) `FeSO_(4) (NH_(4))_(2)SO_(4)6H_(2)O` `underset(("White PPT"))(BaSO_(4))darr HgO underset(("Brown ppt"))(HG(NH_(2))Idarr)` `Fe^(3+)+K_(4) underset((x))([Fe(CN)_(6)])rarrFe_($)[Fe(CN)_(6)]_(3)` (Blue) `{:(Fe^(2+)+K_(3)[Fe(CN)_(6)]rarrFe_(3),[Fe(CN)_(6)]_(2)),(,""darr"Spontaneous"),(Fe^(3+)+[Fe(CN)_(6)]^(4-)overset(DELTA)larrFe_(4),underset(("Prussian Blue"))([Fe(CN)_(6)]_(3))):}` |
|
| 22. |
Two salts A and B both give white ppt. with BaCI_(2), (insoluble in conc. HCI and conc. HNO_(3)) and brown ppt. with alkaline Nessler's reagent. Salt A gives blue colour with reagent X. Reagent X on oxidation gives reagent Y. The reagent Y gives blue colour with salt B. (X and Y are potassium contaning reagents.) The ratio of number of water molecules as crystallisation in salt A and B is: |
|
Answer» 4 (b) `FeSO_(4) (NH_(4))_(2)SO_(4)6H_(2)O` `UNDERSET(("WHITE ppt"))(BaSO_(4))darr HgO underset(("BROWN ppt"))(Hg(NH_(2))Idarr)` `Fe^(3+)+K_(4) underset((x))([Fe(CN)_(6)])rarrFe_($)[Fe(CN)_(6)]_(3)` (Blue) `{:(Fe^(2+)+K_(3)[Fe(CN)_(6)]rarrFe_(3),[Fe(CN)_(6)]_(2)),(,""darr"Spontaneous"),(Fe^(3+)+[Fe(CN)_(6)]^(4-)overset(DELTA)larrFe_(4),underset(("Prussian Blue"))([Fe(CN)_(6)]_(3))):}` |
|
| 23. |
Two salts A and B both give white ppt. with BaCI_(2), (insoluble in conc. HCI and conc. HNO_(3)) and brown ppt. with alkaline Nessler's reagent. Salt A gives blue colour with reagent X. Reagent X on oxidation gives reagent Y. The reagent Y gives blue colour with salt B. (X and Y are potassium contaning reagents.) The reagent X on reaction with conc. H_(2)SO_(4) gives gas. |
|
Answer» `CO` (b) `FeSO_(4) (NH_(4))_(2)SO_(4)6H_(2)O` `underset(("White ppt"))(BaSO_(4))darr HgO underset(("Brown ppt"))(Hg(NH_(2))Idarr)` `FE^(3+)+K_(4) underset((x))([Fe(CN)_(6)])rarrFe_($)[Fe(CN)_(6)]_(3)` (Blue) `{:(Fe^(2+)+K_(3)[Fe(CN)_(6)]rarrFe_(3),[Fe(CN)_(6)]_(2)),(,""darr"Spontaneous"),(Fe^(3+)+[Fe(CN)_(6)]^(4-)overset(Delta)larrFe_(4),underset(("Prussian Blue"))([Fe(CN)_(6)]_(3))):}` |
|
| 24. |
Which of the followingis a polar molecule ? |
|
Answer» `BfF_(3)` LONE pair of electons and hence is polar . All other MOLECULES are symmertical and have ZERO net dipole moment . |
|
| 25. |
Write the structures of major and minor products when 2-bromo-3-methylbutane is heated with potassium hydroxide in ethanol. |
|
Answer» Solution :Dehydrohalogenation OCCURS giving methylbutenes as reaction products. `CH_(3)-OVERSET(CH_(3))overset(|)(CH)-underset(Br)underset(|)CH-CH_(3)overset(alc.KOH)tounderset("2-Methyl-2-butene (MAJOR)")(CH_(3)-overset(CH_(3))overset(|)(C)=CH-CH_(3))tounderset("3-Methyl-1-butene (minor)")(CH_(3)-overset(CH_(3))overset(|)CH-CH=CH_(2))` |
|
| 26. |
Which of the following halides would undergo Friedel-Crafts alkylation without rearrangement? |
|
Answer» `CH_3CH_2Cl` |
|
| 27. |
The value of DeltaH for the reaction X_(2(g))+4Y_(2(g))hArr2XY_(4(g)) is less than zero. Formation of XY_(4(g)) will be favoured at : |
|
Answer» HIGH PRESSURE and LOW TEMPERATURE. |
|
| 28. |
What isthe active mass of water ? |
| Answer» Solution :ACTIVE mass is concentration in MOL `L^(-1).1 L H_(2)O = 1000 g = 1000//18 "mol" = 55.5 ` mol. Hence, active mass of `H_(2)O= 55.5 ` mol `L^(-1)`.. | |
| 29. |
We know that the relationship between K_(c)andK_(p) is K_(p)=K_(c)(RT)^(Deltan) What would be the value of Deltan for the reaction : NH_(4)Cl_((s))hArrNH_(3(g))+HCl_((g))? |
|
Answer» 1 `NH_(4)Cl_((s))hArrNH_(3(g))+HCl_(g)` `Deltan=n_(("gaseous products"))-n_(("gaseous reactants"))=2-0=2` |
|
| 30. |
Which step in the mechanism determines the overall rate of the reaction ? |
|
Answer» FASTEST |
|
| 31. |
Water has high boiling point. |
| Answer» SOLUTION :DUE to INTERMOLECULAR HYDROGEN bonding, water has HIGH boiling point. | |
| 32. |
X -rays are emitted during |
|
Answer» `ALPHA,n" reaction"` |
|
| 33. |
What is the specific in Lewis structure of CO ? |
Answer» Solution :The Lewis structure of CO MOLECULE is as under. Here, C = 4 `bar(e)`and O = 6 `bar(e)`= 10 total ELECTRONS THUS, there is a tripal bond between C and 0 . From which two are covalent bond and one is coordinate covalent bond. In CO one coordinate bond is due to two electron of oxygen. So from three bonds only two electron of oxygen. So from three bonds only two bonds are covalent as electron share by C and O . |
|
| 34. |
Which effect observed when pressure of gas increases ? |
|
Answer» Solution :Fixed amount of any GAS if pressure becomes double than its volume BECOME half. When pressure of gas INCREASES volume decreases. Nature of gas is very compressible if pressure increases gas compressible more. Gases are highly compressible because when a GIVEN mass of a gas is compressed the same number of MOLECULES occupy a smaller space. If increases pressure, gas becomes thick and density increases. |
|
| 35. |
The weight percentage of iron in heamo-globin is 0.33. If the approximate molecular weight of heamoglobin is 68000, how many iron atoms are present in the molecule? (At.wt. of Fe is 56) |
|
Answer» SOLUTION :100 parts of HEAMOGLOBIN =0.33 parts iron 6800 parts of heamoglobin=? WEIGHT of Fe in heamoglobin `=(6800)/(100) xx 1/3 =227g` 56g of Fe =one Fe 227g of Fe=? Numbewr of iron atoms `=(227)/(56)=4` |
|
| 36. |
Which of the following on heating does not given nitrogen gas? |
|
Answer» `NH_(4)NO_(3)` |
|
| 37. |
What are the characters of gasesous state. |
|
Answer» Solution :The characters of gaseous state are : (a) Gases do not have fixed volume and shape (B) They have very low densitites. (c) They exerts pressure on the walls of the continuous (d) They are liquegiable (e) They freely intermix with eacth other (diffusion) (f) Theyare HIGHLY COMPRESSIBLE. |
|
| 38. |
When passed over heated copper at 575 K an organic compound was found to give an ketone. The organic compound is most likely to be a/an |
|
Answer» Primary |
|
| 39. |
What is the radius of sodium atom if it crystallizes in bcc structure with the cell edge of 400 pm ? |
|
Answer» |
|
| 40. |
The value of Bohr radius for hydrogen atom is |
|
Answer» `0.529 XX 10^(-8) CM` |
|
| 41. |
Which resonance form in each of the following sets is the major contributor to the real structure? (i)underset((a))(H_(2)C=CH-ddotunderset(..)CI: )harr underset((b))(H_(2)overset(-)overset(ddot)(C)-CH=overset(-+)underset(..)CI: )harrunderset((c))(H_(2)overset(+)(C)-CH=overset(ddot)underset(..)CI:^(-)) (ii) underset((a))(H_(2)overset(-)(C)-CH=overset(+)(N)H_(2)) harr underset((b))(H_(2)=CH-overset(ddot)(N)H_(2)) |
|
Answer» |
|
| 42. |
What is the half life period of tritium ? |
| Answer» SOLUTION :The half life PERIOD of TRITIUM is 12.33 years. | |
| 43. |
Which of the following fruits contain maximum of potassium? |
|
Answer» Grapes |
|
| 44. |
Which of the following is true about pressures in flasks A and B? |
|
Answer» <P> The pressure in flask `A` is four times that in flask `B`. `(P_(A))/(P_(B))=(n_(A)T_(A))/(n_(A)T_(B))=8XX(300)/(600)=4` `n_(A)=(m)/(2)`, `n_(B)=(m)/(16)`, `(n_(A))/(n_(B))=8` |
|
| 45. |
What is the half-life period of tritium ? |
|
Answer» 12.33 minute |
|
| 46. |
When 40 mL of 0.1 N HCl and 20 mL of 0.1 M H_(2)SO_(4) are mixed together, the normality of the mixture will be : |
|
Answer» `(1)/(5)N` `0.1xx40+0.2xx20=N_(R )(60)` `N_(R )=(8)/(60)=(2)/(15)` |
|
| 47. |
Thiosuphate reacts differently with iodine and bromine in the reaction given below 2S_(2)O_(3)^(2-)+I_(2)rarrS_(4)O_(6)^(2-)+2I^(-) S_(3)O_(3)^(2-)+2Br_(2)+5H_(2)Orarr2sO_(4)^(2-)+2Br^(-)+10H^(+) Which of the following statement justifies the above dual behaviour of thisosuplhate ? |
| Answer» Solution :`BR+_(2)` is a stronger OXIDISING agent than `I_(2)` for details REFER to | |
| 48. |
Which of the following statements are correct about CO_(3)^(2-) ? |
|
Answer» The HYBRIDIZATION of central atom is `sp^(3)` . The hybridization of carbon in`CO_(3)^(2-)` is `sp^(2)`. So, (A) is WRONG.  Due to resonance all C-O bond lengths are equal. Formal charge on each O-atom = `("Total charge")/("Number of O-atoms") = (-2)/(3) = -0.67 `units. ALI C - O bond lengths are equal as mentioned above. |
|
| 49. |
Which of the following atoms ha the highest first ionisation energy- |
|
Answer» Rb |
|
| 50. |
What will be the minimum pressure required to compress 500 "dm"^3 of air at 1 bar to 200 dm^3 temperature remaining constant. |
|
Answer» Solution :According to Boyle.s LAW, at constant temperature, `P_1 V_1 = P_2 V_2` Suppose the required pressure is `P_2`. `:.""1 XX 500 = P_2 xx 200` or `P_2 = (1xx500)/200 = 2.5` bar |
|



