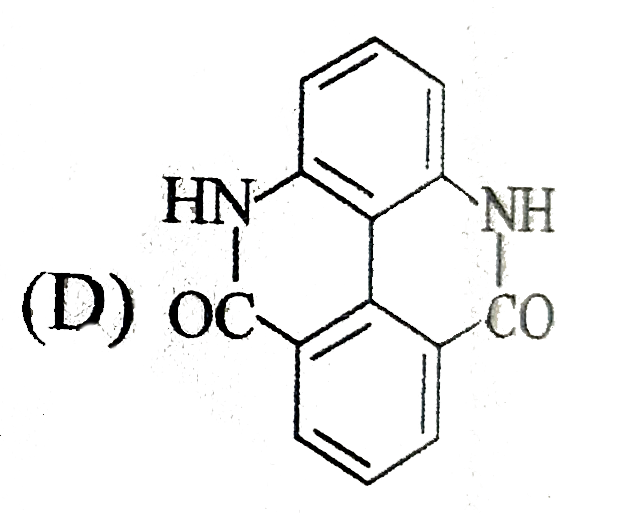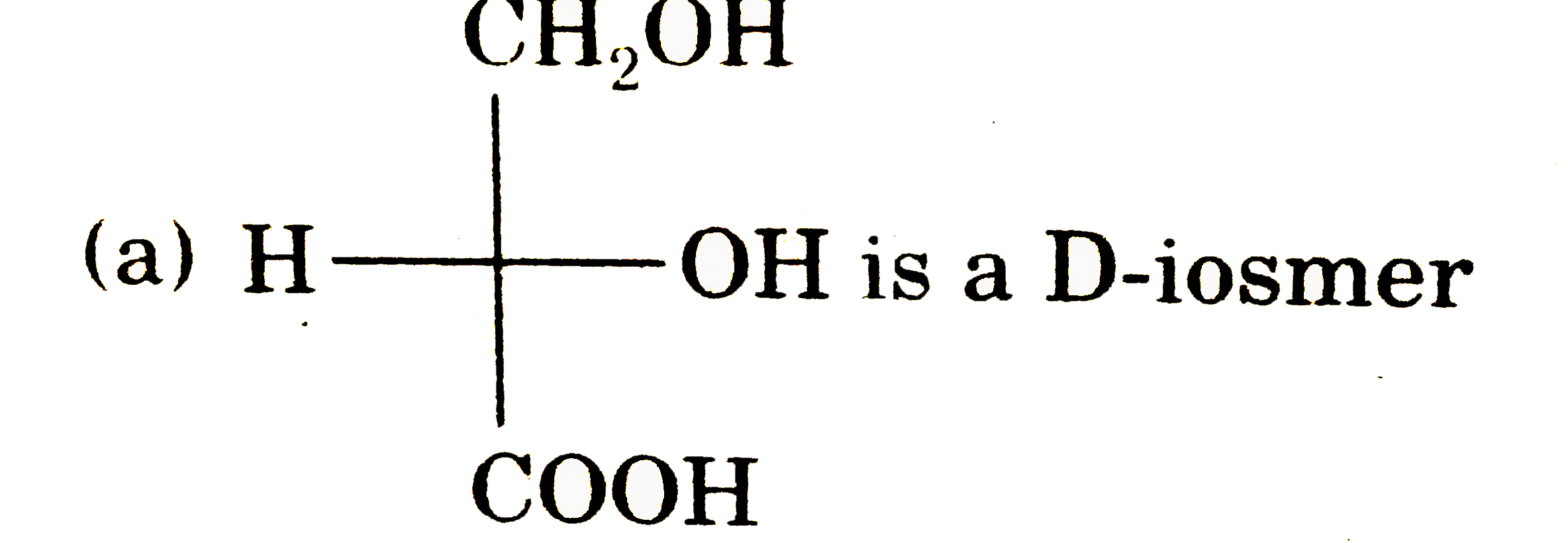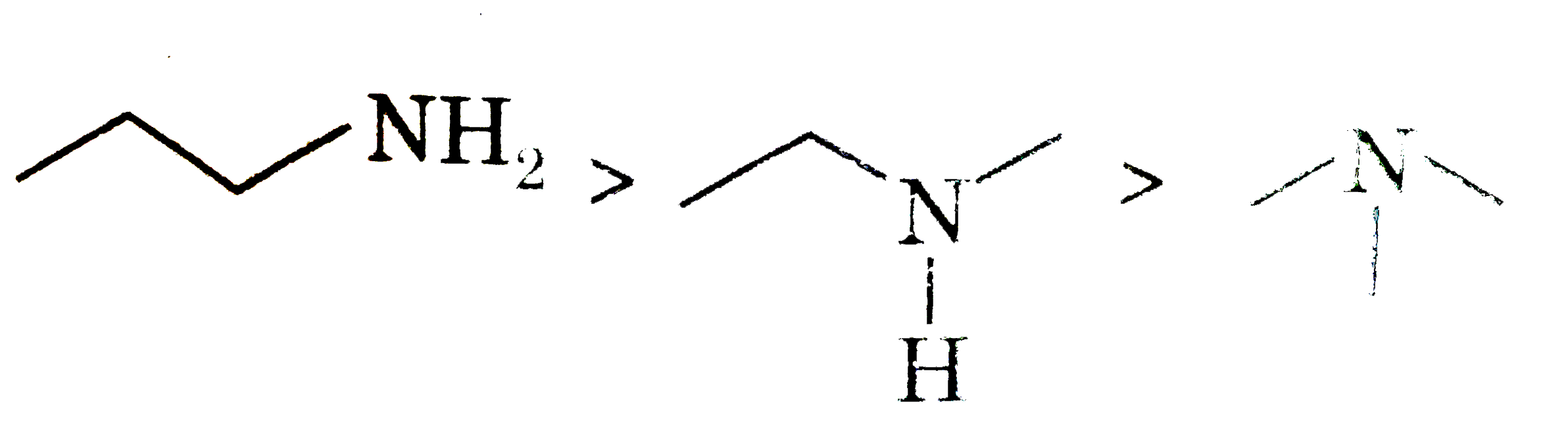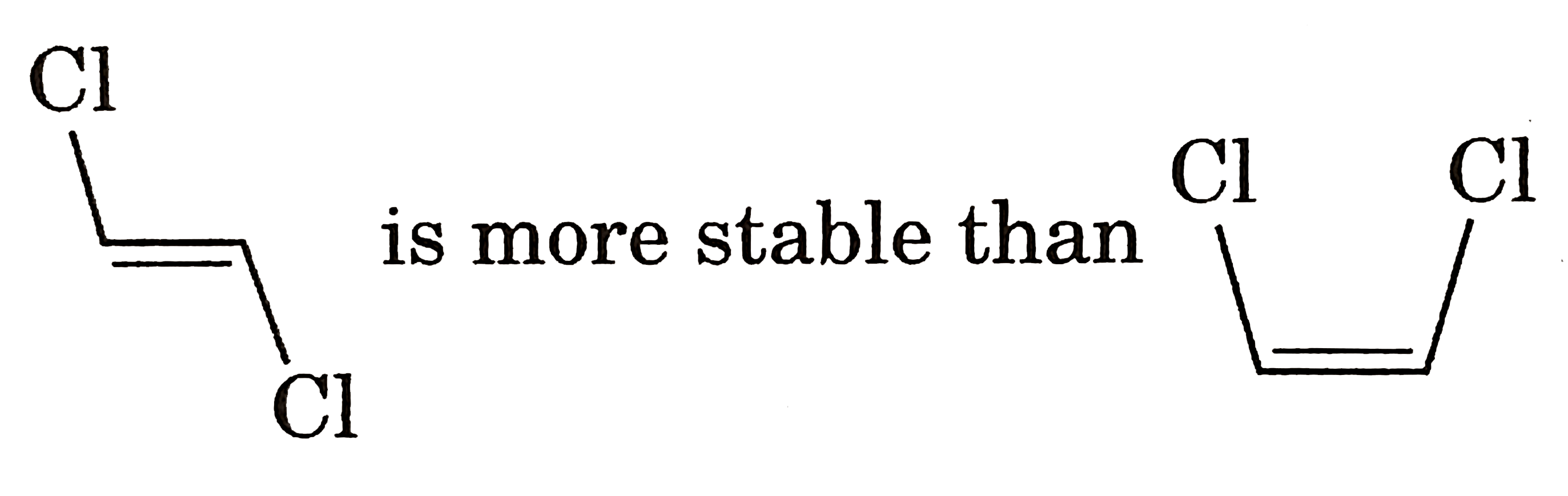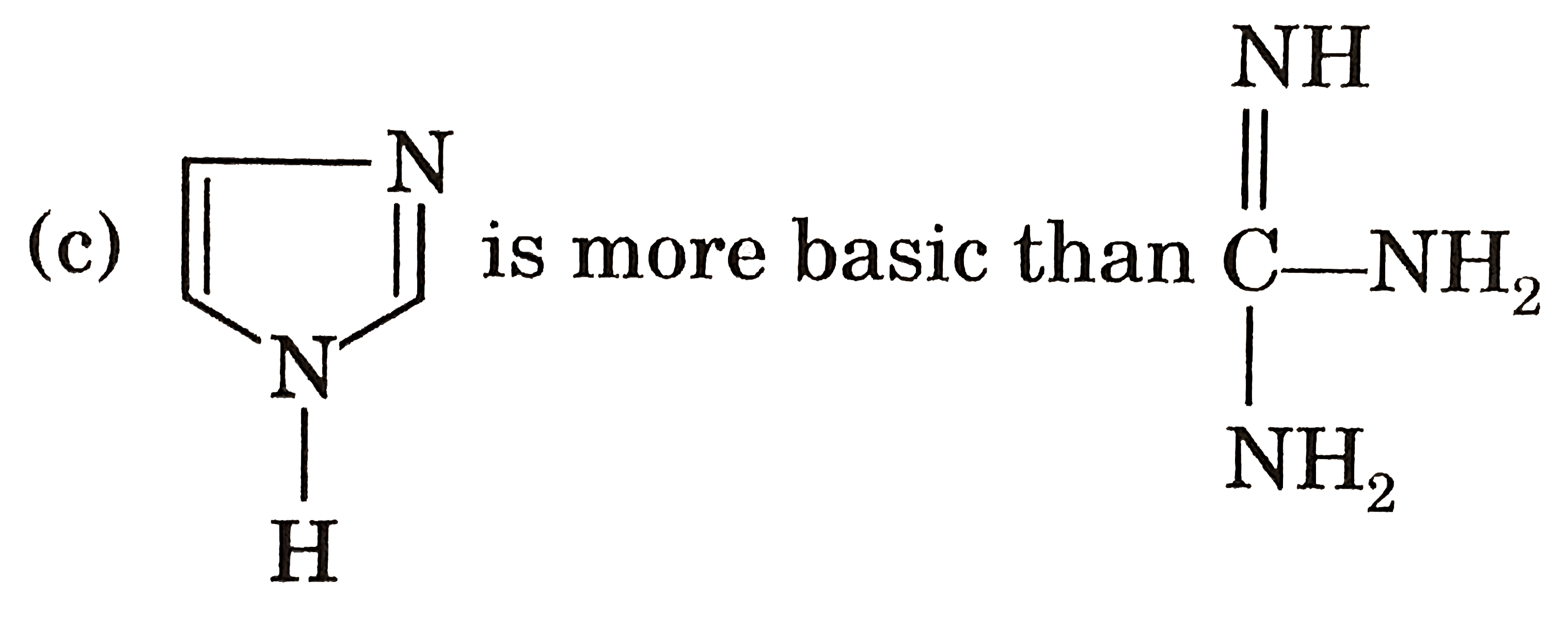Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Separation of petroleum into its components is mostly done by |
|
Answer» CHROMATOGRAPHY |
|
| 2. |
Separate out the following into extensive and intensive : Volume, Temperature, Pressure,Boiling point, Free energy |
| Answer» SOLUTION :Volume and FREEENERGY are EXTENSIVE , OTHERS are intensive | |
| 3. |
Semiconductors possess conductivity in the range ______ to _________ |
| Answer» SOLUTION :`10^(-6)` to `10^4 OHM^(-1) m^(-1)` | |
| 4. |
Select wrong statement:- |
|
Answer» If a very small amount of `AlCl_(3)` is added to gold sl, coagulation occurs, but if a large quantity of `AlCl_(3)` is added, there is no coagulation |
|
| 5. |
Select wrong clientical reaction aniong the following |
|
Answer» `8NH_(3)+3Cl_(2) to 6NH_(4)CL+N_(2)` |
|
| 6. |
Select true statement. |
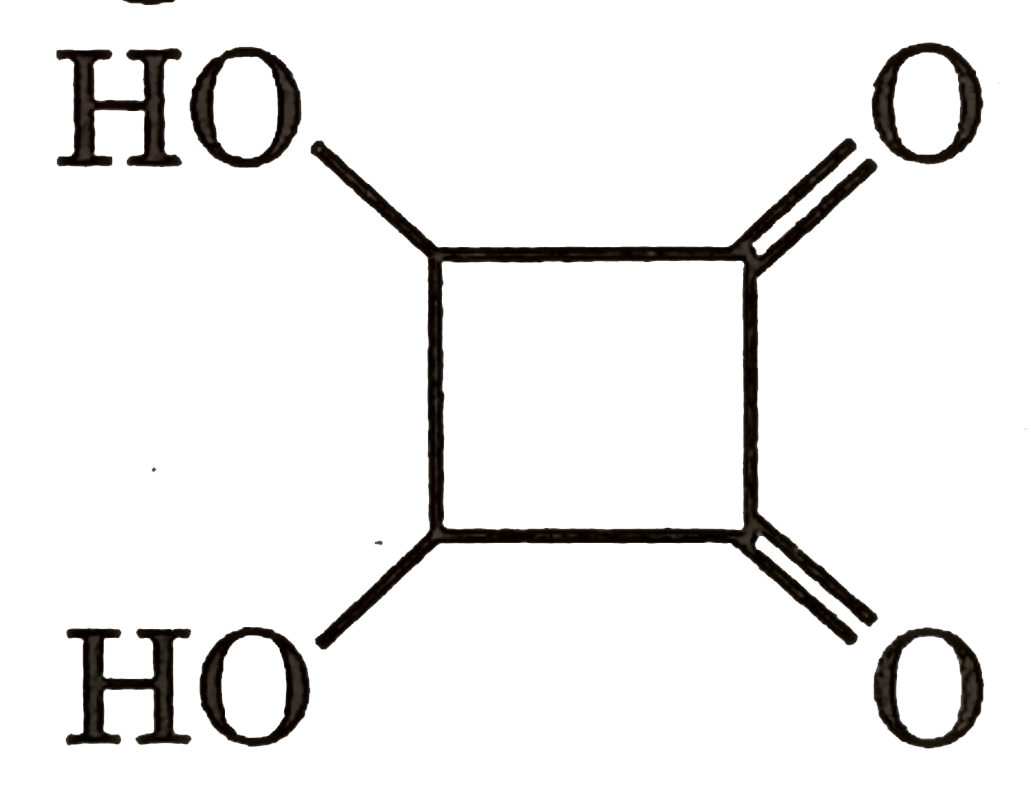
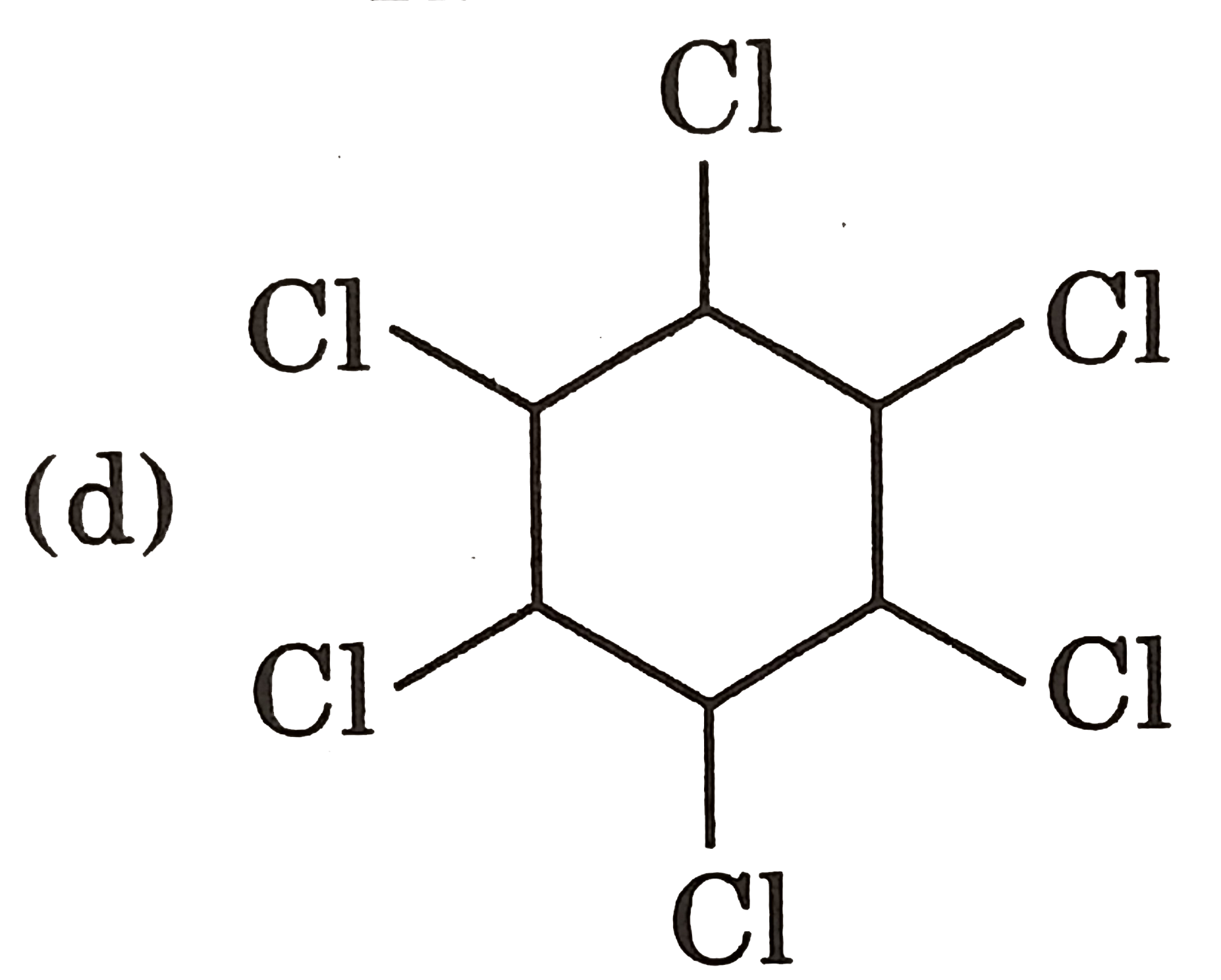
|
Answer» Gauche from of 3-hydroxy propanol is more stable than it's anti form 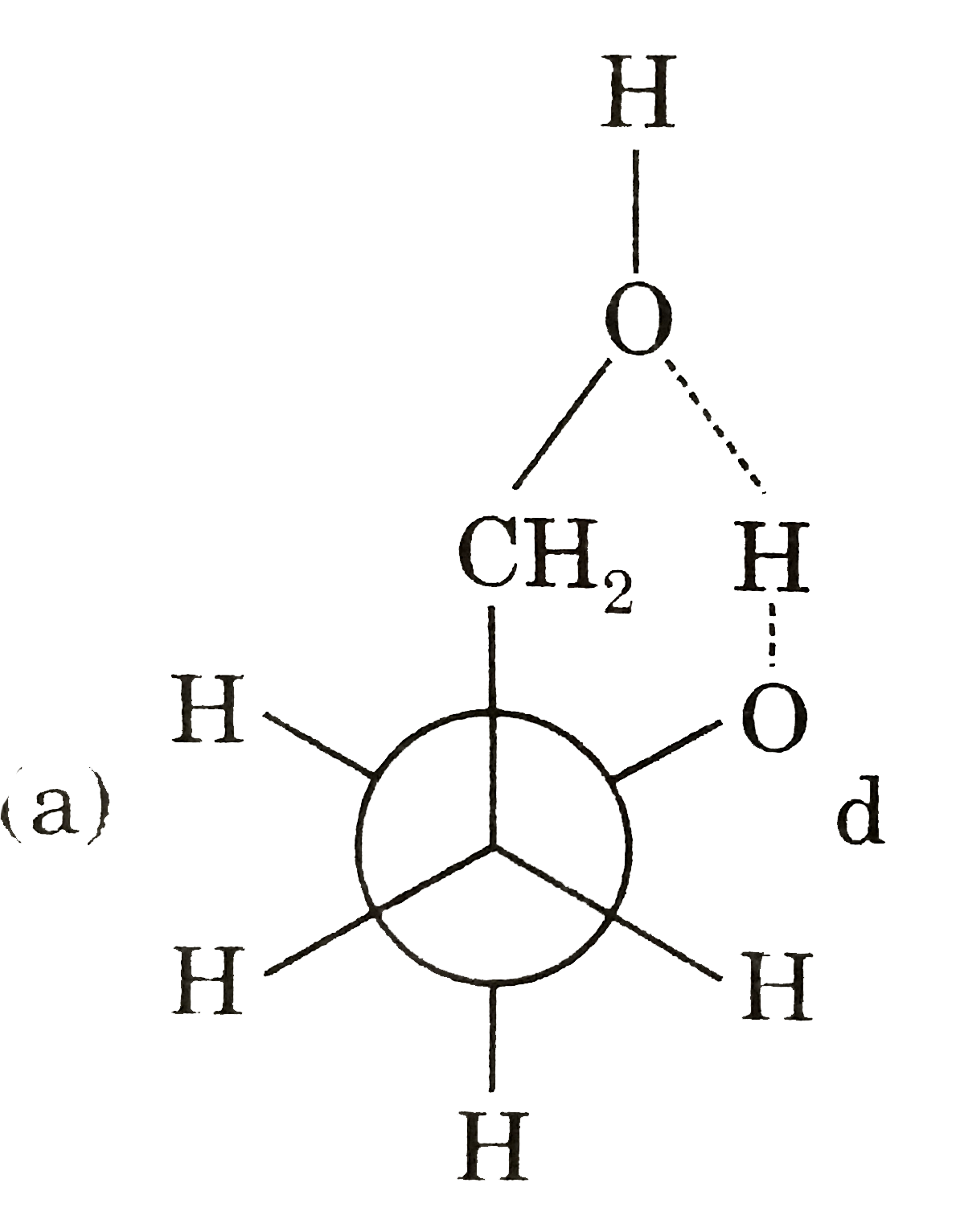 due to INTRAMOLECULAR H-bonding in gauche form due to INTRAMOLECULAR H-bonding in gauche form 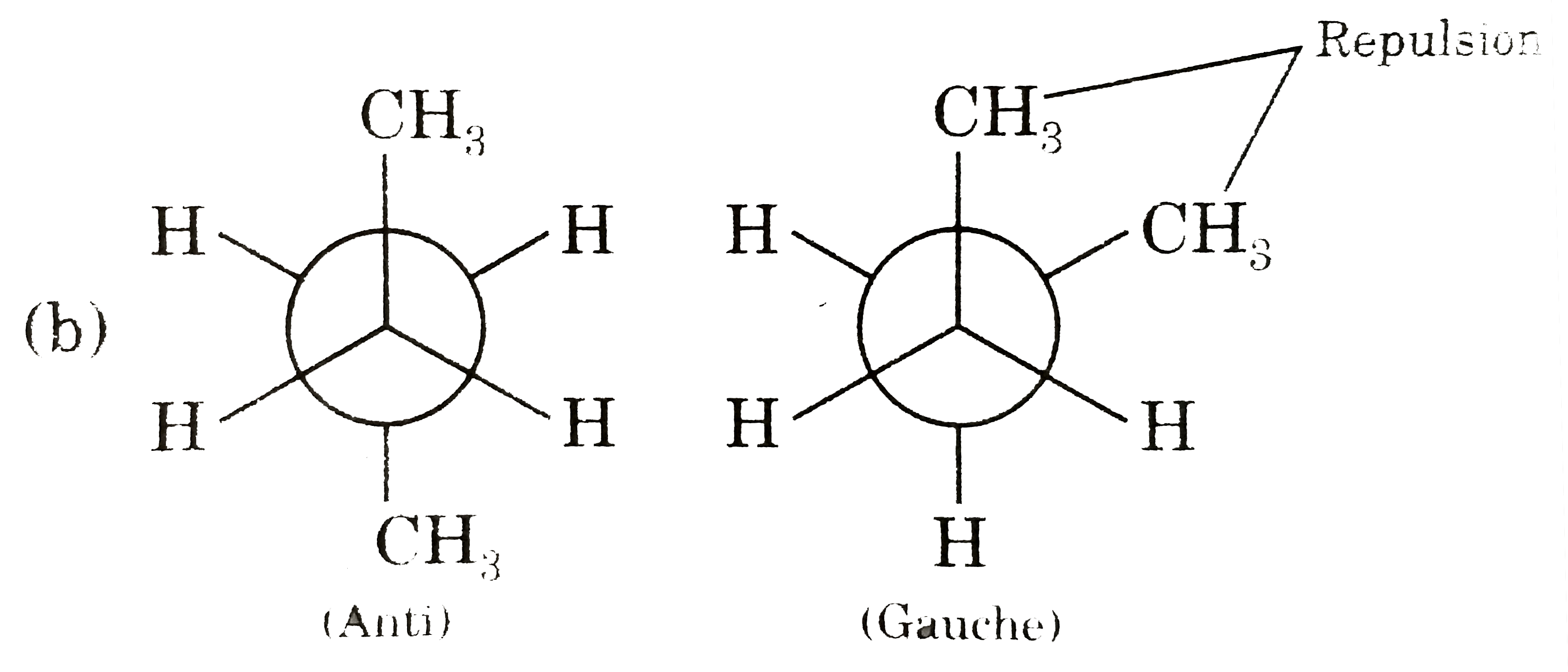 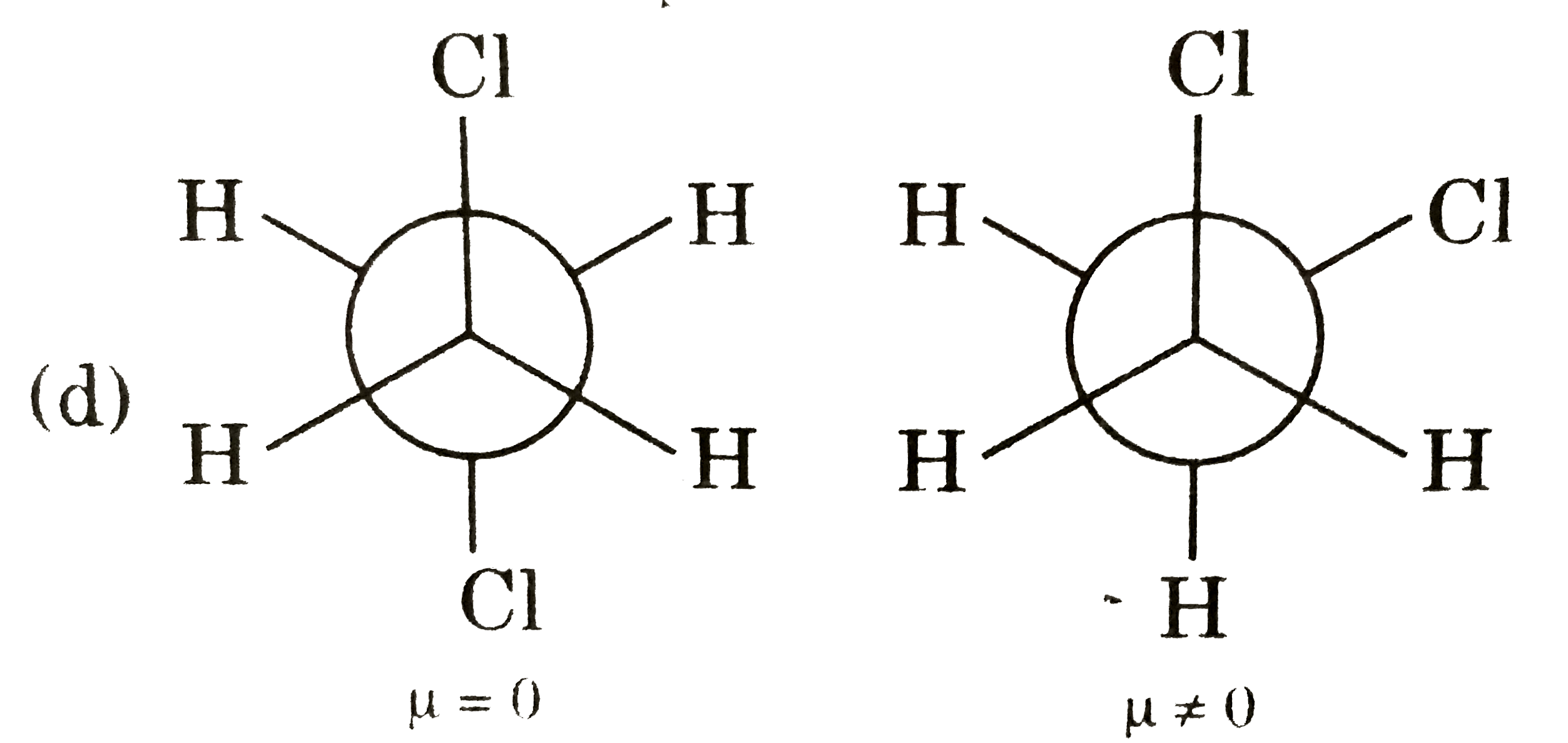 Staability of eclipsed from is less than staggered at room temperature. On heating rate of intercoversion from anti-gauche increases therefore `mu` increases. |
|
| 7. |
Select the types of redox reaction form the following on the basis of type of tedox changes: (a) intermolecular redox, (b) intramolecular redox (c) auto redox. If none, write none. (i)C_(6)H_(5) CHO overset(NaOH)(rarr) C_(6)H_(5)CH_(2) OH + C_(6) H_(5) COONa (iI) Cr_(2)O_(7)^(2-) + 2OH^(-) rarr 2CrO_(4)^(2-)+ H_(2)O (iii) 2Mn_(2)O_(7) rarr 4MnO_(2) + 3O_(2) (iv) NO_(3)^(-) + H_(2)S + H_(2)O + H^(+) rarr NH_(4)^(+) + HSO_(4)^(+-) (v) Fe + N_(2) H_(4) rarr NH_(3) + Fe(OH)_(2) (vi) 2KOH + Br_92) rarr KBr + KBrO (vii) 2Cu^(+) rarr Cu + Cu^(2+) (viii) Ag(NH_(3))_(2)^(+) overset(2H^(+))(rarr) Ag^(+) + 2NH_(4)^(+) (ix) 5KI+ KIO_(3) + 6HCl rarr 3I_(2) + 6KCI + 3H_(2)O |
|
Answer» |
|
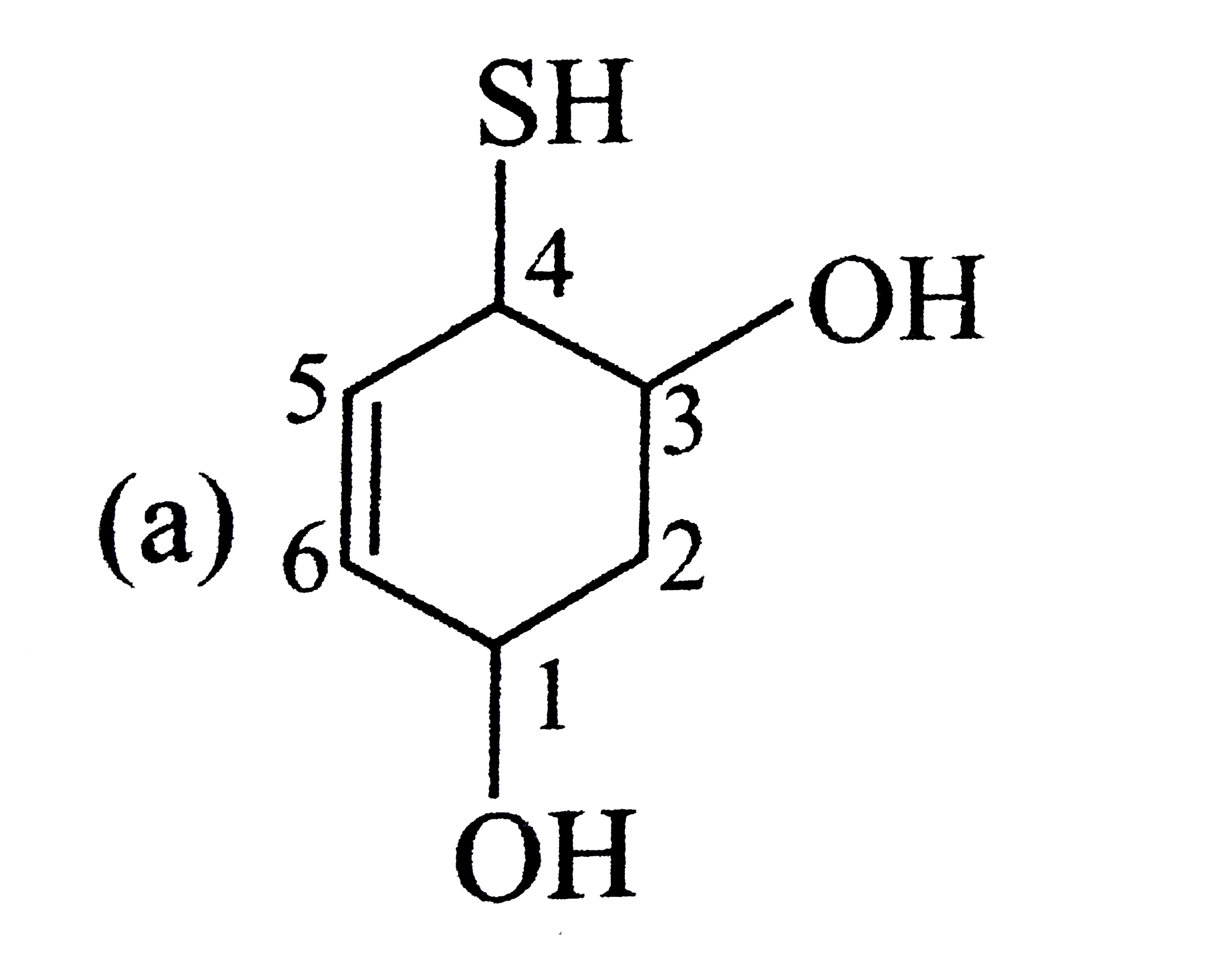
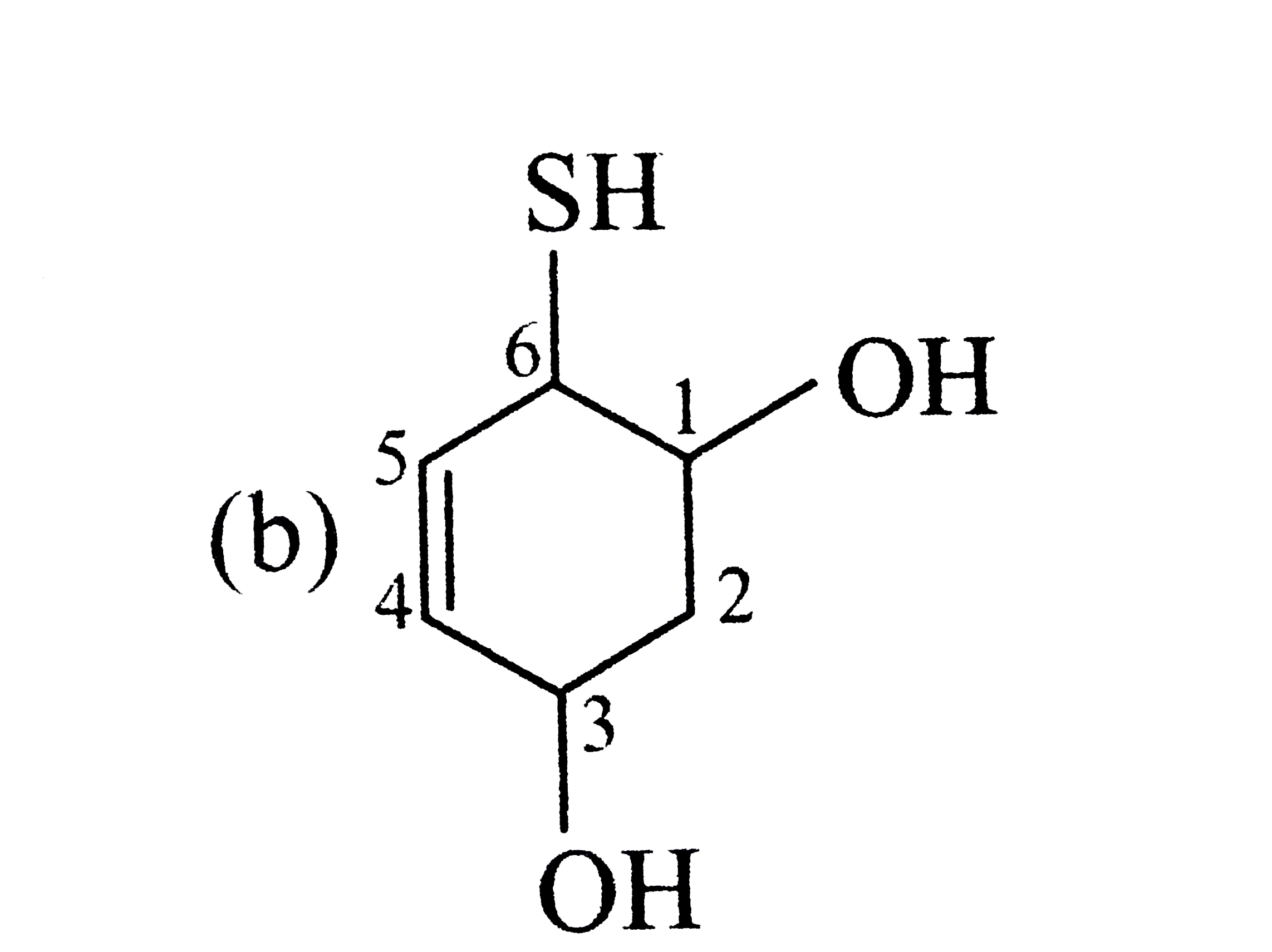
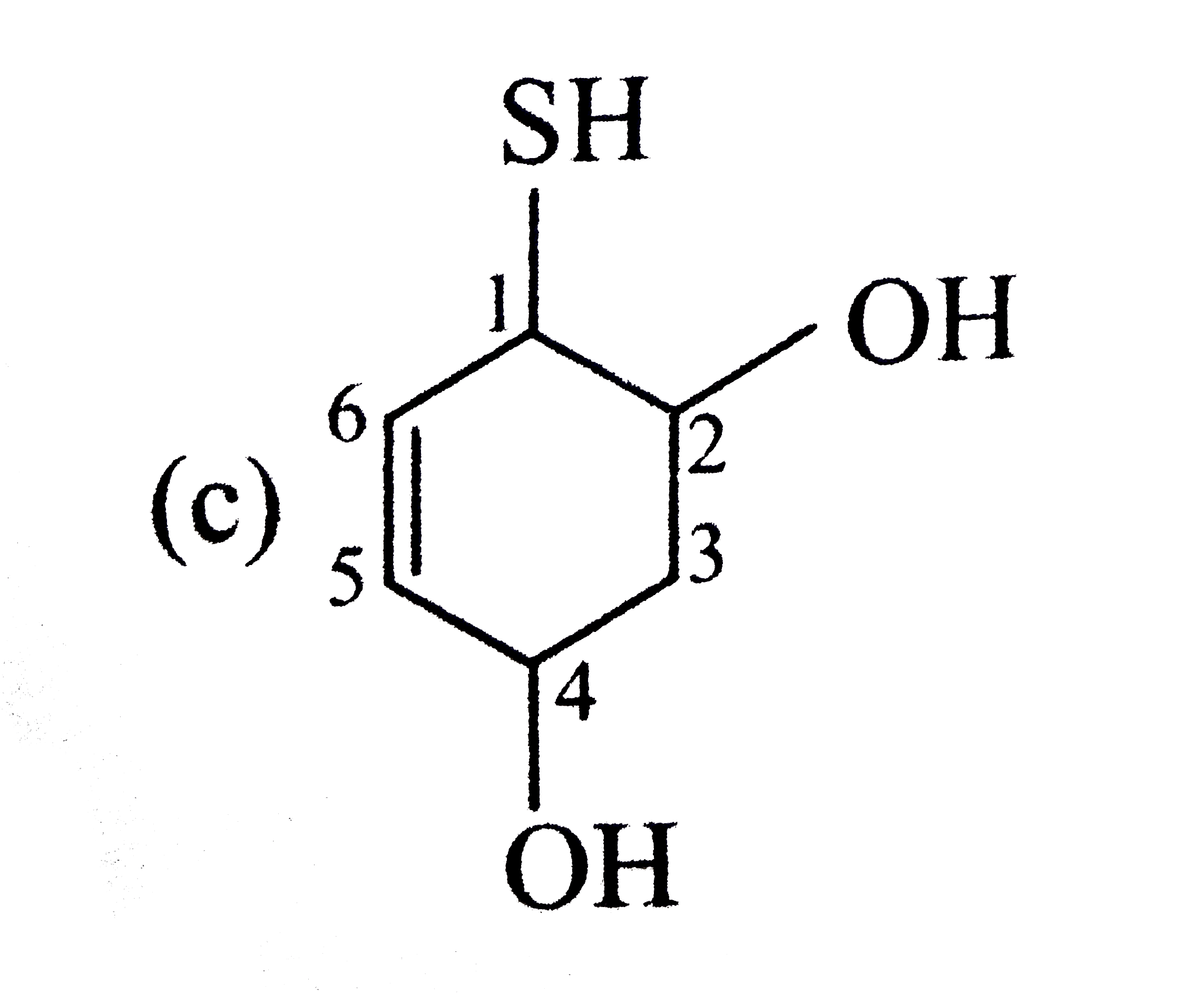
| 8. |
Select the structure with correct numbering for IUPAC name of the compound |
|
Answer»
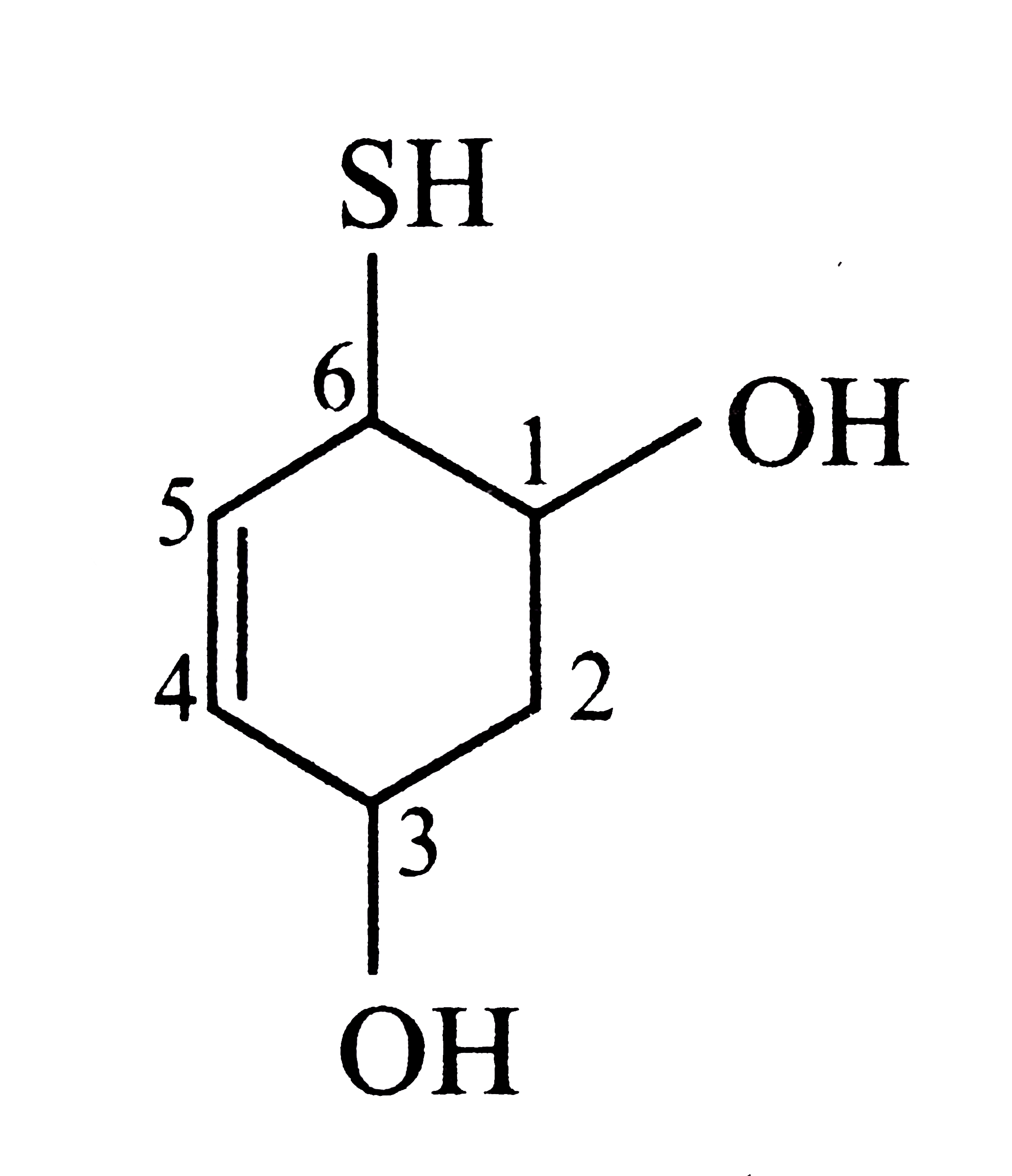
|
|
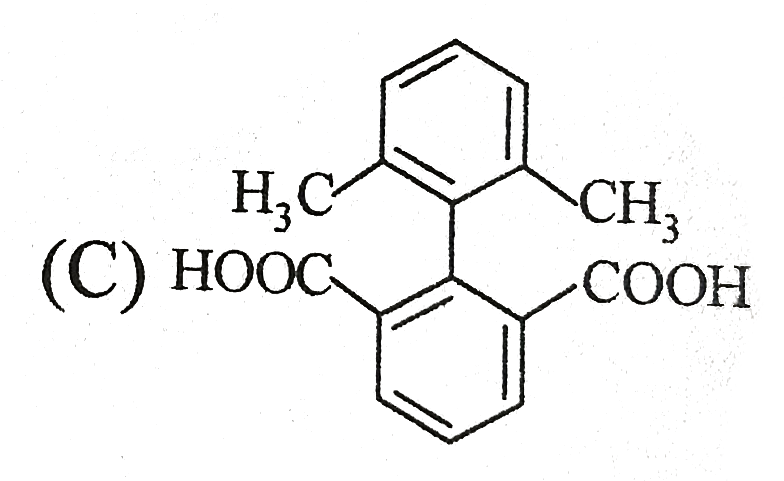
| 9. |
Select the structures that are not resonance structures of the same molecule . |
|
Answer»
|
|
| 10. |
Select the species which is not aromatic. |
|
Answer»
|
|
| 11. |
Select the species having zero oxidation state at the underlined elements : |
|
Answer» `(CH_(3))_(2)ul(S)O` |
|
| 12. |
Select the reactio(s) that would result in the formation 2-bromopropane. (1) CH_(3)CH=CH_(2)+Hbroverset("peroxide")(to)(II) CH_(3)CH=CH_(2)+Hbroverset(C Cl_(4))(to) (III) CH_(3)CH_(2)CH_(3)+Br_(2)overset(hv)(to)(IV) CH_(3)CH=CH_(2)+Broverset((C Cl)_(4))(to) |
|
Answer» I and III `IIIto` Halogenation of ALKANE TAKE PLACE. |
|
| 13. |
Select the reactions in which the correct orientations have been mentioned in the major products . |
|
Answer»
|
|
| 14. |
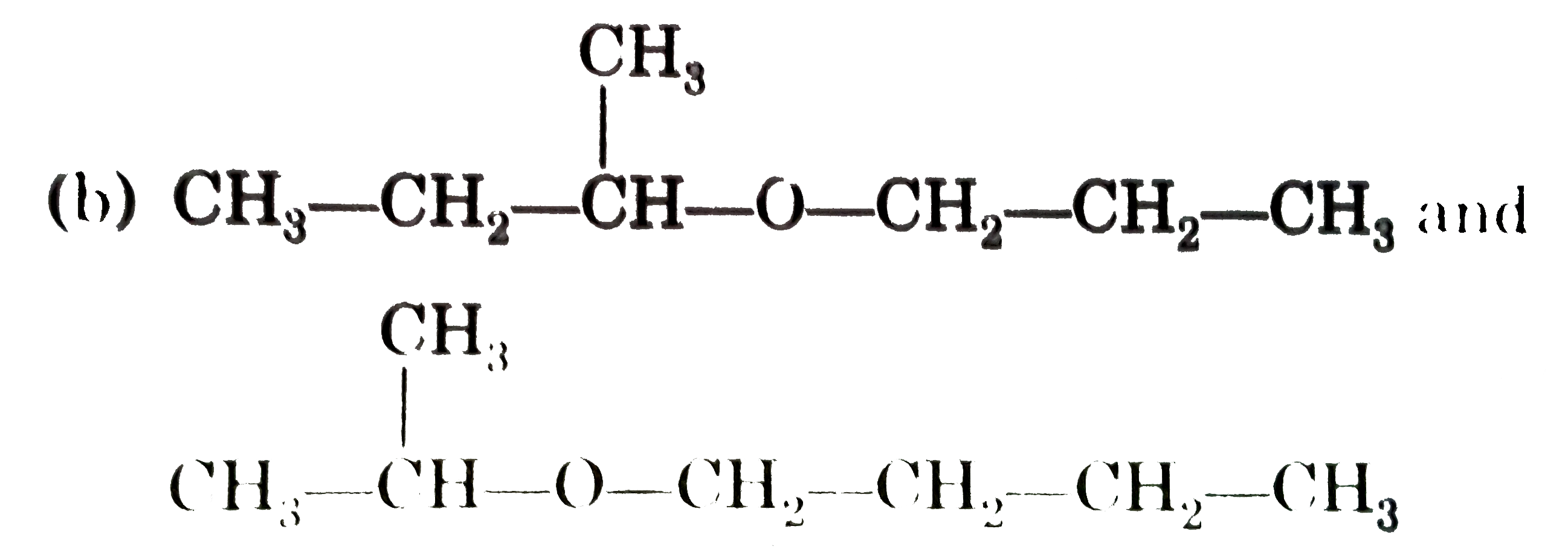
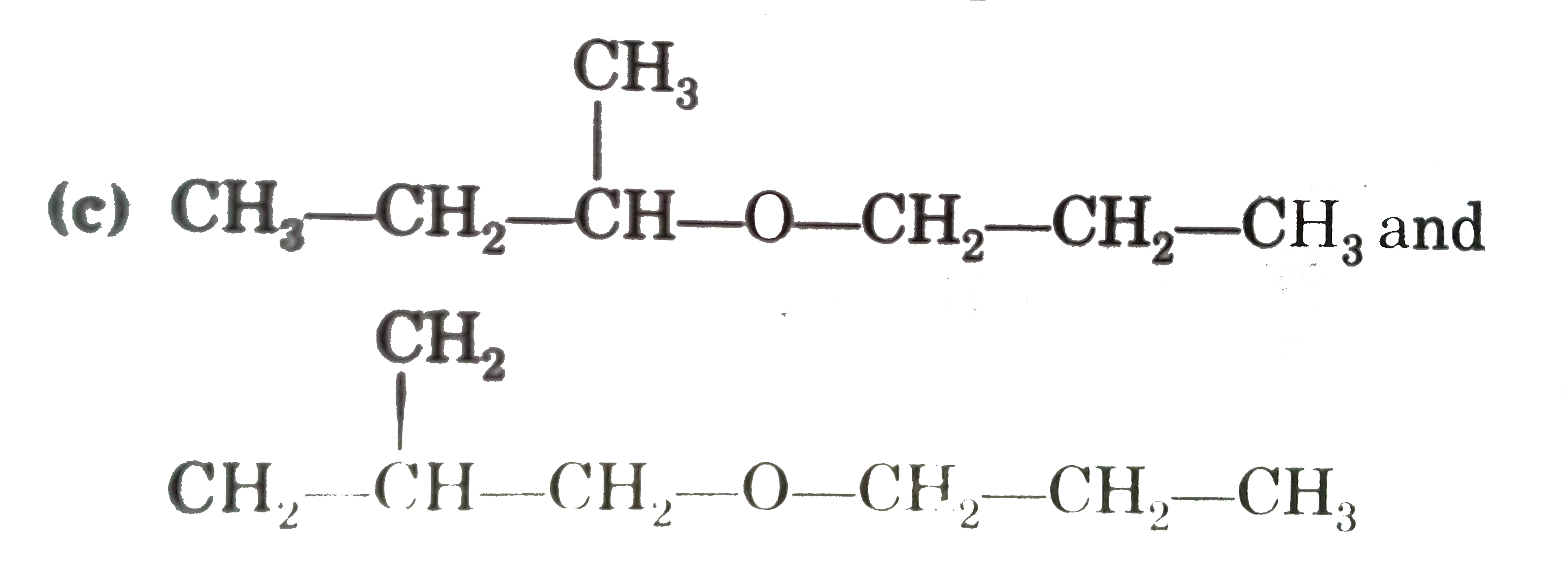
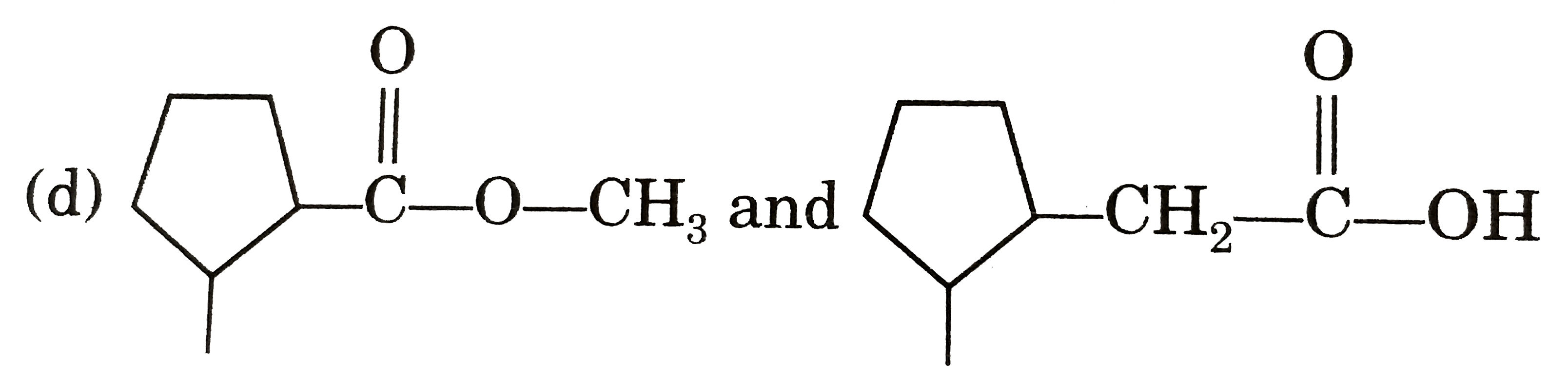
Select the pair of metamers from the following : |
|
Answer»
|
|
| 15. |
Select the pK_(a) value of the strongest acid from the following : |
|
Answer» `1.0` |
|
| 16. |
Select the pair of atoms having the same number of electrons in their outermost shell |
|
Answer» Solution :(a) `Na = [NE] 3s^(1), Ca = [Ar] 4s^(2)` (b) `Mg = [Ne] 3s^(2), Fe [Ar] 3d^(6) 4s^(2)` (c) As and Bi belong to the same group with outer shell E.C. `= ns^(2) NP^(3)` (d) Pb and Sb lie in different groups |
|
| 17. |
Select the oxidant/reducant atoms in the followingchange. Also report the number of electrons involved in redox change. As_(2)S_(3) + HNO_(3) rarr H_(3) AsO_(4) + H_(2)SO_(4) + NO |
|
Answer» |
|
| 18. |
Select the organic compounds, which will give red colour in Lassaigne's test? |
|
Answer» `NaCNS` |
|
| 19. |
Select the option in which all the parametres are intensive : |
|
Answer» PH, volume, Electrodes potential. |
|
| 20. |
Select the optically active compound among the following : |
|
Answer»
|
|
| 21. |
Select the optically active compound among the following: |
|
Answer»
`B to ` NON planar and optically active DUE to absence of POS & COS `C to ` non planar but having POS so , optically inactive. `D to `Planar compound. |
|
| 23. |
Select the natural series among the following: |
|
Answer» `(4N+1)` |
|
| 24. |
Select the non-polar molecules among the following : |
|
Answer» `NO_(2)` |
|
| 25. |
Select the name of mineral which is used for the production of boric acid |
|
Answer» Chromite |
|
| 28. |
Select the most stable intermediate. |
|
Answer»
|
|
| 31. |
Select the most appropriate statemen. In BF_(3) |
|
Answer» all thebonds are COMPLETELY ionic |
|
| 32. |
Select the members of group 14 that (i) forms the most acidicdioxide (ii) is commonly found in +2 oxidation state, (iii)used as semiconductor. |
|
Answer» (ii) Since theinert paireffect becomesmore and more predominant does the group, therefore, STABILITY of +2 oxidation state increasesdown the group. thus, lead is commonly found in +2 oxidation state. (iii) Siliconand germanium are neithertoo electronegativenor too electrpositive. Therefore they act as metalloidsand hence BEHAVEAS semiconductors. |
|
| 33. |
Select the molecule which has only one pi bond. |
|
Answer» `CH_(3)-CH=CH-CH_(3)` |
|
| 34. |
Select the member(s) of group 14 thatused as semiconductor. |
| Answer» SOLUTION :SILICON and GERMANIUM | |
| 35. |
Select the major product obtained from the addition of HBr to 1-methylcyclohexene. |
|
Answer» 1-bromo-2-methylcyclohexane |
|
| 36. |
Select the least basic compound. |
|
Answer»
|
|
| 37. |
Select the least acidic compound. |
|
Answer» `NO_(2)-CH_(2)-OVERSET(overset(O)(||))(C)-O-H` |
|
| 38. |
Select the indicator form the given table for titratioin of 20 mL of 0.02 M CH_(3)COOH with 0.02M NaOH. Given pK_(a)(CH_(3)COOH) = 4.74 |
|
Answer» I |
|
| 39. |
Select the incorrect statements |
|
Answer» KOH is a STRONGER base then NaOH |
|
| 40. |
Select the incorrect statement(s). |
|
Answer» At critical CONDITIONS, VOLUME occupied by the gas 12 times the volume of 1 mole GASEOUS molecuels |
|
| 41. |
Select the incorrect statement(s) : |
|
Answer» KOH is weaker base than NaOH B) `Mg^(+2)` ions are not precipitated as `KIP lt Ksp` C) Peroxides are more soluble than super OXIDES |
|
| 42. |
Select the incorrect statement with respect to sodium peroxide ? |
|
Answer» It decolurises the acidified `KMnO_(4)` solution. |
|
| 43. |
Select the incorrect statement of the following. |
|
Answer»
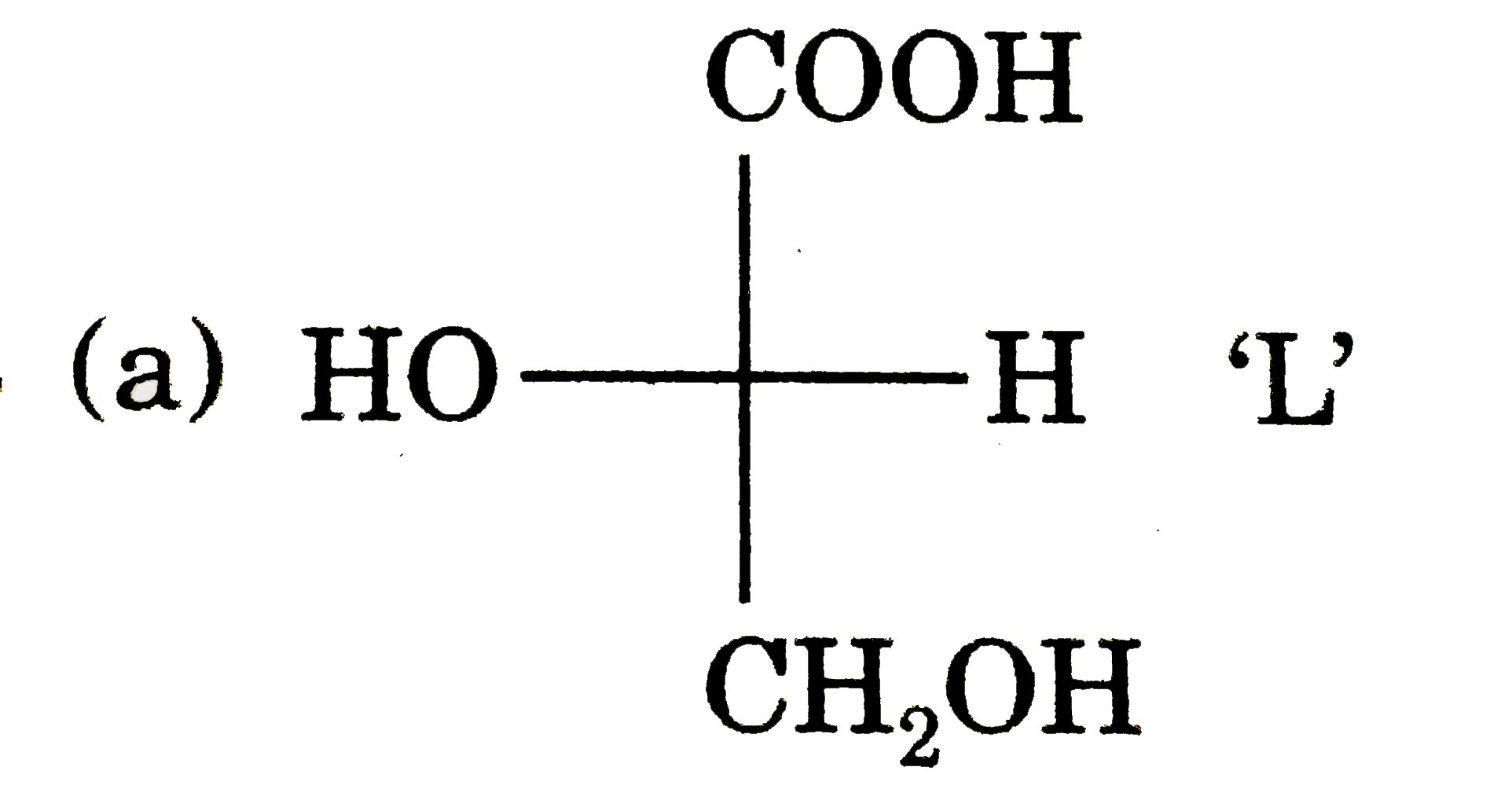 More ACTIVE H-more H- BONDING with MOLECULES. 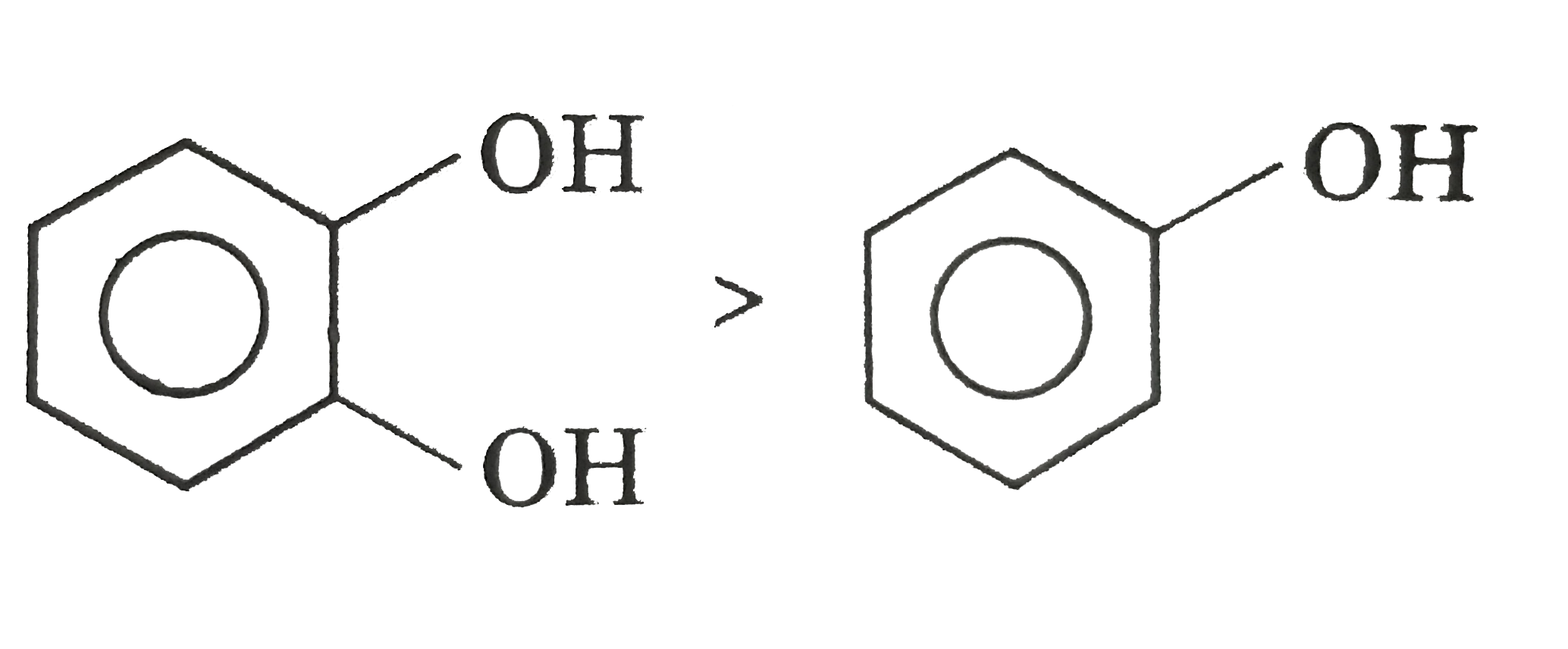 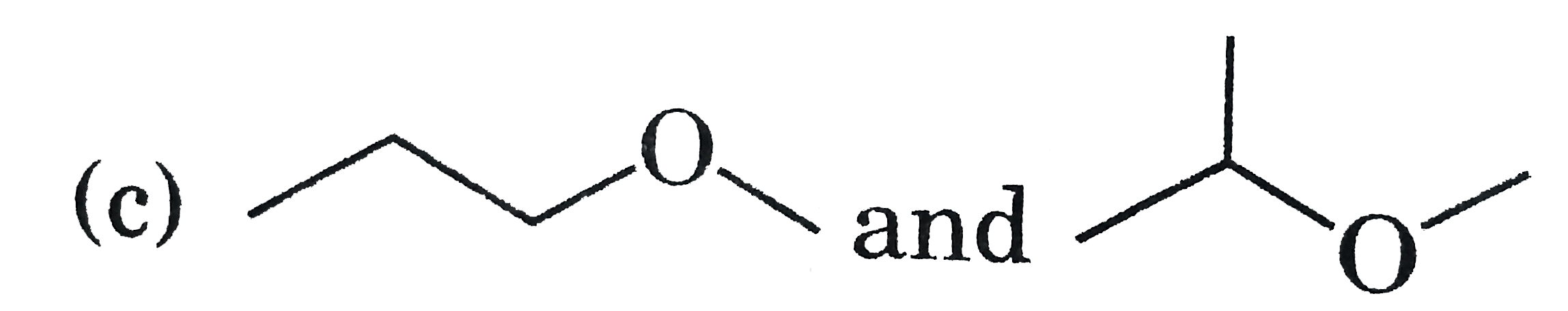
|
|
| 44. |
Select the incorrect statement from the following : |
|
Answer» `CH_(3)-COOH` can give effervescence of `CO_(2)` |
|
| 45. |
Select the incorrect statement for H_2O_2 structure |
|
Answer» It is NON planar |
|
| 46. |
Select the incorrect statement for developing of an exposed camera film involving the reaction |
|
Answer» a. Hydroquinol ACTS as REDUCTANT |
|
| 47. |
Select the incorrect statement about the boron |
|
Answer» PURE form of the elements are obtained by the reduction of `BCl_(3)` with ZINE at `900^(@)C` |
|
| 48. |
Select the incorrect statement about Barium |
|
Answer» It shows photo electric efect |
|
| 49. |
Select the incorrect statement : |
|
Answer» K.E. of photo-electron does not DEPEND upon the WAVELENGTH of INCIDENT radiation We know `lambda alpha 1/(upsilon)therefore KE alpha 1/(Delta lambda)` `therefofe ` KE dependent on `lambda` |
|