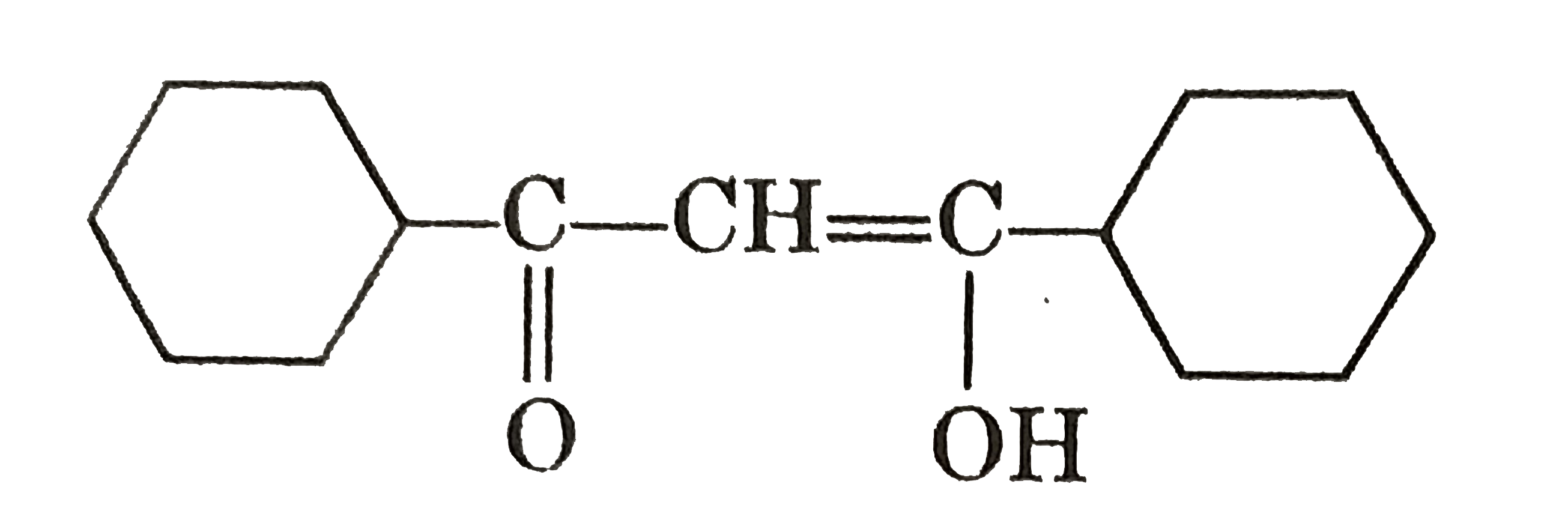Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Temporary hardness of water can be removed (A) By Clark.s process (B) By Boiling water |
| Answer» Answer :B | |
| 2. |
Temporary hardness of water can be removed |
|
Answer» By boiling |
|
| 3. |
Temperature pressure and molar concentration are the three factors which can disturb the equilibrium in areversiblereaction. Their effect is governedby Le Chatelier's principle .Whereas high concentration of the reactants always favours the formation of the products irrespective of its nature the effect of temperature and pressure depends upon the nature of the reaction . pressure does not disturb theequilibrium point in a reaction in which the reactants and products have same number of moles in the gaseous state. For a reaction CO(g) +H_(2)O(g) hArr CO_(2) (g) +H_(2) (g) at a given temperature the equilibrium amount of CO_(2) (g) can be increased by |
|
Answer» adding a catalyst |
|
| 4. |
Temperorary hardness is caused due to the presence of: |
|
Answer» `CaSO_(4)` |
|
| 5. |
Temperature of1 mole of a gas is increased by 1^(@) at constant pressure. Work done is |
|
Answer» 2 cal When temp. is INCREASED by `1^(@)`, at CONSTANT pressure, suppose volume increased by `DELTAV` `:. P ( V+DeltaV) = R (T+1) `or`PV + P DeltaV =RT + R ` But`PV =RT :. P Delta V = R`, i.e., work done `=R= 2 cal`. |
|
| 6. |
Temperature of onemole of helium gas is increased by 1^(@)C. Hence,the increasein its internal energy will be |
|
Answer» 2 CAL But for MONOATOMIC GAS, `C_(v)= (3)/(2) R`. Also `DeltaT =1^(@)` (Given) Hence, `DeltaU = C_(v)DeltaT = (3)/(2) R = (3)/(2) xx2 = 3 cal` |
|
| 7. |
Temperature of an ideal gas increases in: |
|
Answer» Adiabatic expansion Expansion `rArr W = - Ve rArr Delta E lt 0 rArr Delta T lt 0` (b) `q=0, W gt 0, Delta E = W rarr Delta E gt 0 rArr Delta T gt 0` (C ) Isothermal `rArr Delta T = 0` (d) Isobasc expansion `rArr nC_(P) Delta T = nC_(P) Delta T - nR Delta T` `nR Delta T = nDelta T (C_(P) - C_(v)) gt 0 rArr Delta T gt 0` |
|
| 8. |
Temperature of a flask becomes 27^(@)C to 277^(@)C. At 277^(@)C temperature 0.1 dm^(3) gas bubble out from the flask then calculate volume of flask. |
|
Answer» |
|
| 9. |
Temperature of a gas is tK. What would be the temperature at which volume and pressure, both will reduced to half of the initial values |
|
Answer» t/2 |
|
| 10. |
Temperature of 1 mole of a gas is increased by 2^(@)C at constant pressure. Work done is : |
| Answer» Solution :`w=-nR(T_(2)-T_(1)),w=-1R(2)=-2R` | |
| 11. |
Temperature below which the gas does not obey ideal gas law is |
|
Answer» CRITICAL TEMPERATURE |
|
| 12. |
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called |
|
Answer» INVERSION TEMPERATURE |
|
| 13. |
Temperature at the foot of a mountain is 30^@C and pressure is 760mm whereas at the top of the mountain these are 0^@C and 710mm. Ratio of the densities of the air at the foot and the top of the mountain is ____(approx) |
|
Answer» |
|
| 14. |
Temperature at the foot of a mountain is 30^(@)C and pressure is 760 mm whereas at the top of the mountain these are 0^(@)C and 710 mm. Compare the densities of the air at the foot and at the to of the mountain. |
|
Answer» <P> |
|
| 15. |
Teflon is also known as PTFE. |
|
Answer» |
|
| 16. |
Technique of distribution of mixture between two phases, one stationary phase and other mobile phase is |
|
Answer» DISTILLATION |
|
| 17. |
…….technique is applied to separate different fraction of crude oil in petroleum |
| Answer» SOLUTION :FRACTIONATING COLUMN TECHNIQUE | |
| 18. |
Tautomers possess |
|
Answer» same PHYSICAL properties but different CHEMICAL properties |
|
| 19. |
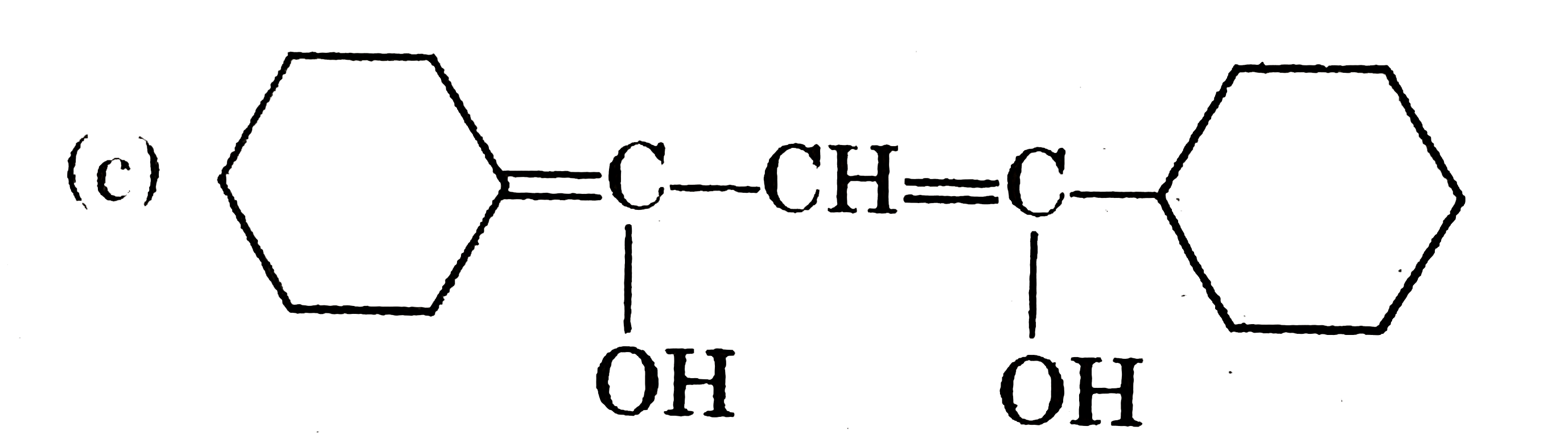
Tautomerism : Structural Isomers that undergo rapid interconversions and exist in dynamic equilibrium are known as Tautomers and relationship between them is known as Tautomerism. Tautomers generally have different functional groups. At equilibrium more stable tautomer is present in higher amount by ratio remains same until and unless change is made externally. Tautomerism actually arises due to rapid oscillation of an atoms between two polyvalent atoms in a molecule underset("Keto form")(underset(H)underset(|)(C)H_(2)-overset(O)overset(||)(CH)) hArr underset("enol form")(CH_(2)=overset(OH)overset(|)(CH) Above is an example of Keto-enole tautomerism. condition for this type of keto enol tautomerism is presence of alpha-H. amount of enol at equilibrium is known as enolic content. it is more if enol is more stable and less if keto is more stable. Which of the following compounds have higher enolic content than Keto content : |
|
Answer» II & IV 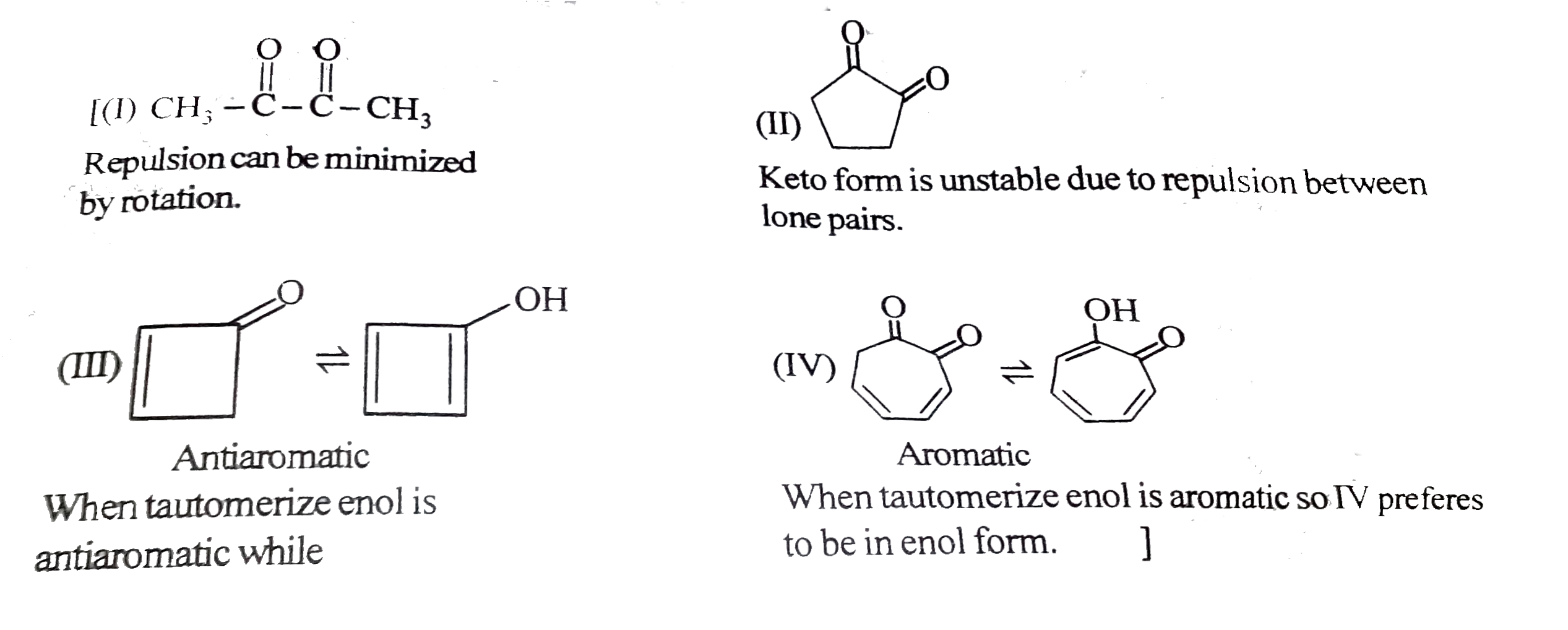
|
|
| 20. |
Tautomerism : Structural Isomers that undergo rapid interconversions and exist in dynamic equilibrium are known as Tautomers and relationship between them is known as Tautomerism. Tautomers generally have different functional groups. At equilibrium more stable tautomer is present in higher amount by ratio remains same until and unless change is made externally. Tautomerism actually arises due to rapid oscillation of an atoms between two polyvalent atoms in a molecule underset("Keto form")(underset(H)underset(|)(C)H_(2)-overset(O)overset(||)(CH)) hArr underset("enol form")(CH_(2)=overset(OH)overset(|)(CH) Above is an example of Keto-enole tautomerism. condition for this type of keto enol tautomerism is presence of alpha-H. amount of enol at equilibrium is known as enolic content. it is more if enol is more stable and less if keto is more stable. Tautomers of which of the following can show geometrical isomerism |
|
Answer» `CH_(3)-CHO` 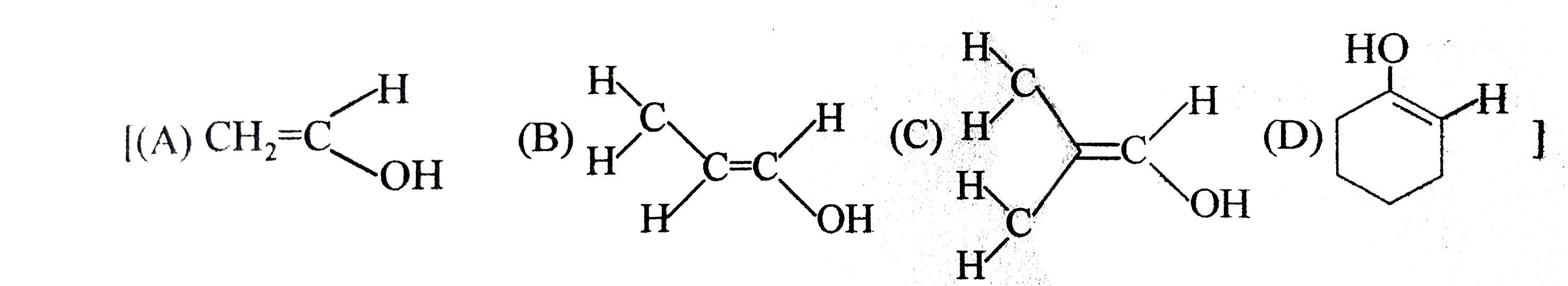
|
|
| 21. |
Tautomerism is exhibited by X number of compounds among the following. What is X. |
|
Answer»

|
|
| 22. |
Tautomerism is not exhibited by : |
|
Answer»
|
|
| 23. |
Tautomerism is exhibited by |
|
Answer» `(Me_(3)C CO)_(3)CH` |
|
| 24. |
Tautomerism is due to spontaneous interconversion of two isomeric forms with differentfunctional groups into each other. The term tautomer means constitutional isomers thatundergo such rapid inter conversion that they cannot be independently isolated. In keto-enol tautomerism although Keto form in general is more stable, but some factors like H-bonding and extended double bonds conjugation may increase the stability of enol form Enolic form of acetyl acelone is stabilized due to (I) resonance as a result of conjugation (II) intra molecular H-bonding(III) dipole- dipole repulsion |
|
Answer» I and III 1) EXTEND of conjugation 2) intra molecular HYDROGEN BOND formation. |
|
| 25. |
Tautomerism is due to spontaneous interconversion of two isomeric forms with differentfunctional groups into each other. The term tautomer means constitutional isomers thatundergo such rapid inter conversion that they cannot be independently isolated. In keto-enol tautomerism although Keto form in general is more stable, but some factors like H-bonding and extended double bonds conjugation may increase the stability of enol form Which of the following statements are correct of the following |
|
Answer» I and II are tautomers |
|
| 26. |
Tautomer of which of the follwing can show geometrical isomerism |
|
Answer» `CH_(3)-CHO` 
|
|
| 27. |
Tautomerism exhibited by |
|
Answer» `(CH_(3))_(3)CNO` 
|
|
| 28. |
Tautomer of which of the following can show geometrical isomerism? |
|
Answer» `CH_3-CHO` |
|
| 29. |
Tautomer of which of the following can show geometrical isomerism |
|
Answer» `CH_3 - CHO` |
|
| 30. |
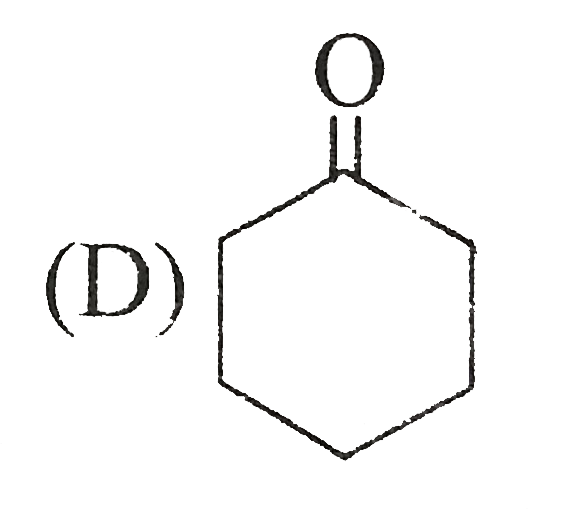
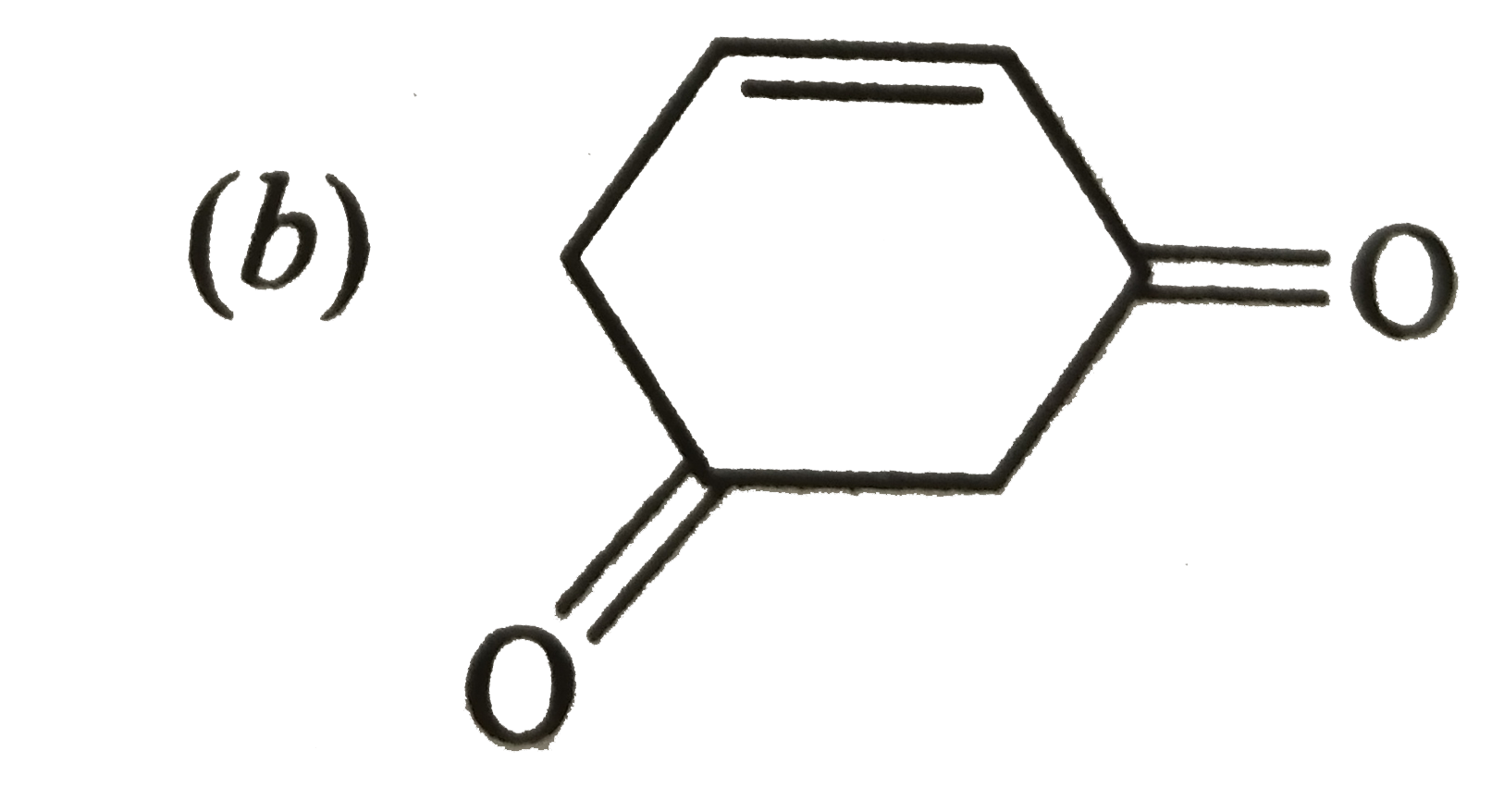
Tautomer of following compound is : |
|
Answer»
|
|
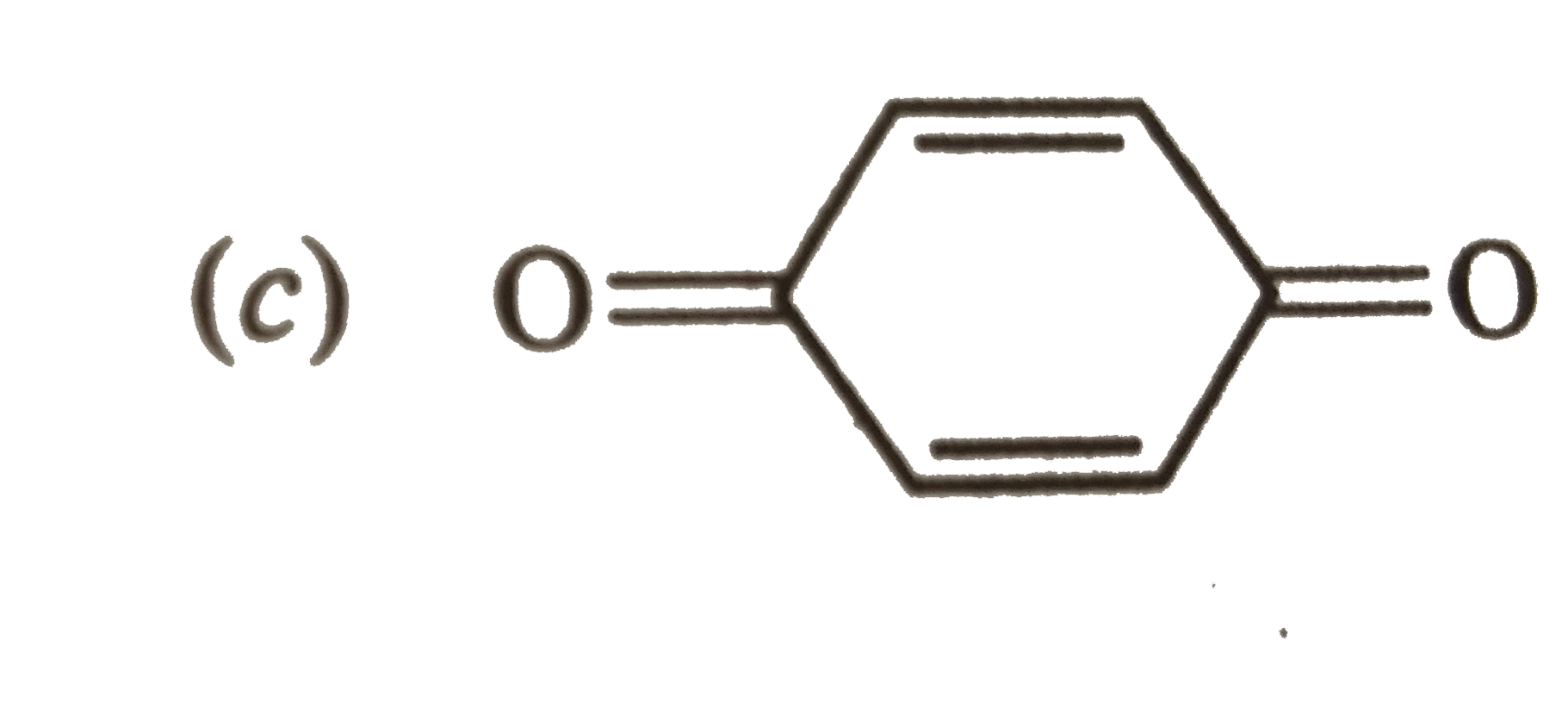
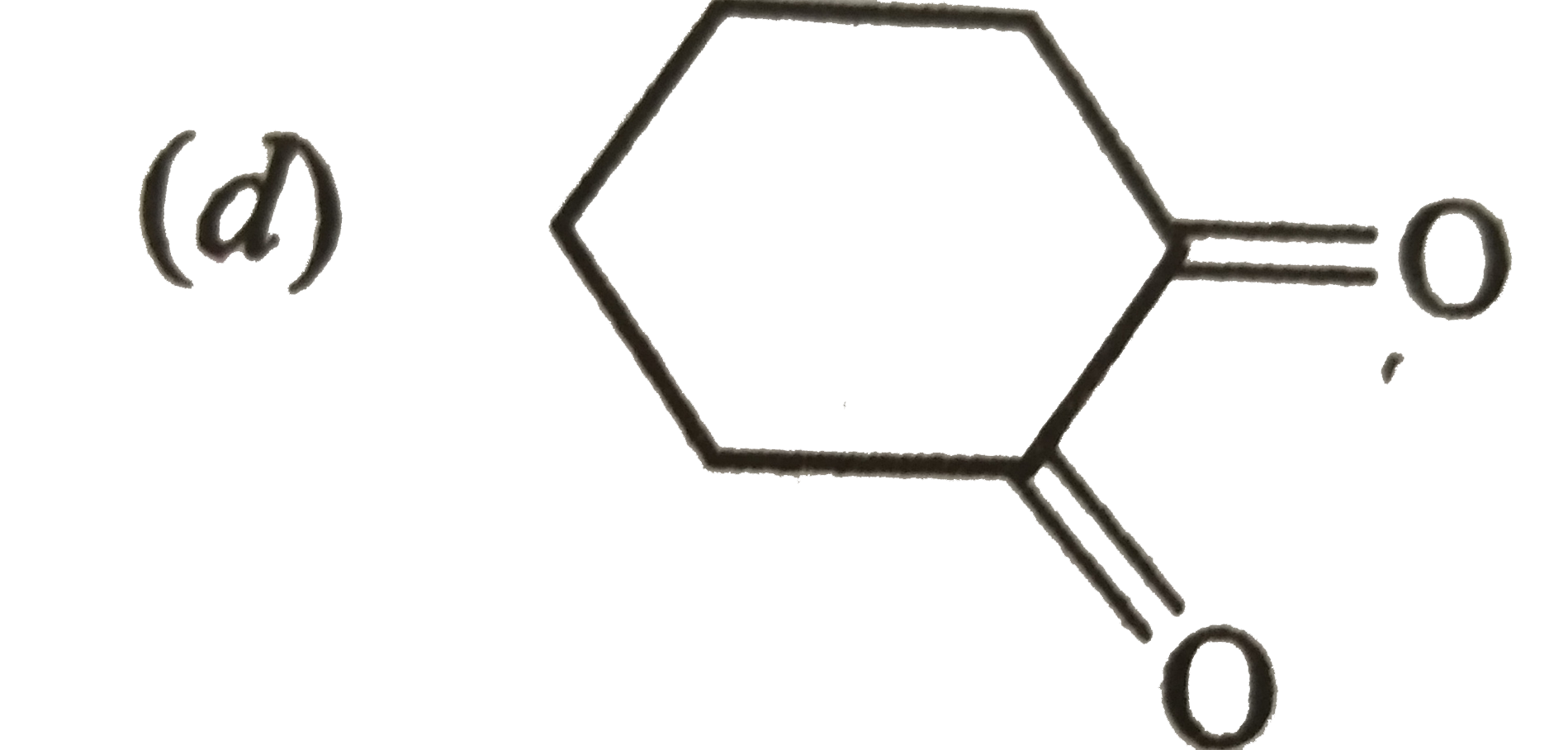
| 31. |
Tautomer of following compound is : |
|
Answer»
|
|
| 32. |
Tautomer in following is Diad system : |
|
Answer» `CH_3COCH_3` |
|
| 33. |
Tartonic acid is a product from tartaric acid formed by treatment with |
Answer» SOLUTION :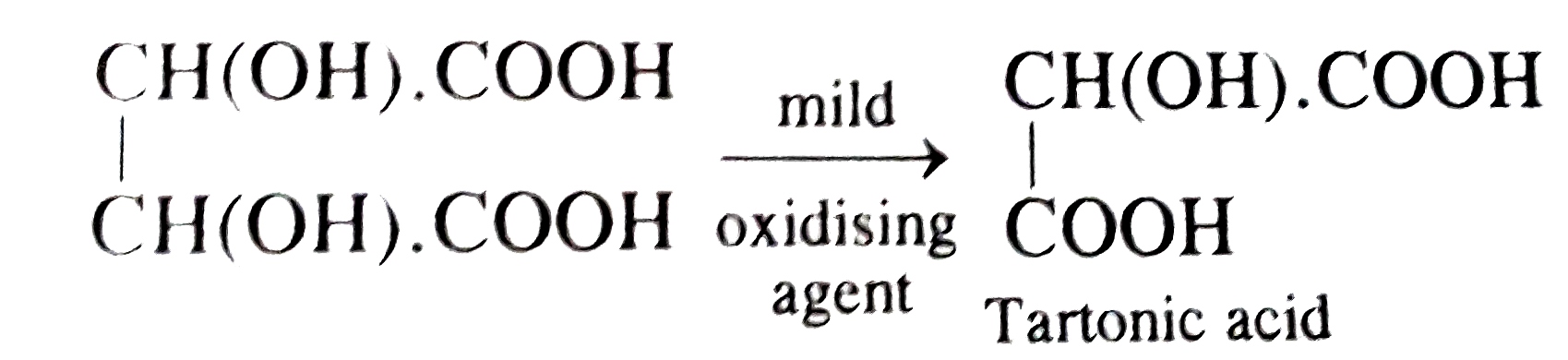
|
|
| 34. |
Tamarind contains |
|
Answer» DEXTRO--TARTARIC ACID |
|
| 35. |
TakingZ-axis as the internuclear axis, explain why2p_(x) or 2p_(y)orbital does not combine with 2s obtial to form molecular orbtals ? |
| Answer» SOLUTION :The SMALL ++ overlap cancels the +- overlap . HENCE , there is not net OVERLAPPING . | |
| 36. |
Taking Z-axis as the intermolecular axis , when two 2p_(x) orbitals of two atoms/ions overlap, the molecular orbitals formed are ………….and ……… |
|
Answer» <P> SOLUTION :`pi_(2_(p_(X))),pi_(2_(p_(x)))^(**)` |
|
| 37. |
Take the following reaction in consederation. In which enthalpy change is positive. 2A_((g)) hArr C_((g))+ D_((g)) . Which of the following will not affect on equilibrium ? |
|
Answer» CHANGE in CONCENTRATION of reactants |
|
| 38. |
Take a quick look at environment pollution. |
|
Answer» Solution :Environmental pollution is the effect of undesirable changes in our surroundings that have harmful effects on plants, animals and human beings. A SUBSTANCE, which causes pollution, is known as pollutant. Pollutants can be solid, liquid or gaseous SUBSTANCES present in greater concentration than in natural abundance and are produced due to human activities or due to natural happenings. Some pollutants can be degredable, like discarded vegetables. But some pollutants which are slowly degradable remain in the environment in an unchanged form for many decades. For examples, plastic MATERIALS, heavy metals, nuclear wastes, DDT and other many chemicals. These types of substanes once released into the environment are difficult ot remove. These pollutants cannot be degraded by natural processes. So they are harmful to living organisms. In the process of environmental pollution, pollutants ORIGINATE from a source and get transported by air or WATER or are dumped into the soil by human beings. |
|
| 40. |
Taj Mahal is threatened by pollution from |
|
Answer» Chlorine |
|
| 41. |
Table-tennis ball has a mass 10g and a speed of 90m/s. If speed can be measured within an accuracy of 4% what will be the uncertainty in speed and position? |
|
Answer» Solution :`m = 10G = 10xx 10^(-3) kg = 10^(-2) kg` Uncertainty in SPEED `(Delta v) = 4% " of "90 ms^(-1) = (4)/(100) xx 90 = 3.6 ms^(-1)` `Delta x xx (m xx Delta v) = (h)/(4PI) :. Delta x = (h)/(4pi m Delta v) = (6.626 xx 10^(-34) kg m^(2) s^(-1))/(4 xx 3.14 xx (10^(-2) kg) (3.6 ms^(-1))) = 1.46 xx 10^(-33) m` |
|
| 42. |
Table-tennis ball has a mass 10 g and a speed of 90 m/s. If speed can be measured within an accuracy of 4% what will be the uncertainty in speed and position |
|
Answer» Solution :The Uncertainty in the speed of ball `=(90xx4)/(100)=(360)/(100)=(360)/(100)=3.6 MS^(-1)` Uncertainty in position = `(H)/(4pi m TRIANGLE V)` |
|
| 43. |
System in which there is no exchange of matter , work or energy from surrounding is ____ |
| Answer» SOLUTION :ISOLATED | |
| 44. |
Synthesis gas is a mixture of |
|
Answer» `CO+H_(2)` |
|
| 48. |
Symbols ._(35)^(79)Br and .^(79)Br can be written whereas symbols ._(79)^(35)Br and ._(35)Br are not acceptable. Answer briefly. |
| Answer» Solution :`._(79)^(35)BR` is not acceptable because atomic number should be written as SUBSCRIPT, while mass number should be written as SUPERSCRIPT. `._(35)Br` is not acceptable because atomic number of an element is fixed. HOWEVER, mass number is not fixed as it depends upon the isotope taken. Hence, it is essential to indicate mass number. | |
| 49. |
Symbols ._(35)^(79)Br and .^(79)Br can be written, where as symbols ._(79)^(35)Br and .^(35)Br are not acceptable. Answer briefly. |
|
Answer» Solution :The compostion of any atom can be represented by USING the NORMAL element symbol X with superscript on the left side as the mass number A and subscript Z on the left side as atomic number (ie) `._(Z)^(A)X`. Hence , the symbols `._(79)^(35)Br and ._(35)Br` are acceptable. |
|
| 50. |
Symbols ._(35)^(79)Br and .^(79)Br can be written, whereas symbols ._(79)^(35)Br and .^(35)Br are not acceptable. Answer briefly. |
| Answer» Solution :Atomic NUMBER of an ELEMENT is FIXED. HOWEVER, mass number is not fixed as it depends upon the isotope taken. Hence, it is ESSENTIAL to indicate mass number. | |