Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following exhibit electromeric effect ? |
|
Answer» Alkanes |
|
| 2. |
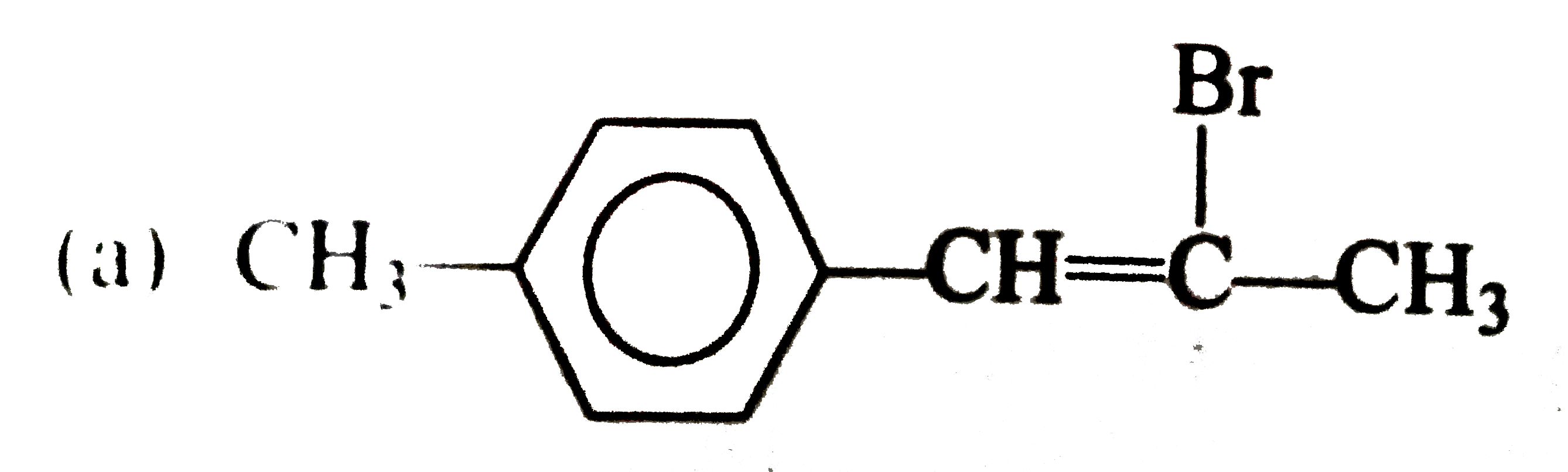
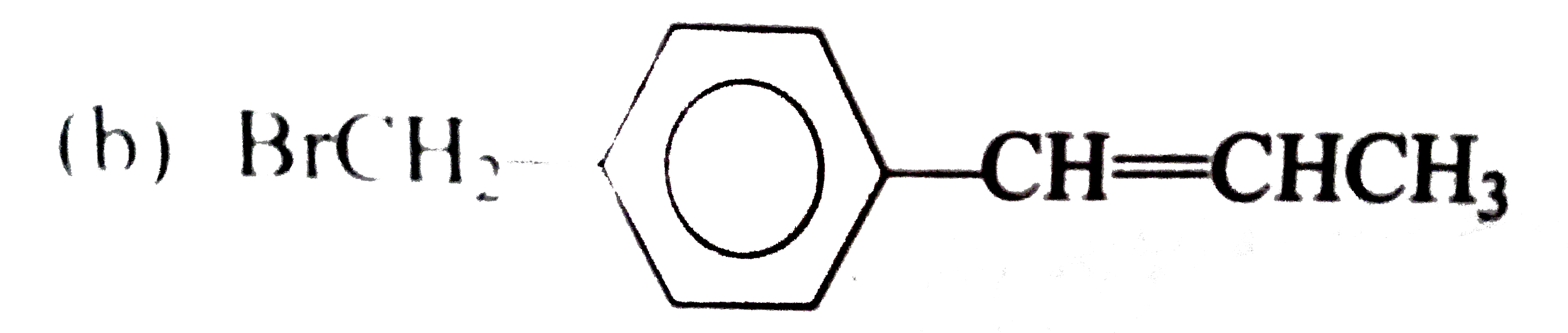
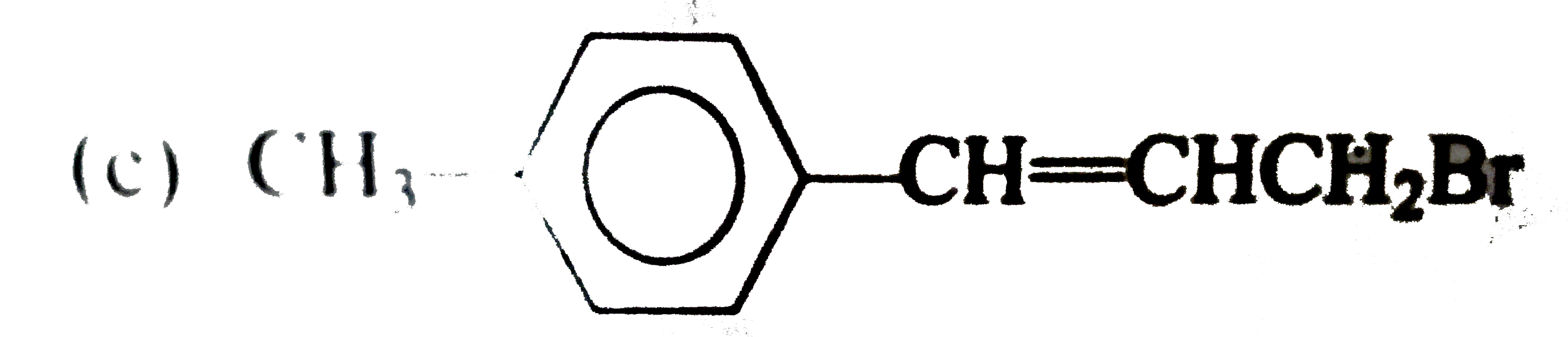
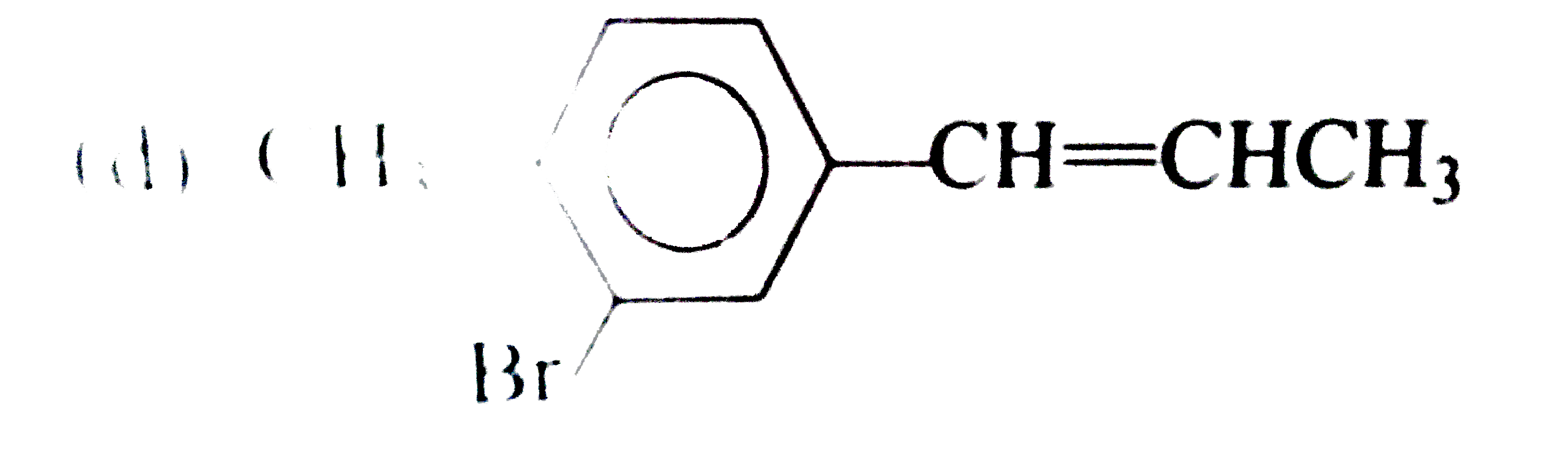
Which one of the following compounds is an allylic halide? |
|
Answer»
|
|
| 3. |
Which of the following are wrong ? |
|
Answer» DE Broglie relation is applicable only to microscopic particles |
|
| 4. |
The temporary effect in which there is complete transfer of a shared pair of pi-electrons to one of the atoms joined by a multiple bond on the demand of an attacking reagent is called |
|
Answer» INDUCTIVE effect |
|
| 5. |
Themajor constituent of natural gas is |
|
Answer» methane |
|
| 6. |
X-ray diffraction studies show that of copper crystallizes in an fcc unit cell with cell edge of 3.608xx10^(-8) cm. In a separate experiment, copper is determined to have a density of 8.92 g//cm^3, Calculate the atomic mass of copper . |
|
Answer» SOLUTION :`RHO=(ZxxM)/(a^3xxN_0)` or `M=(rhoxxa^3xxN_0)/Z` For fcc lattice, Z=4 HENCE, `M=((8.92 G cm^(-3))(3.608xx10^(-8) cm)^3 (6.022xx10^23 "atom mol"^(-1)))/"4 atoms"=63.1 g mol^(-1)` `therefore` Atomic mass of copper =`63.1 g mol^(-1)` |
|
| 7. |
Which of the following represent the correct IUPAC name for the compounds concerned : (i) But-3-yn-1-ol or But-4-ol-1-yne (ii) 4-Chloro-2-methylpentane or 2-Chloro-4-methylpentane (iii) 2, 2,-Dimethylpentane or 2-Dimethylpentane (iv) 2, 3- Dimethylpentane or 3, 4,-Dimethylpentane (v) 2, 4, 7- Trimethyloctane or 2, 5, 7-Trimethyloctane. |
|
Answer» Solution :(i) But -3- yn -1 ol : ` Hoverset(4)C = overset(3)C- overset(2)CH_2 - overset(1) CH_2 OH""`Functional group (-OH) gets preference in numbering the carbon chain. (ii) 2-Chloro--4-methylpentane : `""H_3 overset(1)C -UNDERSET(underset(CI)|)overset(2)CH - overset(3)CH_2 - underset(underset(CH_3)|)overset(4) CH - overset(5)CH_3""`Chloro substituent gets preference in numbering. (iii) 2, 2 -Dimethylpentane : `H_2 overset(1)C - underset(underset(CH_2)|)overset(underset(2)overset(CH_2)|)C - overset(3)CH_2 - overset(4) CH _2 -overset(5) CH_2""` Position of both the SUBSTITUENTS are indicated separately. (iv) 2, 3-Dimethylpentane : `overset(1) CH_3 - overset(2)underset(underset(CH_3)|) CH - overset(3) underset(underset(CH_3)|) CH - overset(4) CH_2 - overset(5) CH_2""` It has lowest set of locants. (v) 2, 4, 7-Trimethyloctane : `overset(1) CH_2 - underset(underset(CH_3)|)overset(2) CH - overset(3)CH_2 - underset(underset(CH_3)|)overset(4)CH- overset(5) CH_2 - overset(6) CH_6 - underset(underset(CH_3)|)overset(7)CH -overset(8) CH_3""` It has lowest set of locants. |
|
| 8. |
Which of the following is correct representation of Lewis structure of given species? |
|
Answer»
|
|
| 9. |
What is the oxidation number of underlined nitrogen in NH_(4)underlineNO_(3) ? |
|
Answer» Solution :`NH_(4)NO_(3)` `NH_(4)+N+3(O)=0` `therefore+1+N+3(-2)=0` `thereforeN=6-1=+5` |
|
| 10. |
What is Mathematical representation of partial gas of Dalton ? |
|
Answer» Solution :`p_("TOTAL")=p_(1)+p_(2)+p_(3)+…….p_(n) ""` …..(i) `= (n_(1)+n_(2)+….) (RT)/(V) ""` …..(ii) |
|
| 11. |
Which has bigger electron affinity in each pair? (a)F or Cl^- ,(b)O or O^- , (c)Na^+ or F ,(d)F or F^- |
|
Answer» |
|
| 12. |
What happens when a mixture of ammonium sulphate and sodium nitrate is heated. Write the equation. |
|
Answer» Solution :Double decomposition first occurs to form ammonium nitrate which then decomposes to yield laughing gas `(N_(2)O)` as SHOWN in above. `(NH_(4))_(2)SO_(4)+2NaNO_(3)tounderset("AMM. nitrate")(2NH_(4)NO_(3))+Na_(2)SO_(4)` |
|
| 13. |
Which of the following regarding 1,2 or 1,4-conjugate addition of HBr to 1,3-butadiene is false? |
|
Answer» the kinetic product 3-bromo-1-butane, ARISES from the most stable carbocation intermediate |
|
| 14. |
Which isotope of hydrogen is used as a tracer in organic reactions. |
| Answer» Solution :Hydrogen has three isotopes , viz, H, D and T . Due to DIFFERENCE in MASSES, the rate constants of these isotopes with the same substrate are different . In other WORDS , both D and T show isotope effect. But since T is not only radioactive but is also least abundant hydrogen isotope, therefore, D is USED as a TRACER to study the mechanism of organic reactions. | |
| 15. |
Which of the following about the electron is incorrect? |
|
Answer» It is negatively charged particle. |
|
| 16. |
Write the Schrodinger wave equation. |
|
Answer» `(del^(2)PSI)/(delx^(2))+(del^(2)psi)/(dely^(2))+(del^(2)psi)/(delz^(2))+(8pi^(2)m)/(h^(2))=(E-V)psi=0` `psi` = amplitude of wave; E = total energy of electron V = POTENTIAL energy; m = mass of electron |
|
| 17. |
Which of the following molecules represents the order of hybridisation sp^2 , sp^2, sp, spfrom left to right atoms? |
|
Answer» `CH_3- CH = CH - CH_3` 
|
|
| 18. |
Which of the following orders of ionic radii is correctly represented ? |
|
Answer» `H^(-) gt H gt H^(+)` |
|
| 19. |
Which one among the following gives a dicarbonyl compound with O_(3) followed by reduction with zinc and water |
| Answer» Answer :D | |
| 20. |
Which metal ions are present in waste of electroplating and metal refining industry? |
|
Answer» Nickel |
|
| 21. |
Write the bond line structure of HC=C-CH=CH_(2) |
Answer» SOLUTION :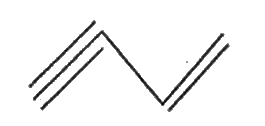
|
|
| 22. |
The weight of 112 ml of oxygen at STP, on liquifaction would be |
|
Answer» 0.32g |
|
| 24. |
Vander Waal's constants for three different gases are given: {:("Gas", a, b),(X, 3.0, 0.025),(Y,10.0,0.030),(Z,6.0, 0.035):} Which is correct? |
|
Answer» maximum CRITICAL TEMPERATURE - Y Min molecular volume = less b = x Max molecular volume = more b = z Max critical temperature = more a/b = y . |
|
| 25. |
Which of the following reagent is helpful to differentiate ethylene dichloride and ethylidene chloride? |
| Answer» Solution :aqueous KOH | |
| 26. |
What is auto - protolysis of water ? |
| Answer» | |
| 27. |
Which one of the following is used as propellant for aerosols and foams? |
|
Answer» FREONS |
|
| 29. |
The sodium salt of acid which does not form an alkane when subjected to electrolysis is |
|
Answer» SODIUM FORMATE |
|
| 30. |
Which one of the following has pentagonal bipyramidal shape? |
| Answer» Solution :`AsF_(5)` | |
| 31. |
Which of the following compounds will give blood red colour while doing the Lassaigne's test for N ? |
|
Answer» `(NH_(2))_(2)CO` |
|
| 32. |
Which d-orbital is involved insp^(3) d hydridisation and why ? |
|
Answer» SOLUTION :` d_(z^(2))` . The THREE PLANAR triangular hybrid orbitals MAY be assmed to be formed form ONE s and two p- orbitals `(sp^(2))`and the remaining p-orbital may combine with ` d_(z^(2))` to form two axial hybrid orbitals. |
|
| 33. |
Which one of the followingionshas thelargestnumberof unpairedd electrons ? |
|
Answer» `Zn^(+2)` `Fe^(+2)(A)3d^(6)` 
|
|
| 34. |
What is molar mass ? |
|
Answer»
|
|
| 35. |
The relative order of acidity of alcohols in comparison to water and ethylene is |
|
Answer» `H_(2)O gt ROH gt HC -= CH` |
|
| 36. |
What is Metamerism ? |
|
Answer» Solution :Metamerism is one kind of structural isomerism which arises due to the unequal distribution of carbon atoms on EITHER side of the functional group or different alkyl groups attached to the either side of the same functional group and having the same molecular FORMULA. e.g, `C_(4)H_(10)O` : (i) `underset("1-methoxypropane")(CH_(3)-O-CH_(2)-CH_(2)-CH_(3))` (II) `underset("2-methoxypropane")(CH_(3)-O-underset(CH_(3))underset(|)(CH)-CH_(3))` (iii) `underset("Ethoxyethane")(CH_(3)-CH_(2)-O-CH_(2)-CH_(3))` |
|
| 37. |
The stable +2 ions of lanthanides in aqeous solution are |
|
Answer» `EU^(2+)` |
|
| 38. |
Which of the following does not have zero electron gain enthalpy? |
|
Answer» Be |
|
| 39. |
The reagent(s) used for softening the temporary hardness of water area) Ca_3(PO_4)_2 b) Ca(OH)_2 c) Na_2CO_3 d) NaOCl |
|
Answer» Only a & B |
|
| 40. |
Which acid gives wine red colour with neutral FeCl_(3) |
|
Answer» PROPANOIC ACID |
|
| 42. |
Which of the following statement are correct ? (A) Sink for CO is ocean (B) Green house effect Causes lowering of temperature of earth's surface (C) To control CO emission by automobiles, usually catalytic converters fitted into exhaust pipes (D) H_(2)SO_(4), herbicides and insecticides form mists |
|
Answer» A, B |
|
| 43. |
The shapes of SF_(4) and XeF_(2)respectively are |
|
Answer» trigonal bipyramidal and trigonal bipyramidal pairs and one lone pair `XeF_(2)` has `sp^(3)d` hybridisation and linear geometry DUE to presence of THREE lone pairs. 
|
|
| 45. |
The SI unit of pressure is pascal and it is equal to the pressure exerted by |
|
Answer» a MASS of 10.2 GRAM on `1.00 cm^2` AREA |
|
| 46. |
When R-Mg - X , Grignard's Reagent is treated with solvents can yield alkanes. . What is B ? |
|
Answer» `CH_4` 
|
|
| 47. |
Zeolites are |
|
Answer» SOLUTION :Zeolites are METAL aluminosilicates with some water of hydration. These can be represented by the general formula `M_("X")[(AIO_(2))_("x")(SiO_(2))_(y)]_(n).zH_(2)O` where n is the charge on metal cation. Permutit `Na_(2)[(AIO_(2))_(2)(SiO_(2))_(2)].xH_(2)O` is an EXAMPLE of zeolite. |
|
| 48. |
Which of the following diagaram correctly represents the Boltzmann distribution of molecular speeds at two temperatures T_1 and T_2 where T_2 >T_1 ? (Proportion of molecules = f, molecular speed = v) |
|
Answer»
|
|
| 49. |
Which one of the following elements is unable to form MF_6^(-3)ion ? |
|
Answer» B |
|