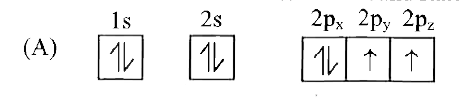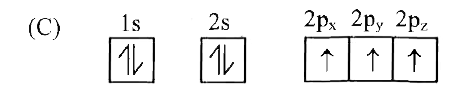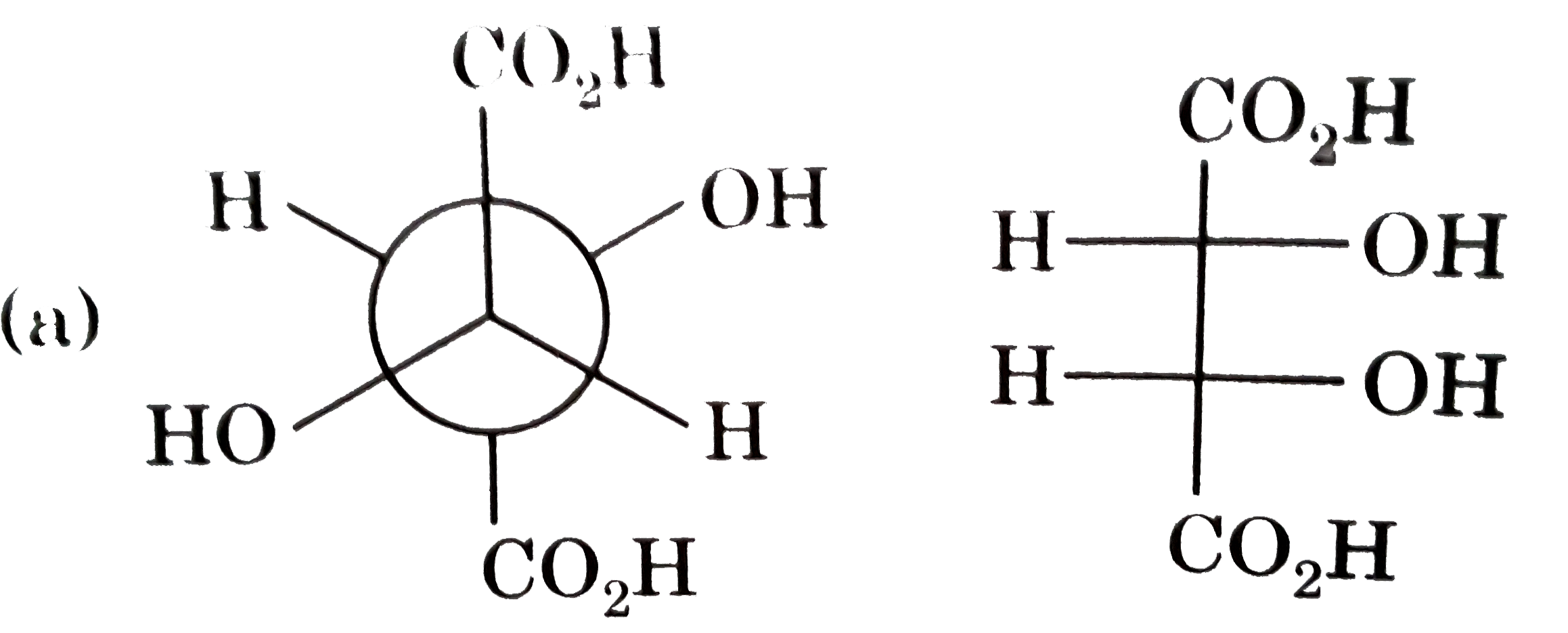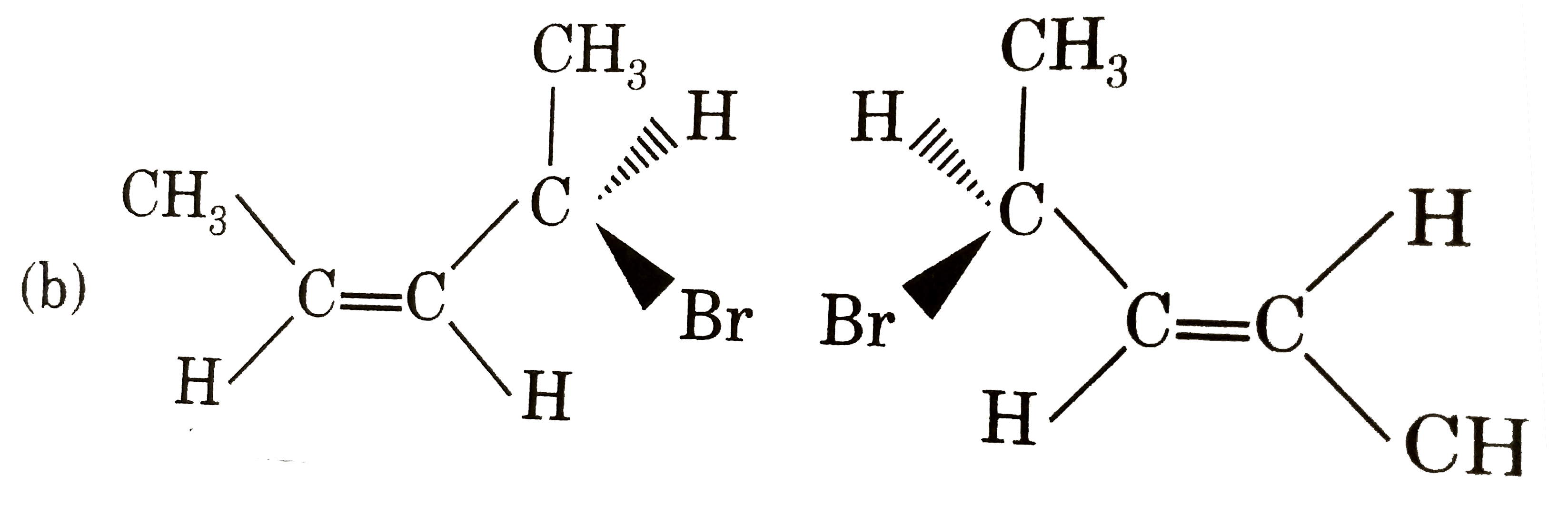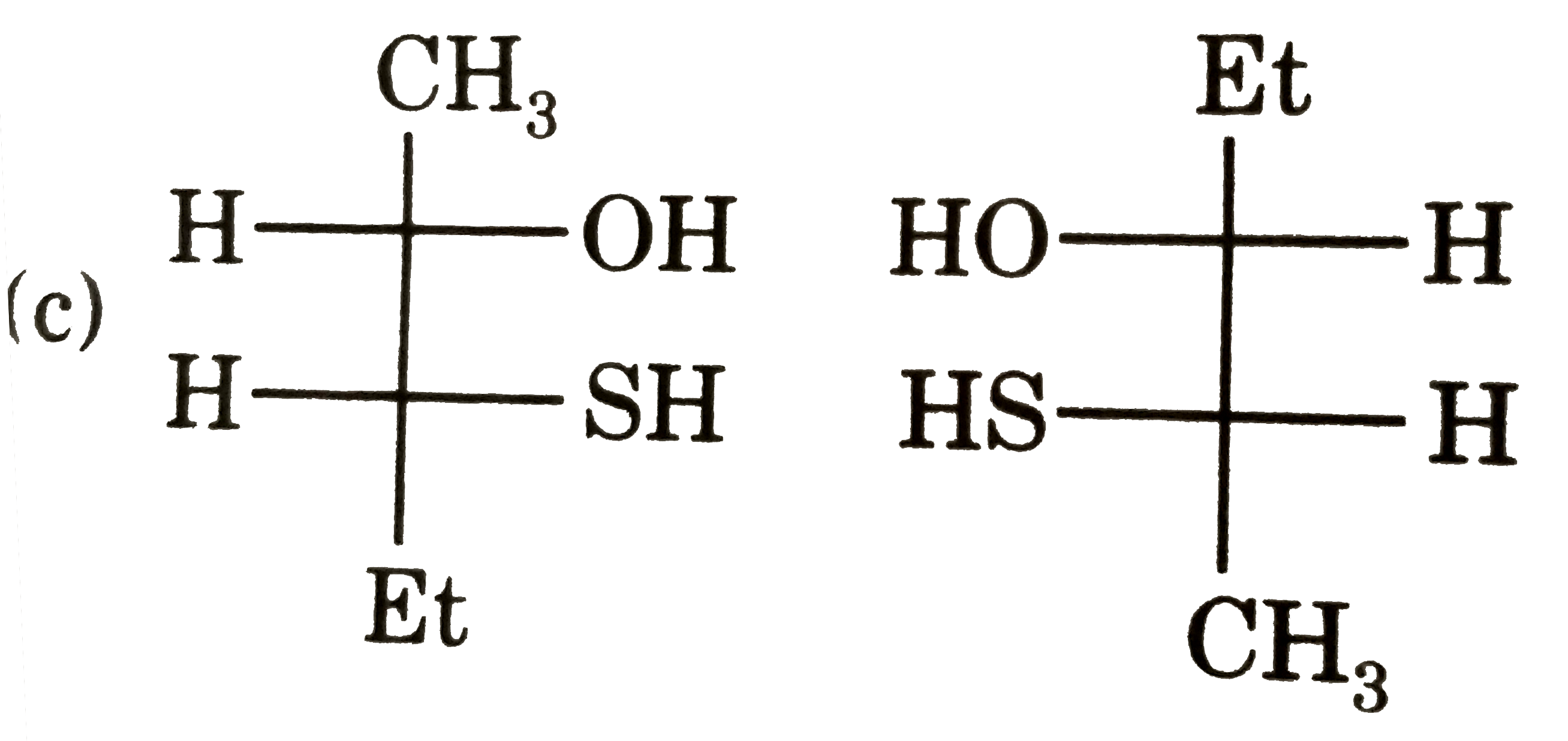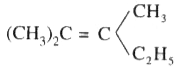Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What isindicatedby n, land m_(l)? |
|
Answer» SOLUTION :(N )Principalquantumnumber(n ) :it givesthat energylevelor energystate. (l ) Angularquantumnumber: itgivesorientationof ORIBITALS `(m_(1)`Magneticquantumnumber: itgivesorientationof oribitals . |
|
| 2. |
To a container containing 3 moles of C_(2)H_(6)further of gram C_(2)H_(6) is added and 2.4 xx 10^(24)molecules of gas are then removed. The left ovr gas is burnt in the presence of excess oxygen . Then: |
|
Answer» 60 gms of `C_(2)H_(6)` are left for combustion |
|
| 3. |
Which positive and negative ion containing solution are acidic (pH lt 7) ? |
| Answer» Solution :If the negative IONS are `CL^(-), NO_3^(-) , SO_4^(2-)`and positive ion is EXCEPT `Na^(+), K^(+) ,BA^(2+)`than the solution of such salt is always acidic. | |
| 4. |
The starting compound A in the reaction is |
|
Answer» Phenol 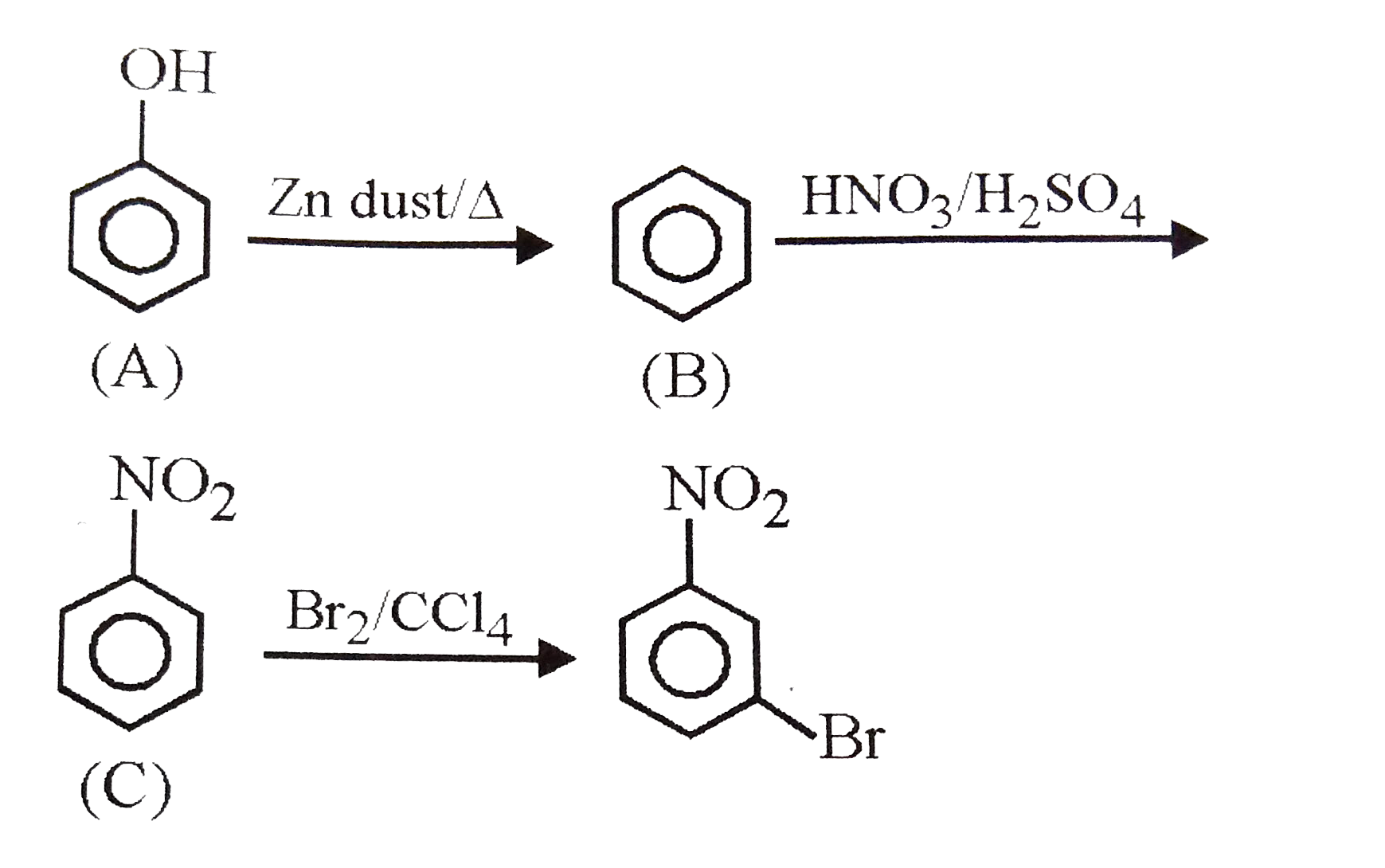
|
|
| 5. |
Which of the following reactions are correctly represented? |
|
Answer» `R - CH = CH_2 + HCL to R- underset(Cl)underset(|)CH-CH_3`<BR>`R-CH= CH_2 + HI underset(hv)overset(H_2O_2) to R-CH_2-CH-2 -I` |
|
| 6. |
Which of the following reagents does not react with acetic acid to form acetyl chloride |
|
Answer» `SO_(2)Cl_(2)` |
|
| 7. |
Variable valency is exhibited by |
|
Answer» NORMAL ELEMENTS |
|
| 9. |
What is metal displacement redox reaction? |
| Answer» SOLUTION :A more electropositive element can displace a less than electropositive METAL IRON from its compounds Most of these reactions are used in METALLURGICAL PROCESSES. | |
| 10. |
Which one has a greater electron affinity, chlorine or fluorine. Explain why? |
| Answer» SOLUTION :CHLORINE. Larger size OFCL can accomodate the extra electron easily. But size of second period element fluorine is very small. Thus difficult to hold 1 extra electron as columbic repulsion will be very high. | |
| 11. |
Which of the following reagent can be used to convert trans-2-butene to racemic 2,3-butanediol |
|
Answer» Cold alk, `KMnO_(4)` 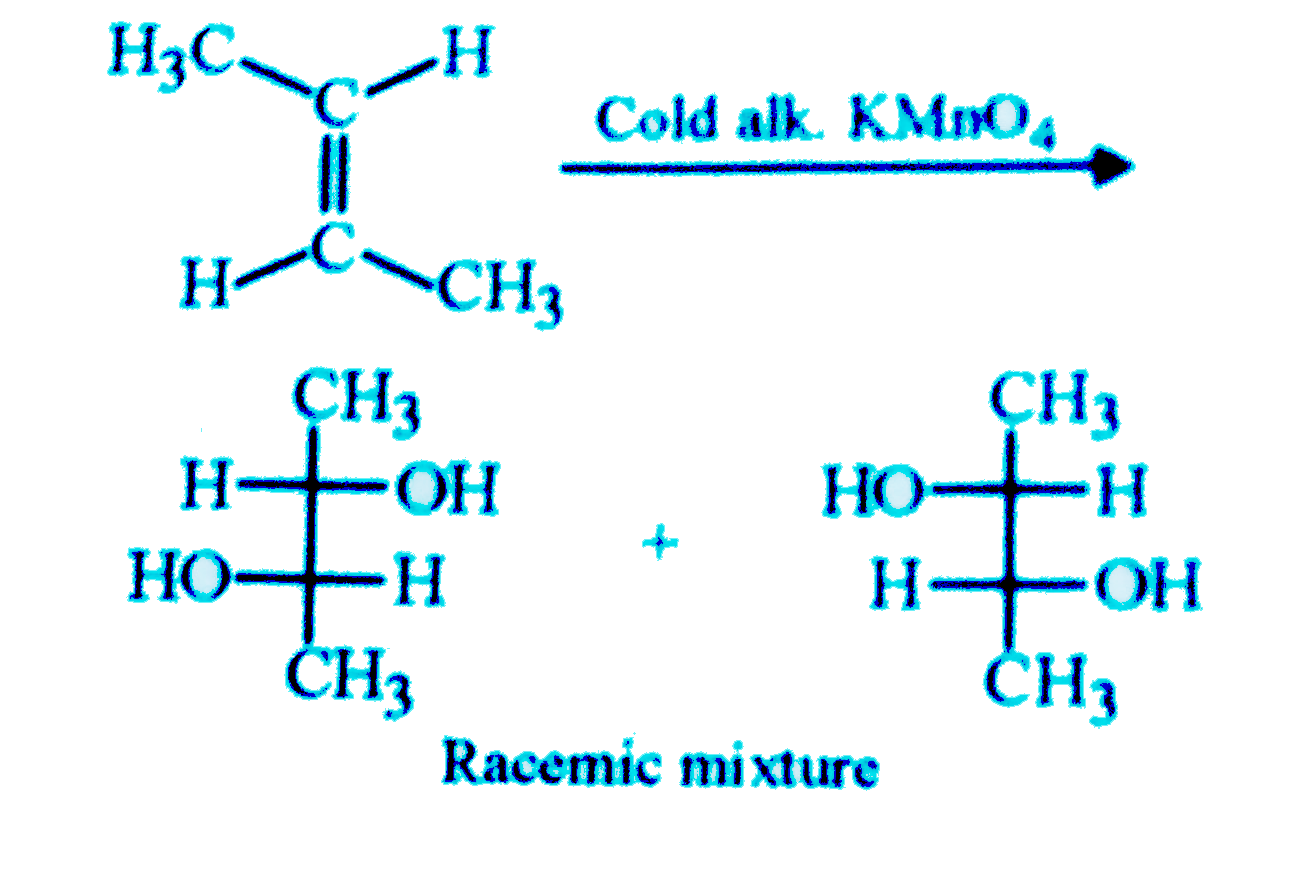
|
|
| 12. |
Which of the followingg elements does not form hydride by direct heating with dihydrogen? |
|
Answer» `Be` `BeH_(2)`, HOWEVER, can be prepared by the reaction of `BeCl_(2)` with `LiAlH_(4)`. `2BeCl_(2)+LiAlH_(4) to 2BeH_(2)+LiCl+AlCl_(3)`. |
|
| 13. |
The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in frequencies (Hz) (1 nm = 10^(-9)m) |
|
Answer» Solution :WAVELENGTH of violet radiation `= 400NM = 400 xx 10^(-9) m` `:.` Frequency `(v) = (c)/(lamda) = (3 xx 10^(8) ms^(-1))/(400 xx 10^(-9) m) = 7.5 xx 10^(14) s^(-1) or 7.5 xx 10^(14)HZ` Wavelength of RED radiation `= 750nm = 750 xx 10^(-9)m` `:.` Frequency `(v) = (c)/(lamda) = (3 xx 10^(8) ms^(-1))/(750 xx 10^(-9)m) = 4.00 xx 10^(14) s^(-1) or 4.00 xx 10^(14)Hz` Thus, in terms of frequency, the range of the visible lights is from `4.0 xx 10^(14) Hz " to " 7.5 xx 10^(14)Hz` |
|
| 14. |
Which orbital are overleap and form pi -bond ? When? |
|
Answer» SOLUTION :`pi`-bond is formed by overlapping of `2p_(X) - 2p_(x) and 2p_(y) -2p_(y)` orbitals. In this the axes of ATOMIC orbitals that are overlapping REMAINS parallel to each other and it is perpendicular to. INTERNUCLEAR axis. |
|
| 15. |
Which of the following statements is/are correct about the reaction. H_(2)SO_(4)+Cu_(3)P+Cr_(2)O_(7)^(2-)toCuSO_(4)+H_(3)PO_(4)+2Cr^(3+) |
|
Answer» The number of moles of `Cr_(2)O_(7)^(2-)` required to oxidise 6 " mol of "`Cu_(3)P` to `CuSO_(4)` and `H_(2)PO_(4)` is 11 mol. `Cr^(1+)to3Cu^(2+)3e^(-)` ("oxidation")` `underline(underset(x=-3)(P^(3-))toundersetunderset(x=5)(x+x-8=0)(H_(3)PO_(4)))` `underline(4H_(2)O+Cu_(3)Pto3Cu^(2+)+H_(2)PO_(4)+11e^(-)+5H^(o+))` REDUCTION: `Cr_(2)O_(7)^(2-)+6e^(2-)+14H^(o+)to2Cr^(3+)+7H_(2)O`..(ii) Multiply equation (i) and 6 and equation (ii) by 11, net redox equation is: 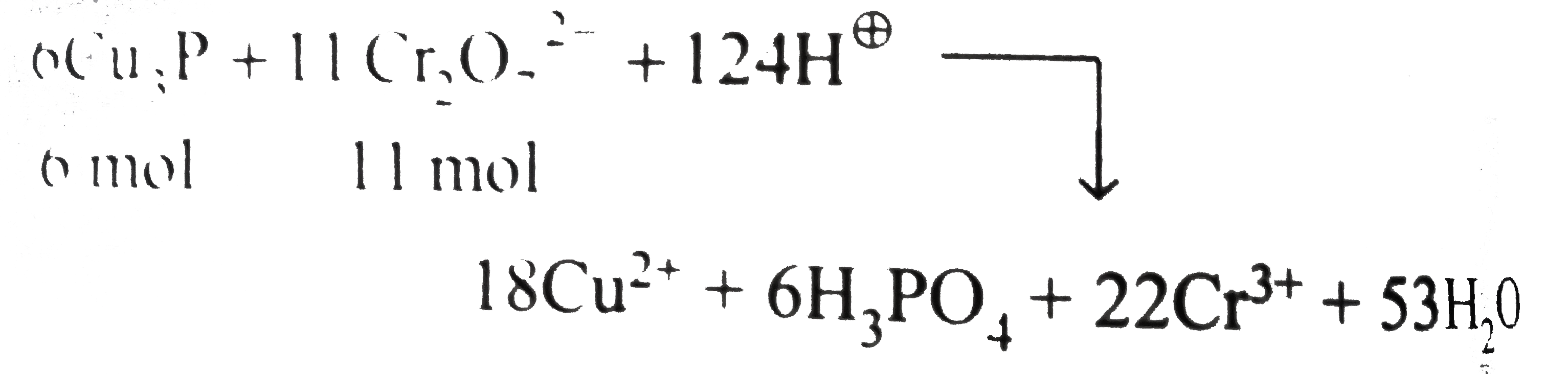 (ii). Number of moles of `H_(2)SO_(4)` used `=62H_(2)SO_(4)` `(=124H^(o+)=62SO_(4)^(2-))` Number of moles of `CuSO_(4)` formed`=18CuSO_(4)(=18SO_(4)^(2-))` ltBrgt Number of moles of `Cr_(2)(SO_(4))_(3)` formed`=11Cr_(2)(SO_(4))_(3)` ltBrgt `(=33SO_(4)^(2-))` Total number of `SO_(4)^(2-)` ION in reactant `=62` Total number of `SO_(4)^(2-)` ion in product`=18+33=51` Rest 11 " mol of "`SO_(4)^(2-)` ion in the reactant react with `(11)/(2mol)` of `K^(o+)` ion to give 11 " mol of "`K_(2)SO_(4)`. Net redox reaction is: `6Cu_(3)P+11K_(2)Cr_(2)O_(7)+62H_(2)SO_(4)to18CuSO_(4)` `+6H_(3)PO_(4)+43H_(2)O+11Cr_(2)(SO_(4))_(3)+11K_(2)SO_(4)` |
|
| 16. |
Which of these will not react with acetylene ? |
|
Answer» NAOH  `HC-=C^-` is a STRONGER BASE than NaOH |
|
| 17. |
Which conformer of ethane has maximum energy |
|
Answer» SKEW |
|
| 18. |
Which is not correctly matched ? {:((a) " Basic strength of oxides",,Cs_(2)O lt Rb_(2)O lt K_(2)O lt Na_(2)O lt Li_(2)O),((b)" Stability of peroxides",,Na_(2)O_(2) lt K_(2)O_(2) lt Rb_(2)O_(2) lt Cs_(2)O_(2)),((c )" Stability of bicarbonates",,LiHCO_(3) lt NaHCO_(3) lt KHCO_(3) lt RbHCO_(3) lt CsHCO_(3)),((d)" Melting point",,NaF lt NaCl lt NaBr lt Nal):} |
|
Answer» a and d `L.E.a(1)/(gamma_(+)+gamma_(_))""`,`""` moreis L.E. more is melting POINT`"":. NaF GT NaCl gt NaBr gt Nal` |
|
| 19. |
Which of the following oxide of nitrogen is ionic- |
|
Answer» `NO` |
|
| 20. |
What is the bond length of C-C in benzene? |
|
Answer» 139pm |
|
| 21. |
Which bond do you expect to be stronger in each of the following cases and why ?(i)H - H , Cl - Cl(ii)O _(2), N_(2)(iii)F - F , Cl - Cl |
|
Answer» Solution :(i) H - His stronger BECAUSEOF smaller size of H - atoms . (ii) ` N-= N` is stronger because it contains a trople bond while ` O_(2)` contains a bouble bond ` (O = O)` . (iii) ` Cl - Cl` is stronger becausa irepulsions betweens the two F-atoms in ` F_(2)`are larger ONO account of greater electrons density around F-atom due to smaller size ANS hence greater repulsions between the twoF-atoms . |
|
| 22. |
Which is the following metallic oxidesexhibit amphoteric nature ? |
|
Answer» `BAO` |
|
| 23. |
What is inorganic benzene ? Why is it so called ? How will you get it from diborane ? |
Answer» SOLUTION :Borazine or borzole `(B_(3)N_(3)H_(6)`) is known as inorganic benzene . It is called because and the structure of borazine is similar to that of benzene. It is also isoelectronic and isosteric with benzene . Like carbon in benzene both N and b B in borazine are `sp^(2)` hybridised. Each N has a p-orbital which is perpendicular to the `alpha`- bonding orbitals and contains a lone pair of electrons. In contrast each B has an empty P - orbital which is also perpendicular to the plane of the ring. THUS the x-bonding in borazine is DATIVE and it arises from the sideways and overlaps the fully filled orbitals of N and empty P-orbitals of B  Preparation of borazine : At low temerature diborane combines with ammonia to form an addition compound. `B_(2)H_(6)+2NH_(3) overset("Low temp ") RARR B_(2)H_(6).2NH_(3)` When this addition compound is heated to 473 K, it decomposes to form a volatile compound called borazine . `3B_(2)H_(6).2NH_(3) overset(473 K) rarr UNDERSET("Borazine" )(2B_(3)N_(3)H_(6))+12H_(2)` |
|
| 24. |
What is meant by binary hydride? Give example. |
| Answer» Solution :A BINARY hydride is a COMPOUND formed by hydrogen with other ELECTRO positive elements including METALS and non-metals, e.g., LiH or `MgH_2` | |
| 25. |
The statementthat isnot correctfor periodic classification element is : |
|
Answer» The propertiesof elementare PERIODICFUNCTION of their atomicnumbers. |
|
| 26. |
What is B? |
|
Answer» `CdS` (D) `Cd(NO_(3))_(2)` (E) `K_(2)[Cd(CN)_(4)]` (F) `[Cd(NH_(3))_(4)]^(2+)` |
|
| 27. |
Under pressure, (with hydrogen) in presenceof nickel catalyst, benzene forms |
|
Answer»
|
|
| 28. |
use the thermodymanmic information : (1)/(2)N_(2)(g)+(1)/(2)O_(2)(g)DeltaH^(@)=90.4Kj//mol (1)/(2)N_(2)(g)+O_(2)(g) DeltaH^(@)=33.8KJ//mol 2NO_(2)(g)toN_(2)O_(4)(g) DeltaH^(@)=-58.0KJ//mol to Calcualate DeltaH^(@) Kj /mol for thereaction : 2NO_(2)(g)+O_(2)(g)toN_(2)O_(4) |
|
Answer» `-171.2` |
|
| 29. |
When KI in acidic medium were mixed in 50ml H_2O_2liberated I_2requires 20ml of 0.1N hypo. What is strength of H_2O_2. |
|
Answer» 6.8 g/litre No of milli equivalents of `H_(2)O_(2)= 20 xx 0.1= 2` No of milli moles of `H_2O_2=1` `:.` 1Lof `H_2 O_2`contain `1000/50 =20`millimoles `:.` strength = `20 xx 34 xx 10^(-3)` g = 0.68 g/L |
|
| 30. |
Write the half eaction for the following redox reac tiojns (a)2Fe^(3+)(aQ)+2I^(-)(aq) rarr 2 Fe^(2+)(aq)+I_(2)(aq) (b)Zn(s)++2 H^(+)(aq)rarr Zn (c )AI(s)+3Ag^(+)(aQ)rarrAI^(3)rarr AI^(3+)(aq)+3Ag (s) |
|
Answer» `therefore E_(Zn,Zn^(2+))=0763 V therefore E_(Zn^(2+)//Zn)^(I@)=-0.763 V "and" therefore E_(CD,Cd^(2+))^(@)=0.403V therefore E_(Cd(2+,Cd))^(@)=0.403V` SINCE `Zn^(2+)//Zn` electrode is at lower potential therefore it acts as the anode while `Cd^(2+)//Cd` electrode with higher potential acts as the cathode In other owrds Zmn loses electrons and `Cd^(2+)` iion ACCEPTS them Therefore cell reaction is `Zn+Cd^(2+)rarrZn^(2+)+Cd` and `E_(Cell)^(@)=E_(Cd^(2+),Cd)^(-E^(@))(Zn^(2+)Zn)=-0.04030-(-0.763)=+0.360V` |
|
| 31. |
What will be the mass of one atom of C-12 in grams ? |
|
Answer» Solution :The MASS of a `""^(12)C` (carbon-12) atom was determined by a mass spectrometer and it was found to be EQUAL to `1.992648xx10^(-23)` g. It is KNOWN that 1 mole of `""^(12)C` weighing 12 g contains `N_(A)` number of ATOMS.Thus, 1 mole of `""^(12)C` atoms `= 12 g = 6.022xx10^(23)` atoms `6.022xx10^(23)` atoms of `""^(12)C` have mass `= 12g` `:.` 1 atom of `""^(12)C` atom will have mass `= (12)/(6.022xx10^(23))g` `= 1.992648xx10^(-23)g ~~1.99xx10^(-23)g` |
|
| 32. |
When thereare two electron is thesame orbitals , they have ……………spins. |
| Answer» SOLUTION :OPPOSITE | |
| 33. |
What is the magnetic nature of oxygen molecule. |
| Answer» SOLUTION :PARAMAGNETIC. | |
| 34. |
Which one of the following metallic hydroxides does not dissolve in sodium hydroxide solution ? |
|
Answer» `Al(OH)_(3)` |
|
| 35. |
Which of the following can restore the colour of the lead paintings |
|
Answer» HYDROGEN |
|
| 36. |
What causes the temporary and permanent hardness of water? |
| Answer» SOLUTION :Temporary hardness of water is due to the presence of bicarbonates of calcium and MAGNESIUM in water i.e., `Ca(HCO_(3))_(2)`, and `Mg(HCO_(3))` in water. Permanent hardness of water is due to the presence of soluble chlorides and sulphates of calcium and magnesium ie,` CaCl_(2)`, `CaSO_(4)`. `MgCl_(2)` and `MgSO_(4)`. | |
| 37. |
Useually ina redox reaction one substance is oxidised and the toher is reduced can you thing of an inorganic compound which undergoes intramolecular redox reaction ? |
|
Answer» Solution :Intramolecularredox reaction can occur if the cation is oxidised nad the anin is reduced for EXAPLE decomposition of ammounium DICHROMATE may be regarded as an intrmolicular redox reaction in which O.N of N INCREASES form -3 to 0 nad that of Cr decreases from +6 to 3 i.e `NH_(4)^(+)` is oxidised to `N_(2)` while `Cr_(2)O_(7)^(2-)` is reduced to `Cr_(2)O_(3)` `(NH_(4))_(2)overset(+6)Cr_(2)O_(7) to overset(0)N_(2)+overset(+3)Cr_(2)O_(3)+4H_(2)O` |
|
| 38. |
Which emits beta-particles ? |
|
Answer» `""_(1)H^(3)` `""_(6)^(14)C to ""_(7)^(14)N + ""_(-1)e^(0)` n/p =1.33 `""` n/p =1 |
|
| 39. |
Which of the following properties of the aqueous solution of AlCl_3 ? |
|
Answer» Amphoteric |
|
| 40. |
What is the relationship between pK_(a) and pK_(b) values where K_(a) and K_(b) represent constants of the acid and its conjugate base respectively ? |
| Answer» SOLUTION :`pK_(a) + pK_(B) = pK_(W)= 14`. | |
| 41. |
whichof thefollowinghas maximumboilingpoint ? |
|
Answer»
|
|
| 42. |
What is the different betweeen distillation distillation under reduced pressure, and steam distillation? |
| Answer» Solution :Disstillation involves the conversion of a liquid into vepour by HEATING followed by the condensation of the vapour thus produced by cooling. This method is commonly used for those LIQUIDS which are SUFFICIENTLY stable at their boiling points and which contain non-volatile impurities for example simple organic liquids such as BENZENE, ethanol ecetone, chloroform carbon, tetrachloride, toluence, etc. , can be purified by simple distillation. Distillation under reduced pressure. On the other hand, is applicable for the purificatgion of liquids with high boiling points and liquids which decompose at or below their boiling. A liquid boils when its vapour pressure becomes equal to the external pressure. Clearly. the same liquid will boil at a lower temperature if the pressure on it is reduced. Since the liquid now boils at a lower temperature, the decomposition of the liquid does not occur. Steam distillation is used for the separation and purification of organic compounds (solid or liquid) from non-volatile organic or inorganic impurities. This method is applicable to only those sompounds which are volatile is steam, insolubl in WATER, possess high vapour pressure, and contain non-volatile impurities. | |
| 43. |
What are the number of bond pairs and lone pairs of electrons of N-atom in NO_(3)^(-) ? |
|
Answer» One O - atom forms a double BOND. Other two O-atoms shared with two electrons of N-atom. `:.` No. of bond PAIRS = 4 No. of LONE pairs = No. of valence electron-Bonding pairs = 4 - 4 = 0 `:.` No. of lone pairs = 0 |
|
| 44. |
What is the resultant solution is called if CC14 is used? |
|
Answer» AQUEOUS SOLUTION |
|
| 45. |
Which one of the following reactions is not a redox reaction ? |
|
Answer» `H^(+)+OH^(-)toH_(2)O` |
|
| 46. |
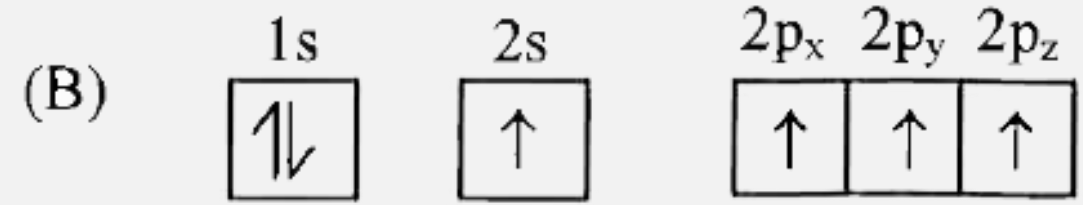
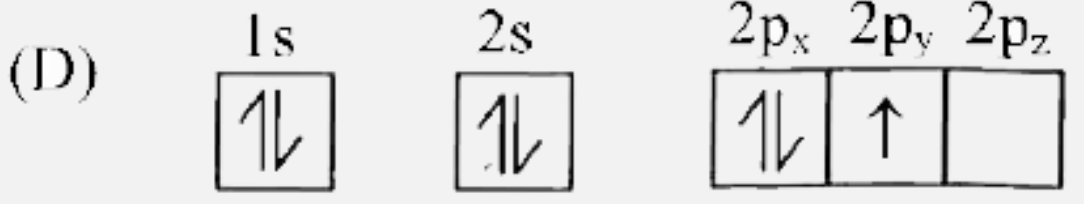
Which of the following is the electronic configuration of nitrogen atom in ground state? |
|
Answer»
|
|
| 47. |
What do you mean by the (i) acceptable values of E and (ii) corresponding wave functions that are obtained by solving the Schrodinger wave equation for H-atom? |
|
Answer» SOLUTION :(i) eiger VALUES (II) EIGEN FUNCTIONS. |
|
| 48. |
What is the normality of 4.9 g/ L solution of H_(2)SO_(4) at 25 ""^(@)C ? |
|
Answer» 0.2 Normality `= ("gm/lit")/("equivalent wt.") = (4.9)/(49) =0.1` |
|
| 49. |
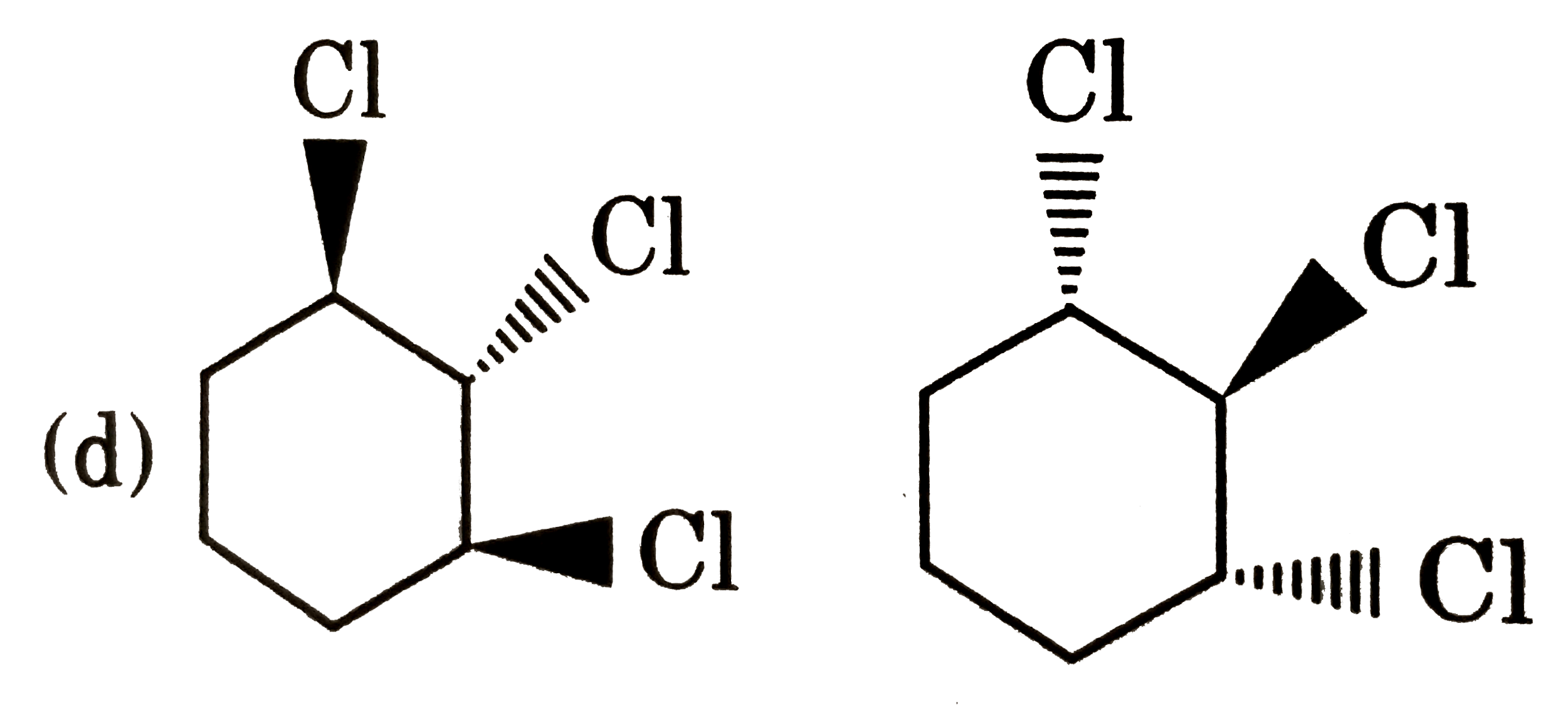
Which of following pair is diastereomers? |
|
Answer»
|
|