Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Photon having energy equivalent to the binding energy of 4^(th) state of He^+ atom is used to eject an electron from the metal surface of work function 1.4 eV. If electrons are further accelerated through the potential difference of 4V then the minimum value of De-broglie wavelength associated with the electron is : |
|
Answer» Now K.E.= 3.4 - 1.4 = 2eV Now , total energy = 2+ 4 = 6 eV i.e. POTENTIAL = 6V For electron `L= sqrt((150)/(V)) ,` So l = 5 Å |
|
| 2. |
Photoelectric emission is observed from a surface for frequencies v_(1) and v_(2) of the incident radiation (v_(1) gtt v_(2)). If the maximum kinetic energies of the photoelectrons in the two cases are in the ratio 1 : k, what will be the theshold frequency (v_(0)) in term of v_(1), v_(2) and k ? |
|
Answer» SOLUTION :In 1st case, K.E. `= h v_(1) - h v_(0) = h (v_(1) - v_(0))` In 2nd case, K.E. `= h v_(2) - h v_(0) = h (v_(2) - v_(0))` GIVEN `(h (v_(1) - v_(0)))/(h(v_(2) - v_(0))) = (1)/(k) or v_(2) -v_(0) = k (v_(1) - v_(0)) = k v_(1) - kv_(0)` or `k v_(0) - v_(0) = kv_(1) - v_(2) or v_(0) (k -1) = kv_(1) = v_(2) or v_(0) = (kv_(1) - v_(2))/(k -1)` |
|
| 4. |
Photoelectric emission is observed from a metal surface with incident frequencies v_(1) and v_(2) where v_(1) gt v_(2). If the kinetic energies of the photoelectrons emitted in the two cases are in the ratio 2 : 1, then the threshold frequency v_(0) of the metal is |
|
Answer» `v_(1) = v_(2)` `hv_(2) = hv_(0) + (1)/(2) E or 2 hv_(2) = 2 hv_(0) + E`...(ii) EQN. (i) - Eqn (ii) gives `hv_(1) - 2hv_(2) = hv_(0) - 2 hv_(0)` or `v_(1) - 2 v_(2) = - v_(0) or v_(0) = 2 v_(2) - v_(1)` |
|
| 5. |
Photoelectric emission is observed from a metallic surface for frequenciesv_(1) and v_(2)of the incident light (v_(1)gtv_(2)).If the maximum values of kinetic energy of the photoelectrons emitted in the two cases are in the ratio 1:n,then the threshold frequency of the metallic surface is |
|
Answer» `(upsilon_2 - upsilon_1)/(K-1)` `(hupsilon_1 - hupsilon_0)/(H upsilon_2 - hupsilon_0) = 1/KimpliesK (upsilon_1 - upsilon_2) = upsilon_2 - upsilon_0` `impliesK upsilon_1 - K upsilon_0 = upsilon_2 - upsilon_0` `impliesupsilon_0 (1-K) = upsilon_2 - K upsilon_1 impliesupsilon_0 = (upsilon_2 - K upsilon_1)/(1-K)` |
|
| 7. |
Photochemical smog was first observed in__________(city) and is formed from _________ and __________ in ________season |
|
Answer» |
|
| 8. |
Photoelectric effect was first observed by Hertz. The number of electrons ejected in the photoelectric effect is proportional to_____of light used (frequency, intensity) |
| Answer» SOLUTION :INTENSITY | |
| 10. |
Photochemical smog occurs in warm, dry and sunny climate. One of the following is not amongst the components of photochemical smog. Identify it. |
|
Answer» `NO_2` |
|
| 11. |
Photochemical smog occurs in warm, dry and sunny climate. One of the following is not amongst the components of photochemical smog, identify it. |
|
Answer» `NO_3` Concentration of PAN, `O_3`, KETONES and aldehydes INCREASES up in the atmosphere. `SO_2` is not responsible for photochemical smog. |
|
| 12. |
Photochemical smog is formed due to presence of : |
|
Answer» OXIDES of sulphur |
|
| 13. |
Photochemical smog is ____in character while classical smog is ____in character. |
|
Answer» OXIDISING, reducing |
|
| 15. |
Photochemical smog is formed in |
|
Answer» SUMMER during DAY time |
|
| 16. |
Photochemical smog is a mixture of oxidising pollutants.(T/F) |
|
Answer» |
|
| 17. |
Photochemical smog is due to the presence of |
|
Answer» OXIDES of SUPHUR |
|
| 18. |
Photochemical smog can be reduced by : |
|
Answer» using catalytic converted in the AUTOMOBILES |
|
| 19. |
Photochemical smog always contains |
|
Answer» `O_(3)` |
|
| 20. |
Photochemical chlorination of alkane is initiated by a process |
|
Answer» PYROLYSIS |
|
| 21. |
Photo chemical smog formed in congested metropolitan cities mainly consists of |
|
Answer» OZONE, `SO_(2)` and hydrocarbons |
|
| 22. |
Photo chemical smog formed in congested metropolination cities mainly consists of …………. . |
|
Answer» Ozone, `SO_(2)` and HYDROCARBONS |
|
| 23. |
Phosphorus trichloride (PCl_(3)) contains 22.57 % of phosphorus, phosphine (PH_(3)) contains 91.18 % of phosphorus. Hydrochloric acid (HCl) contains 97.23 % of chlorine. Show that the data is according to the Law of Reciprocal Proportions. |
|
Answer» Solution :Let US FIX `1g` of phosphorus (P) as the FIXED weight In phosphorus trichloride `(PCl_(3))` Weight of phosphorus `= 22.57 g` Weight of chlorine `= 100 - 22.57` `=77.43 g` `22.57 g` of phosphorus have combined with chlorine `= 77.43 g` `1.0 g` of phosphorushas combined with chlorine `= (77.43)/(22.57)g=3.43g` In phosphine `(PH_(3))` Weight of phosphorus `= 91.18 g` Weight of HYDROGEN `=100-91.18 = 8.82 g` `91.18 g` of phosphorus have combined with hydrogen `=8.82 g` `1.0 g` of phosphorus has combined with hydrogen `= (8.82)/(91.18)g=0.097g` Thus, the ratios by weights of chlorine and hydrogen combining with a fixed weight of phosphorus in the two compounds is : `3.43 : 0.097` (This ratio is not the same)...(i) In hydrochloric acid gas `(HCl)`, Weight of chlorine `=97.23 g` Weight of hydrogen `=100-97.23 = 2.77 g` Thus, chlorine and hydrogen have combined in the ratio `97.23 : 2.77` ....(ii) Let us compare the two ratios. These are RELATED to each other as : `(3.43)/(0.097):(97.23)/(2.77)or35.37:35.10or 1:1("approximately")` Since these two ratios are nearly the same `(1:1)`, the data is according to the Law of Reciprocal Proportions. |
|
| 24. |
Phosphorus has three allotropic forms : (i) white phosphorus (ii) red phosphorus and (iii) black phosphorus. Write the difference between white and red phosphorus on the basis of their structure and reactivity. |
|
Answer» Solution :Structrue. White phosphorus CONSISTS of discrete `P_(4)` molecules in which each P is `sp^(3)`-HYBRIDIZED and lies at the corner of a tetrahedron. Thus, it has a tetrahedral structure with six P-P bonds In contrast, red phosphorus also consists of `P_(4)` tetrahedra but these are joined together through single covalent P-P bonds to form a polymeric structure Reactivity. White phosphours is much more reactive than red phosphorus. This is due to the reason that in white phosphours, the PPP angle is only `60^(@)` which is much smaller than the tetrahdral angle `(109^(@)-28^(@))`. As a result, `P_(4)` tetrahedral molecules have considerable angle strain and hence white P is very reactive. it READILY catches fire in air. Becaues of polymeric structure, red P is HOWEVER, much less reactive than white P. It is quite stable and does not catch fire in air. |
|
| 25. |
Phosphorus has the oxidation state of +3 in |
|
Answer» ORTHO PHOSPHORIC ACID |
|
| 26. |
Phosphorus forms a number of oxoacids. Out of these oxoacids, phosphinic acid has strong reudcing property. Write its structure and also write a reaction showing its redcuing behaviour. |
|
Answer» Solution :The STRUCTURE of phosphinic acid (hypophosphorus acid) is GIVEN on the R.H.S. Since it contains two P-H bonds, it acts as a strong reducing agent. For example, it reduces `AgNO_(3)` to metallic SILVER. `4 AgNO_(3) + H_(3)PO_(2) + 2H_(2)O rarr 4Ag darr H_(3)PO_(4) + 4 HNO_(3)` 
|
|
| 27. |
Phosphorous pentachloride when heated in a sealed tube at 700 K it undergoes decomposition as PCl_(5)(g) hArr PCl_(3)(g)+Cl_(2)(g), K_(p)=38 atm Vapour density of the mixture is 74.25. When an inert gas is added to the given reversible process, then the equilibrium will. |
|
Answer» Be UNAFFECTED |
|
| 28. |
Phosphorous exhibits highest oxidation state in |
|
Answer» `PH_(3)` |
|
| 29. |
Phosphoric acid H_(3)PO_(4) can not be neutralised to : |
| Answer» Solution :N//A | |
| 30. |
Phosphoric acid is preferred over sulphuric acid in preparing hydrogen peroxide from peroxides. Why ? |
|
Answer» Solution :`H_2SO_4` acts as a catalyst for decomposition of `H_2O_2`. THEREFORE, some weaker acids such as `H_3PO_4` is PREFERRED over `H_2SO_4` for preparing `H_2O_2` from PEROXIDES. `2BaO_2 + 2H_3PO_4 to Ba_3(PO_4)_2 + 3H_2O_2` |
|
| 31. |
Phosphine on decomposition produces phosphorus and hydrogen. The change in percentage in volume |
|
Answer» 50ml |
|
| 32. |
Phosphine is prepared by the reaction of water with which reagent ? |
|
Answer» CALCIUM phosphide |
|
| 33. |
Phosphate pollution is caused by |
|
Answer» SEWAGE and agriculture FERTILIZERS |
|
| 34. |
Phosphate containing fertilisers cause water pollution. Addition of such compounds in water bodies causes _______ |
|
Answer» enhanced GROWTH of algae |
|
| 35. |
Phosphate containing fertilisers cause water pollution. Addition of such compounds in water bodies causes..... |
|
Answer» enhanced growth of algae. These plants decompose by micro-organisms consume oxygen. As a result, the amount of dissolved oxygen in the water decreases. This leads to anaerobic CONDITION, COMMONLY with accumulation of obnoxious decay and animal death. Thus bloom infested water inhibits the growth of other living organisms in the water body. This process in which NUTRIENT enriched water bodies support a dense plant population which kills animal life by depriving it of oxygen and RESULTS in subsequent loss of biodiversity is known as eutrophication. |
|
| 36. |
Phosgene gas ("COCl"_(2)) dissociates to carbon monoxide gas and chlorine gas with a change of pressure from 450 mm Hg to 600 mm Hg. If volume and temperature remain constant through out the experiment the value of K_(p) is: |
|
Answer» 400 `450- CANCEL(x)+ cancel(x)+x=600`, x =150 `K_(P)=(PCO.PCl_(2))/(PCOCl_(2)), (150 xx 150)/(300)=75` |
|
| 37. |
Phenylmagnesium bromide reacts with methanol to give |
|
Answer» a mixture of ANISOLE and Mg(OH)BR |
|
| 38. |
Phenyl magnesium bromide reacts with methanol to give |
|
Answer» a mixture of ANISOLE and `MG(OH)BR` |
|
| 39. |
Phenyl acetate on heating with anhy. AlCl_(3) gives |
|
Answer» o & p-Hydroxyacetophenone |
|
| 40. |
Phenols can be converted into salicylic acid using |
|
Answer» `C Cl_(4)` and alkali at 340 K |
|
| 41. |
Phenolphthalein is formed by condensation of phthalic anhydride and C_(6)H_(5)OH (phenol). Which of the following structures shows colour in basic medium ? |
|
Answer»
|
|
| 42. |
Phenolphthalein is not a good indicator for titrating |
|
Answer» NAOH against oxalic ACID |
|
| 43. |
Phenol when it first reacts with concentrated sulphuric acid and then with concentrated nitric acid gives |
|
Answer» nitrobenzene 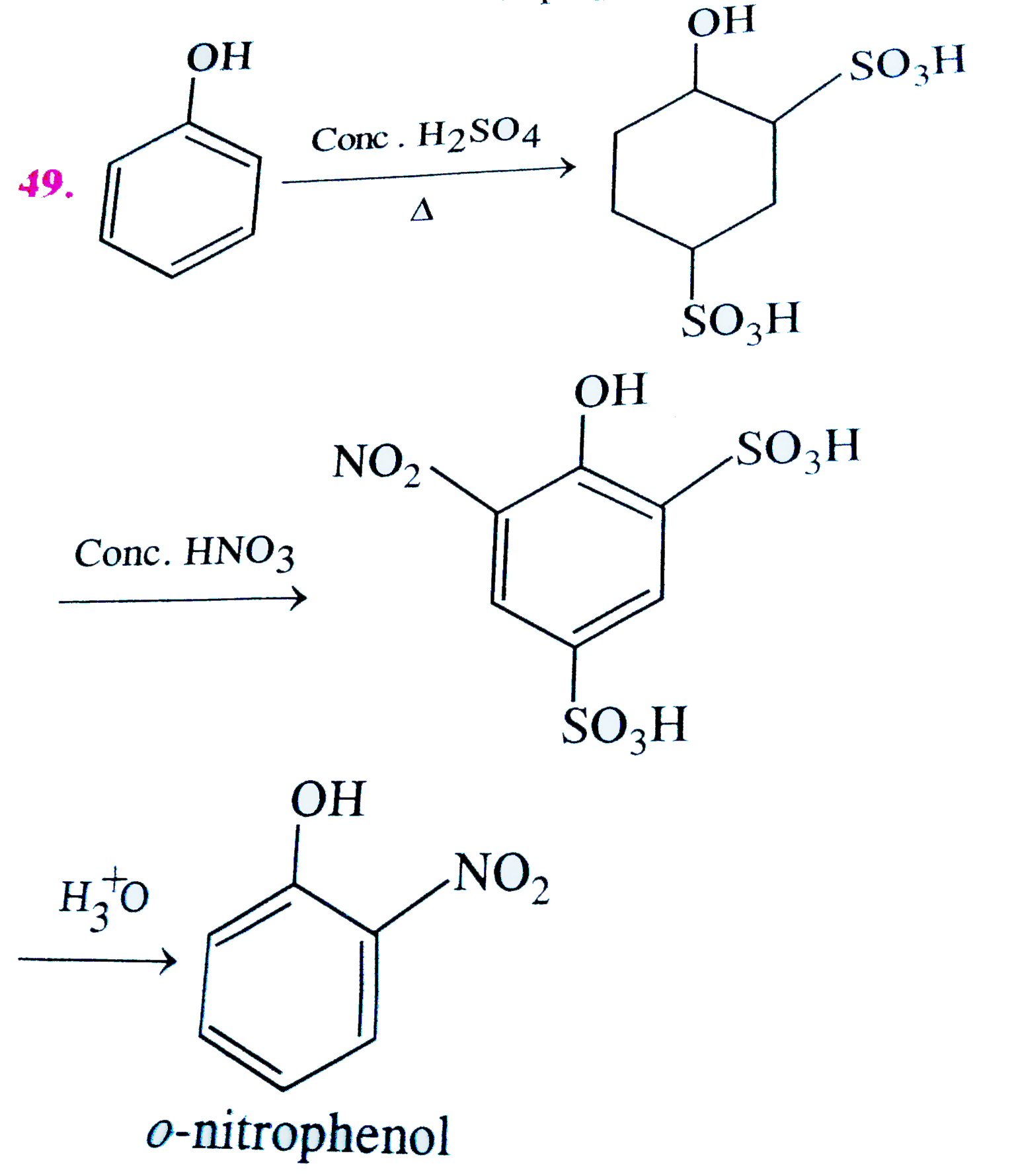 `-SO_(3)H` GROUP are KNOCKED out on boiling with water. |
|
| 44. |
Phenol reacts with which of the following reagents to give salicyladehyde? |
|
Answer» `CH_(3)Cl_(3)+NaOH` |
|
| 45. |
Phenol reacts with bromine in carbon disulphide at low temperature to give : |
|
Answer» o-Bromophenol |
|
| 46. |
Phenol reacts with bromine in chloroform at low temperature to give |
|
Answer» m-bromophenol 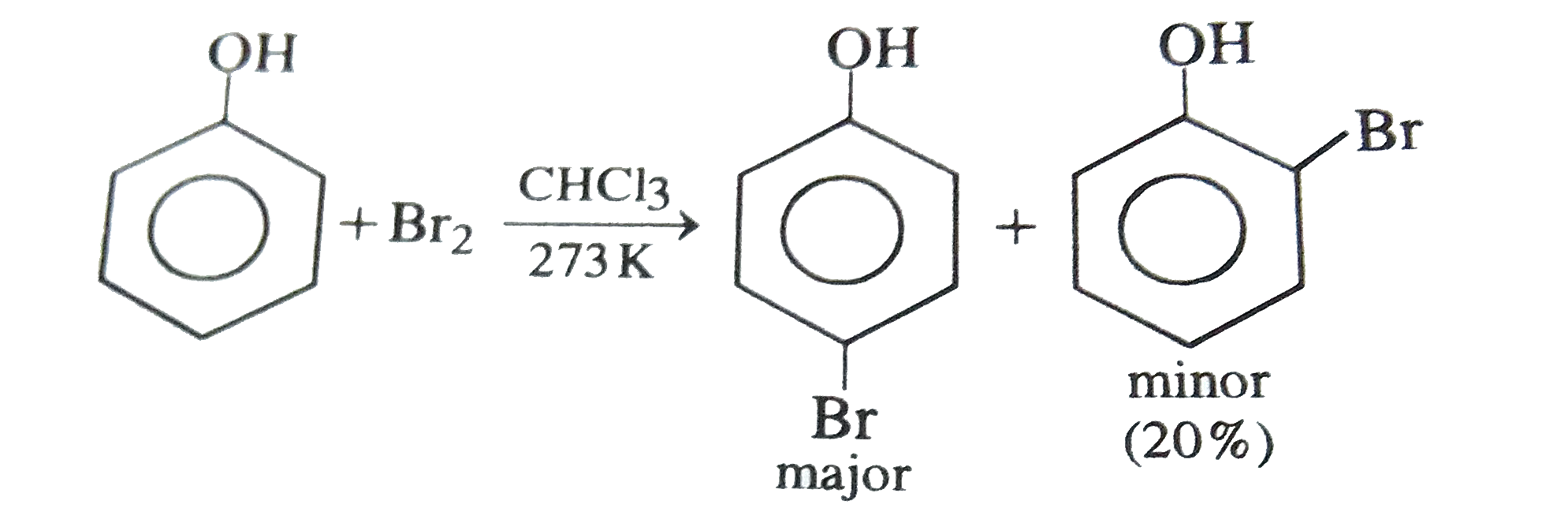
|
|
| 47. |
Phenol, p-Methylphenol, m-Nitrophenol and p-Nitrophenol follows order of increasing acidic strength |
|
Answer» PHENOL, p-Methylphenol, p-Nitrophenol, m-Nitrophenol 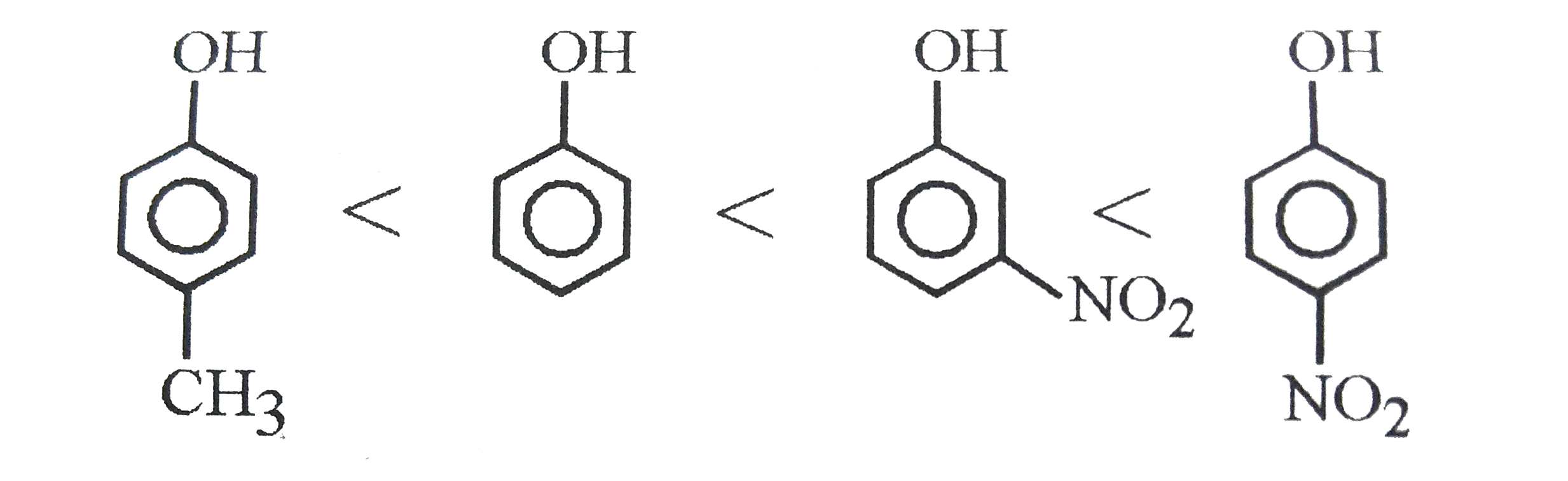
|
|
| 48. |
Phenol overset(X)rarr forms a tribromo derivative. "X" is ...... |
|
Answer» BROMINE in benzene 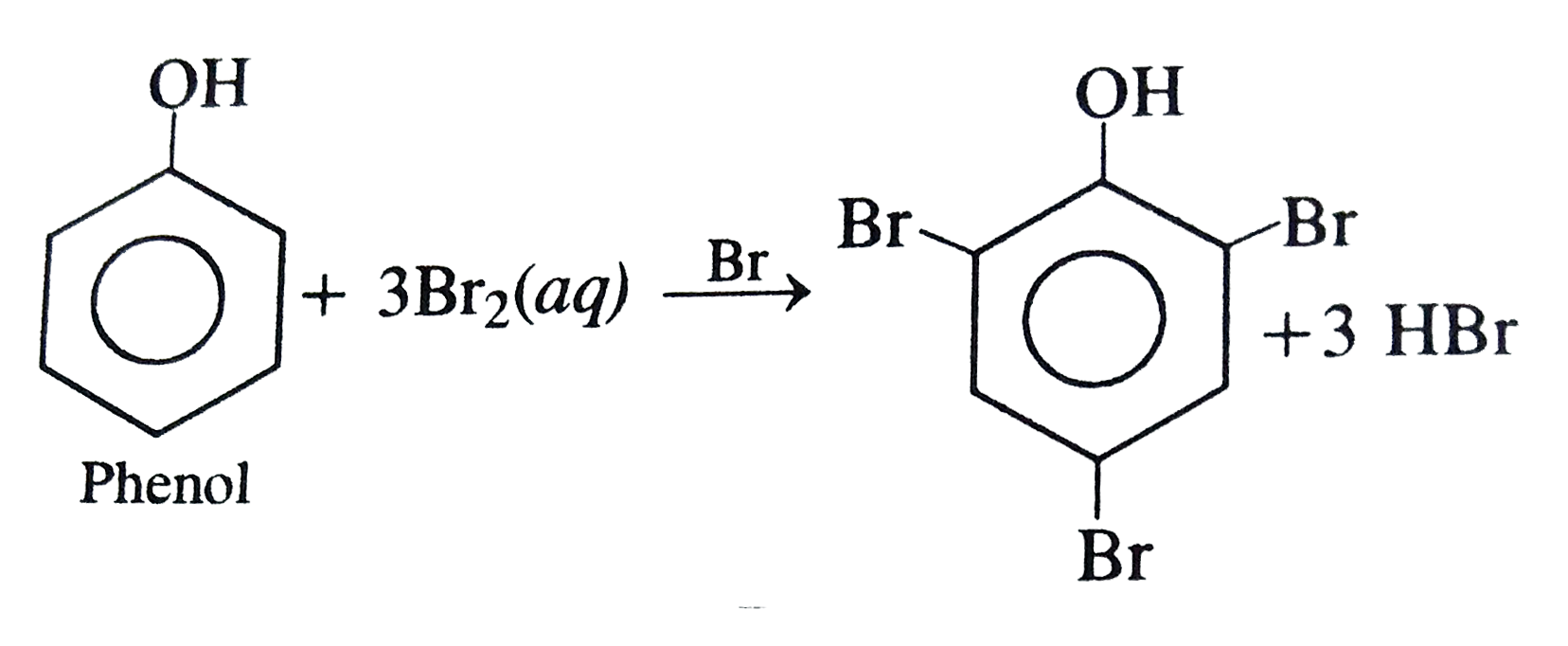
|
|
| 49. |
Phenol on treatment with conc. HNO_(3) gives |
|
Answer» PICRIC ACID 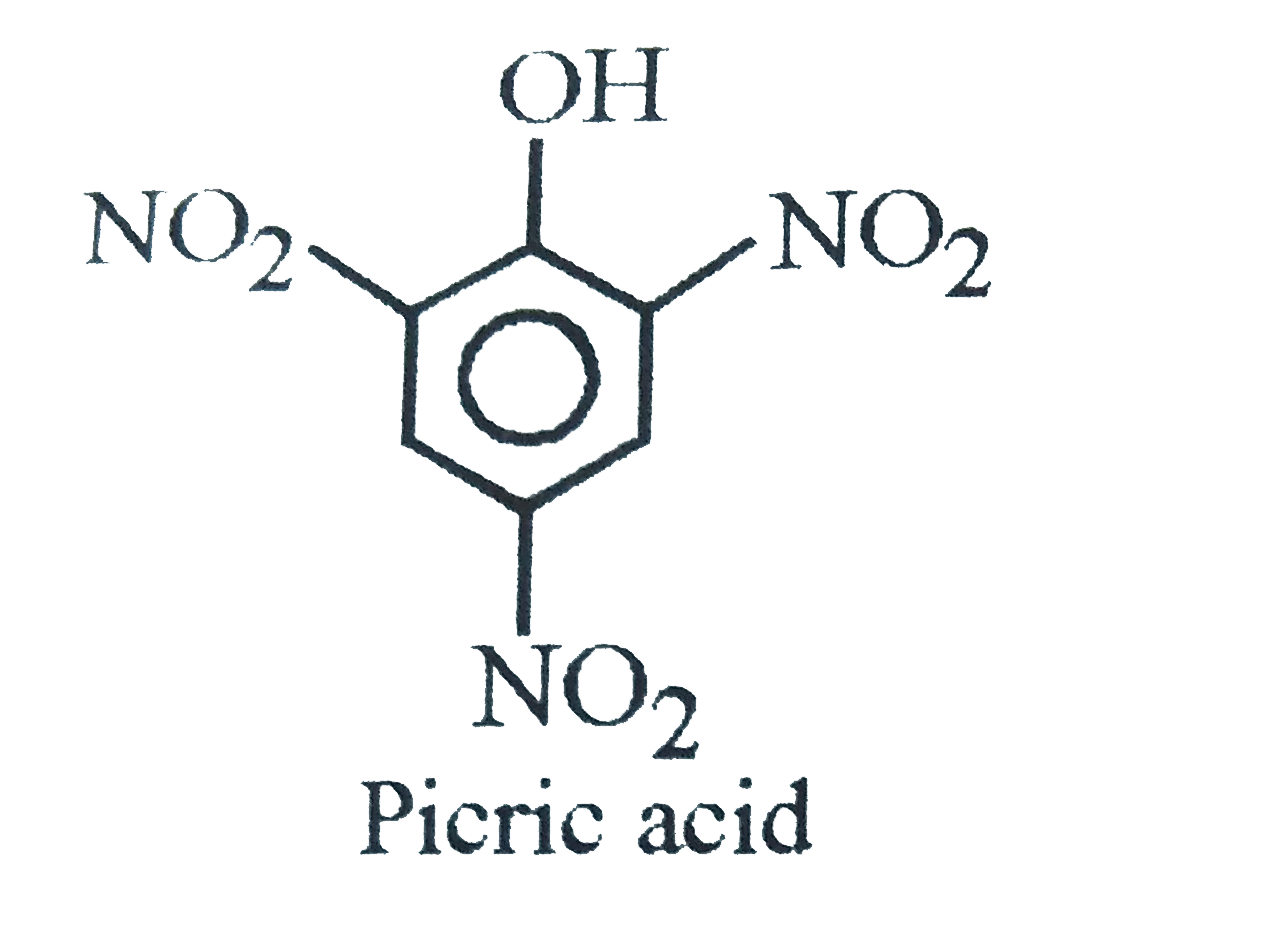
|
|