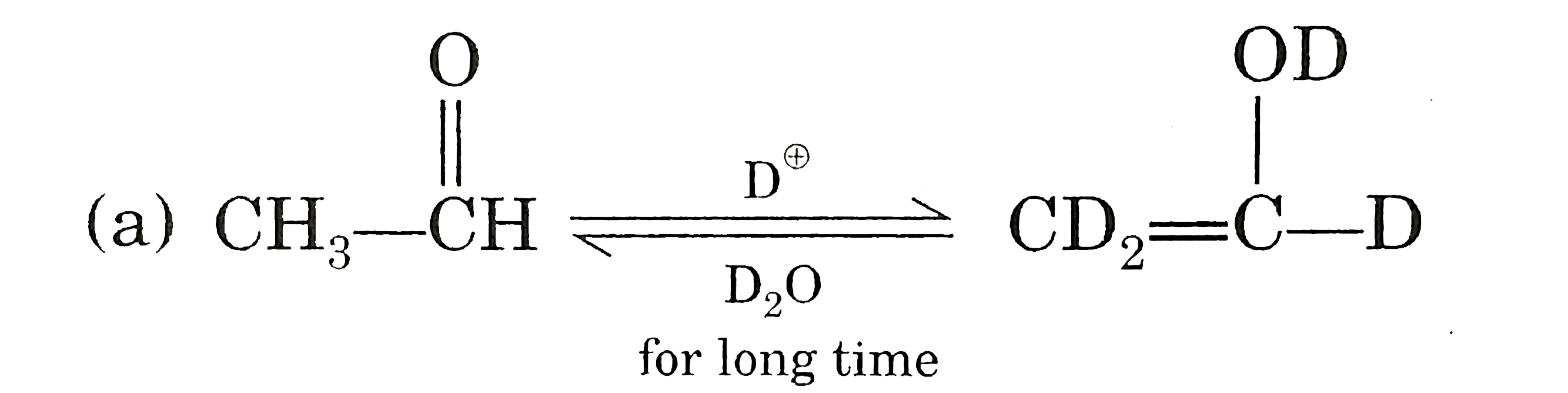Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Vander waal constant 'b' is very high for the following gas |
|
Answer» `Cl_2` |
|
| 2. |
Which of the following gas can be liquified very easily |
|
Answer» `O_(2) (T_(C) = 154.4 K)` |
|
| 3. |
The species Ar, K^(+) " and " Ca^(2+) contain the same number of electrons. In which order do their radii increase ? |
|
Answer» `Ca^(2+) lt AR lt K^(+)` `{:("Radius "alpha 1/(Z_("eff"))" for isoelectronic species "),(""Ca^(2+) ""K^(+) "Ar"),("Z201918"),(ul("No. of" BAR(E) "181818")),( leftarrow " Nuclear charge increses "leftarrow),(.:. to " Their redii increases" to ):}` |
|
| 4. |
Which of the following contents are oxidise when they react with dihydrogen |
|
Answer» CALCIUM |
|
| 5. |
Using MO theory, predict which of the following species has the shortest bond length? |
|
Answer» `O_(2)^(2+)` |
|
| 6. |
Which of the two elements, carbonand silicon, formsmultiple bonds ? |
|
Answer» Solution :Due to SMALLER size and HIGHER electronegatively , carbon UNDERGOES `ppi- ppi`overlop to form multiple bonds but silicondoes not. For EXAMPLE , `CO_(2)`has `C = O`doublebond but `SiO_(2)` has only `Si-O` single bonds. |
|
| 7. |
Which of the following is used in preparation of artifical flavours : |
|
Answer» LOWER alcohol |
|
| 8. |
Which of the following is redox reaction ? |
|
Answer» `NaOH_((aq))+HCl_((aq))toNaCl_((aq))+H_(2)O_((l))` 
|
|
| 9. |
What happens whensodium metal is heated in free supply of air ? |
|
Answer» Solution :On being HEATED in air, sodium reacts VIGOROUSLY with OXYGEN to FORM sodium peroxide. The chemical equation involved in the reaction is : `2Na_((g))+O_(2(g)) to Na_(2)O_(2(g))` |
|
| 10. |
Which one of the following is not prepared from halide by chemical oxidation process |
|
Answer» `F_(2)`<BR>`Cl_(2)` |
|
| 11. |
What is the maximum number of electrons which can be accommodated in an atom in which the highest principal quantum number is 4 ? |
|
Answer» 10 `= [Ar]^(18) 4S^(2) 3D^(10) 4p^(6)`. Total `e^(-) = 36` |
|
| 12. |
The type ofhybridisation of boron is diborane is (a) sp, (b) sp^(2) , (c ) sp^(3), (d) dsp^(2) |
| Answer» SOLUTION :In `B_(2)H_(6), B` is `SP^(3)`-hybridized. THEREFORE , option (C ) is correct. | |
| 14. |
What is the oxidation state of phosphorous element in cyclo meta phosphoric acid ? |
| Answer» ANSWER :B | |
| 15. |
Whichare the drawbacksofRutherfordatomicmodel |
| Answer» SOLUTION :Rutherfordmodelis thatnoinformationabout theelectronicstructureof ATOM can beobtained(II) i.ehowelectronsare distributedin anatom.Thenucleusand energypossessedby thenremainunknown. | |
| 16. |
What is the difference of dipole moment of NH_(3) and NF_(3) molecules ? |
| Answer» Solution :The DIPOLE MOMENT of`NH_(3).(0.80 xx 10^(-30) " CM" )`is less than the dipole moment of `NH_(3) (4.90 xx 10^(-30) "Cm")` | |
| 17. |
Write Lewis Acid-Base principle. |
|
Answer» Solution :Defination : G.N. Lewis in 1923 defined an "acid as a species which accepts electron pair and base which donates an electron pair." Comparison of Bronsted-Lowry and Lewis Base: As far as base are concerned, there is not much DIFFERENCE between Bronsted-Lowry and Lewis concept, as the has provides a lone pair in both the cases. The Lewis base `NH_3` donate electron pair to `H^+` and `NH_4^+` is farm. `NH_3` is act as base in both principle. All Lewis base will be Bronsted-Lowry base and Lewis base. Comparison of Bronsted-Lowry and Lewis Acid : In Bronsted-Lowry acid must containing PROTON. HOWEVER, in Lewis concept many acids do not have proton. Electron deficient species like `BF_3 , AlCl_3 , FeCl_3 , NO_2^(+), Mg^(2+) , Co^(3+)`etc. can be act as Lewis acids. "Thus all Lewis acid not act as Bronsted-Lowry acid." `UNDERSET"Lewis acid"(BF_3) + underset"Lewis base"( :NH_3) to F_3F: NH_3` Species like `H_2O , NH_3 , OH^-` etc. which can donate a pair of ELECTRONS can act as Lewis bases. |
|
| 18. |
Which of the following is correct for acid buiter? [salt = S, acid = A] |
|
Answer» ` P^(K_4) =P^(H) +LOG""([S])/([A]) ` |
|
| 19. |
What is the wavelengthof objecthaving0.1kg ? Whatit indicate ? |
| Answer» Solution :`lambda = 6.626 xx 10^(34) m`it is veryshort.So itdoesnothave WAVE nature . | |
| 20. |
What is the half life period of tritium? How is it undergoes radioactive disintegration? |
|
Answer» Solution :(i) The HALF life of tritium = 12.33 years (ii) Tritium is a beta-emitting radioactive ISOTOPE of HYDROGEN. `"_(1)^(3)Hto_(-1)^(0)e+_(2)^(3)He` |
|
| 21. |
Which of the following molecule acts as a Lewis acid ? |
|
Answer» `(CH_(3))_(2)O` |
|
| 22. |
What will be the mass of one ""^(12)C atom in g ? |
|
Answer» `=(12g)/(6.022xx10^(23))` `=1.9927xx10^(23)g` |
|
| 23. |
The valence shell of transition element consists of |
|
Answer» ND orbitals |
|
| 24. |
Using the standart electrode potential find out the pair between which redox reaction is not feasible E^(@) values : Fe^(3+)//Fe^(2+)=+0.77,I_(2)//I^(-)=+0.54,Cu^(2+)//Cu=+0.34,Ag^(+)//Ag=+0.80V |
|
Answer» SOLUTION :CALCULATE the `E_("cell")^(@)` of the four redox reaction If `E_("cell")^(@)` of a reaction is -ve that reaction will not occur (a) `2Fe^(3+)+2e^(-)rarr2 Fe^(2+),E^(@)=+0.77 V` `2 I^(-)rarrI_(2)+2e^(-),E^(@)=-0.54V` `2Fe^(3+)+2I^(-) rarr 2Fe^(2+)+I_(2),E_("Cell")^(@) =+0.23 v` This reaction is feasible SINCE `E_("cell")^(@)=+0.23V` (b)`CurarrCu^(2+)+2e^(-),E^(@)=-0.34 V` `2Ag^(+)+2e^(-)rarr2A,E^(@)=+0.80 V` `CU+2Ag^(+)rarr2Cu^(2+)+2agE^(@)=+0.46V` This reaction is feasible since `E^(@)` is +ve (c ) `2Fe^(3+)+2e^(-)rarr2Fe^(2+),E^(@)=+0.77V` `CurarrCu^(2+)+2e^(-),E^(@)=-0.34V` `2Fe^(3+)+Curarr2Fe^(2+)+Cu^(2+),E^(@)=+0.43 V` This reaction is Feasible since `E_("cell")^(@)` is +ve (d) `AgrarrAg^(+)+E^(-)E^(2)=-0.80 V` `Fe^(3+)+E^(-)rarrFe^(2+),E^(@)=-0.03 V` This reaction is not feasible since `E_(cell)^(@)` is -ve thus option (d) is correct |
|
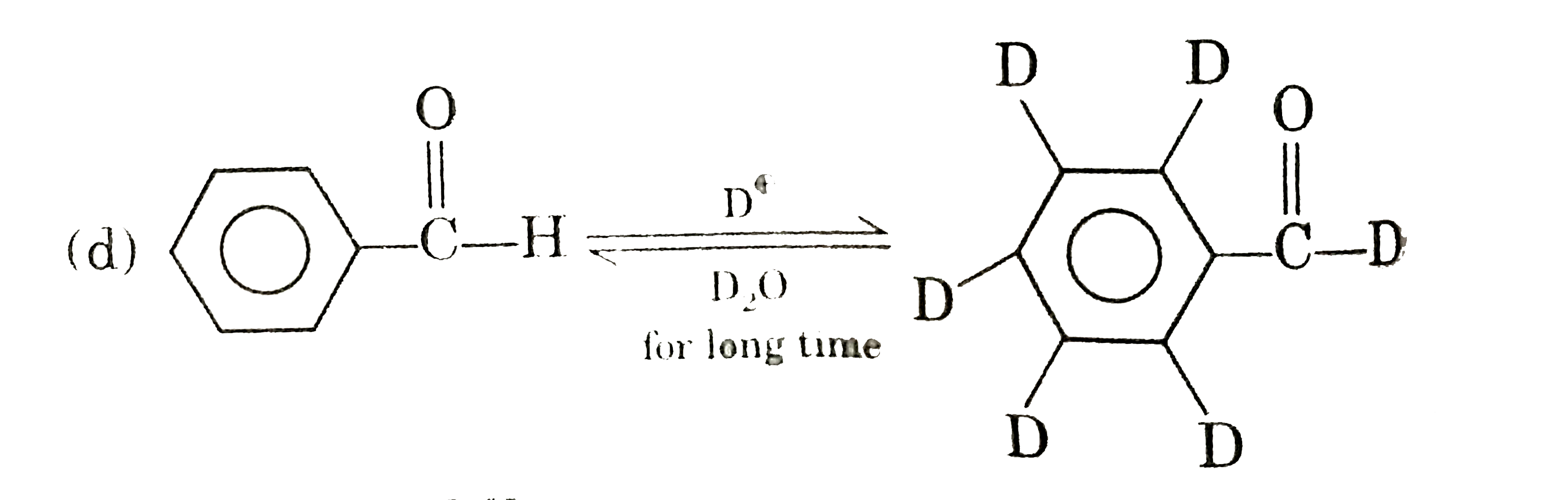
| 25. |
Which one of the following is correct ? |
|
Answer»
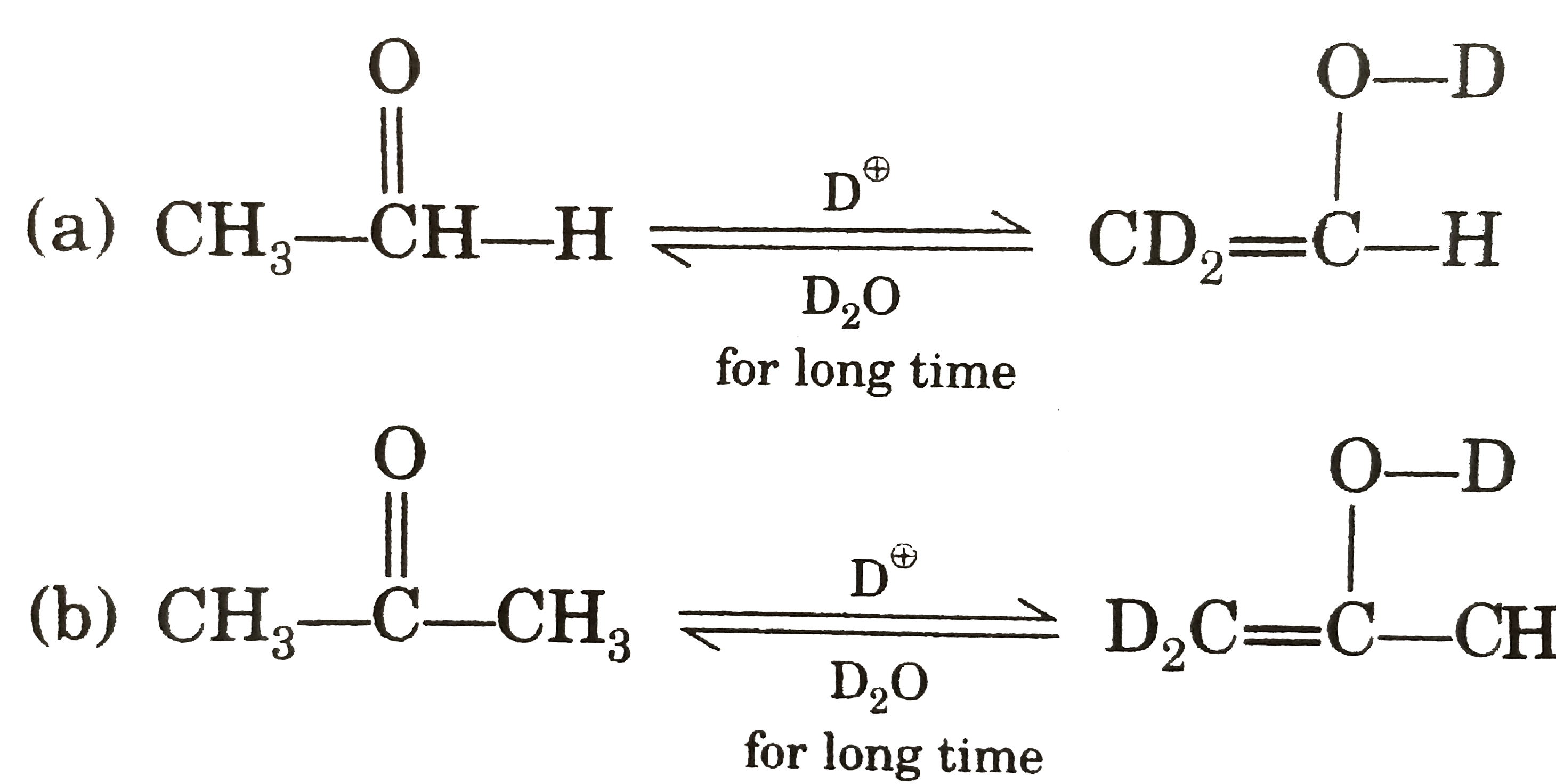 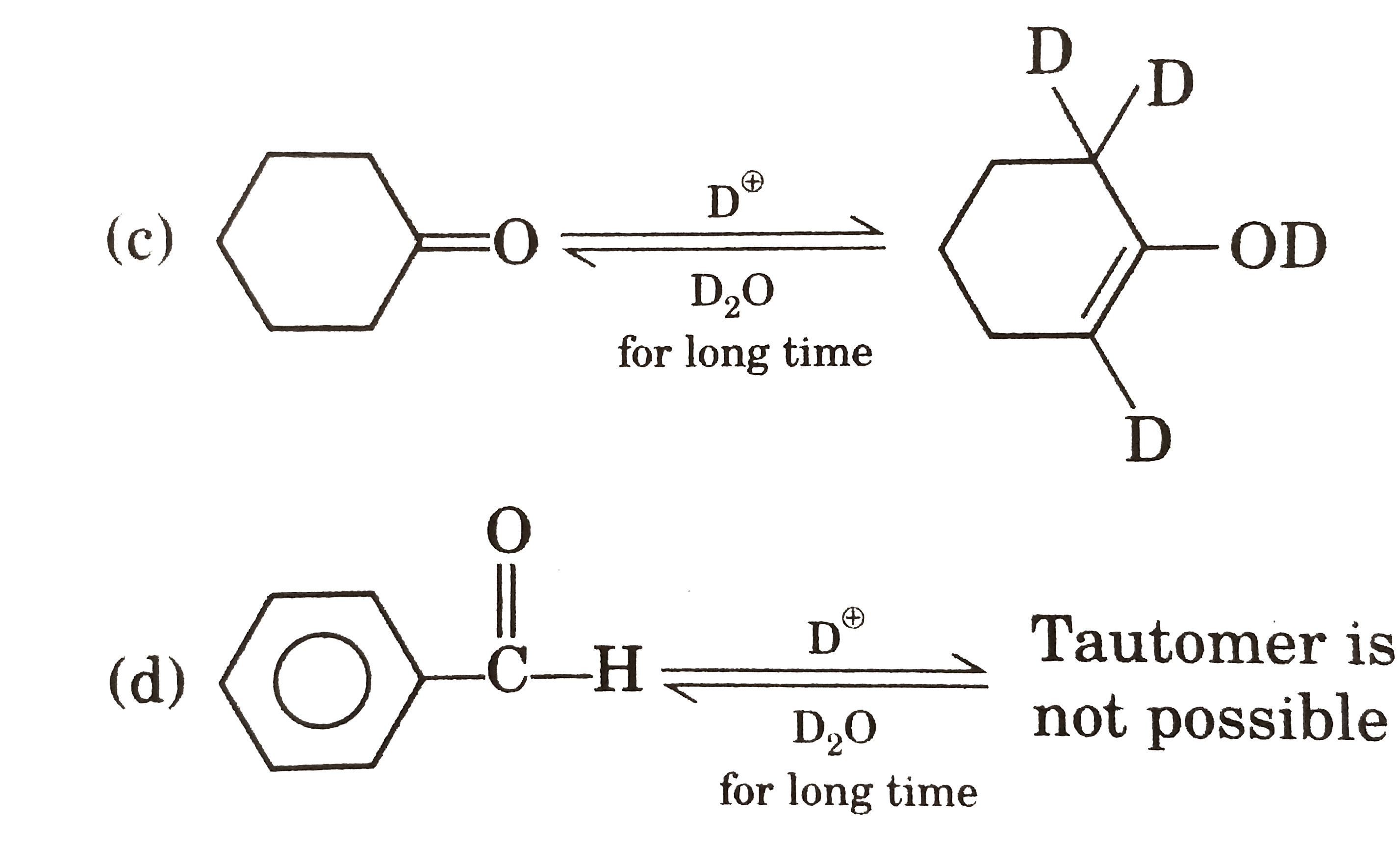
|
|
| 26. |
Treatment of phenol with nitrous acid (NaNO_(2)+HCl) gives mainly |
|
Answer» p-Nitrosophenol |
|
| 27. |
Write short notes on : (a) Resonance (b) Hyperconjuction |
|
Answer» Solution :(a) Resonance : (i) Certain organic compounds can be represented by more than one structure and they differ only in the position of bonding and lone PAIR of electrons.Such structures are called resonance structure and this phenomenon is called as MESOMERISM or mesomeric effect. (ii) For example, the structure of aromatic compounds such as benzene and conjugated system like 1,3 BUTADIENE cannot be represent by a single structure and their observed properties can be explained on the BASE of a resonance hybrid. Resonance structure of benzene.  (I) and (II) are called as resonance hybrids of benzene. (iv) For 1,3 butadine:  (I), (II) and (III) are called as resonance hybrids of 1,3 butadine. (B) Hyperconjugation: (i) The delocalization of electrons of `rho` bond is calledas hyperconjugation.It is a special stabilizing effect that results due to the interaction of electrons of a `rho` bond with the adjacent empty non-bonding P-orbitals resulting in an extended molecular orbital. (ii) Hyperconjugation is a permanent effect. (iii) For example, in propene, the `rho`-electrons of C-H bond of methyl group can be delocalised into the `pi` - orbital of doubly bonded carbon as represented below. 
|
|
| 28. |
The vapour density of a volatile chloride of a metal is 59.5 and the equivalent mass of the metal is 24. The atomic mass of the element will be |
|
Answer» 96 |
|
| 29. |
Which one is precipitated in ionic form on Ptelectode ? |
|
Answer» `Li^(++)` |
|
| 30. |
What do you mean by hydroformylation of olefins ? |
| Answer» | |
| 31. |
Which one of the following is the kinetic energy of a gaseous mixture containg 3 g of hydrogen and 80 g of oxygen at temperature T(K) ? |
|
Answer» 3RT |
|
| 32. |
What are the types of chemical equilibrium? Explain with suitable example. |
|
Answer» SOLUTION :(i) Chemical EQUILIBRIUM is of TWO TYPES: (a) Homogeneous equilibrium (b) Heterogeneous equilibrium. (ii) In a homogeneous equilibrium, all the reactants and products are in the same phase `H_(2)(g) + I_(2)(g) hArr 2HI(g)` In the above equilibrium `H_(2),I_(2)` and HI are in the gaseous state. In the above equilibrium H, I, and HI are in the gaseous state. (iii) If the reactants and products of a reaction in equilibrium are in different phases, then it is called heterogeneous equilibrium. e.g , `CaCO_(3)(s) hArr CaO(s) + CO_(2)(g)` |
|
| 33. |
Which electrons from 2s and 2p experiencing more electron from centre ? |
| Answer» SOLUTION :Electrons of 2s ORBITAL experiencing more ATTRACTION than electrons of 2p-orbital. | |
| 34. |
Which of the following cannot reduce Fehling solution ? |
|
Answer» `HCOOH` |
|
| 35. |
Which of the following electronic configuration belongs to p-block elements ? (i) 1s^(2) 2s^(2) 2p^(6) 3s^(2) "" (ii) 1s^(2)2s^(2) 3p^(6) 3d^(10) 4s^(2) (iii) 1s^(2) 2s^(2) 2p^(5) "" (iv) 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(3) |
| Answer» SOLUTION :(III) and (IV) | |
| 36. |
Which instrument is used to determine amount of K? |
|
Answer» Potentiometer |
|
| 37. |
The source of CO_(2) in the atmosphere is |
|
Answer» COMBUSTION of FUEL |
|
| 38. |
Water has maximum density and minimum volume at 4^(@)C. Explain. |
|
Answer» |
|
| 39. |
Which of the following represents correct order of conductivity in solids ? |
|
Answer» `K_"metals" GT gt K_"INSULATORS" lt K_"semiconductors"` |
|
| 40. |
When neopentyl bromide is subjected to Wurtz reaction, the product formed is: |
|
Answer» 2, 2, 4, 4-tetramethylhexane |
|
| 41. |
What will be the molarity of chloroform in the water sample which contains 15 ppm chloroform by mass? |
|
Answer» `1.25xx10^(-4)` m MOLARITY of `CHCl_3 " solution " =(1.5xx10^(-3))/(100)xx(1000)/(119.5)` `=1.25xx10^(-4)` |
|
| 42. |
Which of the following does not constitute 0.1g mole ? |
|
Answer» `6.022 xx 10^(22)` molecules of BENZENE |
|
| 43. |
Which of the following alkynes is most acidic? |
|
Answer» `CH_(3)C-=CH` |
|
| 44. |
Which is the function of sodium - potassium pump? |
|
Answer» Maintenance of ION balance |
|
| 45. |
Which of thefollowinghave nounit ? |
|
Answer» Electronegativity |
|
| 46. |
Tritium nucleus contains............ |
|
Answer» `1P +0 n` sol : `""_(1)T^(3)( I e^(-), 1p , 2n)` |
|
| 47. |
Which changes can be done due to vaporization of H_(2)O in atmosphere ? |
|
Answer» COOLING INCREASES |
|
| 48. |
The separation of the constituents of a mixture by column chromatography depends upon their |
|
Answer» DIFFERENT solubilities |
|
| 49. |
Which of the following mixture is known as producer gas ? |
|
Answer» `CO+N_2` |
|
| 50. |
Which processes will increase the number of molecules, to have an energy greater than a particular value |
|
Answer» INCREASING the temperature b) when more gas is introduced ,KE remains same but no. of molecules `darr`. C) constant `T implies` KE constant. |
|