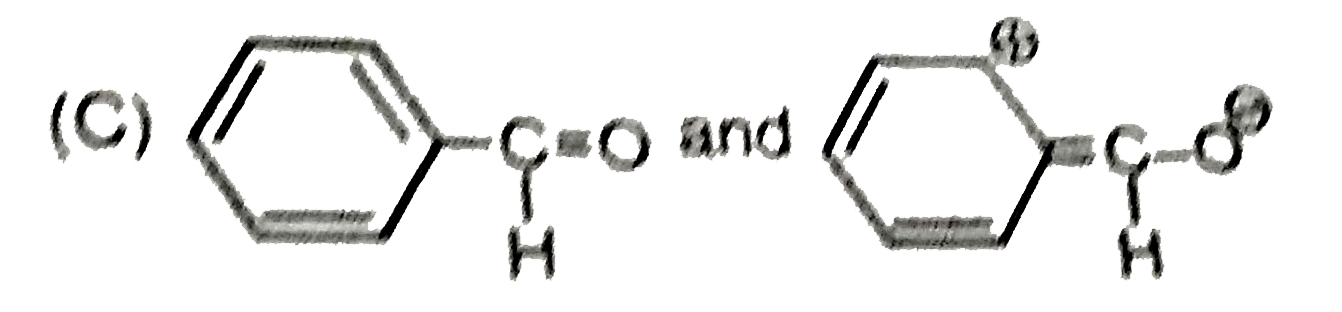Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write down the outermostelectronic configuration of alkali metals. How Justify their placement in group 1 of the periodic table ? |
|
Answer» SOLUTION :All the ELEMNTS of group 1A (alkali metals.) they have same outer electronic configuration .i.e, `ns^(1) ( :. n=no`. Of PRINCIPLE shell.) Electronic configuration is given below : 
|
|
| 2. |
Which among the following compound behave both as an electrophile as well as nucleophile (a) CH_(2) - CH_(2) (b) CH_(2) = CH - CH_(2) (c ) CH_(3) - oversetoverset(O)("||")C-CH_(3) (d) CH_(3) - oversetoverset(O)("||")C-Cl |
|
Answer» Only a |
|
| 3. |
Write Lewis dot symbols for atom of the following elements : Mg, Na , B, O, N, Br |
|
Answer» SOLUTION :`""_(12) MG = 2, 8, 2therefore `Lewis symbol =` overset(cdot )Mg*.""_(11) Na = 2,8, 1 therefore ` Lewis symbol = `overset(cdot )Na ` `._(5)N = 2, 5 therefore ` Lewis symbol= `*overset(cdot)(B )* ."" _(8)O = 2, 6therefore `Lewis symbol = `:overset(cdotcdot)(O):` ` _(7)N = 2, 5therefore `Lewissymbol= `:overset(cdotcdot)underset(cdot)(N):""_(35)Br = 2 , 8 , 28 , 7 therefore ` Lewis symbol=`:overset(cdotcdot)underset(cdotcdot )(Br):` |
|
| 4. |
The shape of acetylene molecule is ……………. |
| Answer» SOLUTION :CYLINDRICAL | |
| 5. |
Which of the following are basic |
|
Answer» `B_(2)O_(3)` |
|
| 6. |
What do the following abbreviations stand for ? (i) dL (ii) dm (iii) mu m (d) nL |
|
Answer» |
|
| 7. |
Which of the following organohalogen compounds when heated with alcoholic potassium hydroxide does not undergo dehydrohalogenation reaction ? |
|
Answer» SECONDARY butyl chloride |
|
| 8. |
What happens when KO_(2) reacts with water ? Write the balanced chemical equation for the reactions |
| Answer» SOLUTION :`2 KO_(2) + 2H_(2)O to 2 KOH+ H_(2)O_(2) + O_(2) or 4KO_(2) + 2H_(2)O to 4 KOH + 3O_(2)`. | |
| 9. |
What are alkali metals? |
| Answer» Solution :1 st GROUP ELEMENTS of periodic table i.e.,lithium, sodium, POTASSIUM, rubidium and CAESIUM are called ALKALI metals. | |
| 10. |
Uses of sodium metal is.... |
|
Answer» as a REDUCING agent |
|
| 12. |
The tautomeric pairs are |
|
Answer» `Me_(2)C=NOH and Me_(2)CH-underset(o+)N=O` |
|
| 13. |
What is the maximinn number of orbitals that can be identified with the following quantum number. |
|
Answer» 1 |
|
| 14. |
Thesize of Cl^(-)=1.81ÅandCl=0.99Å.Explain. |
|
Answer» Solution :`C1-1s^(2)2s^(2)2p^(6)3s^(2)3p^(5),C1^(-)-1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)` GENERALLY anions are larger than their parent atom `C1^(-)`had 18 electrons andC1 has 17 electrons . Thelater experiences an greater effective INWARD PULL by the nucleus. This decreases the SIZE of C1 atom when compared to `C1^(-)` ion. |
|
| 15. |
Write structures for each of the following compounds. Why are the given names incorrect ? Write correct IUPAC names : (i)2-Ethylpentane , (ii)5-Ethyl-3-methylheptane. |
|
Answer» SOLUTION :`(i)CH_3-underset("".^2CH_2overset1CH_3)underset(|)overset3(CH)-overset4CH_2overset5CH_2overset6CH_3` The longest chain here contains six carbon atoms and not five . Therefore, the correct name is 2-methylhexane. (ii)`overset7CH_3-overset6CH_2-undersetunderset(CH_3)|overset5CH-overset4CH_2-undersetunderset(""CH-CH_3)|overset3(CH)-overset2CH_2-overset1CH_3` Since 'e' COMES before 'm' in ALPHABETICAL order , therefore , NUMBERING must be done from right to left. Therefore, the correct name is 3-ethyl-5-methylheptane. |
|
| 16. |
Which one of the following characteristics is not correct for physical adsorption? |
|
Answer» ADSORPTION on solids is reversible |
|
| 17. |
Which type of hydrides are generally non-stoichiometric in nature? |
|
Answer» Metallic hydride |
|
| 18. |
What is the nuclear radius of an atom whose mass number is 125 ? |
|
Answer» SOLUTION :Nuclear radius, `r = R_(0) A^(1//3) " where " R_(0) = 1.4 xx10^(-15)m`, A = MASS NUMBER `:. "" r = (1.4 XX 10^(-15)m) xx (125)^(1//3) = 1.4 xx 10^(-15) xx 5m = 7.0 xx 10^(-15)m` |
|
| 19. |
The value of n,lm and s of 8^(th) electron in an oxygen atom are respectively………… |
| Answer» SOLUTION :2,1,+1,-1/2 | |
| 20. |
Which is least stable? |
|
Answer» `.CH_(3)` |
|
| 21. |
When Sn^(2+) changes to Sn^(4+) in a reaction,then "_____________". |
|
Answer» It is an OXIDATION as `SN^(2+)` LOSES TWO electrons |
|
| 22. |
The treatment of CH_3MgX with CH_3C -= C-H produces |
|
Answer» `CH_4` |
|
| 23. |
Which of the following compounds does not react with acetyl chloride ? |
|
Answer» Methylamine |
|
| 24. |
Which of the following reaction produces hydrogen |
|
Answer» `MG+H_2O` |
|
| 25. |
Which of the following behaves as a saturated compound? |
|
Answer» `C_2H_4` |
|
| 26. |
The size of colloidal particle is in between |
|
Answer» `10^(-7)-10^(-3) cm` |
|
| 27. |
What is responsible for the blue colour of the solution of alkali metali in liquid ammonia? Give chemical equation also. |
|
Answer» Solution :The Solvated electron, `[e(NH_(3))]^(-)` or ammoniated electron is responsible for bios COLOUR of alkali metal solution in `NH_(3)`. It absorbs light from VISIBLE region and radiates COMPLEMENTARY colour. (in the equation am = amnioniated) `Na^(+) (am) + e^(-) (am) + NH_(2) (I) to NaNH_(2) (am) + (1)/(2) H_(2) (g)` |
|
| 28. |
Write the formulae of the first 5 members of homologous series starting with the underlined CH_2=CH_2 |
|
Answer» |
|
| 29. |
Write isotopes of alkaline C_(4)H_(8) and give one examples of it. |
|
Answer» Solution :(a) Position ISOMERS : `CH_(3) - CH_(2) -C -=CH` `CH_(3)-C-=C-CH_(3)` (b) CHAIN isomers : There is no chain isomers of `C_(4)H_(8)` (c) FUNCTIONAL group isomers : `underset("But-1,2-diene")(H_(2)=C=CH-CH_(3))` `underset("But-1,3-diene")(CH_(2)=CH-CH=CH_(2))` (d) Cyclic isomers : 
|
|
| 31. |
What is the oxidation number of sulphur in the following molecules/ ions H_2SO_4 |
|
Answer» Solution :In `H_2SO_4` LET X,+1 ,-2 be oxidation NUMBER of S, H and O Let ON Be  i.e., `(+1 times 2)+ (x times 1)+(-2 times 4)=0` `implies (1 times 2)+x+ (-2 times 4) =0 , 2+ x-8=0, x=+6` Oxidation number of S in `H_2SO_4`is +6. |
|
| 32. |
Which solid is most soluble in water at 25^(@)C? |
|
Answer» `AgNO_(3)` |
|
| 33. |
Which liquid interact each other ? |
|
Answer» WATER - Oil |
|
| 34. |
Which is least stable ? |
|
Answer» `CH_(3)` |
|
| 35. |
Which of the following statements concerning atomic size is correct? |
|
Answer» Atoic size DECREASES down a group |
|
| 36. |
When 5.0 g of a metal is strongly heated, 9.44g of its oxide is obtained. Then the equivalent mass of the metal is ? |
|
Answer» 12 `=(5)/(4.44) xx 8 =(40)/(4.44) =9` |
|
| 37. |
Which is not suitable for atomic radius? |
|
Answer» In each period as GOING from L.H.S. to R.H.S. is decreases. |
|
| 38. |
Whatis atomicnumberand number ofelectronin I and II protonpresent in hydrogenand sodium atomrespectively |
|
Answer» Solution :ATOMICNUMBER (Z ) =NUMBEROF protonsin nucleus ATOMIC numberof hydrogen is I andatomicnumberof sodium is 11 In hydrogen 1 protonand sodium 11 proton 
|
|
| 39. |
The species present in solution when CO_(2) is dissolved in waterare : |
|
Answer» `CO_(2), H_(2) CO_(3), HCO_(3)^(-), CO_(3)^(2-)` |
|
| 40. |
What is the covalence of nitrogen in N_(2)O_(5) ? |
Answer» SOLUTION :Covalency depends upon the number of SHARED pairs of electrons. Now in `N_(2)O_(5)`, each NITROGEN ATOM has FOUR shared pairs of electrons as shown : Therefore, the covalency of N in `N_(2)O_(5)` is 4. 
|
|
| 41. |
Which carboxylic acid(s) gives acetaldehyde on strong heating in presence of catalyst |
|
Answer» `(-)Ph-UNDERSET(CH_(3))underset(|)CH-NH_(2)` |
|
| 42. |
Viscosity of ethanol is 12.0 millipoise. Viscosity of ethanol in S.I system is equal to |
|
Answer» `1.2` 1 poise = CGS unit = `(1 GM)/(Cm - S) = (10^(-3)KG)/(10^(-2) m - s)` `= 0.1 SI UNITS = 12 xx 10^(-3) ` poise ` = 12 xx 10^(-3) xx 0.1 kg//m-s = 1.2 xx 10^(-3)`. |
|
| 43. |
Which of the following compounds is the reactant in Rosenmund's reduction ? |
| Answer» Solution :Refer to comprehensive review | |
| 44. |
Which of the given compound is optically active? |
|
Answer»
|
|
| 45. |
Which of the following is correct comparison for the above effect? Deuterium Kinetic isotopic effect=("Rate of homolytic clevage of C-H bond by Cl")/("Rate of homolytic clevage of C-D bond by Cl") |
| Answer» Answer :A | |
| 46. |
When phenol is treated with excess bromine water, it gives |
|
Answer» m-Bromophenol |
|
| 47. |
What Is bond order ? Explain by example. |
|
Answer» Solution :Bond order according to Lewis : The bond order is given by the number of bonds (SHARED electron pair) between the two atoms in a molecule. e.g. : Bond order in `H_(2), F_(2), Cl_(2)`, HCl is one. In `O_(2) ` bond order two and in`N_(2) ` three because shared electron pairs between two atoms is respectively 2 and 3. According to MO theory bond order : `(1)/(2) (N_(b) - N_(a)) ` where , `N_(b)`= Total no. of boding `e^(-)` of BMO `N_(a)` = Total no. of anti BONDING `e^(-)` of ABMO Bond order : the difference of electron in MO and ABMO orbitals divided by 2 is called Bond order. e.e. :Bond order in`H_(2) = (1)/(2) (N_(b) - N_(a))` `= (1)/(2)( 2 - 0) = 1 ` = `(1)/(2)(2 - 0) = 1 ` Bond order in `F_(2) , Cl_(2) , Br_(2) =(1)/(2) (N_(b) - N_(a))` = `(1)/(2) (10 - 8) = 1 ` Bond order BO in `O_(2) = (1)/(2) (10 - 6 ) = 2 ` Bond order BO in `N_(2) = (1)/(2) = (10 - 4) = 3` |
|
| 48. |
Write the uses of freon. |
| Answer» SOLUTION :(i) Freon gas is USED in refrigerators, air conditioners and in the production of plastic foam. (II) It is used in electronic industry for CLEANING computer parts. | |
| 49. |
Which of the following groups has the highest inductive effect? |
|
Answer» `-CH_(3)` |
|
| 50. |
Writenote on resonance of benzene |
|
Answer» Solution :(i) thephenomenon in which two or morestructures can be written for a substancewithhas identical POSITIONOF atomsis calledresonance (II)Theactualstructureof the molecule is saidto bea resonancehybrid of thevariouspossiblealtemativestructure.  (iii)isbenzeneKekulestructure 1 and iirepresentedteh resonancestructureand structureiii isthe resonancehybridof structuresii and i (iv)the structurei and iiexistonlyin theory.theactualstructuralof BENZENEIS thehythaneof twohypotheticalresomancestructure. |
|