Saved Bookmarks
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Name the non-stoichiometric point defect responsible for colour in alkali metal halides . |
| Answer» Solution :Metal EXCESS or anionic vacancies or F-centres, which is DUE to electrons entrapped in anionic vacancies. | |
| 2. |
Name the most electronegative element. Is it also having the highest electron gain enthalpy? or Why not? |
|
Answer» |
|
| 3. |
Name the monomers which constitute the Teflonpolymers. |
| Answer» SOLUTION :`CF_2 = CF_2` | |
| 7. |
Name the metal which floats on water without any apparent reaction with it . |
| Answer» Solution :Amongst alkali METALS , Li is least reactive and hence does not REACT APPRECIABLY with `H_(2)O` . Further , being smallest , it is the lightest alkali metal EVEN lighter than `H_(2)O` and hence floats over WATER . | |
| 8. |
Name the metal M which is extracted based on the following equation |
|
Answer» Cu |
|
| 9. |
Name the list of elements present in alkali metal group. What is the configuration of them? |
| Answer» SOLUTION :Lithium, sodium, POTASSIUM, RUBIDIUM, caesium and francium are the elemen alkali metal group. Their general ELECTRONIC CONFIGURATION is (noble gas `ns^(1)` | |
| 10. |
Name the first one-toed horse. |
| Answer» Solution : Lithium is the LIGHTEST (density: 0.53g.`CM^(-3)` ) andosmium is the HEAVIEST (density: `22.6 G. cm^(-3))` metal. | |
| 11. |
Name the isotopes of hydrogen. |
| Answer» SOLUTION :PROTIUM `._1H^1` or H, DEUTRIUM or heavy HYDROGEN `._1H^2` or `._1D^2`, Tritium `._1H^3` or `._1T^3` | |
| 13. |
Name the isotope of hydrogen which used in nuclear reactor. |
|
Answer» |
|
| 14. |
Name the industrially important compounds of Sodium ? |
| Answer» Solution : Industrially important COMPOUNDS of SODIUM include sodium CARBONATE, sodium hydroxide, sodium CHLORIDE and sodium bicarbonate. | |
| 15. |
Name the indicator used in the titration of Fe^(2+) against MnO_4^- in an acid medium. |
| Answer» SOLUTION :`MnO_4^-` ACTS as a SELF INDICATOR. | |
| 16. |
Name the indicator and the colour change at the end point in titration of Fe^(2+) against Cr_2O_7^(2-) in an acidic medium. |
| Answer» SOLUTION :Indicator is DIPHENYL AMINE. At the end point, the green solution SUDDENLY turns VIOLET. | |
| 17. |
Name the hydrides which have high potential for hydrogen storage. |
|
Answer» |
|
| 18. |
Name the halogen that does not undergo disproportionation. Write the reason. |
|
Answer» Solution :Fluorine does not UNDERGO disproportionation. Fluorine exhibits only two OXIDATION numbers. Zero in uncombined state and `-1` in COMPOUNDS. |
|
| 19. |
Name the groups in d-block elements which do not form metallic hydrides. |
|
Answer» |
|
| 20. |
Namethe groupsof elements classifiedas s- ,p - andd- blocks. |
|
Answer» <P> Solution :s- block=1,2 ( includingHe ) , p- block= 13 TO18 (3xcept He ) . D- block= 3 to 12. |
|
| 21. |
Name the groups whose elements belong to the p-block of the periodic table. |
| Answer» SOLUTION :GROUPS 13-18 | |
| 23. |
Name the gas law which relates volume and pressure of gas at constant temperature. |
| Answer» SOLUTION :Boyle.s LAW | |
| 25. |
Name the four quantum numbers and mention what they indicate. |
|
Answer» SOLUTION :(i)PRINCIPAL quantum number (n)-INDICATES sizes of SHELL. (ii) Azimuthal quantum number (l)-Indicates the shape and angular momentum. (iii) Magnetic quantum number (m)-Indicates orientation of ORBITALS in spale. (iv) Spin quantum number (s)-Indicates the direction of spin of an electron |
|
| 26. |
Name the following compounds systematically Strategy: Use the following designation of positions in the naphthalence ring system: |
|
Answer» SOLUTION :(i) `1`-Naphthylamine ( or `ALPHA`- naphtlylamine) (ii) `2`-Naphthol (or `beta`-Napthaol) (iii) `1`-Naphthoic acid ( or Nepahehalene-`1`-carboxylic acid) (iv) Naphthalene -1,8-dicarboxylic acid (or Naphthalic acid) (v) `1,4`-Naphthaquinoe ( or `alpha`-naphthaquinone or `1,4`-dihydronaphthelence-`1,4`-DIONE) (VI) `1,2`-Naphthaquinone ( `beta`-naphthaquinone) (vii) `2,6`-Dimethylnaphthalene (viii) `5`-Nitro-`2`-naphthalenesuphonic acid Further fusion can occure in a linear manner to give anthracene, tetracene, pentacene, and so on-a series called acenes. ring system are designated as shown below: 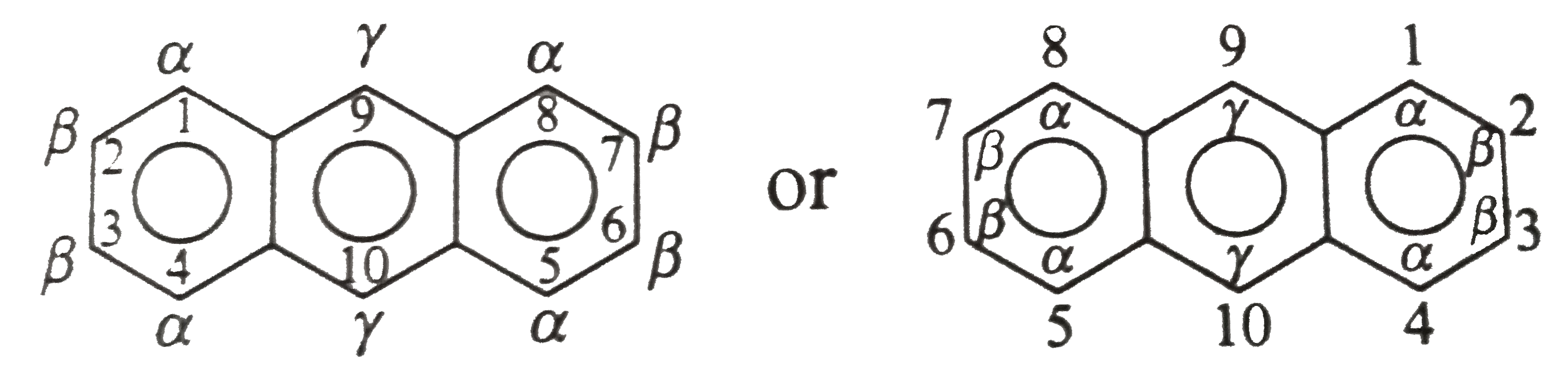 Notice that in anthracene, the positions `1,4,5, and 8` are identical (`alpha`-positions), as position `2,3,6, and 7` (`beta`-positions), and positions `9 and 10` (`gamma`-positions)`. |
|
| 27. |
Name the final product of the following reactions :(i) Benzene is treated with methyl chloride in presence of anhydrous aluminium chloride. (ii) Benzene is treated with acetyl chloride in the presence of anhydrous aluminium chloride. (iii) Phenol is heated with zinc. (iv) Sodium benzoate is heated with soda lime. (v) Benzene is treated with fumining nitric acid in presence of conc. H_(2)SO_(4). (vi) Benzene is catalytically hydrogenated. (vii) Ozone reacts with benzene. (viii) Toluene is treated with a mixture of conc. CNO_(3) and conc. H_(2)SO_(4). (ix) Chlorine is passed through boiling toluene. (x) Toluene is heated with acidic potassium premanganate solution. (xi) Toluene in carbon tetrachloride is oxidised bychromyl chloride. (xii) Ortho-xylene is oxidised with hot acidic K_(2)Cr_(2)O_(7) solution. |
|
Answer» (ii) Methly phenyl ketone (Acetophenone) (iii) Benzene (v) 1.3.5-Trinitrobenzene (vi) Cyclohexane (VII) Ethane-1,2-dial (Glyoxal) (ix) Benzyl CHLORIDE (x) BENZALDEHYDE (xi) Benzaldehyde (xii) Benzene-1,2-dicarboxylic ACID (Phthalic acid) |
|
| 28. |
Name the experiment which shows that light was wave property. |
|
Answer» |
|
| 29. |
name the energy which is released during formation of an ionic crystal? |
| Answer» SOLUTION :LATTICE ENERGY. | |
| 30. |
Name the energy which arises due to motion of atoms or molecules in a body. How is this energy affected when the temperature is increased ? |
| Answer» Solution :The energy which arises due to motion of ATOMS in a body is the thermal energy. It is a MEASURE of the average kinetic energy of the particles and INCREASES with INCREASE in temperature. | |
| 31. |
Name the elements that correspond to the given electronic configurations. Write down their atomic number also. (i) 1s^(2) 2s^(2) 2p^(2) (ii) 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(1) (iii) 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) |
|
Answer» |
|
| 32. |
Name the elements other than carbon in group IVA elements that exhibit catination. |
| Answer» SOLUTION :In group IVA, silicon and germanium ALSO exhibit catenation to some EXTENT. Silanes with the FORMULA `Si_(n)H_(2n+2)` are known where n is one to eight. Germanes `(Ge_(n)H_(2n+2))` with n is one to five are known. | |
| 33. |
Name the elements (alkali metals) Which form superoxide when heated in excess of air. |
| Answer» SOLUTION :POTASSIUM RUBIDIUM and CAESIUM. | |
| 34. |
Name the element which is invariably bivalent and whose oxide is soluble in excess of NaOH and its dipositive ion has noble gas core . |
|
Answer» Solution :Be . It forms bivalent ion which has helium gas CORE . `underset(2,2) (Be) to underset(2) (Be^(2+))` (has He gas ore) `+ 2 e^-` Being slightly acidic , it dissolves in excess of Na OH to form sodium berrylate . `BEO + 2 NaOH to underset("Sod. berrylate") (Na_(2)BeO_(2)) + H_(2)O` |
|
| 35. |
Name the element used in fuel cell for generating electricity. |
|
Answer» |
|
| 36. |
Name the elements of the second period (a) which forms a strongly basic oxide and (b) which forms a strongly acidic oxide. |
|
Answer» Solution :LI, Be,B,C,N,O F and Ne are teh elements of SECOND PERIOD . Metallic nature is period decreases gradually from left to right and non metallic nature increases. Lithium is most metal.ic in the second period. (a) Strongly basic oxide is formed by lithium . The oxide is `Li_2O` (b) Though fluorine and oxygen are highly electro neagtive, they do not form oxiees . Strongly acidic oxide is formed by NITROGEN The oxide is `N_2O_5` |
|
| 37. |
Name the element of group 13 which forms only covalent compounds ? |
| Answer» SOLUTION :Because of small SIZE and highsum of firstthree ionization ENTHALPIES `(Delta_(i)H_(1) + Delta_(i)H_(2) + Delta_(i)H_(3))`, B FORMS onlycovalentcompounds. | |
| 38. |
Namethe elementin thetablewhich hasthe highestand lowestfirstionizationenthalpy. |
| Answer» Solution :Heliumbeing thesmallestinertgas hasthe HIGHEST`Delta_(i) H_(1)` whilefranciumbeing thebiggest alkalimetalhas thelowest`Delta_(i) H` in the periodtable. | |
| 39. |
Name the element in each of the following cases: (i) A bivalent anion of the element having 10 electrons (ii) A trivalent cation of the element having 10 electrons. What is the relationship between the two ions called ? |
|
Answer» Solution :(i) OXYGEN (II) ALUMINIUM The two ions are called isoelectronic. `(O^(2-) = 8 + 2 = 10, Al^(3+) = 13 - 3 = 10)` |
|
| 40. |
Which halogen has highest electron affinity or electron gain enthalpy ? |
|
Answer» |
|
| 41. |
Name the element estimated by Kjeldhal's method. |
|
Answer» |
|
| 42. |
Name the electrophiles in the following reactions. (i) Chlorination (ii) Nitration (iii) Sulphonation (iv) Friedel craft's methylation. |
|
Answer» Solution :(i) CHLORINATION. - `CL^(+)` (II) Nitration - `NO_(2)^(+)` (III) Sulphonation - `SO_(3)` (iv) Friedel Crafts methylation - `CH_(3)^(+)` |
|
| 43. |
Name the distinguishing reaction of alkane from alkene. |
| Answer» Solution :(i) BAEYER TEST (II) Reaction with `Br_(2)` in `C Cl_(4)`. | |
| 44. |
Name the different type of bonds present in NH_(4) Cl after drawing its structure. |
| Answer» Solution :`[H-UNDERSET(H) underset(|)overset(H)overset(|)NtoH]^(+)CL^(-)` . THUS , it has covalent, COORDINATE and ionic bonds. | |
| 45. |
Name the different regions of the atmosphere along with their altitudes and temperature ranges. |
| Answer» SOLUTION :Troposphere (0-11 KM. 15 to `-56^@C`), STRATOSPHERE (11-50 km, -56 to `2^@C`), mesosphere (50-85 km. -2 to `-92^@ C`), thermosphere (85-500 km, -92 to `1200^@C`) | |
| 46. |
Name the different isotopes of hydrogen. Represent them along with their atomic numbers and mass numbers. |
| Answer» Solution :Protium `(._(1)H^(1))`, Deuterium `(._(1)H^(2))`, TRITIUM `(._(1)H^(3))` | |
| 47. |
Name the compounds whose line formulae are given below |
Answer» Solution : `UNDERSET("3-ethyl-4-methylhept-5-en-2-one")(overset(1)(C )H_(3)- overset(overset(O)(2||))(C )-underset(underset(underset(underset(CH_(3))(|))(CH_(2)))(|))overset(3)(CH)- overset(overset(CH_(3))(4|))(CH)- overset(5)(C )H= overset(6)(C )H- overset(7)C )H_(3)` NUMBER the carbon atoms of the longest possible carbon CHAIN in such a WAY that the functional group i.e. `gt C = O` gets the lowest possible locant Name: 3-ethyl-4-methylhept-5-en-2-one  Name: 3-nitrocyclohex-1-ene Name: 3-nitrocyclohex-1-ene Number the carbon atoms of the ring in such a way that double BOND gets the lowest possible coccants followed by the `-NO_(2)` group |
|
| 48. |
Name the compound whose electrolysis in aqueous state, give high purity (99.95%) dihydrogen. |
|
Answer» |
|