Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The average K.E. of an ideal gas per molecule in SI units at 25^@ C is |
|
Answer» `6.17 XX 10^(-21) KJ ` |
|
| 2. |
The average energy of London forces is |
|
Answer» 1 to 2 kJ/mole |
|
| 3. |
The average concentration of SO_(2) in the atmosphere over a city on a certain day is 10 ppm when the average temperature is 298 K. Given that the solubility of SO_(2) in water at 20=98 K is 1.3653 moles "litre"^(-1) andthe pK_(a)of H_(2)SO_(3) is 1.92, estimate the pH of rain on that day. |
|
Answer» Solution :`SO_(2)+H_(2)O rarr H_(2)SO_(3)` As rain water is falling from a great height, each drop of rain water will get saturated with `SO_(2)` before it reaches the earth. Hence, `[H_(2)SO_(3)]=[SO_(2)]=1.3653` moles `"litre"^(-1)` `H_(2)SO_(3) hArr 2H^(+) + SO_(3)^(2-)` SUPPOSE at EQUILIBRIUM, `[H^(+)] = X "mol" L^(-1)` Then `[H_(2)SO_(3)] = (1.3653-(x)/(2)), [SO_(3)^(2-)]=(x)/(2) "mol" L^(-1)` `K_(a) = ([H^(+)]^(2) [ SO_(3)^(2-)])/([H_(2)SO_(3)]) = (x^(2) xx x//2)/((1.3653-x//2))=10^(-1.92)` (`:' pK_(a) = 1.92` means `- LOG K_(a) = 10^(-1.92) or log K_(a) = 1.92 or K_(a) = 10^(-1.92)`) Neglecting x/2 in comparison to 1.3653, we get `(x^(3))/(2xx1.3653)=10^(-1.92)` or `x^(3) = 2.7306xx10^(-1.92)` or ` 3 log x = log 2.7306 - 1.92 = 0 . 4348 - 1.92= - 1.4852 or - log x = 0.485, i.e., pH = 0.485` |
|
| 4. |
The average concentration of Na^(+) ion in human body serum is 3 to 4 gm per litre. The molarity of Na^(+) ion is about |
|
Answer» `0.15M` IMPLIES avg `M=(("3 to 4"))/(23xx1)=0.13M-0.17M` |
|
| 5. |
The average composition of portland cement is |
|
Answer» `CaO:40-50%,SiO_(2):30-40%,Al_(2)O_(3),Fe_(2)O_(3):10-20%` |
|
| 6. |
The attraction that non-polar molecules have for each other is primarily caused by |
|
Answer» Van der WAAL's FORCES |
|
| 7. |
The aufbau principle implies that a new electron will enter an orbital for which |
|
Answer» (N + L) is minimum |
|
| 8. |
The attractive force which holds atoms together in a molecule is called a chemical bond.Explain the formation of a H_2 molecule on the basis of the valence bond theory (VBT) |
| Answer» Solution :Each hydrogen atom CONTAINS one electron in the 1s ORBIT.1s atomic orbital of FIRST hydrogen atom overlap axially with 1s atomic orbit of second hydrogen atom to from a sigma bond. | |
| 9. |
The attacking species in the sulphonation of benzene is SO_3 . |
|
Answer» |
|
| 10. |
The atoms remain indestructible in a chemical reaction. |
|
Answer» |
|
| 11. |
The atoms of 0.004 g magnesium are..... |
|
Answer» `2XX2^(20)` |
|
| 12. |
The atoms/ion listed in correct order of increasing size are |
|
Answer» Na, Si, H |
|
| 13. |
The atomic weights of two elements (A and B) are 20 and 40 respectively. If x % of A contains y atoms, how many atoms are present in 2x g of B? |
|
Answer» Solution :Number of MOLES of `A = X/20` Number of atoms of `A = x/20 xx N` (N is the Av. Const) `therefore y=x/20 xx N , x = (20y)/N` Further, number of moles of B`=(2x)/40 xx N` `=(2N)/40 xx (20 y)/N` =y |
|
| 14. |
The atomic weight of two isotopes of boron are 10.01 and 11.01 . If the atomic weight of natural boron is 10.81, what is the percentage of heavier isotope of boron in nature? |
|
Answer» |
|
| 15. |
The atomic weight of a metal (M) is 27 and its equivalent weight is 9, the formula of its chlroide will be |
|
Answer» MCL |
|
| 16. |
The atomic spectrum of hydrogen is found to contain a series of lines of wavelengths 656.46, 486.27, 434.17 and 410.29 nm. The wavelength (in nm) of the next line in the series will be.... |
|
Answer» `(1)/(lamda) = R_(H) ((1)/(2^(2)) - (1)/(n_(2)^(2)))` `(1)/(410.29 xx 10^(-7) cm) = 109,67 cm^(-1) ((1)/(4) - (1)/(n_(2)^(2)))` This on solving gives `n_(2) = 6` Thus, the NEXT LINE will be OBTAINED for jump from `n_(2) = 7 " to " n_(1) = 2`, so that `(1)/(lamda) = 109,677 cm^(-1) ((1)/(2^(2)) - (1)/(7^(2)))` `= 109,677 xx ((1)/(4) - (1)/(49)) cm^(-1)` `= 109,677 xx (45)/(196) cm^(-1) = 25180.9 cm^(-1)` or `lamda = (1)/(25180.9 cm^(-1)) = 397.1 xx 10^(-7) cm` `= 397.13 nm` |
|
| 17. |
The atomic radii in case of inert gases is |
|
Answer» IONIC RADII |
|
| 18. |
The atomic orbitals involved in hybridisation of SF_(6) molecule is |
|
Answer» `3s, 3_(PX), 3_(PY), 3_(pz), 3d_(z)` |
|
| 19. |
The atomic numbers of vanadium (V), Chromium (Cr), maganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26. Which one of these may be expected to have the highest second ionization enthalpy? |
| Answer» ANSWER :A | |
| 20. |
The atomic numbers of elements A, B, C and D are Z1, Z, Z + 1 and 2 + 2 respectively. If 'B' is a noble gas, choose the correct answers from the following statements Q (1) 'A' has higher electron affinity.Q (2) 'C exists in + 2 oxidation state.Q (3) 'D' is an alkaline earth metal. |
|
Answer» (a) and (B) |
|
| 21. |
The atomic nụmber of the element with the maximum number of unpaired 2p electrons in the ground state is |
|
Answer» 1 |
|
| 22. |
The atomic number of the element which is not included in the main body of the period table |
|
Answer» 43 |
|
| 23. |
The atomic number of Rb is less than that of |
|
Answer» Na |
|
| 24. |
the atomicnumber ofcerium( Ce )is 58 . Thecorrectelectronicconfigurationof Ce^(3+) ion is |
|
Answer» `[Xe] 4F^(1)` E.C.Of Ce (Z= 58) = [Xe] 4f^(1)5d^(1)6s^(2)` `:. ` E.C.Of `Ce^(3+) = [Xe]4f^(1)` Thusoption( a) iscorrect . |
|
| 25. |
The atomic number of an element which shows the oxidation state of + 3 is |
|
Answer» 13 |
|
| 26. |
The atomic number of an element 'M' is 26. How many electrons are present in the M-shell of the element in its M^(3+) state ? |
|
Answer» 11 |
|
| 27. |
Theatomicnumberof an elementis 35. What isthe totalnumberof electrons presentin all thep -orbitalsof the groundstateatom of thatelement |
|
Answer» 6 |
|
| 28. |
The atomic number of an element is 58. it blongs to |
|
Answer» `6^(th)` PERIOD , III A GROUP |
|
| 29. |
The atomic number of an element is 35. what is the total number of electrons present in all the p-orbitals of the ground state atom of that element ? |
|
Answer» 6 |
|
| 30. |
The atomic number at which filling of a g-orbital is likely to begin is : |
|
Answer» 121 |
|
| 33. |
The atomic masses of 'He' and 'Ne' are 4 and 20 a.m.u respectively. The value of the de Broglie wavelength of 'He' gas at -73 .^(@)C is ''M'' times that of the de Broglie wavelength of 'Ne' at 727 .^(@)C. 'M' is |
|
Answer» `:. (lamda_(He))/(lamda_(Ne)) = (m_(He) XX v_(He))/(m_(He) xx v_(He)) = (M_(Ne) xx v_(Ne))/(M_(He) xx v_(He))` As `V = sqrt((3RT)/(M)) , (v_(Ne))/(v_(He)) = sqrt((T_(Ne))/(M_(Ne)) xx (M_(He))/(T_(He)))` `:. (lamda_(He))/(lamda_(Ne)) = (M_(Ne))/(M_(He)) xx sqrt((T_(Ne))/(T_(He)) xx (M_(He))/(M_(Ne)))` `= sqrt((M_(Ne) T_(Ne))/(M_(He) T_(He))) = sqrt((20 xx 1000)/(4xx 200)) = 5` `:. lamda_(He) = 5 lamda_(Ne)` |
|
| 34. |
The atomic masses of two elements A and B are 20 and 40 respectively. If x gm of A contains Y atoms, how many atoms are present in 2x gm of B |
|
Answer» 2y |
|
| 35. |
State whether the given statement is true or false: The atomic mass unit (amu) is defined as one-twelfth of the actual mass of an atom of C^12. |
|
Answer» |
|
| 36. |
The atomic mass of one atom of C^(12)= .......... g. |
|
Answer» `1.992648 XX 10^(-23)` |
|
| 37. |
The atomic mass of an element is double its atomic number. If there are four electrons in the 2p-orbital, the draw the model of the atom showing the arrangement of protons, neutrons and electrons. Give its valence and name the element. |
|
Answer» |
|
| 38. |
The atomic mass of an element is 19. The second shell of its atom contains 7 electrons. The number of neutrons in its nucleus is 2x. The value of x is ______. |
|
Answer» |
|
| 39. |
The atomic mass of a metal M is 56. Calculate the empirical formula of its oxide containing 70 % metal. |
|
Answer» `{:("Element","Percentage","Atomic mass","GRAM ATOMS (Moles)","Atomic RATIO (Molar ratio)","Simplest whole no. ratio"),("M",70.0,56,(70.0)/(56)=1.25,(1.25)/(1.25)=1.0,2),("O",30.0,16,(30.0)/(16)=1.88,(1.88)/(1.25)=1.5,3):}` Empirical formula of METAL oxide `= M_(2)O_(3)`. |
|
| 40. |
The atomic mass is with respect to mass of .......... |
|
Answer» `C^(12)` |
|
| 41. |
Why do ionic radii of alkali metals increase on moving down the group ? |
| Answer» SOLUTION :INCREASE | |
| 42. |
The atomic and ionic radii (M^(3+) ions) of Lanthanide elements decrease with increase in atomic number. This effect is called |
|
Answer» LANTHANOID contraction |
|
| 43. |
The atomic and inoic radii and alkaline earth metals are _______ than the corresponding members of the alkali metals |
|
Answer» larger |
|
| 44. |
The atom without neutron is |
|
Answer» H |
|
| 45. |
The atom of the element having atomic number 14 should have |
|
Answer» One UNPAIRED electron 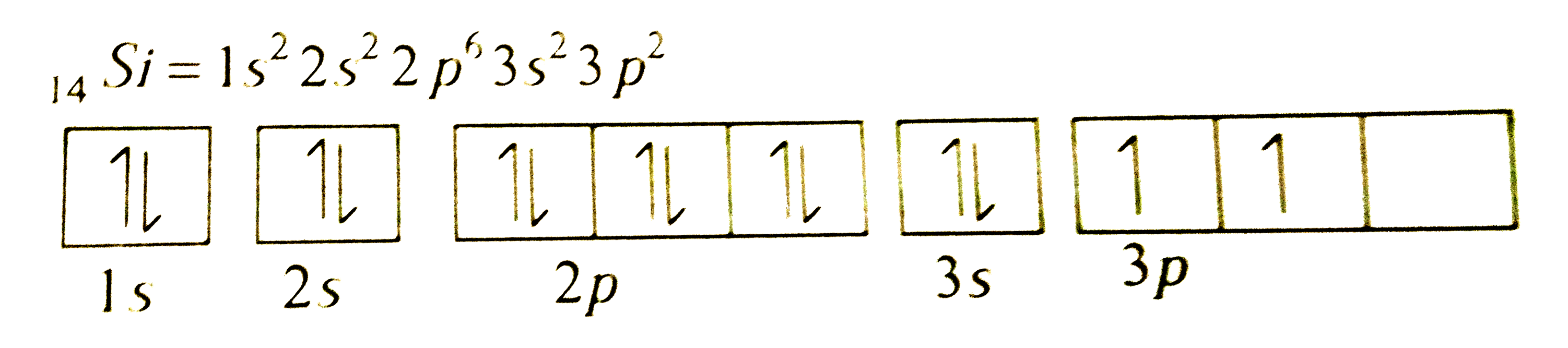
|
|
| 46. |
Theatom ofsmallestatomicradiusamong thefollowingis |
|
Answer» Na |
|
| 47. |
The atmospheric oxidation of NO 2NO (g) + O_(2) (g) hArr 2 NO_(2) (g) was studied with initial pressure of 1 atm of NO and 1 atm of O_2 . At equilibrium , partial pressure of oxygen is 0.52 atm . Calculate K_(p) of the reaction. |
Answer» Solution : As , `1 - x = 0.52` `x = 0.48` = At EQUILIBRIUM , `P_(NO) = 1 - 2x = 1 - 2(0.48) = 0.04` `P_(NO_(2)) = 2x= 2 (0.48) = 0.96` `K_(eq) = (P_(NO_(2))^(2))/(P_(NO)^(2) * P_(O_(2))) = (0.96 XX 0.96)/(0.04 xx 0.04 xx 0.52) = 11.07 xx 10^(2) (ATM)^(-1)` |
|
| 48. |
The atmospheric pressure on the summit of Mt. Everest is 0.333 atmospheres. At what tempperature ("in "^(@)"C") does H_(2)O boil there? (DeltaH_(vap)H_(2)O=40.7 kJxxmol^(-1)) |
| Answer» Answer :A | |
| 49. |
The atmospheric gas which cannot produce green house effect is |
|
Answer» `N_(2)` |
|
| 50. |
The associate wavelength of a particle with mass 1 gram and velocity 100 m/s is ......... |
|
Answer» `6.63 xx10^(-35)` m m=1 GRAM =`1.0 xx10A^(-23)` kg wavelength =`lambda=(?)` `h=6.262 xx10(-34)` JS `lambda=(h )/(p)=(h)/(MV)=(6.626xx10A^(-34) js)/(1.0xx10A^(-3)kg xx100 ms^(-1))` `=6.626xx10^(-33)` m |
|