Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number of sigma-and pi-bonds present in 1, 3-butadiene are respectively |
|
Answer» 9 and 2 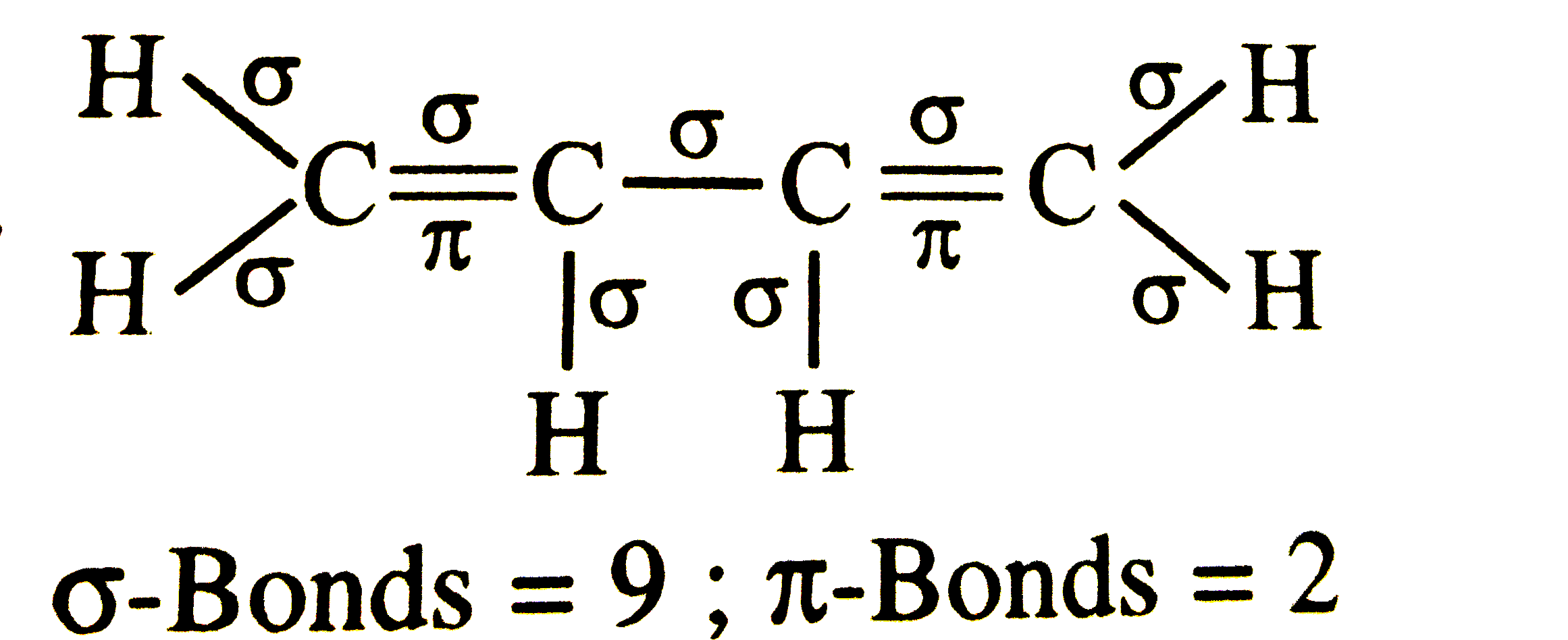 `sigma`-BONDS = 9, `PI`-Bonds = 2 |
|
| 2. |
The number of sigma and pi bonds present in cis-but-2-ene is …… . |
|
Answer» `9 sigma` and `1pi` |
|
| 3. |
The number of sigma and pi bonds in phenol respectively are …… |
|
Answer» 12,3 |
|
| 4. |
The number ofsigma and pi bonds inallyl isocyanide are |
|
Answer» `9 SIGMA, 3pi` |
|
| 5. |
The number of sigma and pi-bonds in 1-butene 3-yne are |
|
Answer» 5 SIGMA and 5 pi |
|
| 7. |
The number of resonance structures for anilinium ion is |
|
Answer» |
|
| 8. |
The number of reagetns among the following which cannot add to propene by free radical mechanism in presence of organic peroxides is (a). HCl (b). HBr (c) HI (d). CH_(3)SH BrC Cl_(3) |
|
Answer» |
|
| 9. |
The number of radial nodes in 3s and 2p respectively are |
| Answer» Solution :Radial nodes `= n - L -1` | |
| 11. |
The number of radial nodes for 3p orbital is... |
|
Answer» 3 |
|
| 12. |
The number of radial nodes for 3p orbital is ..... |
| Answer» Solution :No. of RADIAL nodes in 3P orbital `= n -l-1 =3 -1-1 = 1` | |
| 13. |
The number of protons, electrons and neutrons in ""_(35)Br^(80) are respectively |
|
Answer» 35, 35, 80 |
|
| 14. |
The number of protons, electrons and neutrons in a species are equal to 17,18 and 18 respectively. Which of the following will be the proper symbol of the species |
|
Answer» `""_17^32""CL` |
|
| 15. |
Thenumberof protonneutronandelectronrespectively . ……….. And ………? |
|
Answer» 1,3,1 |
|
| 16. |
The number of properties which are state function amongthe following is Pressure, Volume , Temperature,Heat, Work , Entropy, Enthalpy , Free energy, Internal energy,C_(p) andC_(v). |
|
Answer» |
|
| 17. |
The number of products (stereoisomers) formed in the following reaction is (consider only major product) |
Answer» 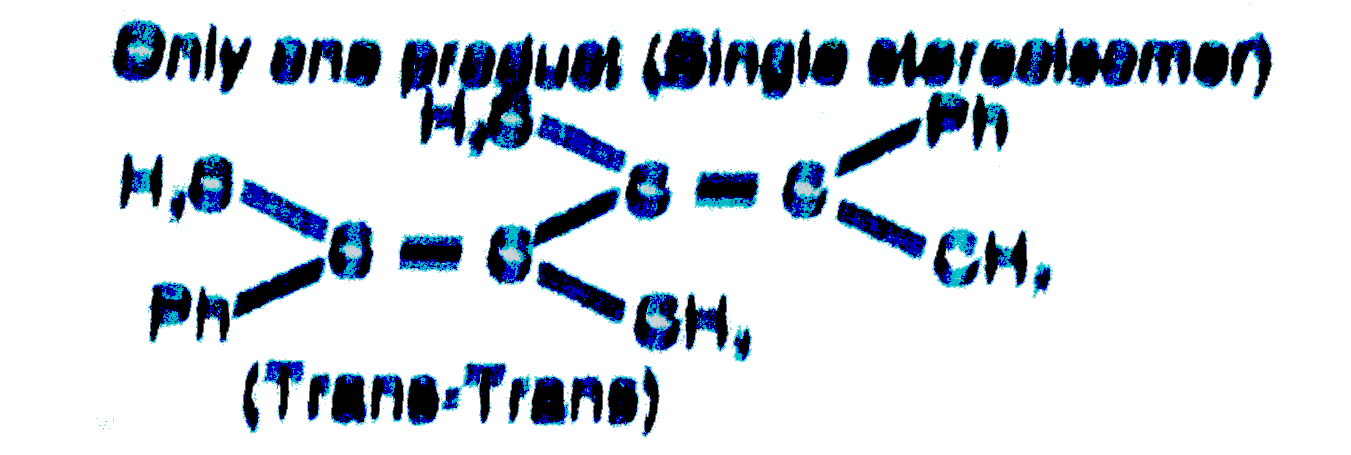
|
|
| 18. |
The number of primary, secondary, tertiary and quaternary carbons in neopentane are respectively |
|
Answer» 4,3,2 and 1 |
|
| 19. |
The number of primary, secondary and tertiary amines possoble with the molecular formula C_(3)H_(9)N respectively : |
|
Answer» 1,2,2 |
|
| 20. |
The number of primary, secondary and tertiary amines possible with the molecular formula C_(3)H_(9)N respectively. |
|
Answer» 1, 2, 2 `C-underset( C )underset(|)N-C""C-underset(NH_(2))underset(|)C-C` `3^(@)`-amines=1 |
|
| 21. |
The number of primary alcohol isomers with the formula C_(4)H_(10)O is |
|
Answer» 1 |
|
| 22. |
The number of possible theoretical conformations of ethane is |
|
Answer» Two |
|
| 23. |
The number of possible structural isomers f~r a compound with the molecular formula C_7H_16 is |
|
Answer» 8 |
|
| 24. |
The number of possible resonance forms for superoxide ion is |
|
Answer» 2 |
|
| 25. |
The number of possible monochloro derivatives of 2,2,3,3-tetramethylbutane is |
|
Answer» 2 |
|
| 26. |
The number of possible isomers of CH_(6)H_(12), is ________ |
|
Answer» 2 |
|
| 27. |
Number of isomers for the compound with the molecular formula C_2BrClFI is ----- |
|
Answer» 3 |
|
| 28. |
The number of possible isomeric structures for the formula C_(4)H_(10) is |
|
Answer» 10 |
|
| 29. |
The number of possible enantiomeric pairs formed bymono chlorination of 2-methylbutane is |
| Answer» Solution :Enantiomeric PAIRS. | |
| 30. |
The number of possible aromatic structure for C_(7)H_(8)O is…………. |
|
Answer» 4 
|
|
| 31. |
The number of pi molecular orbitals completely filled in the ground state of benzene is: |
|
Answer» three |
|
| 32. |
The number of pi bond present in given compound is/are…… CH_(3)-CH=CH-COOH |
|
Answer» 2 |
|
| 33. |
The number of pi and sigma-bonds in toluene is respectively |
|
Answer» `3` and `6` |
|
| 34. |
The number of solutions of sinx=x/10 is |
|
Answer» 10 hcx `therefore 10 ` = nhc`barv` or N `=1(10)/(hcv) = (10)/(hcx)` |
|
| 35. |
The number of peroxy rings in CrO_(5) is ___ |
|
Answer» 2 
|
|
| 36. |
The number of Pentagons and Hexagons, respectively in Co-Fullerence are |
| Answer» ANSWER :D | |
| 37. |
The number of paired electrons in oxygen atom is : |
|
Answer» 6 |
|
| 38. |
The number of p atomic orbitals involved in the formation of a benzene molecule |
|
Answer» <P> SOLUTION :SIX p ATOMIC ORBITALS |
|
| 39. |
The number of oxygen molecules present in 100 grams of limestone is |
|
Answer» `4N_(0)` |
|
| 40. |
The number of oxygen present in 11.2 L of ozone at N.T.P. are______ . |
|
Answer» `1.20xx10^(24)` `-=3xx3.01xx10^(23)" atoms of oxygen "` `-=9.03xx10^(23)" atoms of oxygen "` |
|
| 41. |
The number of oxygen atoms present in 50 g of calcium carbonate is |
|
Answer» `6.023xx10^(23)` |
|
| 42. |
The number of orbitals in the quantum level n = 4 is |
|
Answer» 4 |
|
| 44. |
The number of optically active products obtained form the complete ozonolysis of the given compoundis : |
|
Answer» 0 |
|
| 45. |
The number of optically active isomers possible for the compound CH_(3)-underset(OH)underset(|)(CH)-underset(OH)underset(|)(CH)-underset(OH)underset(|)(CH)-CH_(2)OH |
|
Answer» 3 `:.` The number of ISOMERS `= 2^(n) = 2^(3) = 8`. |
|
| 46. |
The numberof orbitals dependson whichquantumnumberand notdependson whichquantum number ? |
|
Answer» Solution :The numberof orbitalsdependson VALUEOF LAND ITSIS equalto no. of valueof `m_(1)` ITDOES not dependson nand `m_(s)` |
|
| 47. |
The number of of electrons required to balance the following equation NO_3^(-) +4H^(+) + e^(-) rarr 2H_2O +NO is_______ |
|
Answer» In this equation , all the ATOMS are balanced . To balance charge add `3E^(-)` to L.H.S., we have `NO_3^(-) + 4H^(+)+3e^(-) rarr 2H_(2)O+NO` |
|
| 48. |
The number of nodes possible in radial probability distribution curve of 3d orbital is |
|
Answer» 1 |
|
| 49. |
The number of octahedral sites per sphere in fcc structure is |
|
Answer» 8 |
|