Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Name the components of photochemical smog. |
| Answer» SOLUTION :Nitrogen OXIDES, OZONE, organic derivatives. | |
| 3. |
Name the classes of hydrides to which H_2O, B_2H_6and NaH belong |
|
Answer» SOLUTION :`H_2O` :Covalent or molecular HYDRIDE (electron rich hydride). `B_2H_6` : Covalent or molecular Hydride (electron DEFICIENT hydride). NAH : Ionic or SALINE hydride. |
|
| 4. |
Name the classes fo hydrides to which H_(2)O,B_(2)H_(6) and NaH belong. |
| Answer» Solution :`H_(2)O` (covalent hydride , ELECTRON RICH), `B_(2)H_(6)`(Covalent hydride, electron-deficient ), NaH (ionichydride). | |
| 5. |
Name the chief forms of occurrence of magnessium in nature. How is magnessium extracted from one of its ores? |
|
Answer» Solution :Magnesium is QUITE ABUNDANT in the earth crust and occupy sixth position in order of their abundance. Ores of magnesium: The important ores of magnesium are as follows: (i). Carnallite: `(2KCl.MgCl_(2).6H_(2)O) or K_(2)MgCl_(4).6H_(2)O` (ii). MAGNESITE: `MgCO_(3)` (iii). Dolomite: `MgCO_(3).CaCO_(3)CaCO_(3)` (iv). Epsom salt: `MgSO_(4).7H_(2)O` The chlorides and sulphates occur in SEA water. |
|
| 6. |
Name the chief factor responsible for the anomalous behaviour of lithium. |
| Answer» SOLUTION :SMALL SIZE of its ATOM and ION | |
| 7. |
Name the chief factor responsible for the anomalous behaviour of lithium . |
| Answer» SOLUTION :HIGH POLARIZING POWER (charge/size RATIO) . | |
| 8. |
Name the catalyst [X] for the reaction, CO_((g))+H_(2(g))overset([x])toHCHO_((g)) |
|
Answer» `NI` |
|
| 11. |
Name the bonding which links the two boron atoms in a diborane molecule. |
| Answer» SOLUTION :3C- 3E BONDING | |
| 12. |
Name the apparatus used for the measurement of the pressure of a gas. |
| Answer» SOLUTION :MANOMETER | |
| 13. |
Name the anomalous pairs of elements in the Mendeleev's periodic table. |
|
Answer» |
|
| 14. |
Name the anions responsible for permanent hardness of water |
|
Answer» sulphite |
|
| 15. |
Name the allotropic form of carbon whose structure resembles soccer ball. |
|
Answer» |
|
| 16. |
Name the alkyl groups derives from isobutane. |
| Answer» SOLUTION :(i) `(CH_(3))_(2)CHCH_(2)` - 2-Methylpropyl (ISOBUTYL) and `(CH_(3))_(3)C` - 1, 1-Dimethylethyl (tert-butyl). | |
| 17. |
Name the alkanes with octane numbers 0 and 100. |
| Answer» SOLUTION : n-heptane, iso-octane | |
| 18. |
Name the alkaline earth metal which forms covalent compounds . |
| Answer» Solution :Due to high sum of FIRST TWO IONIZATION enthalpies (i.e., `Delta_(i) H_(1) + Delta_(i)H_(2))` and high electronegativity , Be FORMS covalent compounds . | |
| 19. |
Name the alkaline earth metal hydroxide which is amphoteric is nature. |
|
Answer» `Be(OH)_2` |
|
| 20. |
Name the alkaline earth metal hydroxide which is amphoteric . |
|
Answer» Solution :`Be(OH)_(2)` dissolves both in acids as well as bases and hence is amphoteric in nature . `UNDERSET("Basic")(Be(OH)_(2)) + 2 HCL to BeCl_(2) + 2H_(2)O , underset("Acidic")(Be(OH)_(2)) + 2 NaOH to underset("Sod. Berrylate") (Na_(2)BeO_(2)) + 2 H_(2)O` |
|
| 21. |
Name the alkali metals , why is lithium regarded as most apt reducing agent in aqueous solutions? |
| Answer» Solution :SINCE smaller cations` (Li^(+) , Na^(+))` stabilize smaller anions `(O^(2-))` and bigger cations `(K^(+) , Rb^(+)` and `Cs^(+)`) stabilize bigger anions `(O_(2)^(-))` , therefore, Li and Na form only oxides when HEATED in AIR but K , Rb and Cs FORMS SUPEROXIDES . | |
| 22. |
Name the alkali metals which shows diagonal relationship with magnesium . |
| Answer» SOLUTION :LI RESEMBLES MG . | |
| 23. |
Name the alkali metals which form superoxides when heated in excess of air. |
| Answer» SOLUTION :K , RB and CS | |
| 25. |
Name the adsorbent commonly in adsorption chromatography. |
| Answer» SOLUTION :ALUMINA `(Al_(2)O_(3))`. | |
| 26. |
Name othe products of the following reactions. (I). C_(6)H_(6) reacts with methyl chloride in presence of AlCl_(3). (II) C_(6)H_(6) reacts with acetyl chloride in presence of AlCl_(3). (III) C_(6)H_(6) reacts with fuming nitric acid in presence of conc. H_(2)SO_(4). (IV). C_(6)H_(6) is catalytically hyddrogenated. |
|
Answer» I-Chloromethane, II-Toluene, III-Nitrobenzene, IV-n-Hexane 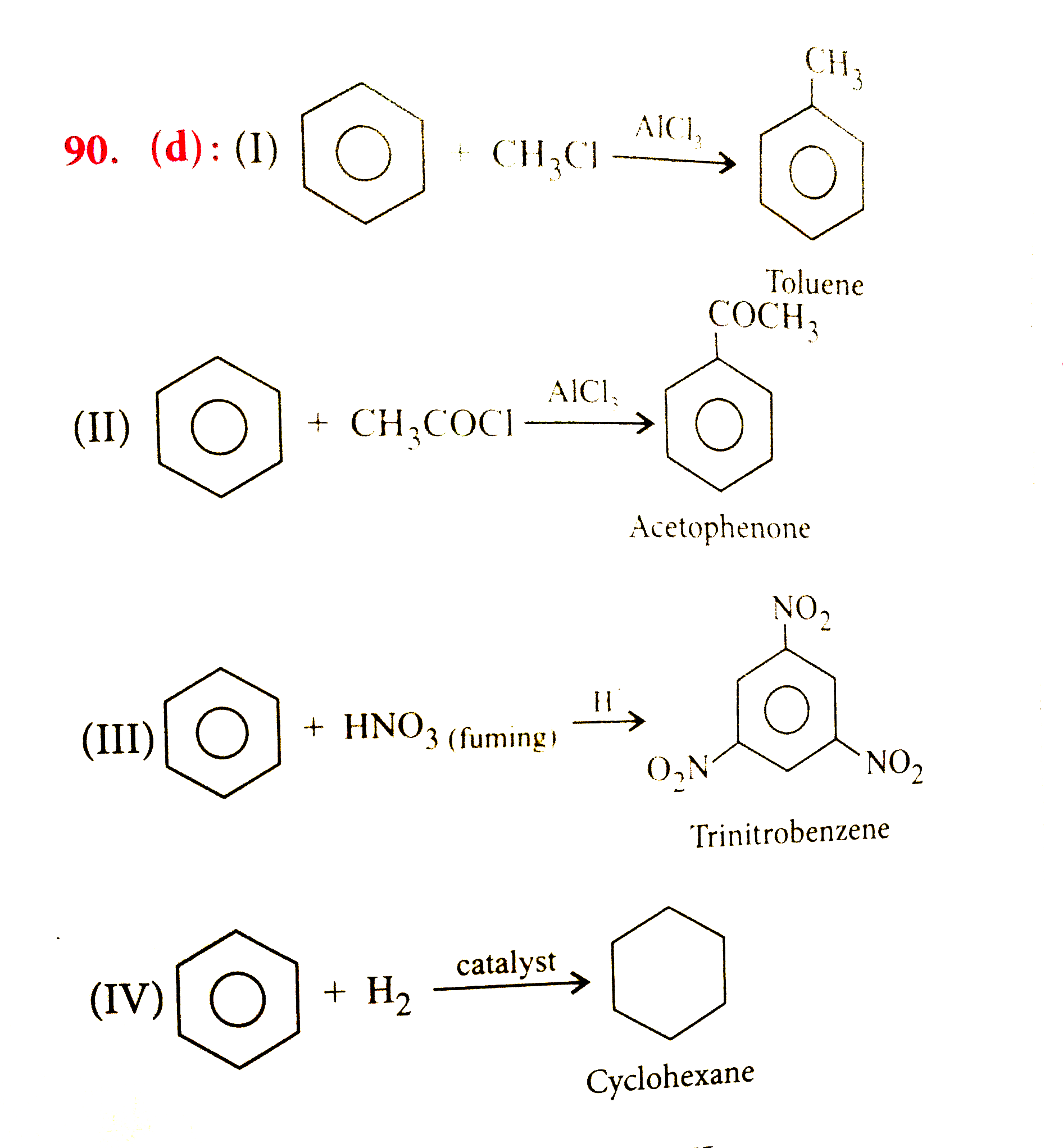
|
|
| 27. |
Name radioactive alkali and alkaline earth metals. |
| Answer» Solution :RADIOACTIVE alkali METAL is FRANCIUM (Fr) and alkali earth metal is RADIUM (Ra) | |
| 28. |
Name one solid which has both Schottky and Frenkel defects |
| Answer» SOLUTION :SILVER BROMIDE (AGBR) | |
| 29. |
Name one solid which has both schottky and Frenkel defects. |
| Answer» SOLUTION :SILVER bronmide (AGBR). | |
| 30. |
Name one reagent or one operation to distinguish between (i) BeSO_(4) and BaSO_(4) (ii) Be(OH)_(2) and Ba(OH)_(2) (iii) Na_(2)CO_(3) and NaHCO_(3) |
|
Answer» Solution :(i) WATER : `BeSO_(4)` is SOLUBLE in `H_(2)O` while `BaSO_(4)` is not (ii) Alkali : `Be(OH)_(2)` dissolves in alkali but `Ba(OH)_(2)` does not (iii) Heat : `NaHCO_(3)` on heating gives `CO_(2)` which turns lime water milky . In constrast , `Na_(2)CO_(3)` does not decompose on heating to form `CO_(2)`. |
|
| 31. |
Name one reaction in which water acts (i) as an oxidising agent and (ii) as a reducing agent. |
|
Answer» Solution :(i) Oxidising agent. Water oxidises a number of highly electropositive METALS whose ELECTRODE potential is less than -0.83 V liberating `H_(2)` gas. For examples, sodium with `E^(@)=-2.71` reduces `H_(2)O` to `H_(2)` gas `underset("(Reductant)")(2na(s))+ underset("(Oxidant)")(2H_(2)O)(l) to 2Na^(+) (aq) + 2OH^(-)(aq)+ H_(2)(g)` (ii) Reducing agent. Water reduces a number of electronegative elements whose electrode potential is higher than +1.23 V liberating either `O_(2)` or `O_(3)` gas. For EXAMPLE, FLUORINE with `E^(@)=+2.87 V ` oxididses `H_(2)O`to `O_(2)` gas `underset("(Oxidant)")(2F_(2)(g)) + underset("(Reductant)")(2H_(2)O(l)) to O_(2)(g) + 4H^(+) (aq) + 4F^(-) (aq)` |
|
| 32. |
Name one example of a reaction in which dihydrogen acts (i) as an oxidising agent(ii) as a reducing agent. |
|
Answer» SOLUTION :OXIDISING AGENT : `2Na + H_(2) to 2Na^(+)H^(-)`. Here, NA is oxidised to `Na^(+)` Reducing agent `CuO + H_(2) overset("Heat")to Cu + H_(2)O`. Here, CuO is reduced to Cu. |
|
| 33. |
Name one example of a reaction in which dihydrogen acts (i) as an oxidising agent and (ii) as a reducing agent. |
|
Answer» Solution :(i) In the reaction of dihydrogen with metals of form METAL hydrides, it acts as an oxidising agent `2NA (s) + H_(2) (g) overset("Heat")to 2Na^(+)H^(-) (s)` Here, Na has been oxidised to `Na^(+)` while dihydrogenhas been reduced to hydride `(H^(-))` ion. (ii) In the reaction of HEATED CUPRIC oxide with dihydrogen to form `H_(2)O` and copper metal, dihydrogen acts as a redcuing agent. `CuO(s) + H_(2)(s) overset("Heat")to Cu(s) + H_(2)O(g)` Here, CuO is reduced to Cu while dihydrogen is oxidised to `H_(2)O` |
|
| 34. |
Name one compound each involving sp^(3) , sp^(2) andsphybridisation. |
| Answer» Solution :`sp^(3) = CH_(4), sp^(2) = C_(2)H_(4), sp = C_(2) H_(2).` | |
| 35. |
Name one compound each in which hydrogen exists in (i) +1 and (ii) -1 and (iii) zero oxidation state. |
| Answer» SOLUTION :`(i) HCL ` or `H_(2)O` or `NH_(3)` (II) NaH or `CaH_(2)` (iii) `H_(2)` | |
| 36. |
Name one Carcinogenic compound. |
| Answer» SOLUTION :ANTHRACENE of Benzanthracene. | |
| 37. |
Name of method by which Halogen present in an organic compound is estimated ? 0.1 g of an organic compound gives 0.08 g of silve bromide, Calculate the percentage of bromite. Atomic masses : Ag=108, Br=80 |
|
Answer» Solution :Carius method Percentage of halogen `=("atomic mass of halogen" XX "mass of silver halide" xx 100)/("moleculer mass of silver halid" xx"mass of organic COMPOUND TAKEN") ` MOLECULAR mass of `AgBr=108+80=188` Mass of silver bromide `=0.09g` Mass of organic compound `=0.1g` % of bromine`=(80xx0.08xx100)/(188xx0.1)=34.04%` |
|
| 38. |
Name of elements with atomic numbers greater than 100 are given by IUPAC Why is potassium considered as an s - block element ? |
| Answer» Solution :The electronic configuration of potassium is : `K(19):1s^2 2s^2 2p^26 3s^2 3p^6 4s^1 or [Ar] s^1` since the last electron ENTERS the s-orbital of the OUTERMOST ENERGY level, potassium is considered as an s-block ELEMENT | |
| 39. |
Name of elements with atomic numbers greater than 100 are given by IUPAC the first inoisation enthalpies of second period element generally increase from lewft to right along the period. Give reason for this general trend. |
| Answer» Solution :As we move from LEFT to right in the second PERIOD, neclear charge incraeses and electrons are ADDED to the same shell so the ELECTORN swill experince greater nuclear attraction and the atomic size will decreases regularly. | |
| 40. |
Name of abovereaction is |
|
Answer» WURTZ reaction |
|
| 41. |
Name of elements with atomic numbers greater than 100 are given by IUPAC The atomic number of the elment with IUPAC name Ununbium is _____ |
|
Answer» 112 |
|
| 42. |
Name each of the following substituted biphenyls : |
|
Answer» |
|
| 44. |
Name any two gases responsible for Green house effect. |
|
Answer» |
|
| 45. |
Name any three quantum numbers and write their significance. |
|
Answer» SOLUTION :a. Principal quantum no (n)-Energy level of an electron. B. Azimuthal quantum No. (l)-Shape of an orbital. C. SPIN quantum No. (s)-Spin of an electron. |
|
| 46. |
Name any one gas pollutant that can pollute environment. |
| Answer» SOLUTION :`CO_(2)` or CO or NO or `NO_(2)` or `SO_(3)` any EXAMPLE. | |
| 48. |
Name and//or draw the structures of the following compounds: (i) Cinnamaldehyde (beta-phenylacraldehyde) (ii) Anisaldehyde (p-methaoxybenzaldehyde) (iii) 4-Bromocyclohexanone (iv) (v) (vi) 3-Hydroxybutanal Strategy: Writing the structures of specific names is a bit problem as they have no relationship with the strycture. Thus, we must be familiar with these names and structures. |
|
Answer» Solution :(i) `C_(6)H_(5)overset(3)(CH)underset("3-Phenylpropenal")(=overset(2)(CH)overset(1)(CH)O)` (ii) 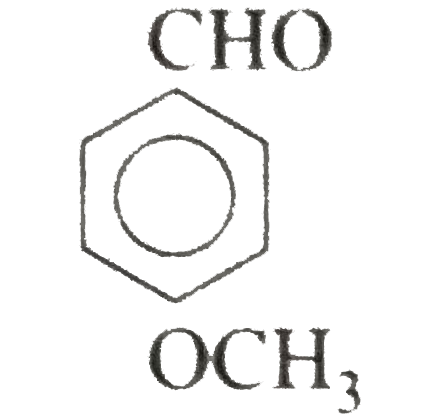 (III) 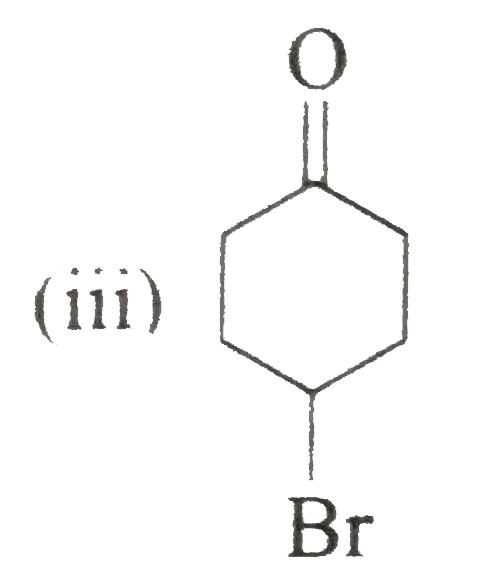 (iv) `2`-CYCLOHEXANONE (v) `overset(8)(CH_(3))overset(7)(CH_(2))overset(6)(CH_(2))overset(5)(C)-=overset(4)(C)-overset(O)overset(3||)(C)-overset(2)(CH_(2))overset(1)(CH_(3))` or 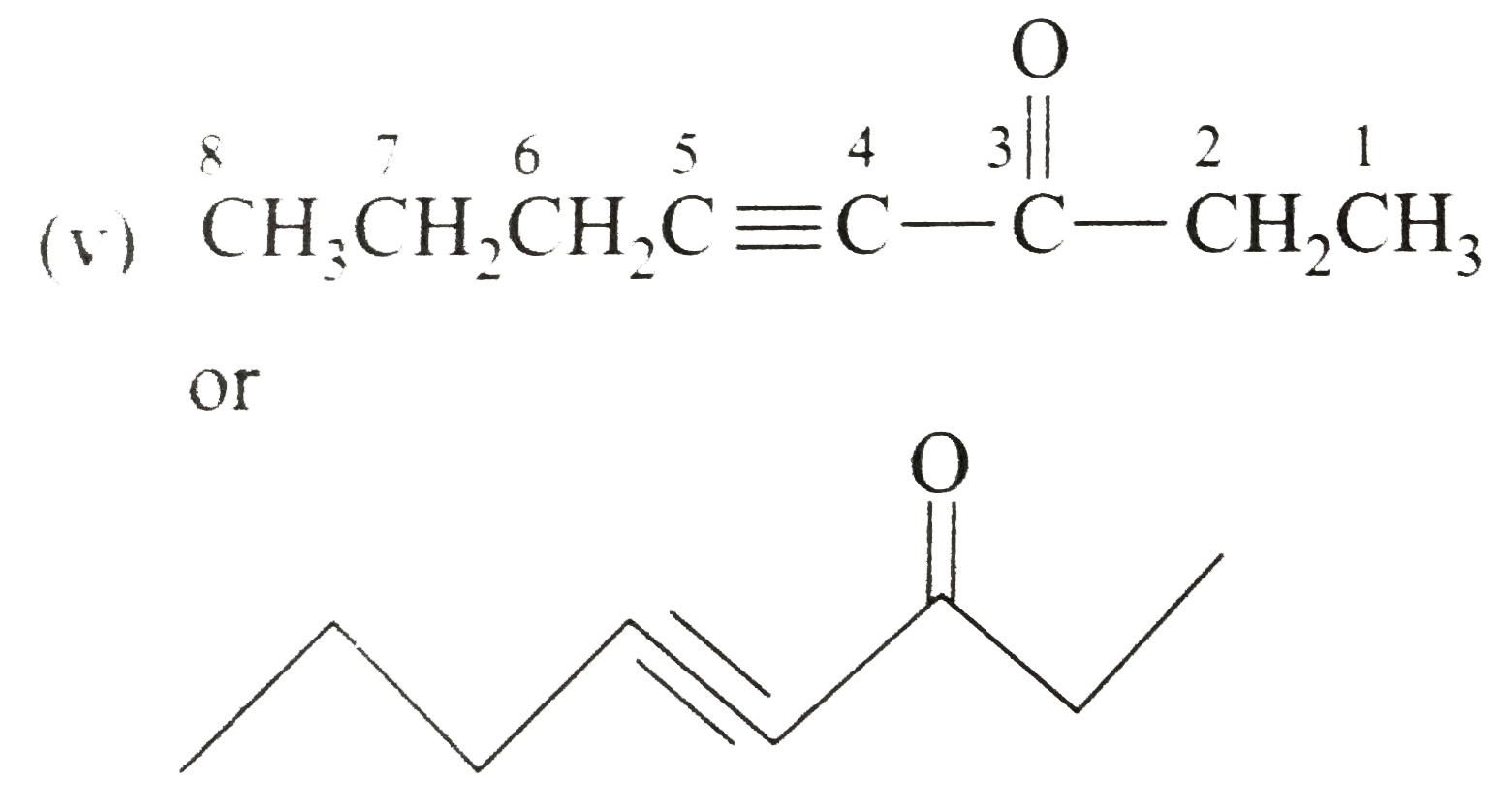 (vi) `CH_(3)-underset(OH)underset(|)overset(3)(CH)-overset(2)(CH_(2))-overset(1)(CHO)` or 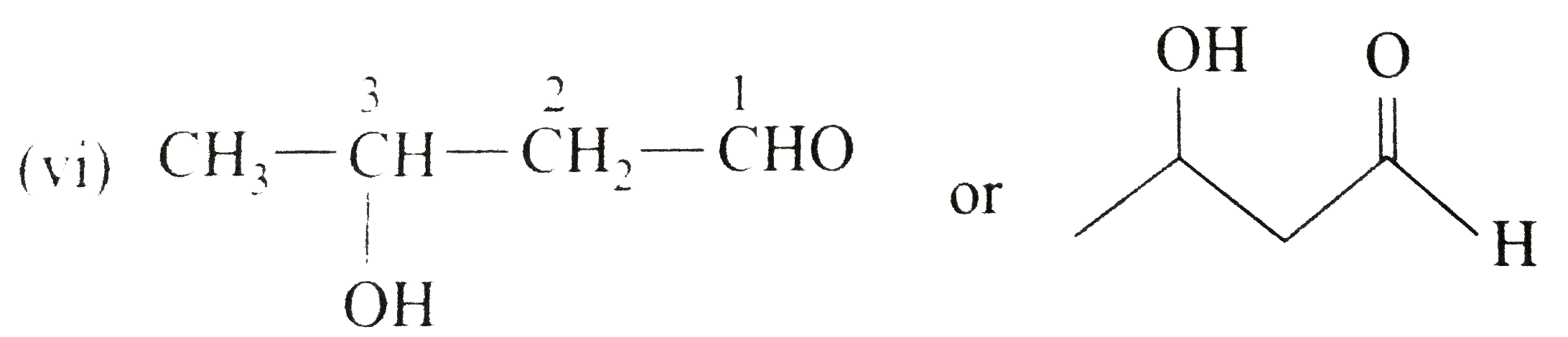 (vii) cis-`4`-Methylhex-`4`-ENAL |
|
| 49. |
Name and represent the type of bonds presentin CuSO_(4). 5H_(2)O . |
Answer» Solution :` CuSO_(4). 5H_(2)O` .Has ionic , COVALENT , coordinate and hydrogen bonds as SHOWN below :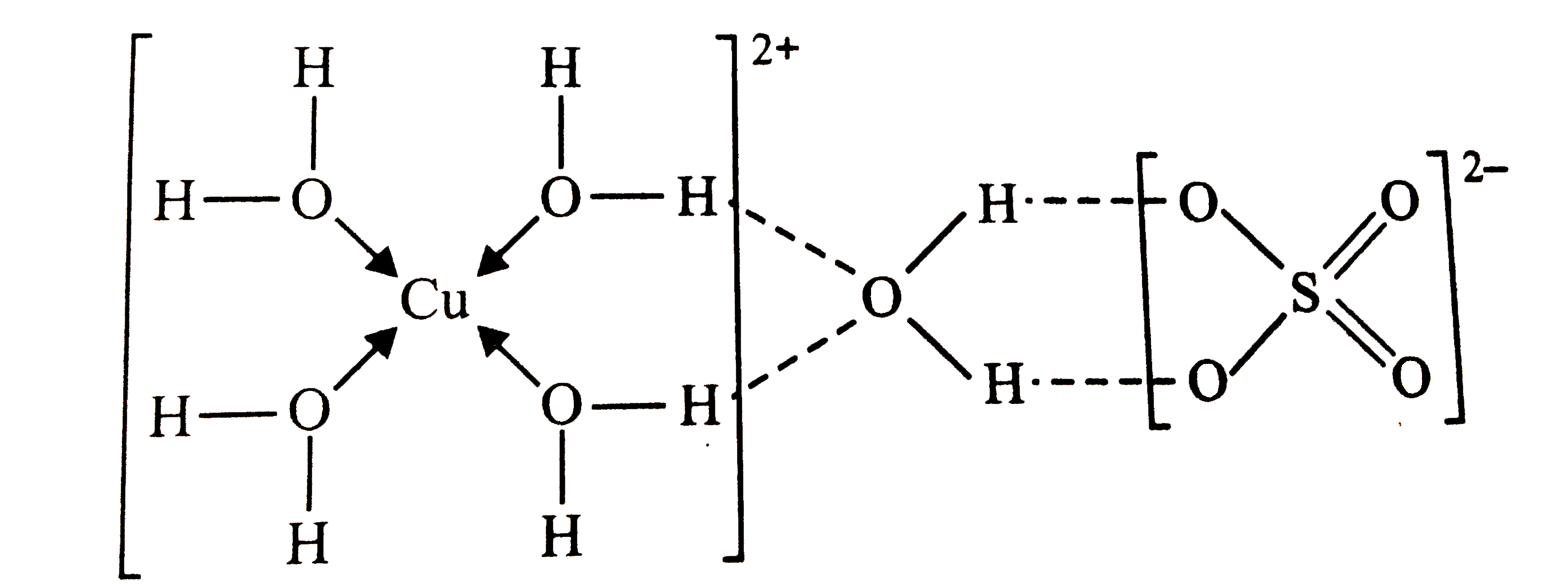 Note that four `H_(2)O`MOLECULES are linked to `Cu^(2+)` cationby coordinate bonds whereas fifth `H_(2)O`molecule is linked by hydrogen bonds between coordinated `H_(2)O` molecules and sulphate ion . |
|
| 50. |
Name an element from Group-2 which forms an amphoteric oxide and a water soluble sulphate. |
|
Answer» SOLUTION :Beryllium is an element from group 2 which gives an amphoteric oxide Bed and water soluble SULPHATE`BeSO_(4)`. `Ba(OH)_(2) +2OH^(-) to [Ba(OH)_(4)]^(2-)` `Ba(OH)_(2)+2HCl +2H_(2)O to [Ba(OH)_(4)]Cl_(2)` `BeSO_(4)` is soluble in water due to highest HYDRATION energy. It is much more than lattice energy. |
|