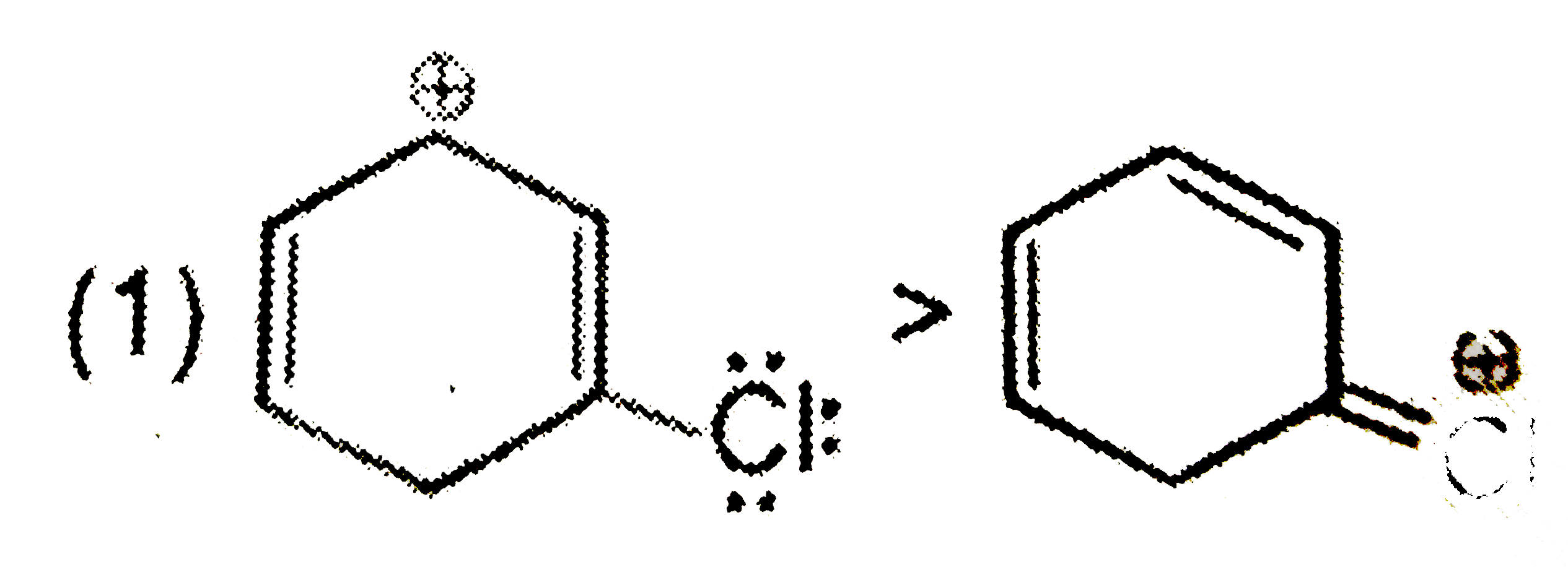Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
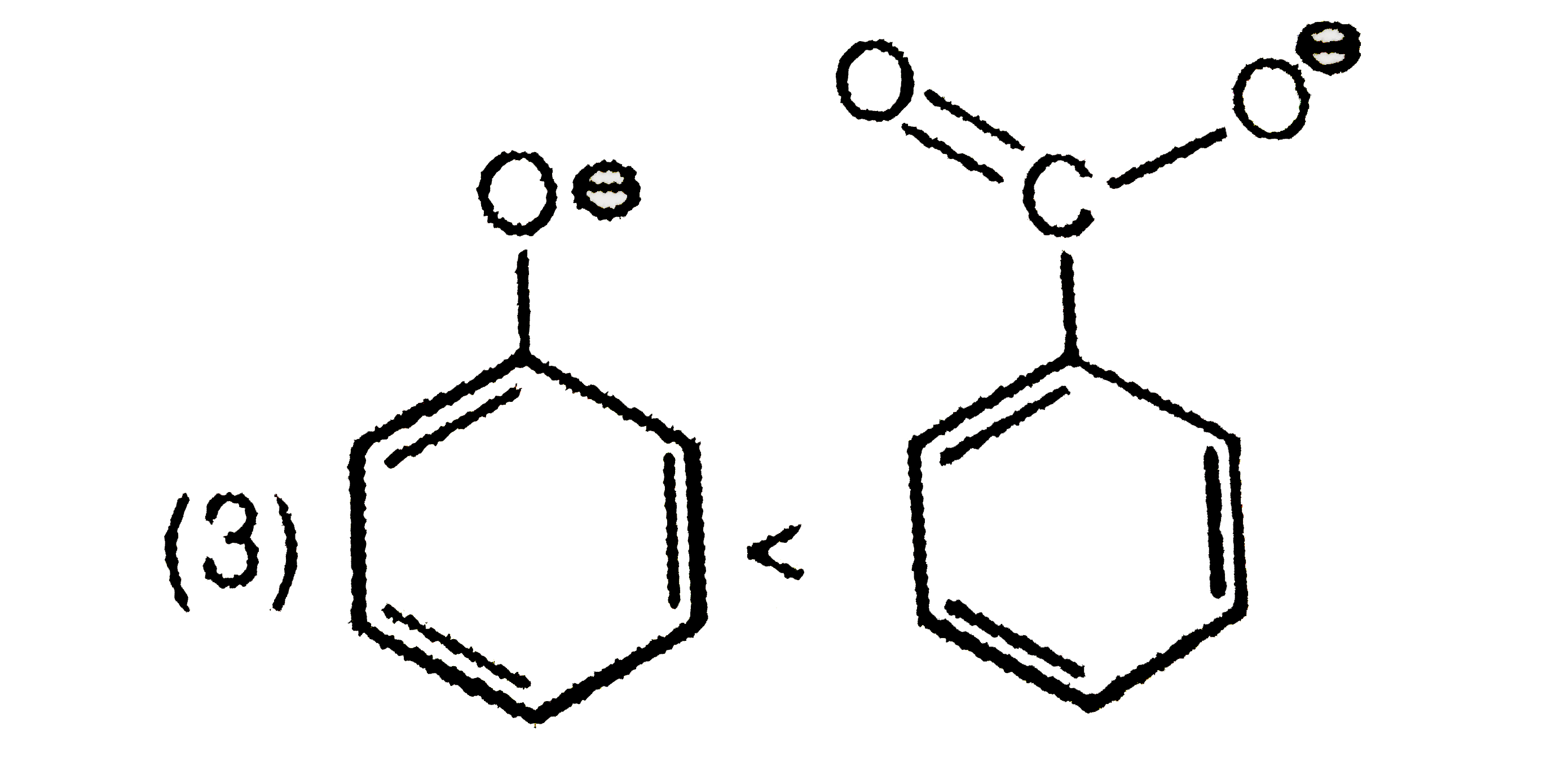
Select the incorrect option of the following statements. |
|
Answer» BIMOLECULAR elimination of ALKYL halides is a stereospecific reaction. |
|
| 2. |
Select the incorrect option for van der Waal's gas. |
|
Answer» He has smaller 'a' (van der Waals' constant) than Ne,Kr,Ar,`CO_(2),CH_(4)` and `NH_(3)` |
|
| 3. |
Select the incorrec option: |
|
Answer» Specific volume and molar heat capacity are intensive PROPERTIES:Change in internal energy for an ideal gas forand isobaric process is EXPECTED as `nC_(V)(T_(2)-T_(1)).` |
|
| 4. |
Select the false statements from the following and try to justify your assertion: (a) Like carbon, silicon also existas in the free state. (b) B(OH)_(3) is acidic in nature. (c) Boron dissovles in hydrochloric acid. (d) The composition of common glass is Na_(2)O,CaO.6SiO_(2). (e) Silica contains SiO_(2) molecules with O=Si=O bonds. (f) BF_(3) is a strnger Lewis acid than BCl_(3) (f) Water glass is sodium silicate. (h) diborane cannot have ethane like structure because it is not a covalent molecule. |
|
Answer» Solution :(a) SILICON does not exist in free state becaue it is more reactive than carbon due to bigger size andalso lesser ionisation enthalpy. (C) Boron dissolves only in oxidising acids like `H_(2)SO_(4)` and `HNO_(3)` to form boric acid. As HCl is not strong oxidising agent, boron does not dissolve in HCl. (e) In silica O=Si=O cannot be present due to the different in the energy state of 2p orbital of oxygen and 3porbial of silicon which are to participitate in sidewise overlapping. (f) `BF_(3)` is a weaker Lewis acid than `BCl_(3)` because of greater magnitude of `p pi- p pi`back bonding, resulting in more ELECTRON density on the boron atom. (h) dibronae cannot have a ethane like structure becasue boron atom is trivalent and more than three hydrogen ATOMS cannot be linked to it. thus `H_(3)B-BH_(3)` structure is not POSSIBLE in this case. |
|
| 5. |
Select the corrrect statement with prespect to the deep blue solution of a n alkali metal in liquid ammonia. |
|
Answer» Its paramagnetism DECREASES with increasing concentration. |
|
| 6. |
Select the correct statements number for the following statements in sequence. (i) IUPAC naem of propyl cyanide is propane nitrile (ii) IUPAC name od diethylether is ethoxy ethane (iii) Ethanol and vinylalcohol are tautomers (iv) Methoxy propane and ethoxyethane are metamers (v) 2, 3-dimethyl-but-2-ene is more stable than 2-methyl-but-2-ene (vi) Stability order of carbocation is 1^(@) lt 2^(@) lt 3^(@) (vii) Hybridization of carbon atom does not change in elimination reaction. (viii) Nucleophilie is lewis acid |
| Answer» Answer :D | |
| 7. |
Select the correct statement(s) from the following |
|
Answer» GRAPHIT is thermodynamically more UNSTABLE FORM of carbon |
|
| 8. |
Select the correct statements for hydrogen like atoms or ions. |
|
Answer» RADIUS of an ORBIT is INVERSELY proportional to Z |
|
| 9. |
Select the correct statement(s) among the following (i) Total number of orbitals in a shell with principal quantum number 'n' is n^(2) (ii). Total number of subshells in the n th energy level is n (iii). The maximum number of electrons in a subshell is given by the expression (4l+2) |
|
Answer» m=l+2 where la nd ma re azimuthal and magnetic QUANTUM numbers. |
|
| 10. |
select the correct statements among the following : |
|
Answer» MILK is emulsion of fat in water. |
|
| 11. |
Select the correct statements about the wave function Psi. |
|
Answer» `Psi` must be REAL |
|
| 12. |
Select the correct statements about the following : NO[BF_(4)] |
|
Answer» It has `5sigma` and `2pi` BONDS |
|
| 13. |
Select the correct statement(s) about chemical reaction in a closed container. |
|
Answer» TOTAL MASS REMAINS conserved. |
|
| 14. |
Select the correct statements about diborane |
|
Answer» `B_2H_6` has three centre TWO electron bond  `B_(2)H_(6)`has three centre two electron bond, each boron UNDERGOES `sp^3`hybridisation only 4 hydrogens and 2 borons are present in the same plane |
|
| 15. |
Select the correct statement(s), |
|
Answer» An electron near the nucleus is attracted by the nucleus and has a low potential ENERGY |
|
| 16. |
Select the correct statements |
|
Answer» <P>At high pressure REAL gases are less COMPRESSIBLE in comparison to ideal GAS b) `H_2`, He have Z > 1 c) At low, `P ,Z < 1`= mole compressible d) Z depends on T. |
|
| 17. |
Select the correct statement(s): |
|
Answer» ALKALINE solution of POTASSIUM tetraiodomercurate (II) gives brown ppt. with ammonium chloride solution. |
|
| 18. |
Select the correct statements: |
|
Answer» Compound (A) is BASIC copper carbonate |
|
| 19. |
Select the correct statements: |
|
Answer» Eclipsed and STAGGERED ethanes given differet produces on reaction with chlorine in presence of light. |
|
| 20. |
Select the correct statement(S) |
|
Answer» Chlorophyllconyains magneium |
|
| 21. |
Select the correctstatements. |
|
Answer» In a mixture of `KHC_(2)O_(4) " and "H_(2)C_(2)O_(4), KMnO_(4)` decolourises FASTER at higher temperature than lower temperature. |
|
| 22. |
Select the correct statement(s) : |
|
Answer» Clay and lime on STRONG HEATING produces a fused mass KNOWN as 'cement clinkers' |
|
| 23. |
Select the correct statements : |
|
Answer» Stability of peroxides and SUPEROXIDES of alkali metals increase with increase in size of the cation . |
|
| 24. |
Select the correct statementns (s) |
|
Answer» SUPER oxide ion contains a three electron bond |
|
| 25. |
Select the correct statement with respect to carbon monoxide. |
|
Answer» It combines with WATER to GIVE carbonic acid |
|
| 26. |
Select the correct statement/s |
|
Answer» `H^(+)` can exist as `H_(9)O_(4)^(+)`in water |
|
| 27. |
Select the correct statement out of the following |
|
Answer» CaF is soluble in WATER |
|
| 28. |
Select the correct statement out of following: |
|
Answer» `NO_(3)^(-)` gives brown RING test in PRESENCE of dil. `H_(2)SO_(4)` |
|
| 29. |
Select the correct statement. In the gas equation, PV=nRT |
|
Answer» n is the number of MOLECULES of a gas |
|
| 30. |
Select the correct statement. (i) Delocalisation of sigma-electron is hyperconjugation. (ii) Delocalisation of pi-electron is resonance. (iii) Partial displacement of sigma-electron is inductive effect. |
| Answer» Answer :D | |
| 31. |
Select the correct statement for the following species. |
|
Answer» (I) and (III) are geometrical isomers. 
|
|
| 32. |
Select the correct statement about the compound NO[BF_(4)] |
|
Answer» It has `5 sigma` and `2pi`bonds |
|
| 33. |
Select the correct statement about harium : |
|
Answer» it SHOWS photoelectric effect |
|
| 34. |
Select the correct statement. |
|
Answer» Among the alkali METALS, only lithium reacts witjh NITROGEN DIRECTLY at ROOM temperature of from nitride. |
|
| 35. |
Select the correct statemens for following equilibrium: |
|
Answer» On INCREASING PRESSURE, melting point decreases while boiling point of `CO_2` INCREASES. |
|
| 36. |
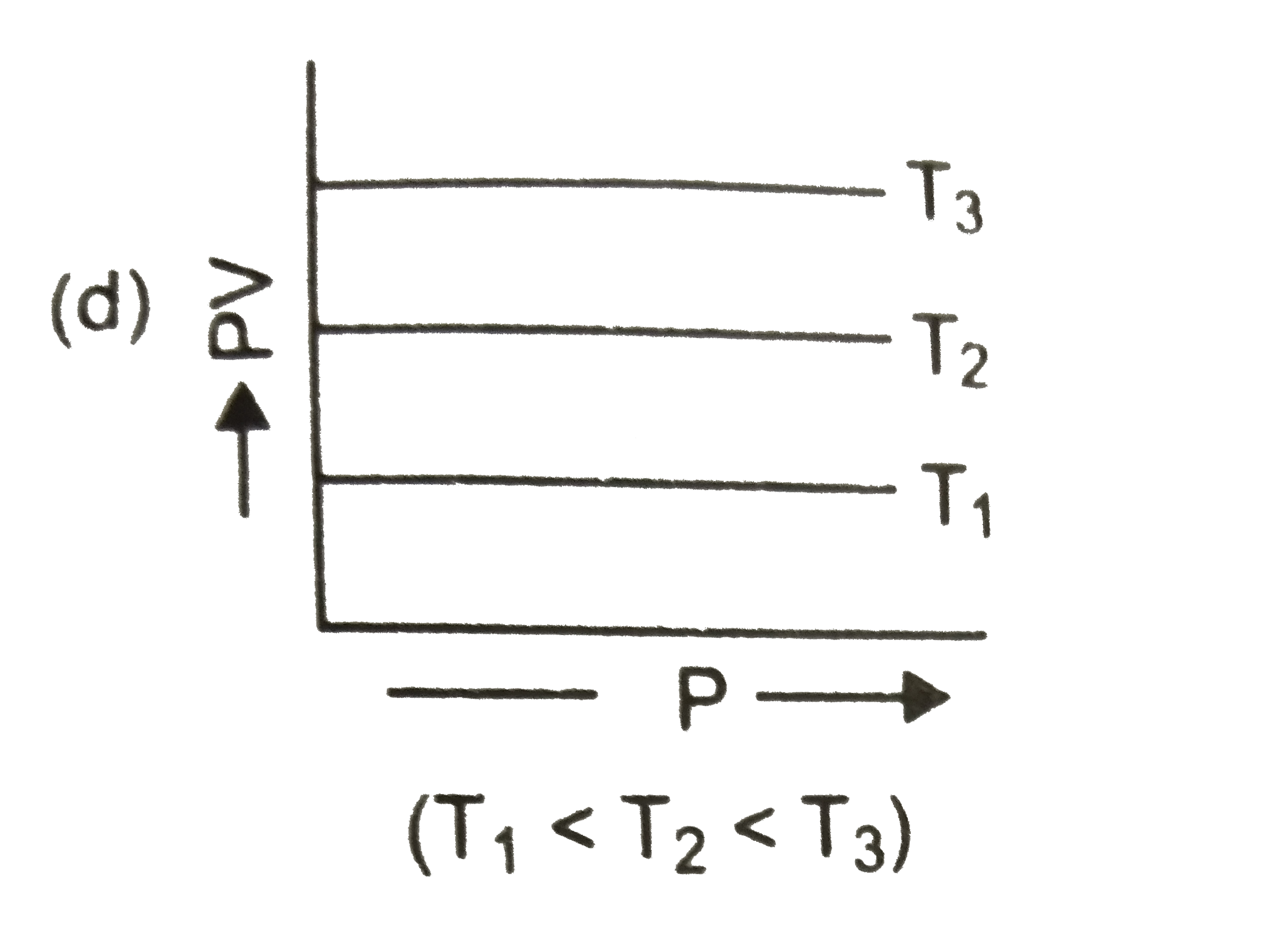
Select the correct set of statement/s: I. Work done by the surrounding in case of infinite stage expansion is more than single stage expansion II. Irreversible work is always greater than reversible work. III. On an ideal gas in case of single stage expansion and compression system as well as surrounding are restored back to their original states IV. If gas is in thermodynamic equilibrium is taken from A to state B, by four successive single stage expansions. Then we can plot 4 points on the P-V indicator diagram. |
|
Answer» II FOUR STEPS `rArr` four points |
|
| 37. |
Select the correct satement regarding the following compouns : |
|
Answer» II has a GREATER DIPOLE moment than I 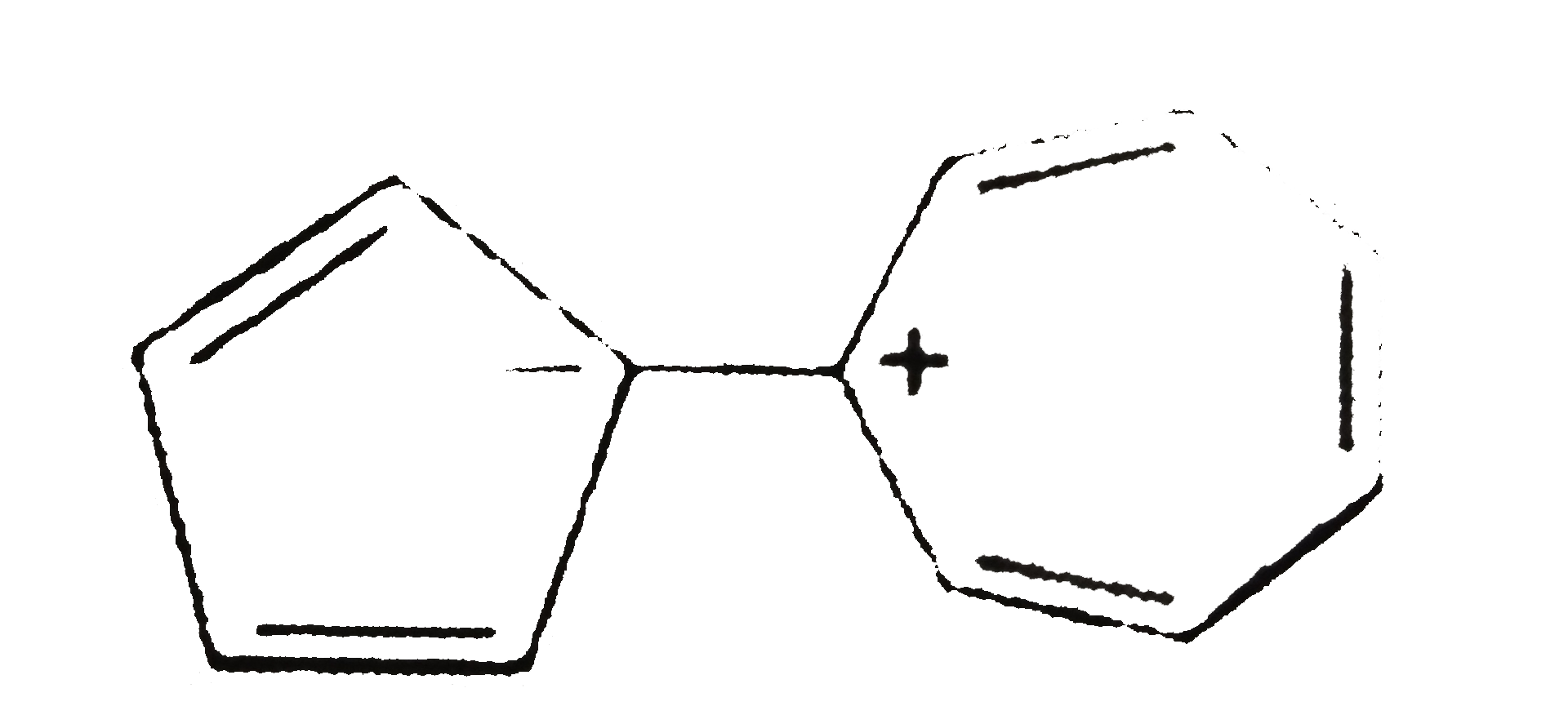 both rings are aromatic so, it will be polar in NATURE. |
|
| 38. |
Select the correct order of thermal stability ? |
|
Answer» `HI GT HBR gt HF gt HCl` |
|
| 39. |
Select the correct order of heat of hydrogenation? |
|
Answer» `IgtIIgtIIIgtIV` |
|
| 40. |
Select the correct option related to stability of following structures. |
|
Answer»
|
|
| 41. |
Select the correct expressions among the following |
|
Answer» `(DELTA G - Delta H)/(T) = ((del Delta G)/(del T))_(P)` `rArr ((del Delta G)/(del T))_(P) = (Delta G- Delta H)/(T)` `Delta G = - n FE_("cell")` `rArr ((del Delta G)/(del T))_(P) = nF ((del E_("cell"))/(del T))_(P) = - Delta S` `rArr (Delta S)/(nF) = ((del E_("cell"))/(del T))_(P)` `((del T)/(del P))_(H) = mu_(JT) = ((-del H)/(del P))_(T) XX (1)/(C_(P))` |
|
| 43. |
Select the correct conclusion (s) about averagelife: |
|
Answer» Average LIFE `= 1//lambda` |
|
| 44. |
Select the correct choice for given statements. T for true and F - for false (i) The physical properties of hydrogen are like those of metals. (ii) Some chemical properties of hydrogen are those of metals and of non-metals. (iii) Metals of groups 7, 8 and 9 form nonstoichiometric hydrides. (iv) The percentage of water in natural sources like rivers is 2.04. |
|
Answer» i-F,ii-T,iii-F,iv-F |
|
| 45. |
Select the correct choice for given reactions: (i) 2Na +2H_(2)O to X (ii) 2F_(2)+2H_(2)O to Y (iii) H_(2)S +2H_(2)O to Z |
|
Answer» `X=2Na_(2)+H_(2)` (ii) `2F_(2)+2H_(2)O to underset(Y) ubrace(4H^(+)+4F^(-)+O_(2))` (III) `H_(2)S+2H_(2)O to underset(Z) ubrace(2H_(3)O^(+)+S^(2-))` |
|
| 46. |
Select the correct choice for given reaction after balancing. 4HgS+4 CaO rarr 4Hg+ x + CaS |
|
Answer» `x=HgSO_(4)` `{:(4HgS + 4CaO RARR 4 Hg + CaSO_(4) + 3 CAS),(""x):}` |
|
| 47. |
Select the compound in each of the following pairs that can be converted to corresponding alkyl bromide more rapidly on being treated with hydrogen bromide : l(i) 1-butanol or 2-butanol (ii) 2-methyl-1-butanol or 2-butanol (iii) 2-methyl-2-butanol or 2-butanol |
| Answer» SOLUTION :(i) 2-Butanol (II) 2-Butanol (III) 2-Methyl-2-butanol | |
| 48. |
Select the compound which can undergo intramolecular cannizaro reaction in basic medium |
|
Answer» A,B,D |
|
| 49. |
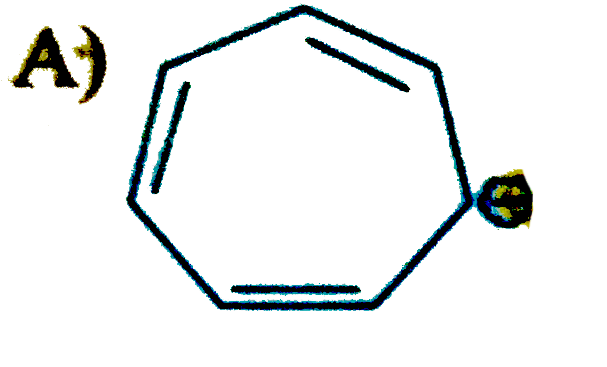
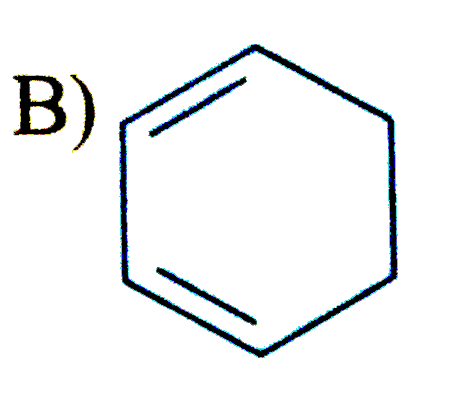
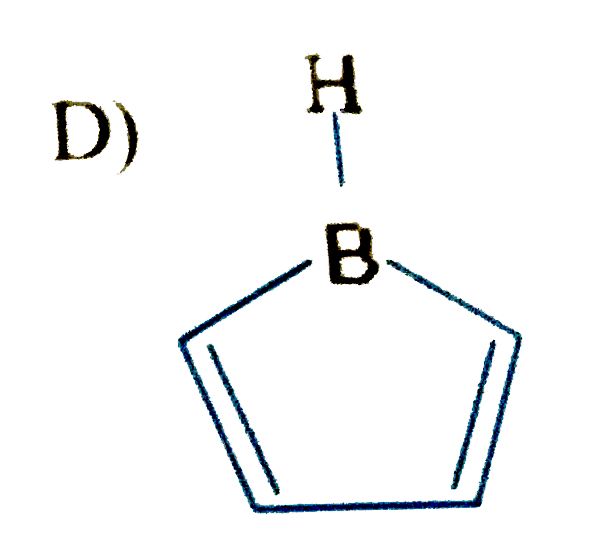
Select the compound/ions which is/are anti-aromatic |
|
Answer»
Hence compound A and D has anti-aromatic CHARACTER. |
|
| 50. |
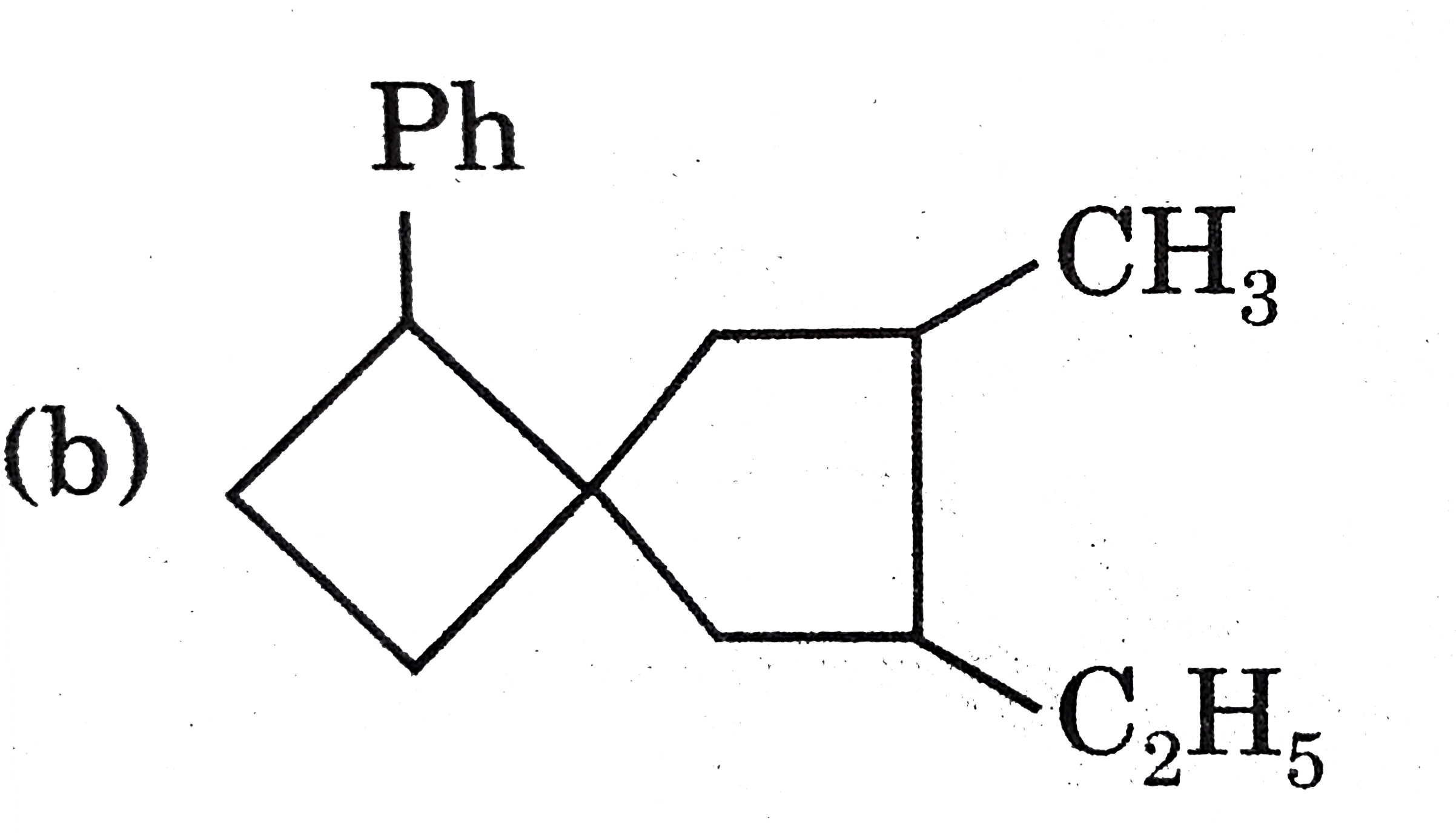
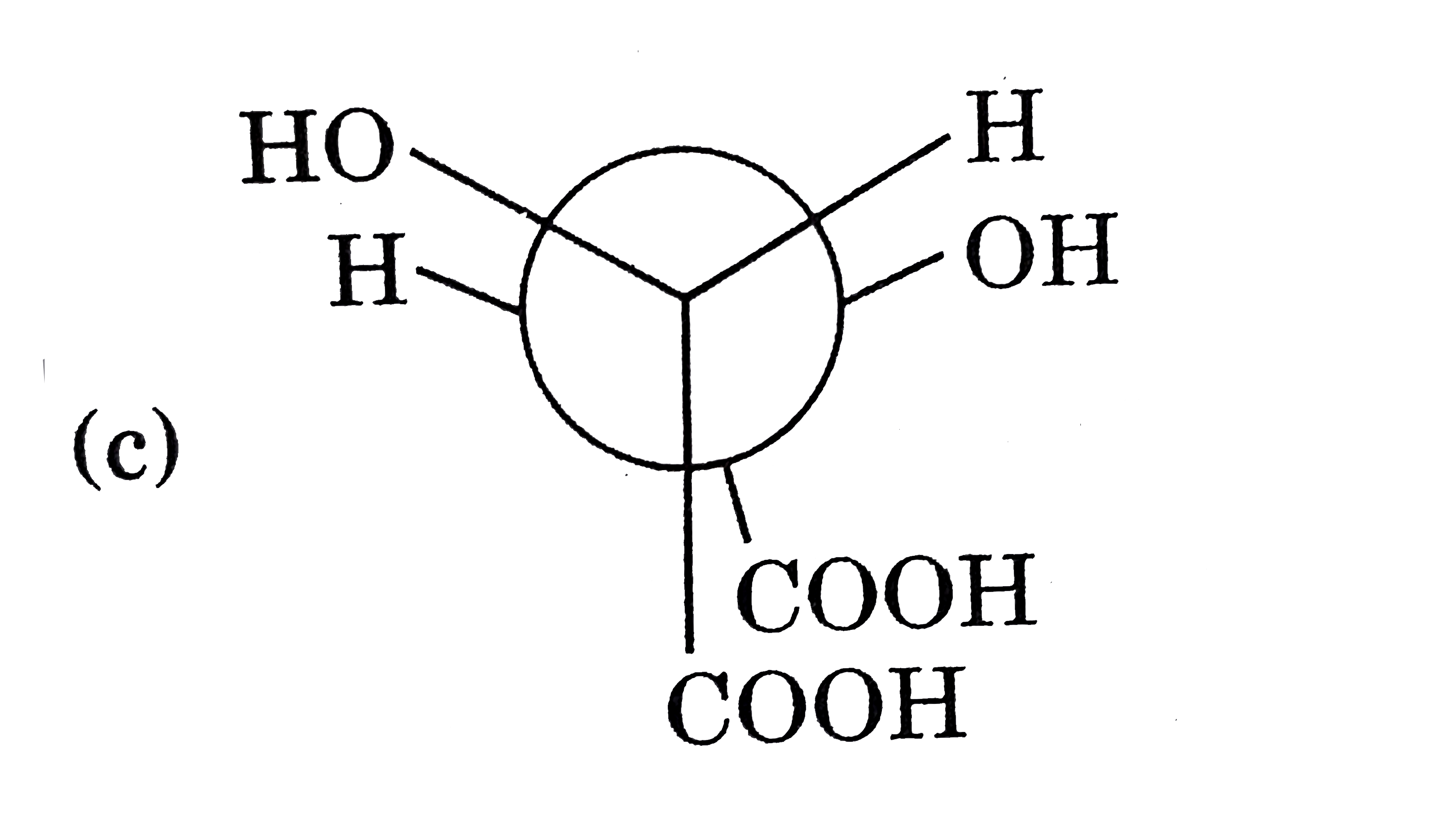
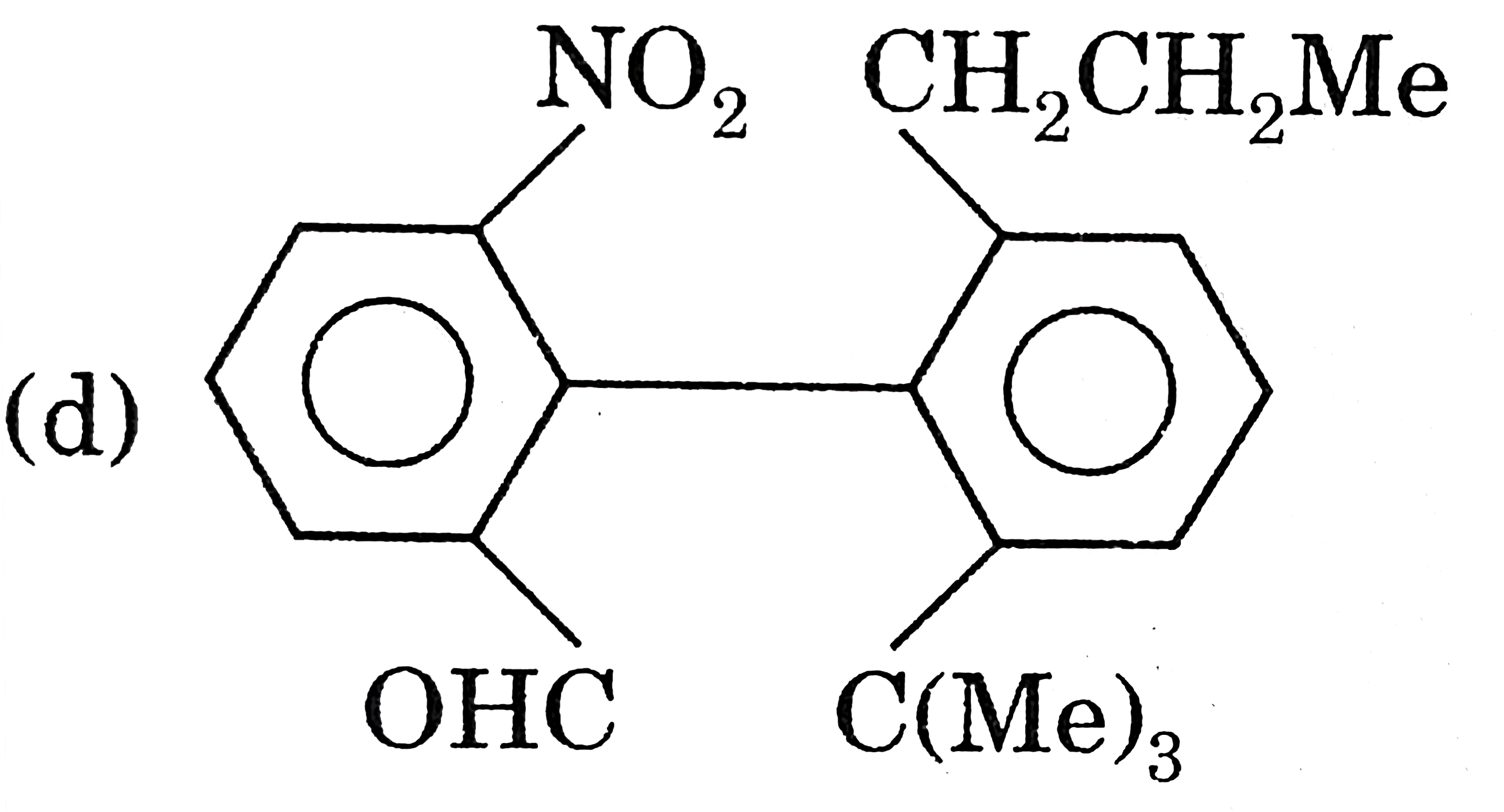
Select the chiral molecule. |
|
Answer»
|
|