Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The absolute entropies of H_(2(g)), O_(2(g)) and H_2O_((l)) are respectively 130.6, 205.1 and 69.9 JK-mol^(-1). Calculate the value of DeltaS for the formation of one mole liquid water. |
|
Answer» |
|
| 2. |
The absolute configuration of is |
|
Answer» S, S  Both `C_(1) and C_(2)` have R-configuration. |
|
| 3. |
Process A rarr B represents |
|
Answer» enthalpy for EXOTHERMIC REACTIONS |
|
| 4. |
The ability of ion to bring about coagulation of a give colloidal solution depends upon |
|
Answer» the size of its ion |
|
| 5. |
The ability of an element to form long chains or rings by linking its atoms is called ......... |
| Answer» SOLUTION :CATENATION | |
| 6. |
The ability of a given substance to assume two or more crystalline structure is called |
|
Answer» POLYMORPHISM |
|
| 7. |
The ability of a catalyst to accelerate the chemical reaction is known as |
|
Answer» Selectivity |
|
| 8. |
The 8:8 type ofpacking is present in |
|
Answer» CsCl |
|
| 9. |
The 8:8 type of packing is present in: |
|
Answer» CsCl |
|
| 10. |
The 6th period of the periodic table contains |
|
Answer» TWO s-block and six p-block ELEMENTS |
|
| 11. |
The 4f subshell of an atom contains 10 electrons. What is the maximum number of electrons having spin in the same direction ? |
Answer» SOLUTION :SEVEN 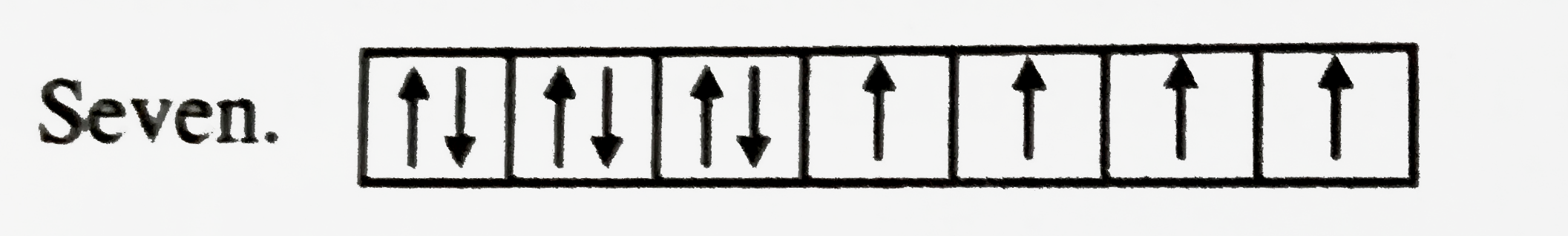
|
|
| 12. |
The 4 F level is successively filled up in |
|
Answer» Rare earths |
|
| 13. |
The 4.0 pH containing solution, concentration of [H^+]is increased 5 times then what is the pH of resulting solution ? |
|
Answer» `5.0` |
|
| 14. |
The 2D unit cell of an element is shown. The two layers are placed one over the other and touching each other. Find the effective number of atoms in the unit cell |
|
Answer» |
|
| 15. |
The 2.901 L saturated solution is formed by 0.08 g CaF_2 at 298 K temp. then calculate K_(sp)(Molecular moles of CaF_2 is 78 g "mol"^(-1)) |
| Answer» SOLUTION :`1.715xx10^(-10)` | |
| 16. |
The ………. Radius of chlorine is99 pm whileits …….. Radius is 180pm. |
| Answer» SOLUTION :Covalentvander WAALS | |
| 17. |
The [111] plane is parallel to |
|
Answer» XY-PLANE |
|
| 18. |
The +1oxidation state in group 13 and +2 oxidationstate in group 14 becomesmore and morestable with increasing atomic number. Explain |
|
Answer» Solution :Theouter electronic configurationof GROUP 13 element is `ns^(2) NP^(1)` and hence their group oxidation state is `+3`. Both B and Al showan oxidationstate of `+3`. Howover , from GA to Tl.The elementhave either `d-` or `d-` andf-electronsbesidess- andp-electrons. Since d- and f-electronshave poor shieldingeffect,THEREFORE , they do not screen s-electronof the valencesheleffectively from the attraction of the nucleus. Consequently,two s-electrons of the valenceshell remain attractedby the nucleusand hence do notparticipat in bond formationwhile only the remaining one p-electron of the valenceshell participatesin bondformation.Further, as the number of d- or d- and f-electronincreasesdown the group, the inert pair effect becomes more nad more prominent. Consequently, the solubility of +3 oxidationstate decreaseswhile that of +1oxidation state increasesdown the group. The outer electronicconfiguationof group 14 element in `ns^(2) np^(2)` and hencetheir group oxidationstate is+4. Both C and Si show an oxidation state of +4. However, from Ge to Pb , the elements have eitherd- or both d- and f-electrons, in addition to s- and p-electrons. Due topoor shielding of s-electrons of the valenceshell bythese d- and f-electrons , the inertpair effectcomes into play. As a result, theelementsfrom Ge toPb showoxidationstates of +2 and +4 . Further , as the number ofd- or d- and f-electronsincreasesdown thegroup, the inert pair effect becomes more andmore perdominant. Consequently, the stabilityof +4 oxidationstate decreases while that of +2 oxidation stateincrease down thegroup from Ge to Pb. |
|
| 19. |
The +1 oxidation state of thalliumis more stable than its +3 oxidationstatebecause of |
|
Answer» its ATOMIC size |
|
| 20. |
The +1 oxidation state is more stable than the +3 oxidation state for thallium. Explain why ? |
| Answer» Solution :The OUTER electronic configurationof thalium is `6s^(2)6p^(1)`.Therefore, it can show oxidationstates of `+1` and`+3` But DUE to inertpair effect,the `6s^(2)` electronsare more stronglyattractedby the nucleus than `6p^(1)`electron. Therefore, `6s^(2)` electronsdo not participatein BOND formation.Insteadonly `6p^(1)` electrontakes PART is more stable than its `+3`state. | |
| 21. |
The +1 - oxidation state in group-13 and +2 oxidation state in group-14 becomes more and more stable with increasing atomic number. Explain. |
|
Answer» Solution :On moving down, in group-13 and 14, lower oxidation state becomes more stable as compared to higher oxidation state, because of inert pair effect. In inert pair effect, s electrons of valence shell do not participate in bonding only .p. electrons participate in bonding. As the SIZE of atom INCREASES, more ENERGY is needed by .s. electrons to participate in bonding from valence shell. In group 13 valence shell CONFIGURATION is `ns^2 np^1` (n = 2 to 6), when electrons of both .s. and .p. ORBITALS participate they show +3 oxidation state but, if only .p. electrons participate then they show +1 oxidation state. In group-14 valence shell configuration is `ns^2 np^2` (n = 2 to 6), when electrons of both .s. and .p. orbitals participate then +4 oxidation state and if only .p. electrons participate, then they show +2 oxidation state. |
|
| 22. |
The 0.2 g of anhydrous organic acid gave on combustion 0.04 g of water and 0.195 g of CO_2The acid is a dibasic acid and 0.5 g of its silver salt leaves on ignition 0.355 g of silver. The percentage of hydrogen in the compound is |
|
Answer» 90 |
|
| 23. |
The 0.2 g of anhydrous organic acid gave on combustion 0.04 g of water and 0.195 g of CO_(2) The acid is a dibasic acid and 0.5 g of its silver salt leaves on ignition -.355 g of silver.The percentage of carbon in the compound is |
|
Answer» `5.6` `=(0.04)/(0.2).(2)/(18).100=2.22%` |
|
| 24. |
That type of crystal defect is indicated in the diagram below |
|
Answer» Frenkel defect |
|
| 25. |
Thallous chloride is more stable than thallic chloride because of |
|
Answer» More ionic character |
|
| 26. |
Thallium shows different oxidation states because |
|
Answer» It is transition element |
|
| 27. |
Thallium chloride (TICI) crystallizes in a cubic lattice whose edge length is found to be 385 pm. If the density of the solid is found to be 7.0 g cm^(-3), predict the type of lattice to which the crystals of TICI belong (Atomic mass of TI = 204, Cl=35.5) |
|
Answer» |
|
| 28. |
Thallium chloride (TICI)crystallizes in a cubic lattice whose edge length is found to be 385 pm. If the density of the solid is found to be7.0 " g cm"^(-3), predict the type of lattice to which the crystals of TICI belong . (Atomic mass of TI = 204, Cl = 35.5) |
|
Answer» |
|
| 31. |
Tetracyline is called a broad spectrum antibiontic because it active against a wide variety of becteria. How many chirality centre does tetracyline have ? |
Answer» 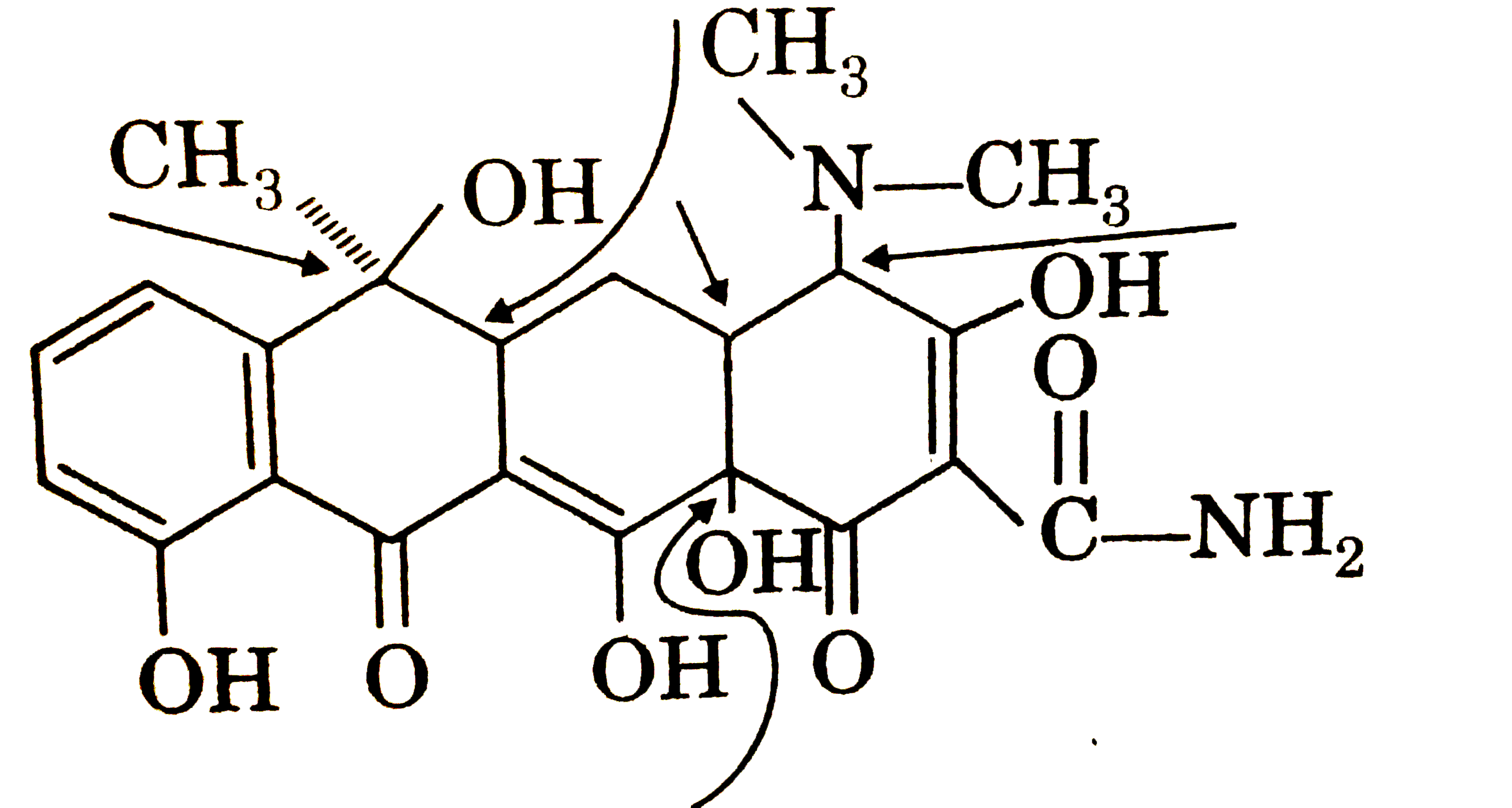
|
|
| 33. |
Tetra halide of group 14 elements possess ...... shape. |
| Answer» Answer :D | |
| 34. |
Terylene is formed by the reaction of one of the following alcohols |
|
Answer» 2-Chloroethanol |
|
| 35. |
Tertiary butyl bromide reacts with aqueous KOH follows ……………….. . |
| Answer» SOLUTION :`S_(N^2)` | |
| 36. |
tert-Butyl chloride on treatment with LiAlH_4 gives 2-methylpropene but with NaBH_4 it gives 2-methylpropane . Why so ? Explain. |
Answer» Solution :Al-H bond is more polar (EN DIFFERENCE =2.1-1.5=0.6) than almost non-polar B-H bond (EN difference =2.1-2.0=0.1) .As a result , due to weak Al-H bond, `[AlH_4]^-` breaks to give `H^-` ION which ACTS a strong BASE and hence brings about dehydrohalogenation of `3^@` alkyl halides to form alkenes  In contrast, due to strong B-H bond, `[BH_4]^-` acts only as a nucleophile and brings about a SUBSTITUTION reaction. 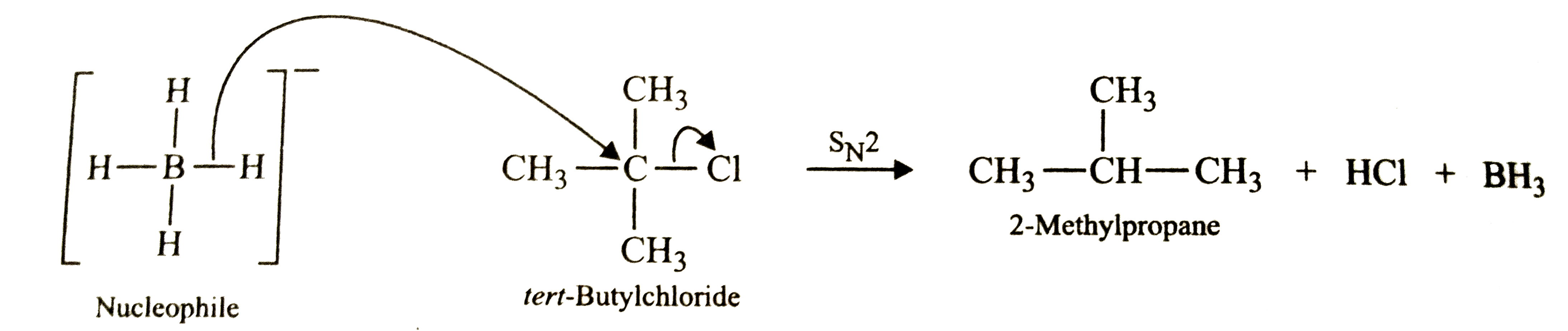
|
|
| 37. |
Tert-Butyl bromideis treated withsodiummetalin dryether. |
|
Answer» Solution :Tertiary alkyl halides donot undergoWurtzreactionbu insteadundergoderhydrohalogeN/Ationto yield alkenes . THUS. `CH_(3) -overset(CH_(3))overset(|)underset(CH_(3))underset(|)(C)-Broverset(" N/A,ether ")underset(Delta)(to) underset(("ISOBUTYLENE"))underset(2-"Methylpropene")(CH_(3)-overset(CH_(3))overset(|)(C)=CH_(3))+ HBR` |
|
| 38. |
Terrestial hydrogen contains 0.0156% of deuterium mostly in the form of |
|
Answer» HD |
|
| 39. |
Terminal alkynes hafvfe a acidic hydrogn/s. sodium salt of terminal alkynes behave as nucleophile as well as strong base. For primary alkyl halides it well as strong base. For primary alkyl halides it behave as nucleophile. Thus primary alkyl halides give SN reaction with its salt. Alkynes undergo give SN reaction with its salt. Alkynes undergo electrophilic as well as nucleophilic addition reaction. they also undergo hydroboration, oxidation and ozonolysis. Q. Compound , Ozonolysis products (A) Acetylene , 1 HCOCH & CH_(3)CHO (B) Ethylene , (2) CH_(3)CHO (c) Benzene (3) One mole of (CHO)_(2) (d) 2-Butene , (4) 2 moles of (CHO)_(2) (5) CH_(2)O |
|
Answer» `{:(A,B,C,D),(3,2,5,4):}` |
|
| 40. |
Terminal alkynes hafvfe a acidic hydrogn/s. sodium salt of terminal alkynes behave as nucleophile as well as strong base. For primary alkyl halides it well as strong base. For primary alkyl halides it behave as nucleophile. Thus primary alkyl halides give SN reaction with its salt. Alkynes undergo give SN reaction with its salt. Alkynes undergo electrophilic as well as nucleophilic addition reaction. they also undergo hydroboration, oxidation and ozonolysis. Q. |
|
Answer» <P>`{:(A,B,C,D),(PT,P,QS,RS):}` |
|
| 41. |
Terminal alkynes hafvfe a acidic hydrogn/s. sodium salt of terminal alkynes behave as nucleophile as well as strong base. For primary alkyl halides it well as strong base. For primary alkyl halides it behave as nucleophile. Thus primary alkyl halides give SN reaction with its salt. Alkynes undergo give SN reaction with its salt. Alkynes undergo electrophilic as well as nucleophilic addition reaction. they also undergo hydroboration, oxidation and ozonolysis. Q. The correct match is |
|
Answer» `{:(A,B,C,D),(3,1,5,2):}` |
|
| 42. |
Terminal alkyne is very weak acid, it forms salt with very strong base such as NaNH_(2) and sodium metal. R-C-=C-Hunderset(NH_(3)(l))overset(NaNH_(2))toR-C-=overset(ө)(C)overset(o+)(N)a+(1)/(2)H_(2) Sodium salt of alkyne is known as shown alkynide. sodium alkynide is hydrolysed with water because it is salt. Sodium salt behaves as nucleophile as well as strong base. for P-alkyl halides it behaves as a cucleophile. thus primary alkylhalides gives SN reaction halids it behaves as strong base hence they undergo elimination reaction. Q. CH_(3)-overset(Br)overset(|)(C)H-underset(Br)underset(|)(C)H_(2)underset((ii)NaNH_(3)Delta)overset((i)C_(2)H_(3)ONa,Delta)toVunderset((ii)H_(2)O)overset((i).CO_(2))toW is |
|
Answer» `CH_(3)-CH_(2)CH_(2)COOH` |
|
| 43. |
Terminal alkyne is very weak acid, it forms salt with very strong base such as NaNH_(2) and sodium metal. R-C-=C-Hunderset(NH_(3)(l))overset(NaNH_(2))toR-C-=overset(ө)(C)overset(o+)(N)a+(1)/(2)H_(2) Sodium salt of alkyne is known as shown alkynide. sodium alkynide is hydrolysed with water because it is salt. Sodium salt behaves as nucleophile as well as strong base. for P-alkyl halides it behaves as a cucleophile. thus primary alkylhalides gives SN reaction halids it behaves as strong base hence they undergo elimination reaction. Q. CH_(3)-overset(Br)overset(|)(C)H-CH_(3)overset(R-C-=overline(C)overset(+)(N)a)toS+T+Br^(-) S and T are |
|
Answer» `CH_(3)-CH_(2)-CH_(3),R-C-=CH` |
|
| 44. |
Terminal alkynes hafvfe a acidic hydrogn/s. sodium salt of terminal alkynes behave as nucleophile as well as strong base. For primary alkyl halides it well as strong base. For primary alkyl halides it behave as nucleophile. Thus primary alkyl halides give SN reaction with its salt. Alkynes undergo give SN reaction with its salt. Alkynes undergo electrophilic as well as nucleophilic addition reaction. they also undergo hydroboration, oxidation and ozonolysis. Q. CH_(3)C-=CH+HOX(2equ.)toY. Y is |
|
Answer» `CH_(3)COCHX_(2)` |
|
| 45. |
Terminal alkynes hafvfe a acidic hydrogn/s. sodium salt of terminal alkynes behave as nucleophile as well as strong base. For primary alkyl halides it well as strong base. For primary alkyl halides it behave as nucleophile. Thus primary alkyl halides give SN reaction with its salt. Alkynes undergo give SN reaction with its salt. Alkynes undergo electrophilic as well as nucleophilic addition reaction. they also undergo hydroboration, oxidation and ozonolysis. Q. When 1-pentyne is treated with dil. H_(2)SO_(4) and HgSO_(4) the product formed is |
|
Answer» 1-pentanol |
|
| 46. |
Terminal alkyne is very weak acid, it forms salt with very strong base such as NaNH_(2) and sodium metal. R-C-=C-Hunderset(NH_(3)(l))overset(NaNH_(2))toR-C-=overset(ө)(C)overset(o+)(N)a+(1)/(2)H_(2) Sodium salt of alkyne is known as shown alkynide. sodium alkynide is hydrolysed with water because it is salt. Sodium salt behaves as nucleophile as well as strong base. for P-alkyl halides it behaves as a cucleophile. thus primary alkylhalides gives SN reaction halids it behaves as strong base hence they undergo elimination reaction. Q. R-CH_(2)-C=ClIunderset(NH_(3)(l))overset(Na)toPoverset(CH_(3)-CH_(2)Br)toR. R is |
|
Answer» `R-C-=C-CH_(2)-CH_(2)-CH_(3)` |
|
| 47. |
Term catalyst was given by |
|
Answer» Rutherford |
|
| 48. |
Term 'Transuranic' means, elements with atomic number |
|
Answer» `gt 92` |
|
| 49. |
ter-Butyl benzene can be obtained by |
|
Answer»
|
|
| 50. |
Tendency to form pentahalides decreases down the group 15 of the periodic table. Account for this observation. |
|
Answer» <P> Solution :Due to basence of d-orbitals, N does not form a pentahlide but FORMS olny a TRIHALIDE due to the presence of three half-filled 2p-orbitals. All the remaining elements (i.e., P, As, Sb, Bi, howere form both tri-and pentahlides due to presence of three p-orbitals as well as vacant d-orbital in the valence shell). But due to inert pair efferct, tendency of As. Sb and Bi to form pentahalides decreases while that of trihalides incerases. In other words, the tendency to form pentahlides decrases down the GROUP, i.e., `PX_(5)gtAsX_(5)gtSbX_(5)gtBiX_(5)`. Further, the stability of pentahalides of any particular element decreases as the size of the hologen (X) increases from F to I. Thus, the only stable pentahalide of Bi is `BiF_(5)`. |
|