Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Prove that the degree of dissociation of a weak monoprotic acid is given by alpha = (1)/(1+ 10^((pK_(a)-pH))) where K_(a) is the dissociation constant of the acid. |
|
Answer» Solution :Suppose we start with C mol `L^(-1)` of the weak monoprotic ACID HA. Then ` {:(,HA,HARR,H^(+),+,A^(-)),("INITIAL molar conc.",C,,,,),("Molar conc.",C-C alpha,,C alpha,,C alpha),("after dissociation",=C (1-alpha),,,,),(,,,,,):}` Thus, `K_(a) = (C alpha. C alpha)/(C (1-alpha))=(C alpha^(2))/(1-alpha) or C = (K_(a)(1-alpha))/(alpha^(2)) ...(i)` Also, `[H^(+)]=C alpha` ...(ii) Substituting the value of C from eqn. (i) , we get `[H^(+)]=(K_(a)(1-alpha))/(alpha^(2)) xx alpha=(K_(a)(1-alpha))/(alpha)` `:. -log (H^(+)]=-[log K_(a) + log (1-alpha)-log alpha]or pH = pK_(a) - log (1-alpha) + log alpha` or `log.(1-alpha)/(alpha) = pK_(a) - pHor (1-alpha)/(alpha) = 10^(pK_(a)-pH) or (1)/(alpha)-1=10^(pK_(a)-pH)` or `(1)/(alpha) = 1 + 10^(pK_(a)-pH) or alpha= (1)/(1+10^(pK_(a)-pH))` |
|
| 2. |
Prove that the halogens, chlorine, bromine and iodine follow the law of triads. |
Answer» Solution :When the halogens, chlorine, bromine and IODINE are placed on below the OTHERS, they had SIMILAR properties. The atomic weight of bromine was close to the average of the atomic WEIGHTS of chlorine and iodine. 
|
|
| 3. |
Prove that states of matter are interconvertible. |
|
Answer» Solution :States of MATTER are interconvertible by CHANGING temperature and PRESSURE `"solid" underset("HEAT")overset("cool")(hArr) "LIQUID" underset("heat")overset("cool")(hArr) Gas`. |
|
| 4. |
Prove that the atomic radii is a periodic property. |
|
Answer» Solution :Atomic radius is the distance between the centre of its nucleus and the outermost shell containing the electron. Atomic radius is a periodic property. (i) VARIATION in periods: The atomic radius decreases while going from left to right in a period. As we move from left to right in a period, the nuclear charge increases by one unit in each succeeding element. But the NUMBER of the shell remains same. Hence, the electrons are attracted strongly by the nucleus. Hence the atomic radius decreases along the period. In 2nd period `r_(Li)gtr_(Be)gtr_(B)gtr_(C)gtr_(N)gtr_(O)gtr_(F)` (ii) Variation in a group: The atomic radius of ELEMENTS increases with increase in atomic number as we move from top to bottom in a group. The attraction of the nucleus for the electrons decreases as shell number increases. Hence atomic radius increases along the group. In `1^(st)` group `r_(Li)ltr_(NA)ltr_(K)ltr_(Rb)ltr_(Cs)` Hence, atomic radii is a periodic property. |
|
| 5. |
Prove that reaction between fluorine and ice is disproportionation reaction : H_(2)O_((s))+F_(2(g))toHF_((g))+HOF_((g)) |
|
Answer» Solution :`OVERSET(+1)(H_(2))overset(-2)(O)+overset(0)(F_(2))tooverset(+1)(H)overset(-1)(F)+overset(+1)(H)overset(-2)(O)overset(+1)(F)` In above reaction oxidation and reduction of F OCCURS. Therefore given reaction is disproportionation reaction. |
|
| 6. |
Prove that relative lowering of vapour pressure is a colligative property. |
|
Answer» Solution :The VAPOUR pressure of a solution containing a nonvolatile , non-electrolyte solute is ALWAYS lower than the vapour pressure of the pure solvent . Consider a CLOSED system in which a pure solvent is in equilibrium with its vapour At equilibrium the molar Gibb's free energies of solvent in the liquid and gaseous phase are equal ` (Delta G=0 ) ` When a solute is added to this solvent , the dissolution takes place and its free energy (G) decreases due to increase in entropy .In order to maintain the equilibrium the free energy of the vapour phase must also decrease . At a given temperature ,the only way to lower the free energy of the vapour is to reduce its pressure Thus the vapour pressure of the solution must decrease to maintain the equilibrium. ` (##SUR_CHE_XI_V02_C09_E05_007_S01.png" width="80%"> We know that from the RAOULT's law the relative lowering of the vapour pressure is equal to the mole fraction of the solute From the above equation , it is clear that the relative lowering of vapour pressure depends only on the mole fraction of the solute ` (X_B) ` and is independent of its nature . Therefore , relative lowering of vapour pressure is a colligative property. |
|
| 7. |
Prove that pH+pOH=pK_(w) at 298 K. |
|
Answer» Solution :The ionic PRODUCT of water is given by `[H^(+)][OH^(-)]=K_(w)=1.0xx10^(-14)` at 298 K TAKING LOG on both sides, `log_(10)[H^(+)]+log_(10)[OH^(-)]=log_(10)K_(w)=log_(10)1.0xx10^(-14)` Multipling by -ve SIGN, `-log_(10)[H^(+)]-log_(10)[OH^(-)]=log_(10)K_(w)=-14.0000` wkt by definition, `pH+pOH=pK_(w)=14`. |
|
| 8. |
Prove that ionization energy is a periodic property. |
|
Answer» SOLUTION :Variation in a PERIOD: On moving across a period from left to right, the ionization enthalpy VALUE increases. This is due to the FOLLOWING reasons: • Increase of nuclear charge in a period • Decrease of atomic size in a period Because of these reasons, the valence electrons are held more tightly by the nucleus, thus ionization enthalpy increases. Hence, ionization energy is a periodic property. Variation in a group: As we move from top to bottom along a group, the ionization enthalpy decreases. This is due to the following reasons: • A gradual increase in atomic size • Increase of SCREENING effect on the outermost electrons due to the increase of number of inner electrons. Hence, ionization enthalpy is a periodic property. |
|
| 9. |
Prove that Hydrogen peroxide is a vigorous oxidising agent and the solution of H_(2)O_(2) is slightly acidic. |
|
Answer» Solution :`H_(2)O_(2(aq))+2H_(2)O_((l))leftrightarrowH_(3)O_((aq))^(+)+HO_(2(aq))^(-)` `H_(3)O^(+)` Hydronium ion formation PROVES that solution of H, 0, is acidic. Because it donates `H_(2)O` to `H_(2)O` to form `H_(3)O^(+)` ion. `H_(2)O^(+)` oxidises Ferrous SULPHATE to Ferric sulphate in acidic medium `2FeSO_(4(aq))+H_(2)SO_(4(aq))+H_(2)O_(2(aq))toFe_(2)(SO_(4))_(3(aq))+2H_(2)O_((l))` |
|
| 10. |
Prove that H_(2)O_(2) act as reducing agent in alkaline medium. |
|
Answer» Solution :In alkaline CONDITIONS, `H_(2)O_(2)` act as a reducing AGENT. `2KMnO_(4(aq))+3H_(2)SO_(4(aq))+5H_(2)O_(2(G))toK_(2)SO_(4)+2MnSO_(4)+8H_(2)O_((L))+5O_(2(g))` |
|
| 11. |
Prove that gypsum is a natural insulator |
|
Answer» Solution :Gypsum have low THERMAL conductivity. (ii) It won.t allow the ELECTRIC current to PASS through it. So it is known as natural INSULATOR |
|
| 12. |
Prove that f the uncertainty in position of a moving electron is equal to its de-Brogile wavelength, then its velocity is completely uncertain |
|
Answer» Solution :Here, we are given that `Delta X = lamda` By de-Brogile equation, `lamda = (h)/(p) or p = (h)/(lamda) = (h)/(Delta x) or Delta x = (h)/(p)` By uncertainty principle, `Deltax. Delta p GE (h)/(4pi) :. (h)/(p). Delta p. ge (h)/(4pi) or (Deltap)/(p) ge (1)/(4pi) or (m Deltav)/(mv) ge (1)/(4pi) or (Delta v)/(v) ge (1)/(4pi) or Delta v ge (v)/(4pi)` THUS, uncertainty in velocity is so LARGE that its velocity is UNCERTAIN |
|
| 13. |
Prove that calcium oxide is basic oxide. |
|
Answer» Solution :CALCIUM oxide is a basic oxide. It combines with acidic oxides at high temperature 1. `underset("Basic oxide")(CaO) + underset("SILICA")(SiO_(2)) rarr underset("Calcium silicate")(CaSiO_(3))` 2 `6CaO + underset(underset("pentoxide")("PHOSPHOROUS"))(P_(4)O_(10)) rarr underset("Calcium phosphate")(2Ca_(3) (PO_(4))_(2))` |
|
| 14. |
Resistance of the wire is inversely proportional to length of the wire. |
|
Answer» SOLUTION :(i) Bond ORDER `prop(1)/("Bond length")` (ii) An EXAMPLE for illustrating the above equation is: Carbon-carbon single bond length `(1.54A)`is LONGER than the carbon-carbon DOUBLE bond length (1.34A) and the carbon carbon triple bond length (1.20A). |
|
| 15. |
Prove that calcium oxide is a basic oxide |
|
Answer» SOLUTION :Calcium oxide is a BASIC oxide. It combines with acidic oxides at high temperature. `underset("Basic oxide")(CaO) +underset(SILICA )(SiO_(2)) tounderset("Calcium silicate")(CaSiO_(3))` `6CaO+underset("PENTOXIDE")underset("Phosphorous")(P_(4)O_(10))to2Ca_(3)underset("Calcium phosphate")((PO_(4))_(2)` |
|
| 16. |
Prove Hess.s law of constant heat summation. |
|
Answer» Solution :If a reaction takes place in several steps then its standard reaction enthalpy is the sum of the standard enthalpies of the intermediate reactions into which the overall reaction may be DIVIDED at the same temperature. e.g. : `C_((s)) + (1)/(2) O_(2(g)) to CO_((g)) , Delta H^( Theta) = (?)` Although `CO_((g))` is the major product, some `CO_2` gas is always produced in this reaction. Therefore, we cannot measure enthalpy change for the above reaction directly. Let us consider the following reactions : `C_((s)) + O_(2(g)) to CO_(2(g)) , Delta_(r) H^( Theta) = -393.5 kj//mol ""...(i)` `CO_((g)) + (1)/(2) O_(2(g)), Delta_(r) H^( Theta)= -283.0 kj//mol ""...(ii)` We can combine the above two reactions in such a way so as to obtain the desired reaction. We reverse equation (ii), `CO_(2(g)) to CO((g)) + (1)/(2) O_(2(g)), Delta_(r) H^( Theta) = + 283.0 kj//mol ""...(iii)` In GENERAL, if enthalpy of an overall reaction `Ato B` along one route is `Delta_(r) H and Delta_(r) H_(1), Delta_(r) H_(2), Delta_(r) H_(3) .....` representing enthalpies of reactions LEADING to same product, B along ANOTHER route, then we have, `Delta_(r) H= Delta_(r) H_(1) + Delta_(r) H_(2) + Delta_(r) H_(3)` |
|
| 17. |
Prove -All the six hydrogens in benzens are identical . |
|
Answer» Solution :Benzene reacts with bromine in the presence of `AlCl_(3)` to form mono bromobenzene. `C_(6)H_(6)+3Br_(2)overset(AlCl_(3))(to)underset("Bromobenzene")(C_(6)H_(5)Br+HBr)` FORMATION of only one monobromo compound indicates that all the SIX HYDROGEN atoms in benzene were indentical. This is possible only if it has a CYCLIC structure of six carbons each containing one hydrogen. |
|
| 19. |
Proton is ……… |
|
Answer» an ionisdedhydrogen ATOM |
|
| 20. |
Proton accelerate the hydrolysis of esters. This is an example of |
|
Answer» A geterogenous catalysis |
|
| 21. |
Propyne overset("Na(l))underset(Delta)rarrX+1/2H_(2),Xoverset(CH_3)underset(Delta)rarrY Which of the following reagents can be used to distinguish Y from propyne? |
|
Answer» `NH_(4) OH + AgNO_(3) " (or) cold alk " KMnO_(4)` |
|
| 23. |
Propyne on passing through red hot iron tube gives …………. . |
|
Answer»
|
|
| 24. |
Propyne is formed by reaction of |
|
Answer» ` CH_3Br` with SODIUM acetylide |
|
| 25. |
Propyne and propene can be distinguished byconc. H_2SO_4Br_2 in "CCl"_4dil. KMnO_4AgNO_3 in ammonia |
|
Answer» `CONC. H_2SO_4` |
|
| 26. |
Propyne and propene can be distinguished by |
|
Answer» Conc. `H_(2)SO_(4)` |
|
| 27. |
Propyneand propene can be distinguished by : |
|
Answer» Conc. `H_(2)SO_(4)` `CH_(3)-C-=CH+NH_(3)+AgNO_(3) rarr underset("white ppt.")(CH_(3))-C-=C.Ag+NH_(4)NO_(3)` while propene does not give any reaction with ammonical `AgNO_(3)` due to ABSENCE of acidic hydrogen. |
|
| 28. |
Propylene is subjected to hydroboration oxidation reaction. The product formed would be |
|
Answer» Propanol `underset("Propylene")(CH_(3)-CH=CH_(2))underset("oxidation")overset("Hydroboration")rarrunderset("1-Propanol")(CH_(3)CH_(2)CH_(2)OH)` |
|
| 29. |
Propylene contains .....sigma and .......pi bonds |
|
Answer» 8,1 |
|
| 30. |
Propyen when passed through a hot iron tube at400^(@)C produces |
|
Answer» BENZENE |
|
| 31. |
Proportion of Na and K like metals are measured by which instrument? |
|
Answer» Spectrometer |
|
| 32. |
Property to be determined Method used for determination |
|
Answer» |
|
| 33. |
Property of all the alkali metals that decreases with their atomic number is // are I) Solun=bility of their hydroxides II) Thermal stability of their carbonates III) SoftnessIV) Hydration energy |
|
Answer» III,IV |
|
| 34. |
Property of the alkaline earth metals that increases with their atomic number is |
|
Answer» solubility of their hydroxide in water Among sulphates , since the size of `SO_(4)^(2-)` ion is very big as compared to the metal cation , therefore , lattice enthalpy remains almost constant but their hydration enthalpy decreases down the group . Thus , the solubility of sulphates decreases down the group . Of course , ELECTRONEGATIVITYAND ionization enthalpy both decrease down the group as the atomic size increases . |
|
| 35. |
Property of alkaline earth metals that increases with their atomic number is.... |
|
Answer» IONIZATION energy |
|
| 36. |
Proper management of disposal of household and industrial wastes can be done by |
|
Answer» Recycling the WASTE material to give useful products again |
|
| 37. |
Propene + X overset((C_(6)H_(5)CO)_(2)O O)rarr 1-halopropene, then what is 'X' ? |
|
Answer» HI |
|
| 38. |
Propene reacts with HBr in the presence of peroxide to form …………………… |
| Answer» SOLUTION :`1-`BROMOPROPANE | |
| 39. |
Propene reacts with alkaline KMnO_4 in cold condition gives |
|
Answer» propan -1 ,2-diol |
|
| 40. |
Propene on reaction with chlorine water gives |
|
Answer»
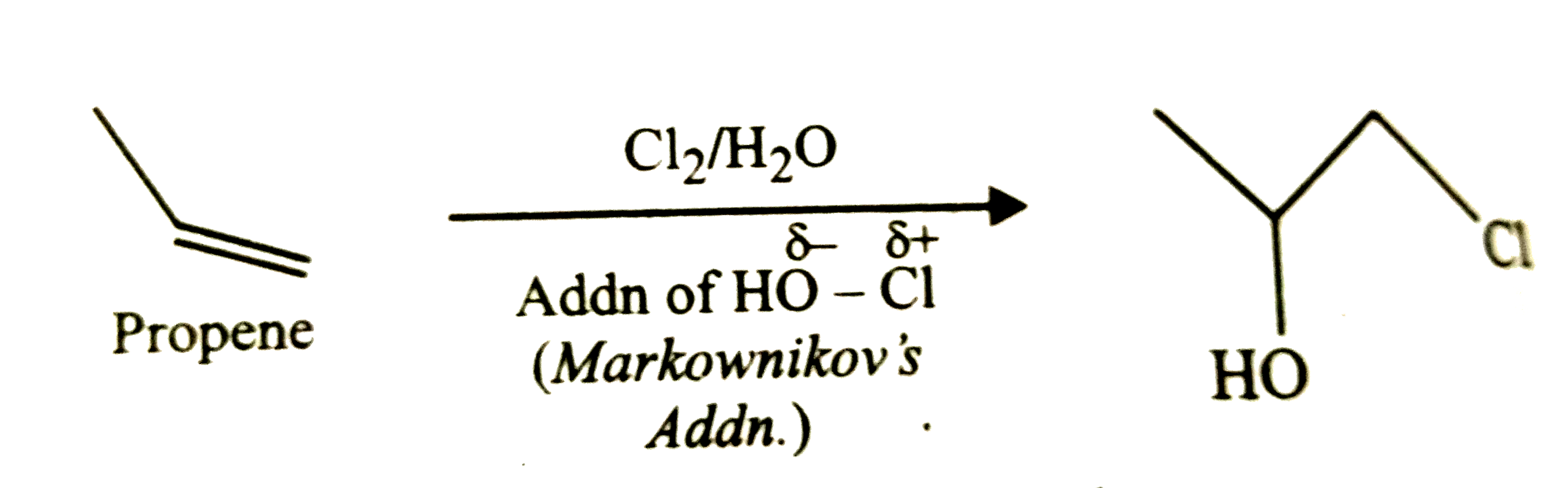
|
|
| 41. |
Propene on hydroboration-oxidation produces |
|
Answer» `CH_(3)CH_(2)CH_(2)OH` |
|
| 42. |
Propene is thetreadted witBrC CI_(3) is presence ofperoxides. |
Answer» Solution :In PRESENCEOF peroxides `Br C CI_(3)` undergoeshomolyticfissionto FORM `C CI_(3)` radicalwhichthenattacks thepropenemolecule at the`CH_(2)` GROUP to forma `2^(@)` alkyl freeradical . Thisradicalsubsequentlyreacts with`BrCCI_(3)` tocompletethe addition. )
|
|
| 43. |
Propene+HBr overset(C_6H_5COOOCO-C_(6)H_(5))rarrA. A is ? |
|
Answer» n-Propyl BROMIDE |
|
| 44. |
Propene (CH_(3)-CH=CH_(2)) can be transformed to compounds (a to j) listed in the left-hand column. Write letter designating the reagent, you believe desired transformation. In the case of multi-step sequence write the reagent in the order they are to be used. |
|
Answer» (b)Oxymercuration-De-mercuration reaction (OMDM reaction) (c) Hydroboration-oxidation reaction (HBO reaction) (d) `CH_(3)CH=CH_(2)OVERSET(OMDM)(to)CH_(3)-overset(O)overset(||)(C)-CH_(3)overset(HOBr)underset("oxidising agent")(to)CH_(3)-CH_(2)-CHO` (h) Halohydrin formation (g) Oxidation reaction (i) `CH_(3)-CH=CH_(2)overset(HBO)(to)CH_(3)-CH_(2)-CH_(2)-OHoverset(SOCl_(2))underset("Darzene process")(to)CH_(3)-CH_(2)-CH_(2)-Cl+SO_(2)+HCl` (J) Halogenation and elimination |
|
| 45. |
Propanone is the product obtained dehydrogenation of |
|
Answer» 2-Propanol 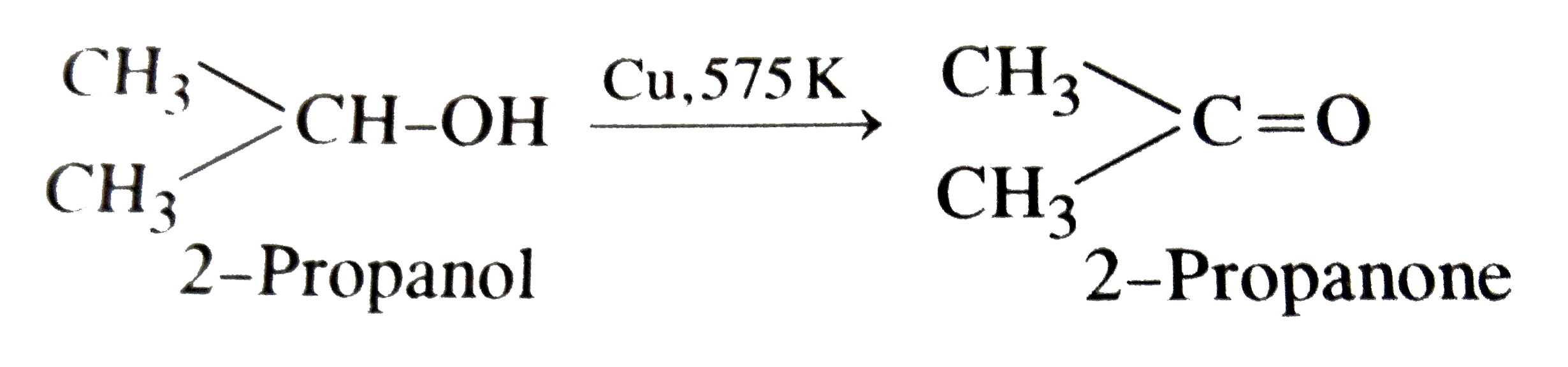
|
|
| 46. |
Propanoic acid with Br//P yields a dibromo product. Its structure would be |
|
Answer» `CH_(3) - underset(Br)underset(|)OVERSET(Br)overset(|)(C) - COOH` |
|
| 47. |
Propane has the structure H_(3)C - CH_(2) - CH_(3) . Calculate the change inenthalpy for the reaction : C_(3) H_(8) (g) + 5O_(2)(g) rarr 3 CO_(2)(g) + 4H_(2)O(g) Given that average bond enthalpiesare : {:(C-C,C-H,C=0,O=O,O-H),(347,414,714,498,464kJ mol^(-1)):} |
|
Answer» Solution :`Delta_(r)H= [B.E. (H-underset(H)underset(|)overset(H) overset(|)(C ) - underset(H) underset(|) overset(H) overset(|) (C) - underset(H)underset(|) overset(H)overset(|)(C)-H)+5 xx B.E. ( O= O) ]= [3 xx B.E. ( O= C=O) + 4B.E. ( H- O-H)]` `= [2 B.E. ( C-C) + 8 B.E. (C-H) + 5 B.E. ( O=O) ] = [ 6 xx B.E. ( C= O )+8B.E. ( O- H) ]` `= 2 ( 347) + 8 ( 414) + 5 ( 498) ]- [ 6 ( 741) +8 ( 464) ] kJ MOL^(-1)` ` = [694+ 3312+2490 ]-[4446 +3712]= - 1662k J mol^(-1)` |
|
| 48. |
Propane can be best prepared by the reaction |
|
Answer» `CH_3 CH_2 I + CH_3 I UNDERSET(DELTA)OVERSET("Na/ether")to ` |
|
| 49. |
(Propanal, Propanone) and (Pentane-3-one, Pentane-2-one) represent the isomerism of which type respectively? |
|
Answer» Metamerism, Position They have same MF but DIFFERENT functional group and functioal group isomerism (II) Pentane-3-one `CH_(3)CH_(2)COCH_(2)CH_(3)` Pentane-2-one `CH_(3)COCH_(2)CH_(2)CH_(3)` `therfore` They are METAMERS |
|
| 50. |
Propanal and pentane-3-one are the ozonolysis products of an alkene ? What is the structural formula of the alkene ? |
Answer» SOLUTION :Structure of PROPANAL and pentane-3-one from ozonolysis is as FOLLOWS :  In product (A) and (B), C = O is replaced by C=C, on ozonolysis we get product (X), name is 3-Ethyl-hex-3-ene. |
|