Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Propanal-1 and pentan-3-one are the ozonolysis products of an alkene. What is th structural formula of alkene? |
|
Answer» `CH_(3)CH_(2)-underset(CH_(2)CH_(3))underset(|)(C)=CH-CH_(2)CH_(3)` |
|
| 2. |
Propanal and pentan-3-one are the ozonolysis products of an alkene ? What is the structural formula of the alkene ? |
Answer» Solution :(i)WRITE the structures of PROPANAL and pentan-3-one with their oxygen atoms facing each other. Remove oxygen atoms and join the TWO fragments by a DOUBLE bond, the structure of the alkene is 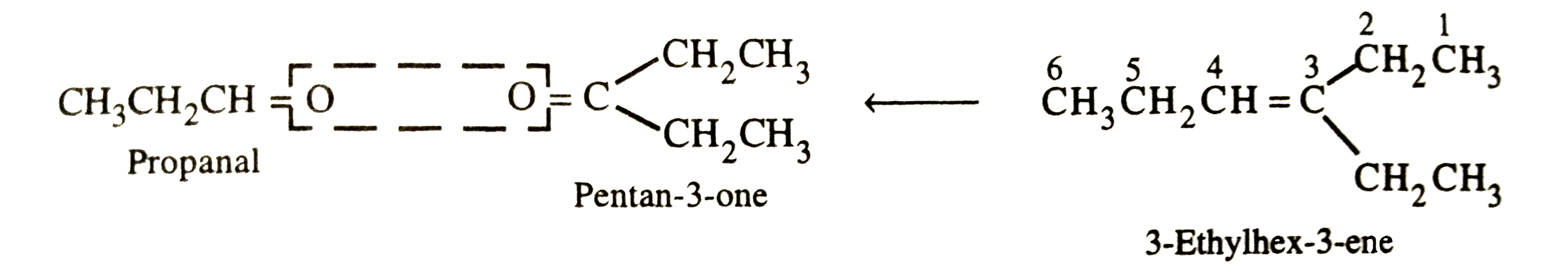
|
|
| 3. |
Propanal and pentan-3-one are the ozonolysis products of an alkene? What is the structural formula of the alkene? |
Answer» SOLUTION :The PRODUCTS of OZONOLYSIS are
|
|
| 4. |
Propan-1-ol can be prepared from propene by |
|
Answer» `H_(2)O //H_(2)SO_(4)` |
|
| 5. |
Propan-1-ol and ethanol can be distinguished by |
|
Answer» LUCAS test |
|
| 6. |
Propadiene overset(HCl(1eq))to A overset(O_2// LiAlH_4)to B + C (B and C are organic products ). If molecular mass of B is 32 then approximate molecular mass of C is |
|
Answer» 98 |
|
| 7. |
Proof spirit contains about |
|
Answer» `40%` alcohol by weight |
|
| 8. |
Proof spirit (by volume) is a mixture of |
|
Answer» `67.1%` ETHYL Alcohol `+32.9%` WATER |
|
| 9. |
Prolonged oxidation of Ethane-1,2,-diol using periodic acid gives : |
|
Answer» ACETIC acid `UNDERSET("Further")(HCHO overset([O])RARR HCOOH)` |
|
| 10. |
Prof. Jonh gave a task to synthesize to three of his stundent ali, divakar and micheal. Difference routes were adopted by them. Ali's method Which of the following statements is correct? |
|
Answer» Ali and DIVAKAR will GET DESIRED product but YIELD will be better in divakar method |
|
| 11. |
Products. The total number of pi bonds in the products is |
|
Answer» |
|
| 12. |
Products of the following reaction Me_2C=CHCH_3 underset((ii)(CH_3)_2S)overset((i)O_3)to ? Are |
|
Answer» `CH_3CHO+CH_3COOH` |
|
| 13. |
Products obtained in above Wurtz reaction is/are . |
|
Answer»

|
|
| 16. |
products formed are |
|
Answer»

|
|
| 17. |
Products A and B formed in the following reactions are respectively: |
|
Answer»
|
|
| 18. |
Products = ? |
|
Answer»
|
|
| 19. |
Product will be |
|
Answer»
|
|
| 20. |
Product/s. The products formed are |
|
Answer» `CH_3 - OVERSET(O)overset(||)C-COOH` |
|
| 21. |
Product of the reaction is |
|
Answer» RACEMIC |
|
| 22. |
Product of the given reaction CH_(3)-CH=CH-Choverset(O_(3)//CH_(2)Cl_(2))underset(-78^(@)C)rarr will be : |
|
Answer» `CH_(3)-CHO` 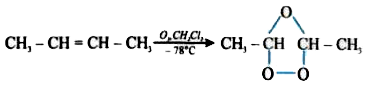
|
|
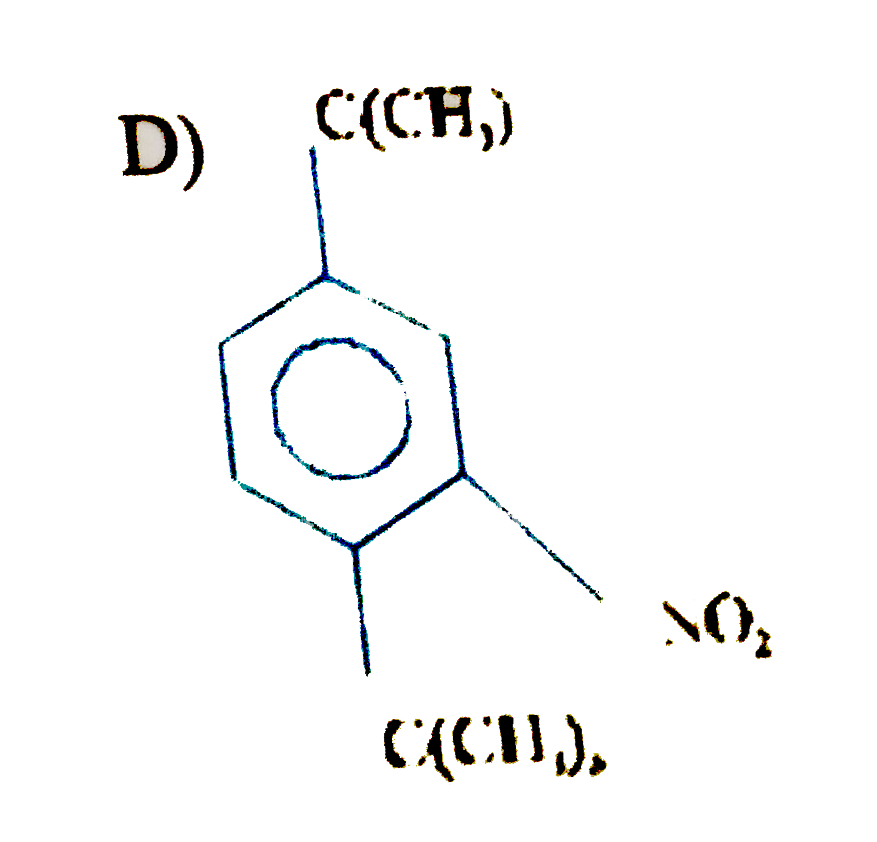
| 23. |
Product of the reaction is |
|
Answer»
Tert-Butyl group was replaced by `NO_(2)`, then it makes benzene ring as deactivating group. |
|
| 25. |
Product of addition of HBr on the alkene C_(6)H_(5) - CH = CH - CH_3 is |
|
Answer» `C_6H_5-UNDERSET(BR)underset(|)(CH)-CH_2-CH_3` |
|
| 28. |
Product obtained in above reaction are : |
|
Answer»
|
|
| 29. |
…………product obtained from the chlorination of benzene sulphonic acid in presence of FeCl_(3). |
| Answer» SOLUTION :m CHLOROBENZENE sulphonic ACID. | |
| 30. |
product not dormed is |
|
Answer»
|
|
| 31. |
Product M cannot respond with : |
|
Answer» 2,4 -DNP |
|
| 32. |
product is |
|
Answer»

|
|
| 33. |
Product is: |
|
Answer» |
|
| 34. |
Product formed is |
|
Answer»

|
|
| 35. |
Product formed on heating Pb(NO_(3))_(2) are |
|
Answer» `PBO,N_(2),O_(2)` |
|
| 36. |
Product can be |
|
Answer»
Due to Aldol CONDENSATION as WELL as dispropornation REACTION. |
|
| 39. |
Product C is : |
|
Answer»
|
|
| 40. |
Product (B) in this reaction is , |
|
Answer»
 CYCLOPENTANONE |
|
| 41. |
Product (A) undergoes how many stroctoral isomers ? |
Answer»  (2) those are (i) positinal isomers.(II) FUNCTIONAL GROUP isomerism. |
|
| 43. |
Product (A) is: |
|
Answer»
|
|
| 44. |
product A is : |
|
Answer»
 (L.D.A = LITHIUM di Isopropyl Amide) L.D.A is sterically hindred BASE, so removes proton from less SUBSTITUTED `alpha-c` of ketone |
|
| 45. |
Product (A) is , |
|
Answer»
|
|
| 46. |
product (A) and (B) in above reactionis : |
|
Answer» `O^(_)-overset(O)overset(||)S-O-H,O^(-)-overset(O)overset(||)S-O-CH_(3)` 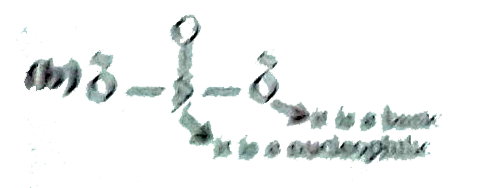 ( sulphur is better nucleophoile than OXYGEN )(but oxygen is better base than sulhur) (A) Is FORMED by ACID -base reaction therefore oxygen will REACT. (B) oid formed by electrophioe and nueophile reaction therefore sulphur wuill react. |
|
| 48. |
Which of the following method of separation can be applied to the mixture of liquids having different boiling points ? |
|
Answer» SIMPLE DISTILLATION |
|
| 49. |
Among the reactions, F_(2(g)) + 2e^(-) to 2F_((g))^(-) and Cl_(2(g)) + 2e^(-) to 2Cl_((g))^(-) which is more feasible ? Give the reason. |
|
Answer» Solution :`F_(2(g)) + 2E^(-) to 2F_((g))^(-)` is easy. Though electron gain ENTHALPY of `Cl_((g))` to give `Cl_((g))^(-)` is more than that of `F_((g))` to give the bond DISSOCIATION of `F_(2(g))` is very less than that of `Cl_(2(g))`. |
|
| 50. |
Process (A) : F_(2(g)) + 2e^(-) rarr 2F^(-)_((g)) , Process (B) : Cl_(2(g)) +2e^(-) rarr 2Cl_(g)^(-) which of these process is easy ? Why ? |
|
Answer» SOLUTION :`F_(2(g)) + 2e^(-) rarr 2F_((g))^(-)` is EASY. Though electron GAIN enthalpy of `Cl_((g))` to give `Cl_((g))^(-)` is more thab that of `F_((g))`to give `F_((g))^(-)` , the bond dissociation of `F_((2(g)))` is very less than that of `Cl_(2(g))` |
|


















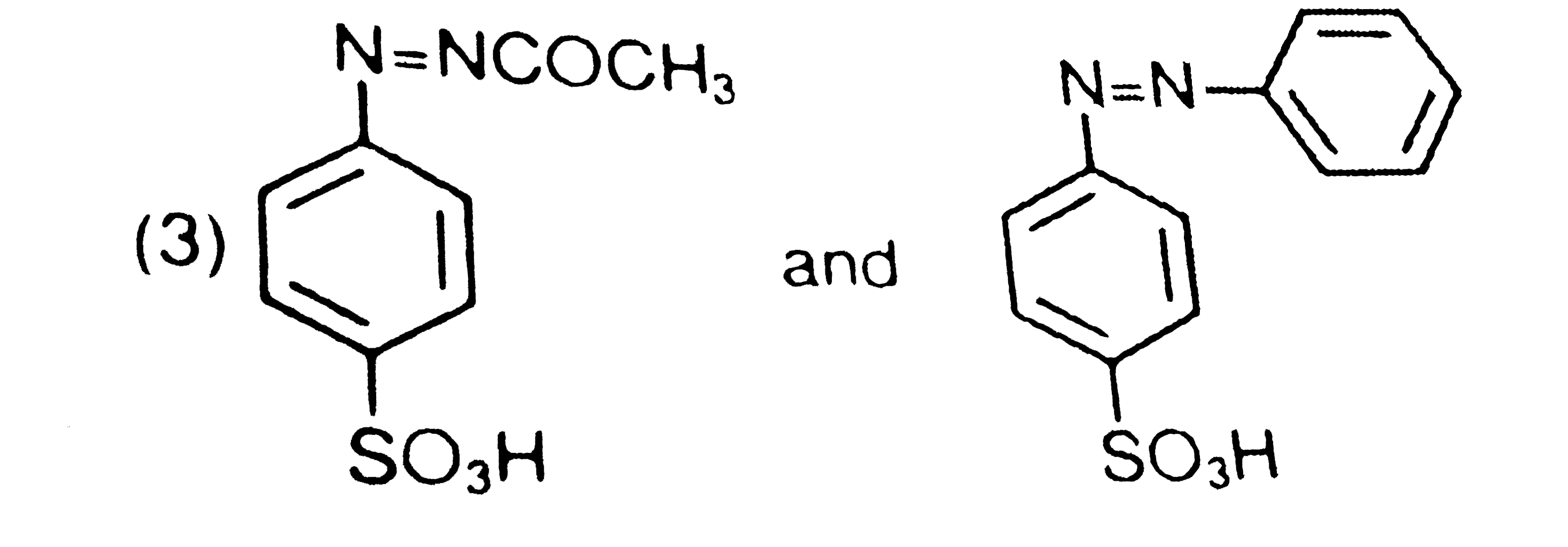
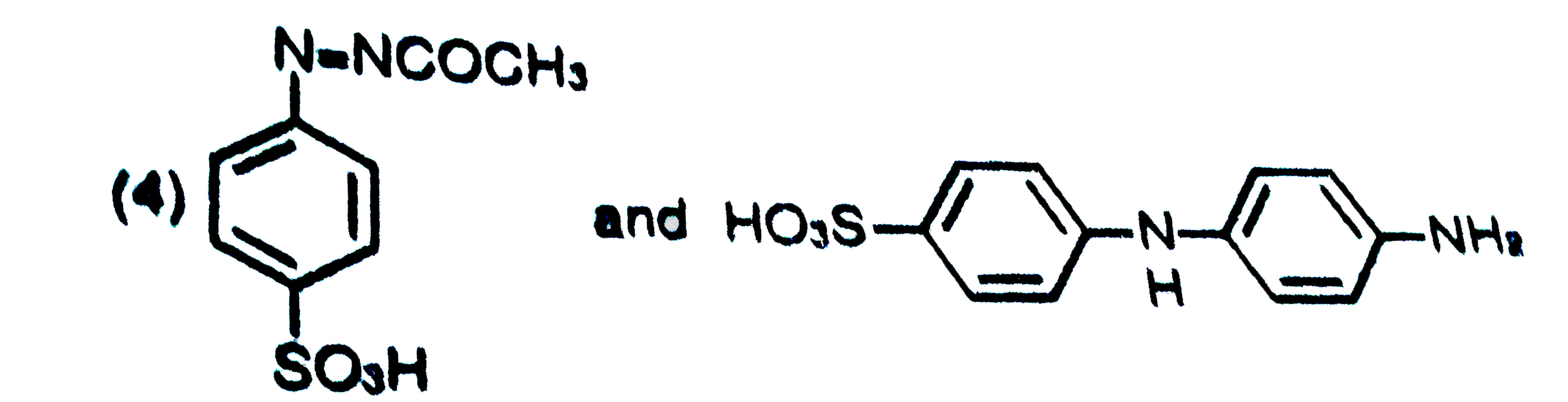




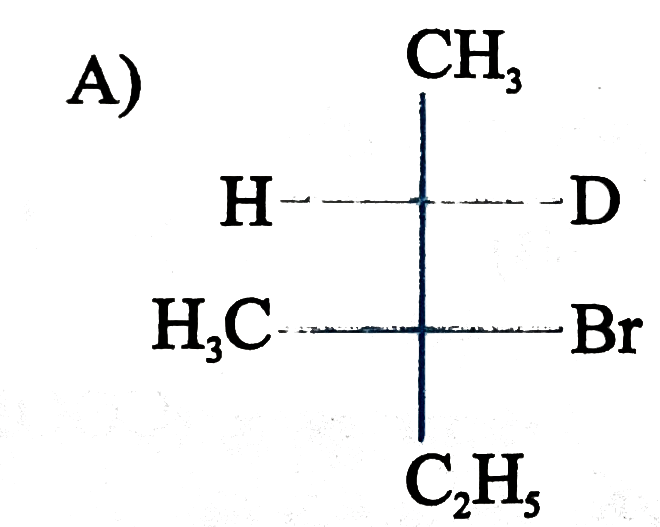
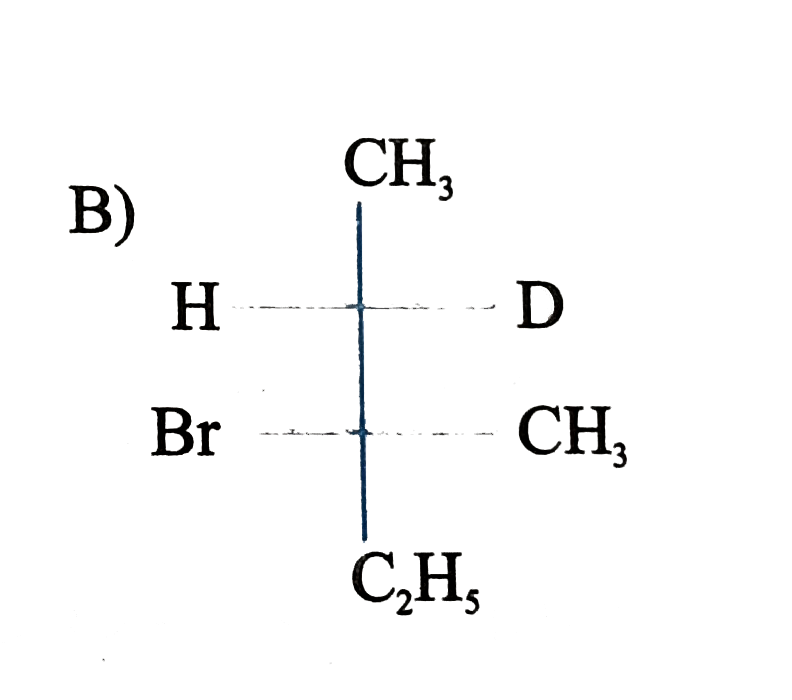
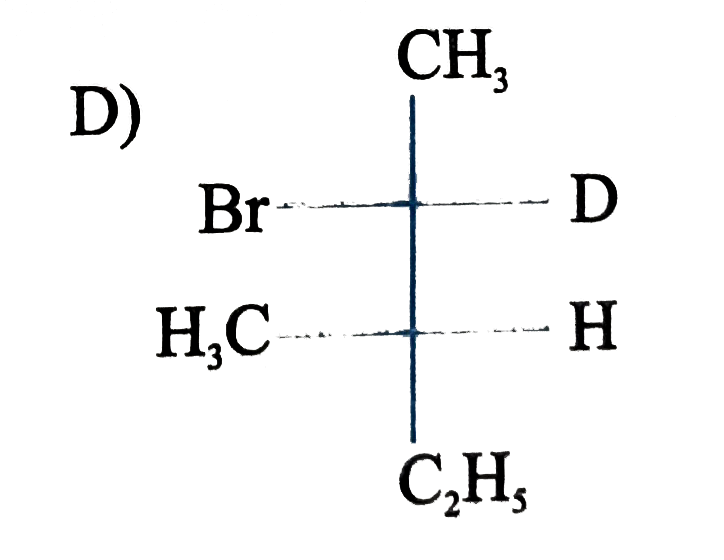
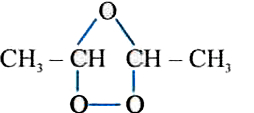
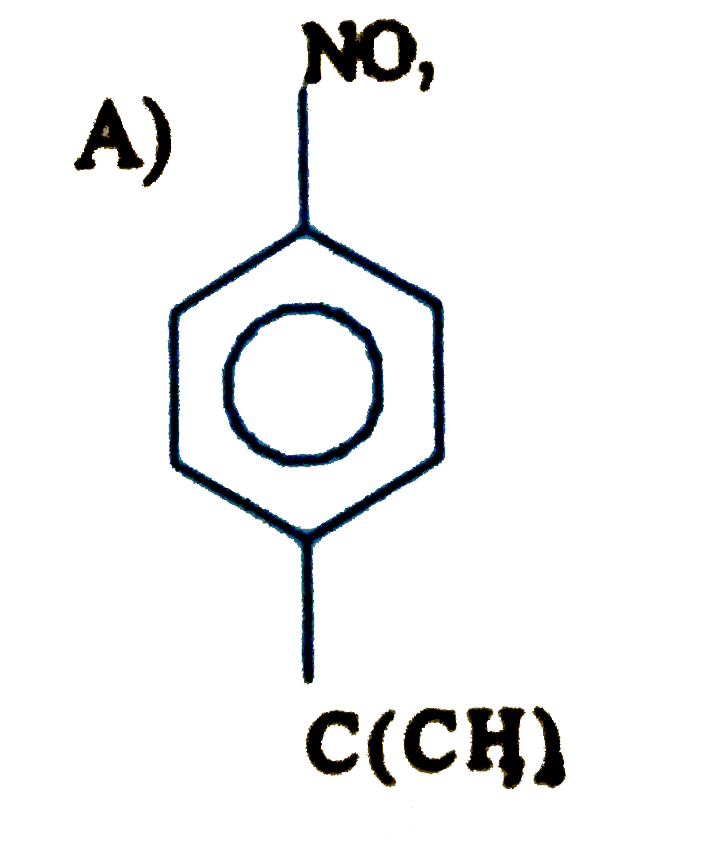
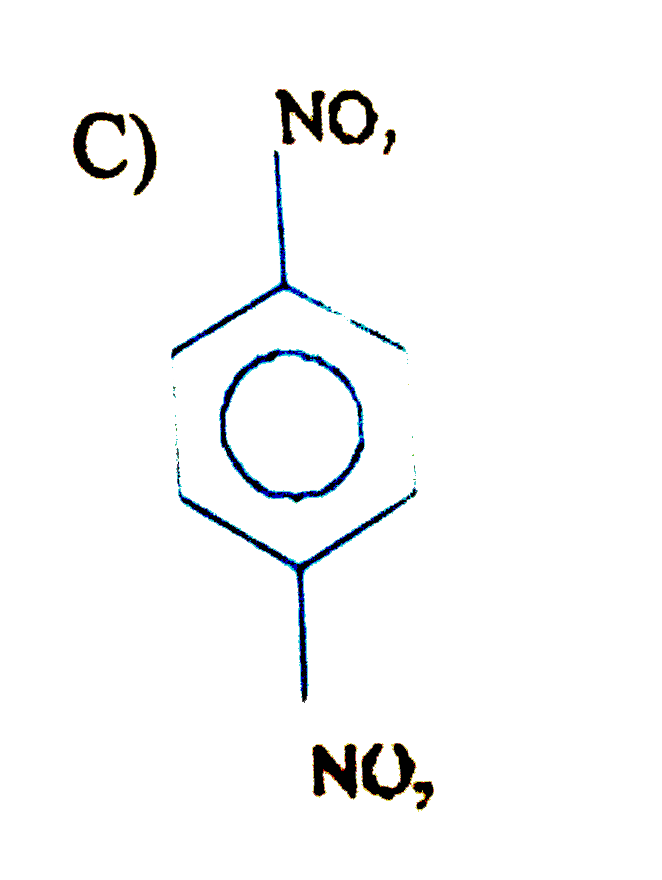





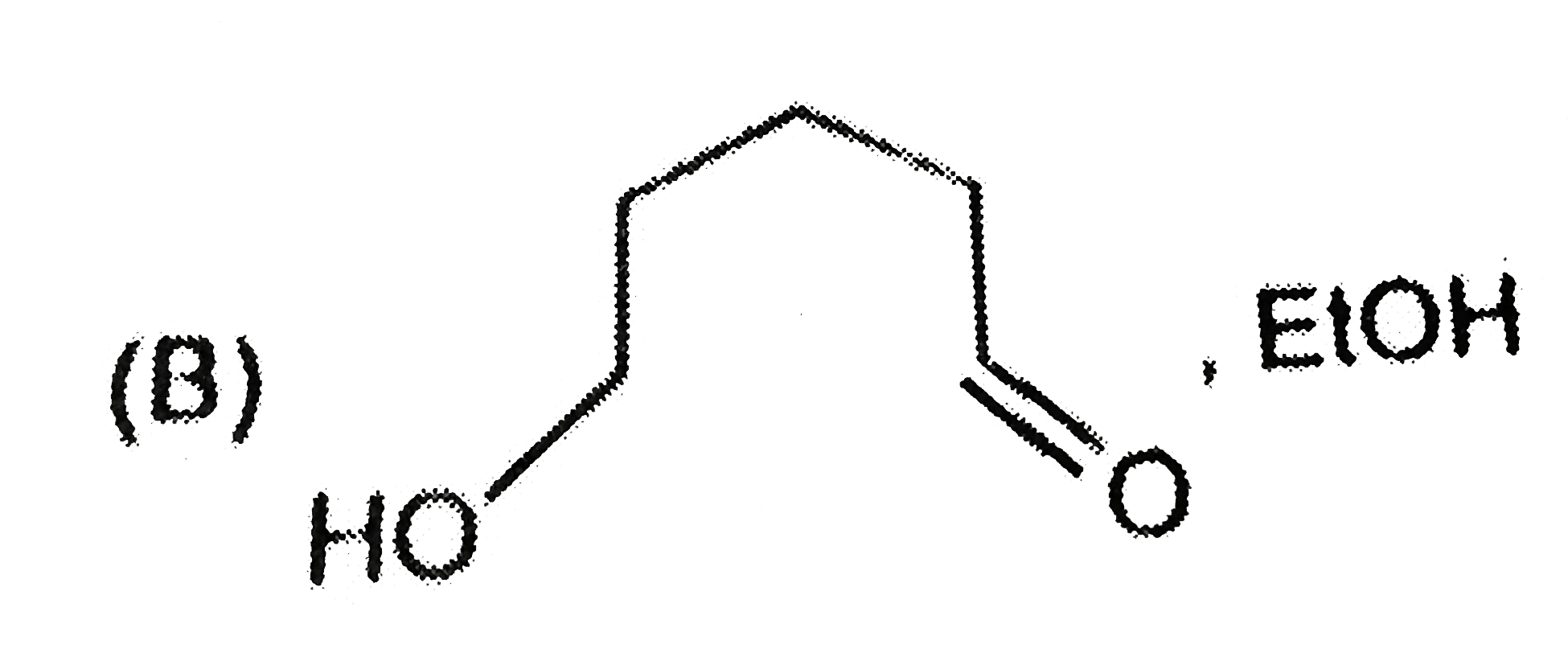
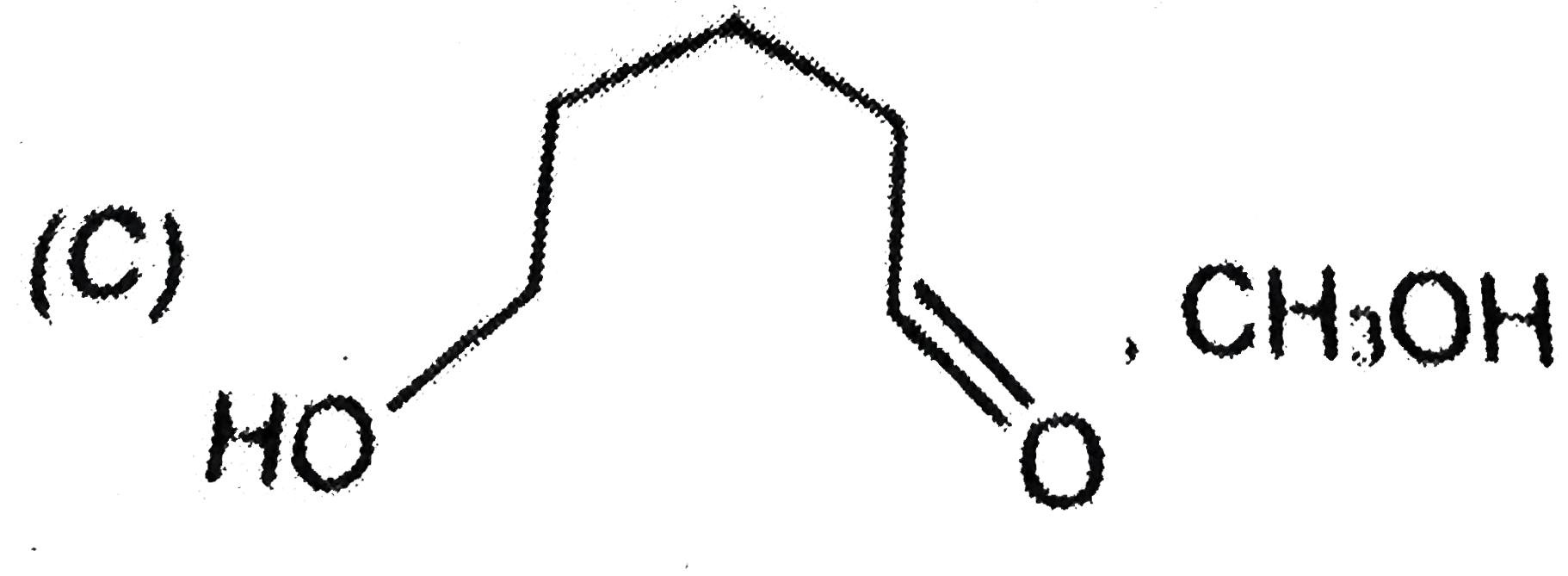
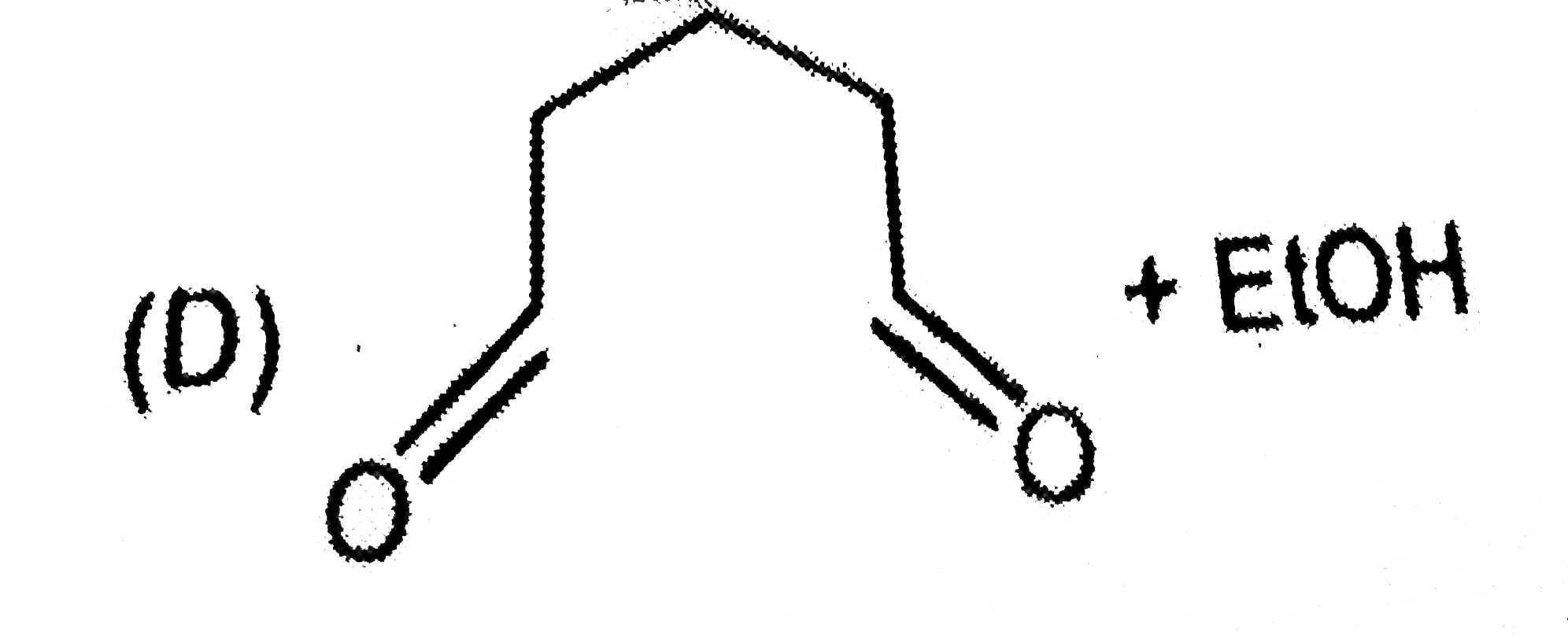
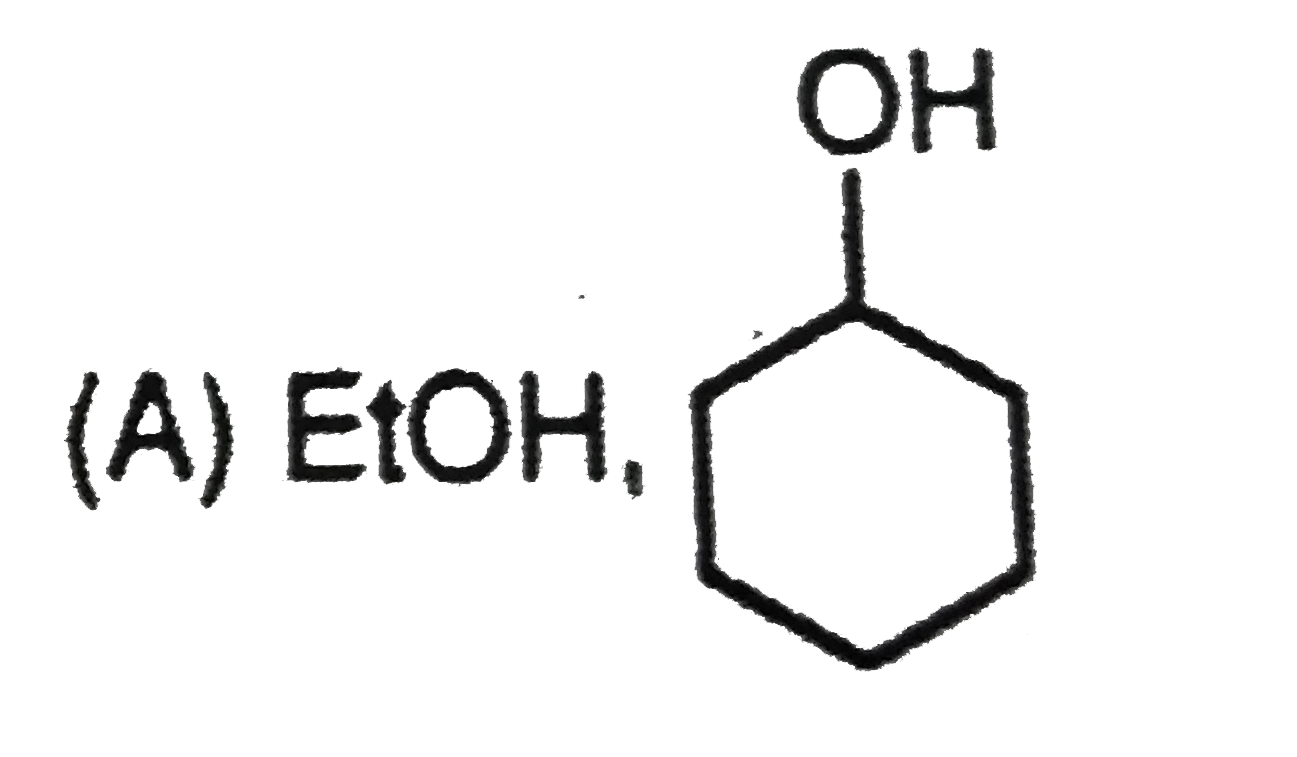
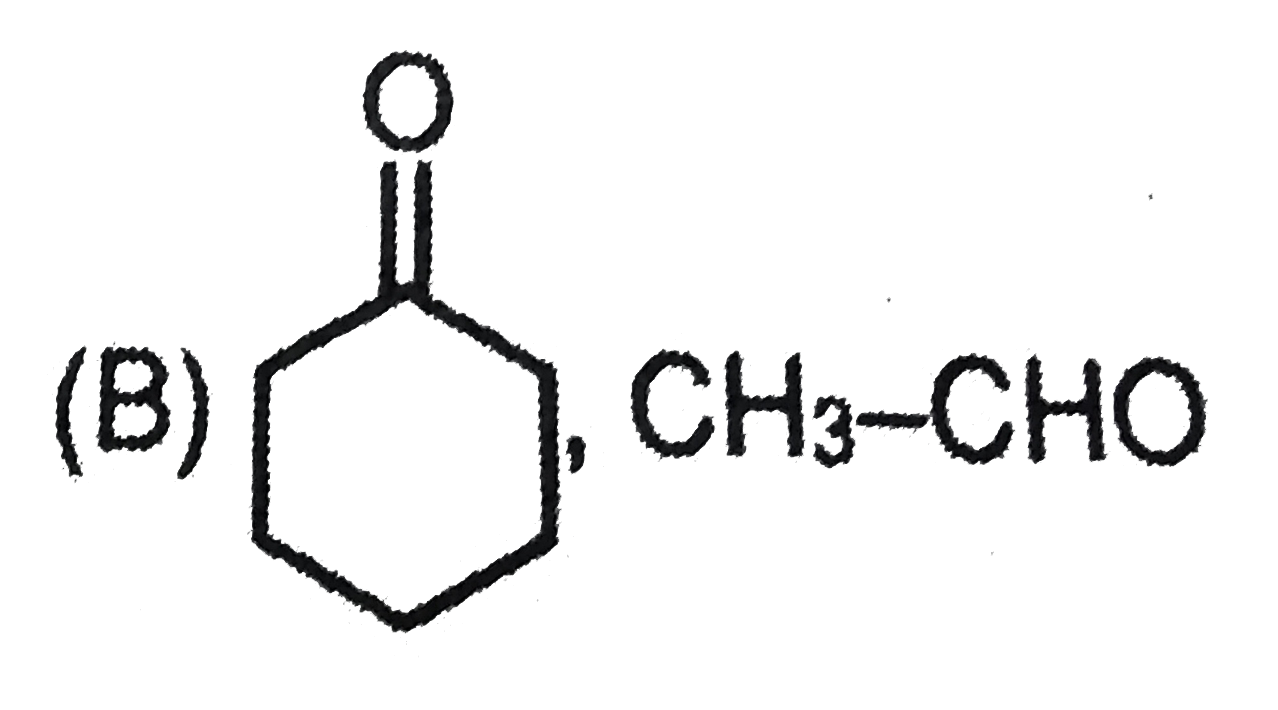
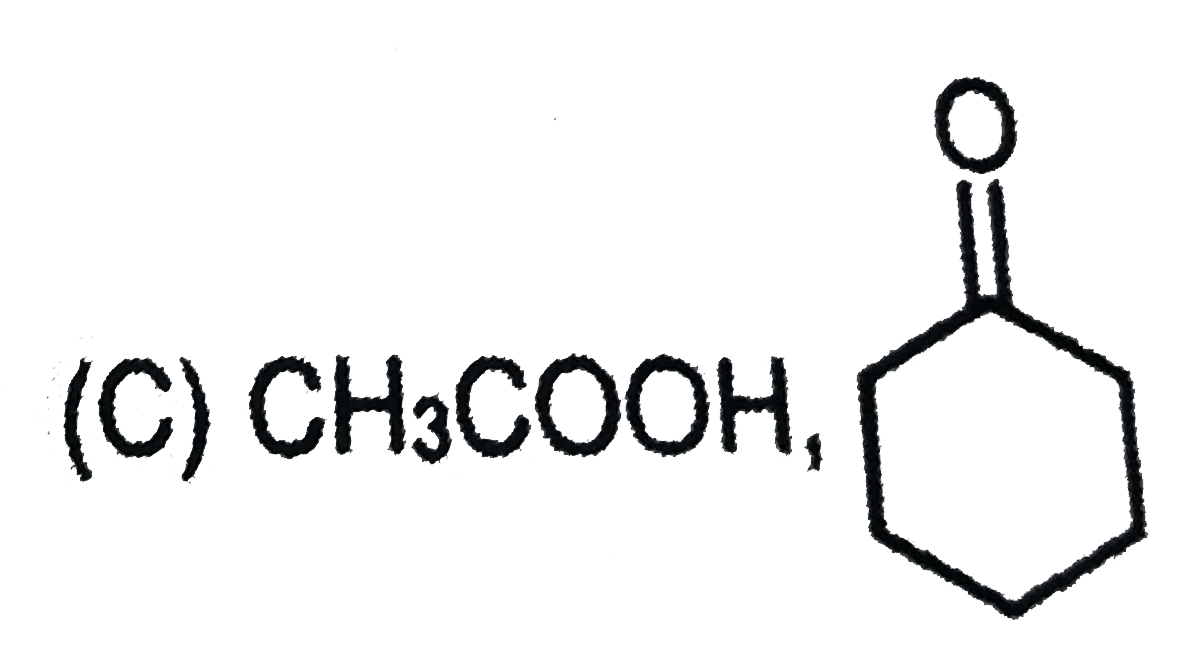
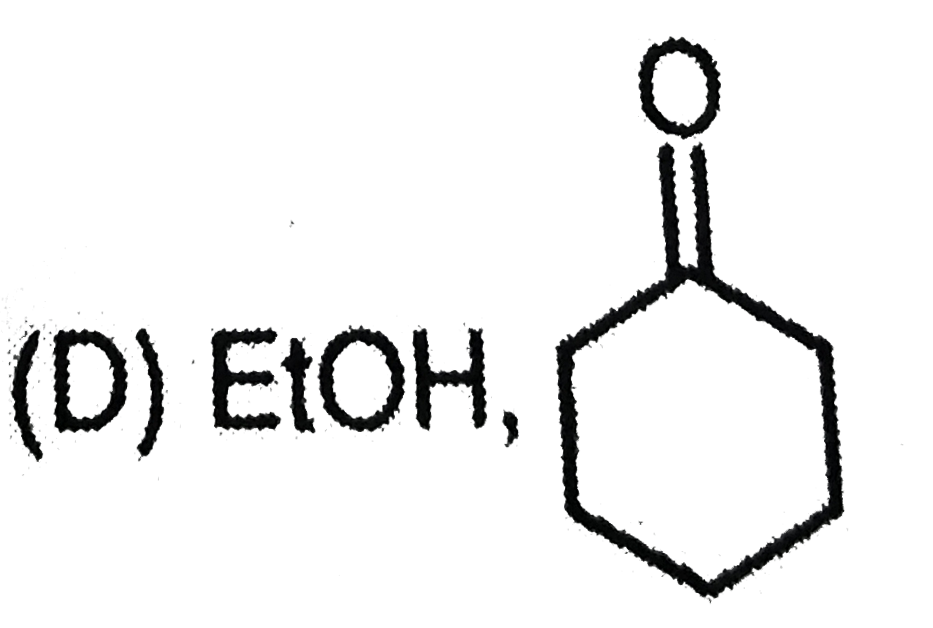




















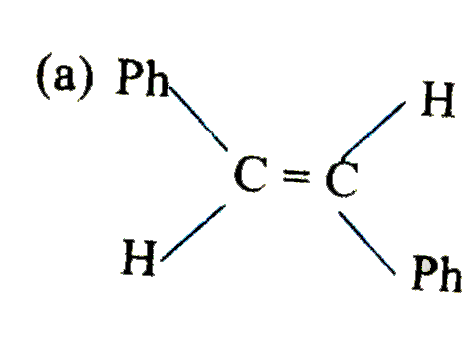
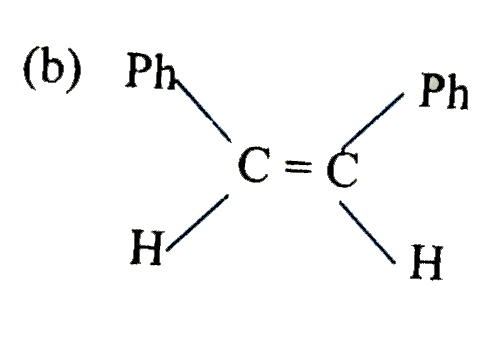
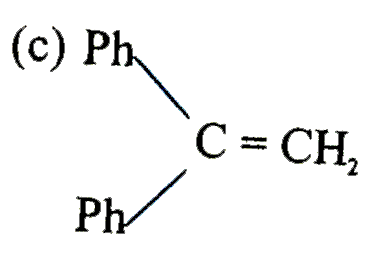
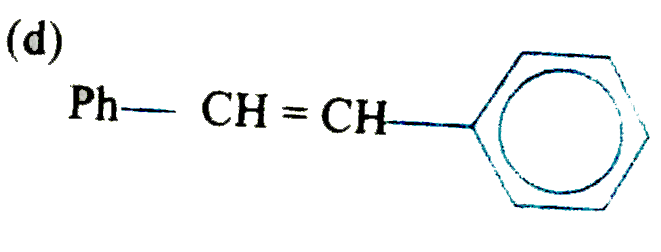
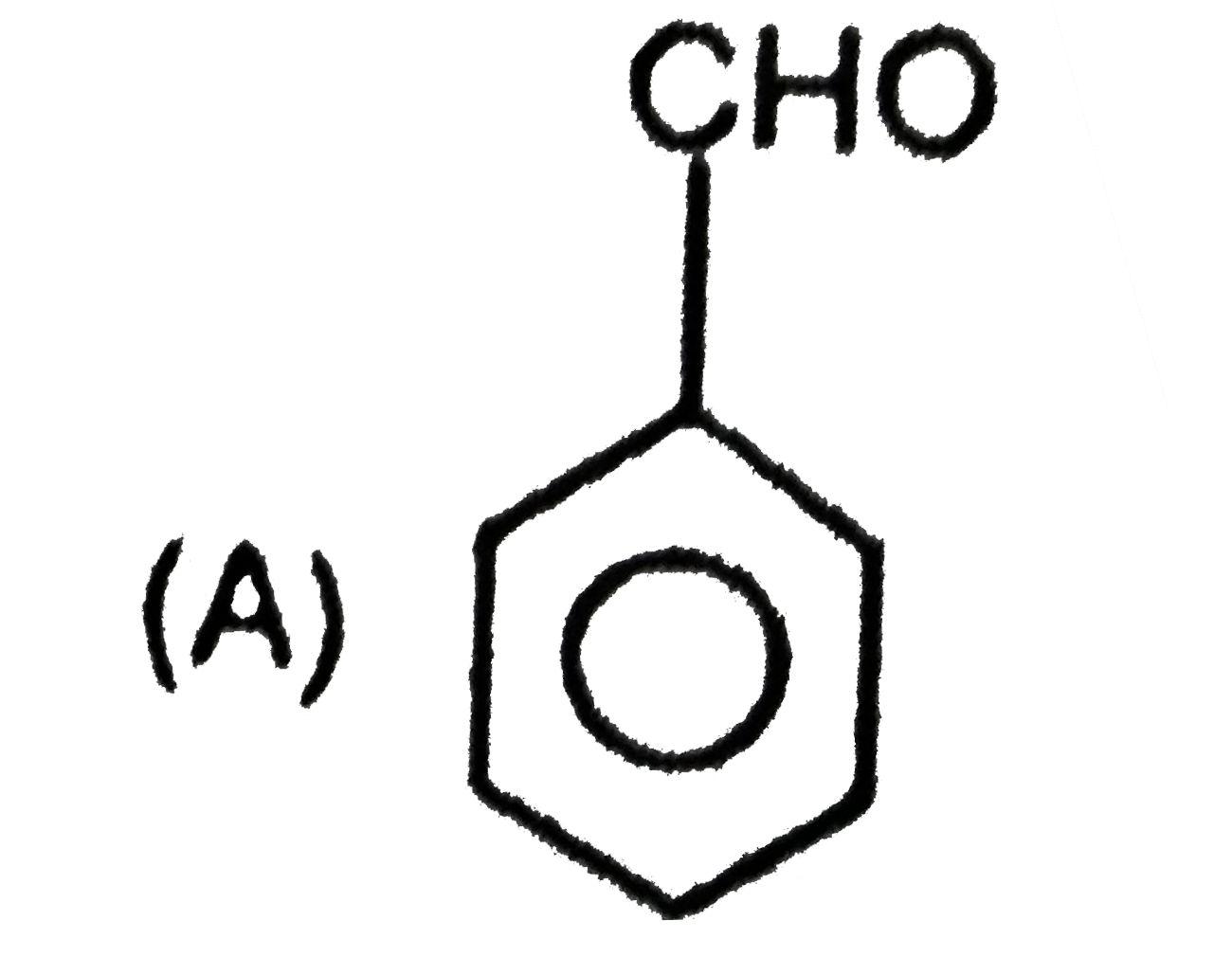
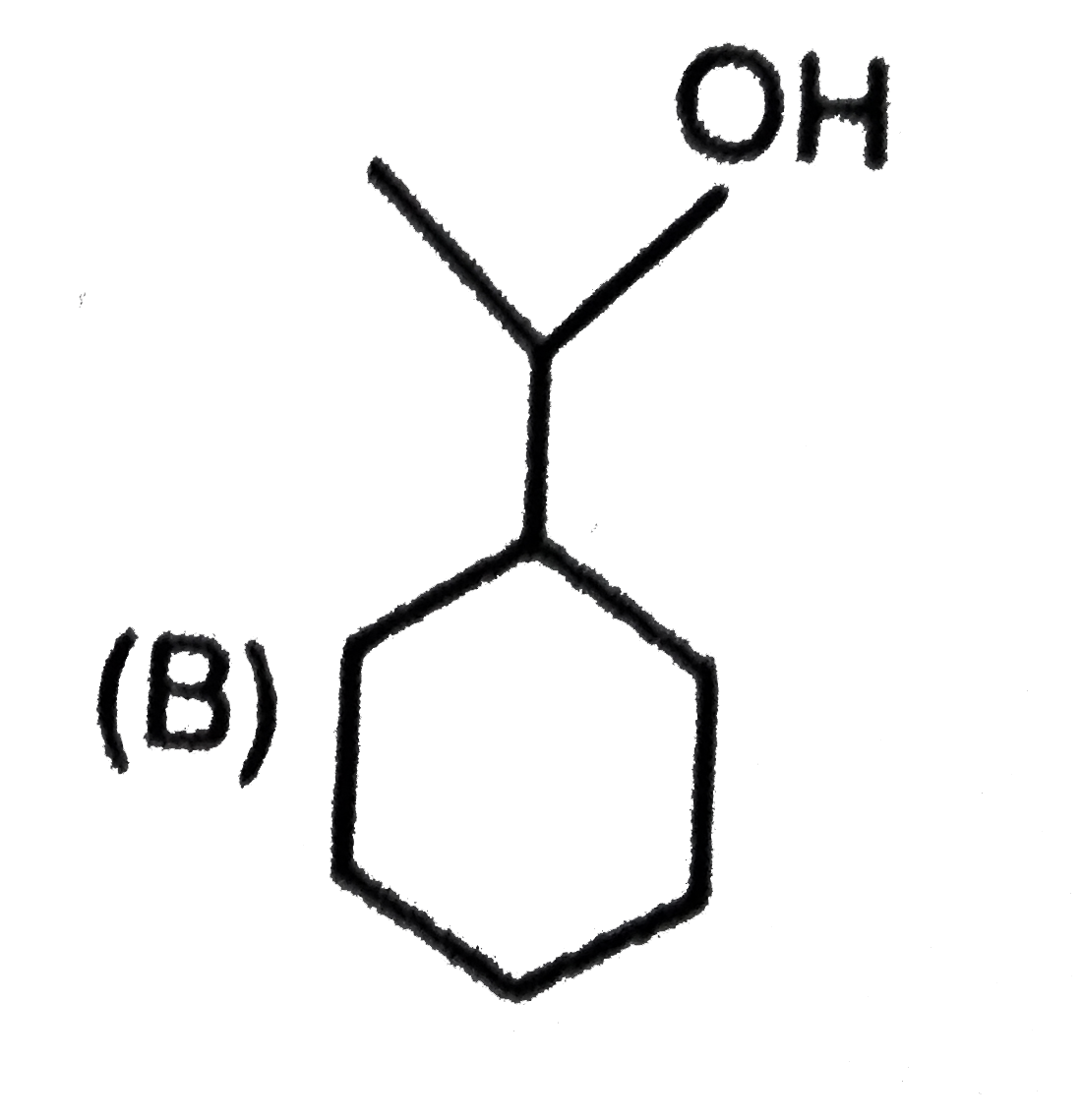
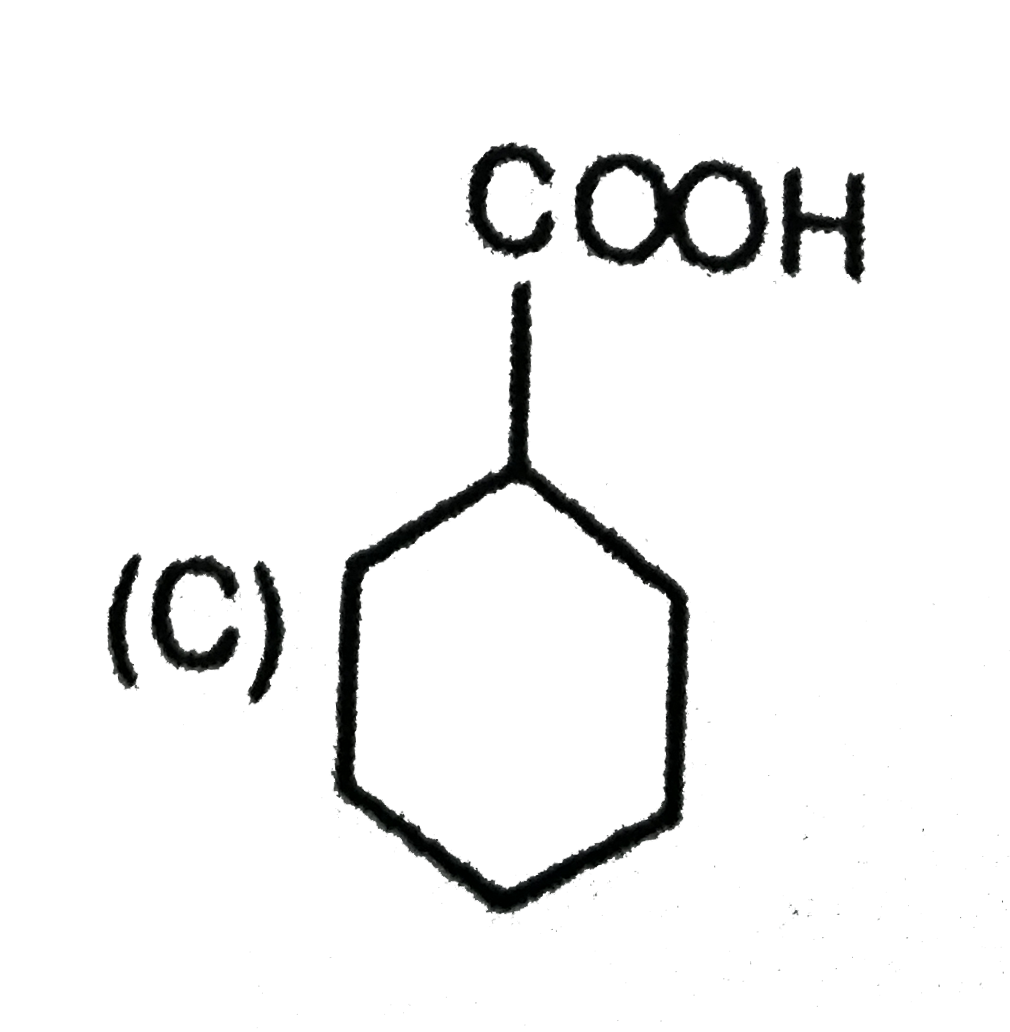
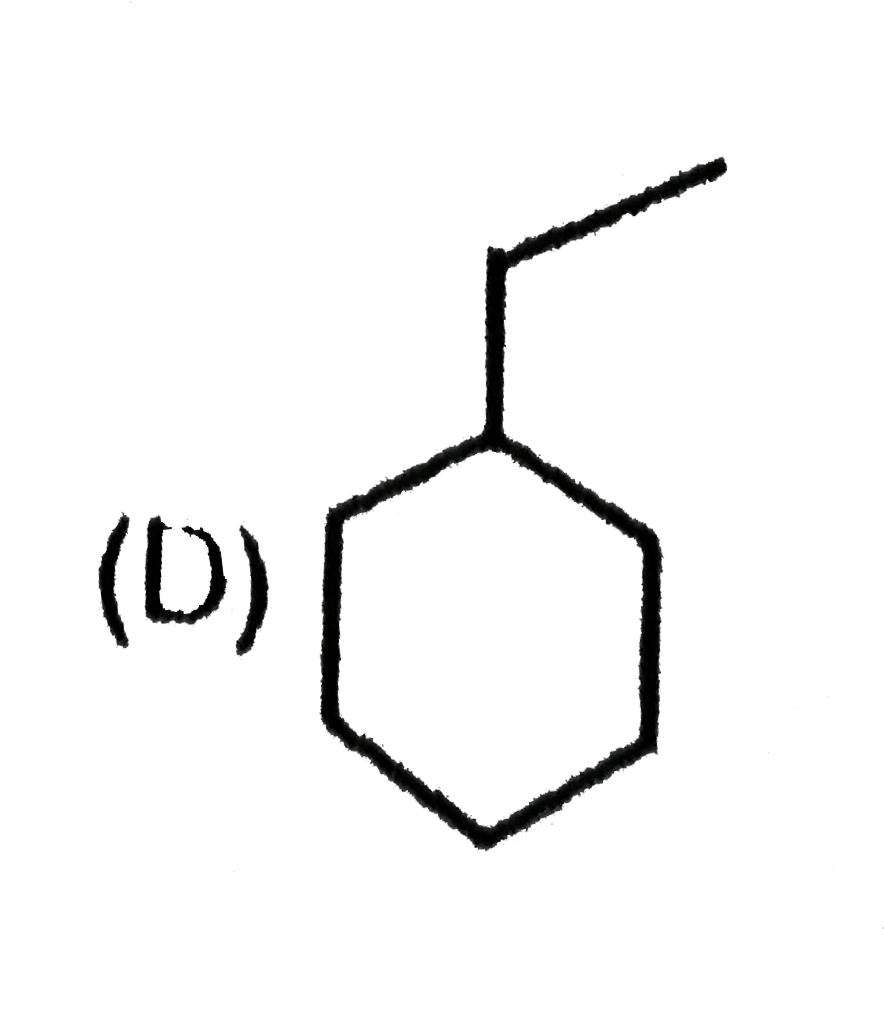








 is
is