Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The increasingorder of atomicradii of the followinggroup 13 element is |
|
Answer» `Al lt GA lt In lt Tl` |
|
| 2. |
The increasing order of basicity of the following compounds is : |
|
Answer» `(B) LT(a) lt (d) lt (C)` |
|
| 3. |
The increasing order of acidic nature among ZnO, Na_(2) O_(2) , P_(2) O_(5) and MgO is |
|
Answer» <P>`ZnO LT P_(2) O_(5) lt Na_2 O_(2) lt MgO` |
|
| 4. |
The increasing basicity order of the following compounds is : (A) " " CH_(3)CH_(2)NH_(2)""(B) {:(""CH_(2)CH_(3)),(""|),(CH_(3)CH_(2)NH):} ""(C) H_(3)C-overset(CH_(3))overset(|)N-CH_(3)""(D) Ph-overset(CH_(3))overset(|)N-H |
|
Answer» `(A) LT (B) lt (C) lt (D)` |
|
| 5. |
The increase in the temperature of the aqueous solution placed in a closed vassel will result in its |
|
Answer» MOLALITY to increase |
|
| 6. |
The incorrectly metched pair among the following is : |
|
Answer» `{:(" MoleculeShape "),(BrF_(5)"Trigonal bipyramidal "):}` |
|
| 7. |
The incorrect statements (S) is/are |
|
Answer» `BeCl_(2)` is an IONIC compound |
|
| 8. |
The incorrect statement regarding the formation of ionic bond |
|
Answer» It INVOLVES electrostatic attraction |
|
| 9. |
The INCORRECT statement is |
|
Answer» LITHIUM is least reactive with water AMONG the alkali metals |
|
| 10. |
The incorrect statement is : |
|
Answer» Lithium is least REACTIVE with WATER AMONG the alkali metals. `4LiNO_3 overset(Delta) to 2Li_2O + 4NO_2 + O_2 ` |
|
| 11. |
The incorrect statement in respect of chromyl chloride test is |
|
Answer» formation of red vapour |
|
| 12. |
The incorrect statement are |
|
Answer» `CH_3 COOH ` is a weak acid |
|
| 13. |
Select the incorrect statement |
|
Answer» `CH_3 COOH ` is a weak acid |
|
| 14. |
The incorrect statement among the following is |
|
Answer» The first ionisation potential of AL is less than the first ionisation potential of Mg |
|
| 15. |
The incorrect statement among the following is : |
|
Answer» CRYSTALLINE boron is obtained by the reduction of boric anhydride with AL powder |
|
| 16. |
The incorrect statement among the following are |
|
Answer» `AlCl_(3)` exists as `Al_(2)Cl_(6)` in vapour state |
|
| 17. |
The incorrect statement according to second law of themodynamics is |
|
Answer» HEAT cannot flow from colder body to a hotter body of its own |
|
| 18. |
The incorrect statement about step-1 is : |
|
Answer» It is `S_N2` REACTION |
|
| 19. |
The incorrect relations (s) about the velocity of photon is (are) |
|
Answer» Dependent on its WAVELENGTH |
|
| 20. |
The incorrect order of second ionization energies in the following is |
| Answer» Answer :A | |
| 21. |
The incorrect option regarding the chemical rection is |
|
Answer» Process is called Lane's process |
|
| 22. |
The incorrect match is |
|
Answer»
|
|
| 23. |
The incorrect IUPAC name is |
|
Answer» `{:(""O),("||"),(CH_(3)-C-CH-CH_(3):" 2-Methyl-3-butanone"),("|"),(""CH_(3)):}` |
|
| 24. |
The incorrect expression among the following is |
|
Answer» in isothermal process, `W_("reversible") = - nRT"In (V_(f))/(V_(i))` `DeltaG^(@) = DeltaH^(@) -TDeltaS^(@)` `RT In K= DeltaH^(@) -TDeltaS^(@)` `therefore In K = -(DeltaH^(@) -TDeltaS)/(RT)` |
|
| 25. |
The incorrect expression among the following is : |
|
Answer» `K= e^(- DeltaG^(@) //RT)` or `LNK = - (DeltaG^(@))/( RT) - ( DeltaH^(@) - T DeltaS^(@))/( RT)` Hence, (d) is incorrect. |
|
| 26. |
The inceasing order of the acidity of the following carboxylic acids is : |
|
Answer» `I LT III ltII lt IV` |
|
| 27. |
The inceasing order of reactivity among group 1 elements is Li lt Na lt K lt Rb lt CS whereas that among group 17 is F gt Cl gt Br gt I. Explain . |
| Answer» Solution :GROUP I elements are electropositive in which IE DECRASES down the group.Therefore reactivity increases down the group. Group 17 elements are ELECTRONEGATIVE which show strong electron accepting capacity. This property depends on their elctrode potentials which very as `F gt CL gt Br gt I`. So the reactivity follows the same order. | |
| 28. |
The impossible set of quantum number is |
|
Answer» n=2, l=0, m=0, s = +1/2 |
|
| 29. |
The impossible set of quantum numbers is |
|
Answer» N = 2 , L= 0 , m =0 , s =+1/2 |
|
| 30. |
The important oxide ore of iron is |
|
Answer» Sidherite |
|
| 31. |
The III A group element that does not displace hydrogen from hydrochloric acid is |
|
Answer» B `therefore` Boron does not displace HYDROGEN from HCl. |
|
| 32. |
The IIIA group element that does not displace hydrogen from hydrochloric acid is |
|
Answer» Solution :Boron REMAINS uneffected by non-oxidising ACIDS like HCI `:.` Boron does not dispalce hydrogen from НСІ |
|
| 33. |
The II A carbonate that decomposes at room temperature of 25^(@) C is |
|
Answer» `CaCO_(3)` |
|
| 34. |
The ignition mixture used in alumino-thermy is a mixture of |
|
Answer» Mg powder `+Na_(2)O_(2)` |
|
| 38. |
The hyperconjugative stabilities of tert-butyl cation and 2-butene, respectively, are due to |
|
Answer» `SIGMA-p` (empty) and `sigma-pi^(**)` electron delocalisations 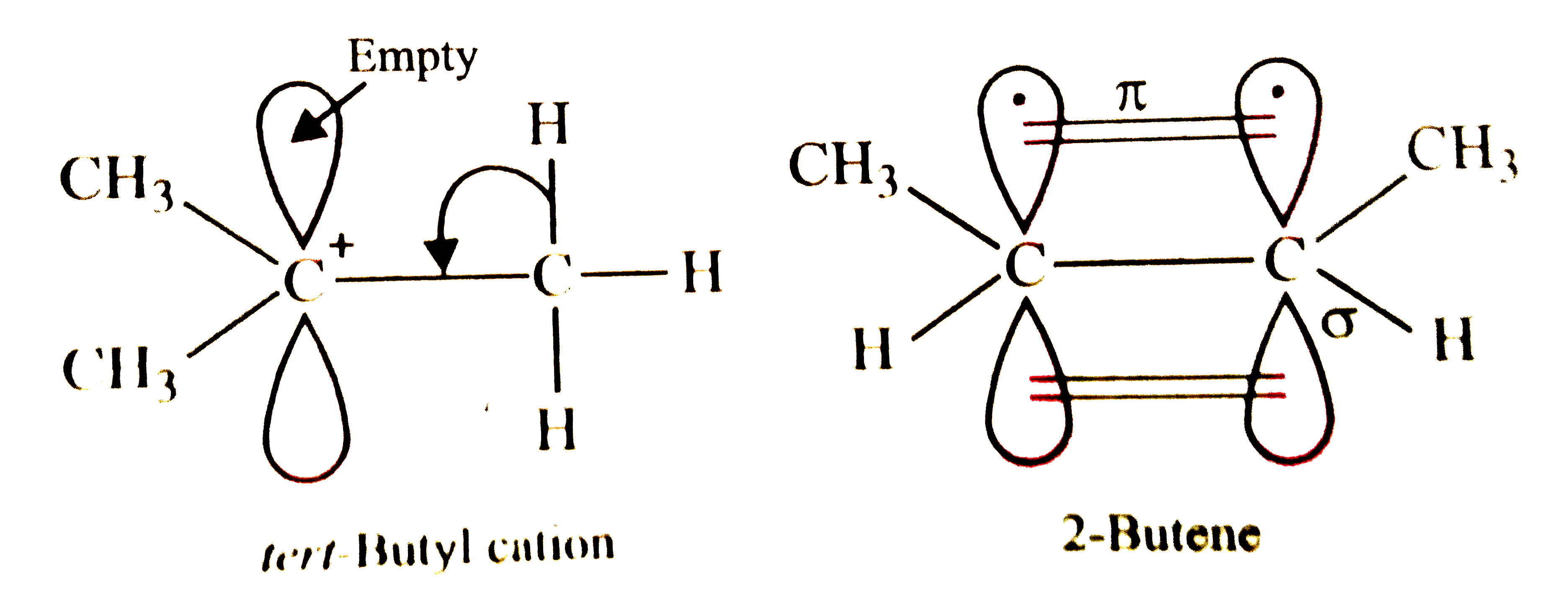 In 2-butene, `pi`-bond contains two electrons, one electron from each of the two carbon atoms, i.e., `pi`-MO contains a PAIR of electrons, but `pi^(**)` MO is empty and hence electrons of the `sigma` C-H bond can move only into the `pi^(**)` orbital. Thus, option (a) is correct. |
|
| 39. |
The hyparconjugative stabilities of tert-butyl cation and 2-butene, respectively ,are due to |
|
Answer» <P>`sigma to p("sigma") ("empty ") and sigma to pi^("*")`ELECTRON delocalization. |
|
| 40. |
The hydroxides and carbonates of sodium and potassium are easily soluble in water while the corresponding salts of magnesium and calcium are sparingly soluble in water . Explain . |
| Answer» Solution :Due to larger size of Na and K as compared to that of Mg and Ca , the lattice ENTHALPIES of hydroxides and CARBONATES of sodium and potassium are MUCH lower than those of the hydroxides and carbonates of magnesium and calcium . As a RESULT , the hydroxides and carbonates of Na and K are EASILY soluble in water while the corresponding salts of magnesium and calcium are sparingly soluble in water . | |
| 41. |
The hydroxides and carbonates of sodium and potassium are easily soluble in water while the corresponding salts of magnesium and calcium are sparingly soluble in water. Explain. |
| Answer» Solution :SODIUM and potassium ions are larger in size than magnesium and calcium ions respectively. DUE to larger size, the lattice energies of hydroxides and CARBONATES of sodium and potassium are much lower than those of magnesium and calcium. This is why the hydroxides and carbonates of sodium and potassium are easily soluble in water, whereas the CORRESPONDING salts of magnesium and calcium are sparingly soluble in water. | |
| 42. |
NaOH, KOH, Na_2CO_3, K_2CO_3 are easily soluble in water while the corresponding salts of magnesium and calcium are sparingly soluble in water. Explain |
| Answer» Solution :Alkali metals form MONOVALENT cations (such as `Na^(+), K^(+)`) while alkaline earth metals form DIVALENT cations (such as `Mg^(2+), Ca^(2+)`). Due to increase in charge of the cations, the LATTICE energy of the corresponding salt increases. For this reason, hydroxides and carbonates of sodium and potassium have LOWER lattice enthalpy values than the hydroxides and carbonates of magnesium and calcium. As hydration enthalpies of the hydroxides and carbonates of sodium and potassium are greater than their lattice enthalpies, these salts readily dissolve in water. HOWEVER, in case of the hydroxides and carbonates of calcium and magnesium, the lattice enthalpy values is greater than that of hydration enthalpy and consequently these salts are less soluble in water. | |
| 43. |
The hydroxides and carbonates of sodium and potassium are easily solub while the corresponding salts of magnesium and calcium are sparingly soluble in waterExplain |
| Answer» Solution :Since group 1 hydroxides and CARBONATES DUE to large size CONTAIN higher hydration en the lattice energy so, they are easily soluble in water. Whereas, in magnesium and calci to small size their lattice energy dominates over hydration energy they are sparingly sol water. | |
| 44. |
The hydroxides and carbonates of Na and K are solube in the water while those of Mg and Ca are spairingly soluble .Explain. |
| Answer» SOLUTION :Lattice energies of the hydroxides and CARBONATES of Na and K are much smaller than those compounds of and MG.(This is due to the LARGER size of Na and K compared to Mg and Ca.) | |
| 45. |
The hydroxide which sublime on heating are i)LiOH ii)KOH iii)RbOHiv) mg(OH)_(2) |
|
Answer» All undergoes sublmation |
|
| 46. |
Give reasons for the following. Sodium and potassium are stored in kerosene. |
| Answer» SOLUTION :Since group 1 hydroxides and carbonates due to LARGE size contain HIGHER hydration EN the lattice energy so, they are easily soluble in water. Whereas, in magnesium and calci to small size their lattice energy dominates over hydration energy they are SPARINGLY sol water. | |
| 47. |
The hydroxide of which metal ion(s) is/are soluble in excess of NaOH solution |
|
Answer» `Al^(3+)` `Zn+2NaOHrarrNa_(2)Zn_(2)_+H_(2)` |
|
| 48. |
The hydrolysis reaction of ethyl acetate CH_3COOC_2H_(5(l)) + H_2O_((l)) hArr CH_3COOH_((l)) + C_2H_5OH_((l)) Is this reaction get equilibrium in open vessel ? |
| Answer» SOLUTION :YES, because the GAS is not in this. | |
| 49. |
The hydrolysis of NCl_(3)" by "H_(2)O produces |
|
Answer» `NH_(2)OHandHOCl` |
|
| 50. |
The hydrolysis constant ofNaX (K_a of HX is 2xx 10 ^(-6))is |
| Answer» Solution :` K_h =(K_W)/(K_a)=(10 ^(-14))/(2 xx 10 ^(-6))= 5 xx 10 ^(-9) ` | |



