Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number of waves make by a Bohr electron in an orbit of maximum magnetic quantum number +2 is . |
|
Answer» NUMBER of WAVES in a SHELL = n=3 . |
|
| 2. |
The number of waves in an orbit are |
|
Answer» `N^(2)` |
|
| 4. |
The number of water molecules present in a drop of water (volume 0.0018 ml) density = 1 gmL^-1 at room temperature is |
|
Answer» `1.084xx10^(18)` |
|
| 5. |
The number of water molecules present in a drop of water (volume = 0.0018mL) at room temperature is |
|
Answer» `1.084 xx 10^(18) `=(0.0018)/(18) = 1 xx 10^(-4)` [`therfore` 0.0018 mL = 0.0018g] Number of water MOLECULES `=1 xx 10^(-4) xx 6.023 x 10^(23)` `=6.023 xx 10i^(19)` |
|
| 6. |
The number of water molecules is maximum in : |
|
Answer» 18 GRAM of WATER `THEREFORE` 18 mole water `= 18xx6.02xx10^(23)` molecules so, 18 mole water has maximum number of molecules. |
|
| 7. |
The number of water molecules in one litre of water is: |
|
Answer» 18 |
|
| 8. |
The number of water molecules in a drop of water weighing 5 mg is |
|
Answer» `6.023 XX 10^(72)` |
|
| 9. |
The number of water molecules in a drop of water weighing 0.018 g is |
|
Answer» `6.022xx10^(26)` No. of moles of water in the drop=Mass of water/molar mass =0.018/18=`10^(-3)` mole No of water molecules present in 1 mole of water =`6.022xx10^(23)` No. water molecules in one drop of water (`10^(-3)` mole)=`6.022xx10^(23)xx10^(-3)` `=6.022xx10^(20)` |
|
| 10. |
The number of water molecules are associated with washing soda is X, then X-l= |
|
Answer» therefore X=10` and X-1 =9 |
|
| 11. |
The number of water molecule (s) directly bonded to the metal centre in CuSO_(4) .5 H_(2) O is |
|
Answer» molecular are directily BONDED to Cu ) |
|
| 12. |
The number of unpaired electrons in carbon atom in excited state is |
|
Answer» One `=1s^2 ,2s^1 , 2p_x^1 2p_y^1 2p_z^1` (Excited state) In excited state no. of UNPAIRED ELECTRON is 4 |
|
| 13. |
The number of unit cells present in 1 mole of NaCl crystal is |
|
Answer» `6.023 GT O^(-2) gt N^(-3)` |
|
| 14. |
The number of types of bonds between two carbon atoms in calcium carbide is |
|
Answer» ONE SIGMA , TWO pi |
|
| 15. |
The number of three centred, two electron bonds in diborane is |
|
Answer» 2 |
|
| 16. |
The number of tetrahedral voids per unit cell in NaCl crystal is ____ |
|
Answer» 4 |
|
| 17. |
The number of tetrahedral voids per unit cell in NaCl crystal is …….. |
|
Answer» 4 NO. of tetrahdral voids is DOUBLE the number of octahedral voids. Hence, tetrahedral voids = 4. |
|
| 18. |
The number of tetrahedral voids in the unit cell of a face- centred lattice of similar atoms is |
|
Answer» 4 |
|
| 19. |
The number of tetrahedral voids in the unit cell of a face centred cubic lattice of similar atoms is |
|
Answer» 4 |
|
| 20. |
The number of tetrahedral voids in the unit cell of a face-centred cubic lattice of similar atoms is |
|
Answer» 4 |
|
| 21. |
The number of tertiary carbons and quaternary hydrogens in neopentane respectively are |
|
Answer» 1 and 1 |
|
| 22. |
The number of teritary carbon atoms in tertiary butyl alcohol is |
|
Answer» 3 |
|
| 24. |
The number of sulphur atoms present in 0.2 mole of sodium thiosulphate is (N=Avogadro number) |
|
Answer» 4N |
|
| 26. |
The number of sub levles in the quantum level n = 3 is |
|
Answer» 1 |
|
| 27. |
The number of structural isomers possible for C_(7)H_(16) is __________ |
|
Answer» `C-UNDERSET( C )underset(|)C-C-underset( C )underset(|)C-C""C-underset( C )underset(|)C-C-C-C-C` `C-C-underset( C )underset(|)OVERSET( C )overset(|)C-C-C""C-C-underset( C )underset(|)C-C-C-C` `C-C-underset( C )underset(|)underset( C )underset(|)C-C-C""C-underset( C )underset(|)overset( C )overset(|)C-C-C-C` `C-overset( C )overset(|)C-overset( C )overset(|)underset( C )underset(|)C-C""C-underset( C )underset(|)C-underset( C )underset(|)C-C-C` |
|
| 28. |
The number of structural isomers obtained by mono-halogenation of propane is |
| Answer» SOLUTION :C - C - C - CL`C- undersetoverset(|)(Cl)( C) - C ` | |
| 29. |
The number of structural isomers for C_6H_14 is |
|
Answer» 3 |
|
| 30. |
The number of structural and configurational isomers of a bromo compound, C_(5)H_(9)Br, formed by the additionn of HBr to 2-penthyne respectively are |
|
Answer» 1 and 2 `CH_(3)-C-=C-CH_(2)CH_(3) OVERSET(HBr) underset((I))(CH_(3)C(Br)=CHCH_(2)CH_(3))+underset((II))(CH_(3)CH=C(Br)CH_(2)CH_(3))` Each one of these will EXIST as a PAIR of geometrical isomers as shown below: 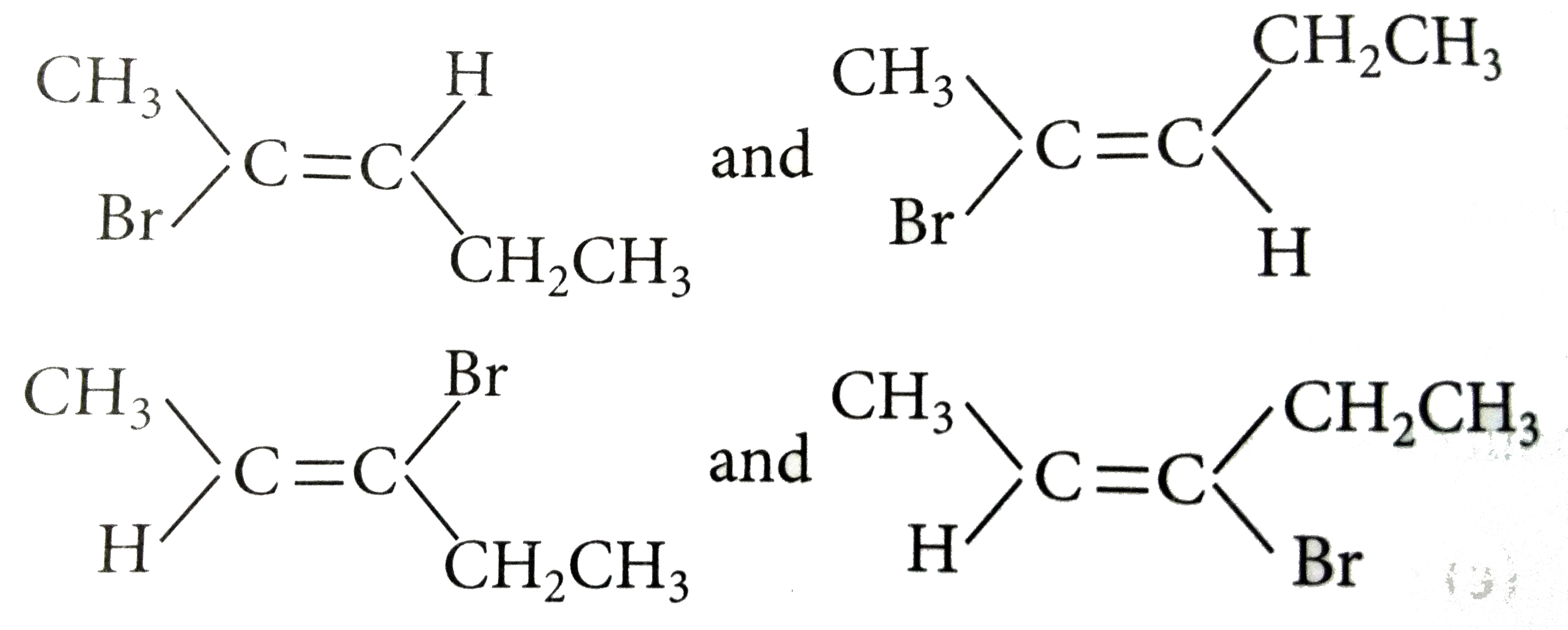 thus, there are two structural and four configurational isomers. |
|
| 31. |
The number of structural and configurational isomers ofa bromo compound , C_5H_9Br formed by the addition of HBr to 2-pentyne respectively are : |
|
Answer» 1 and 2 Each of these two isomers can exist as a PAIR of cis, trans-isomers and HENCE there are four configurational isomers. |
|
| 32. |
The number of stereoisomers possible for a compound of the molecular formula CH_3 - CH = CH - CH (OH)- Meis |
|
Answer» 2  TOTAL POSSIBLE ISOMERS are four. CIS - R , TRANS - R , cis - S , trans - S |
|
| 33. |
Thenumber of stereoisomers of 1,2-dihydroxycylopentane is …. |
|
Answer» 1 |
|
| 34. |
The number of vertebrates is ... |
|
Answer» 1 |
|
| 35. |
The number of stereoisomers obtained by bromination of trans-2-butene is : |
|
Answer» 1 |
|
| 36. |
The number of stereoisomeric products obtained by the addition of HBr to 2-butene |
| Answer» SOLUTION :ONE | |
| 37. |
The number of spherical nodes and planar nodes present in 4d_(x^(2) - y^(2)) and ......and ........respectively. |
|
Answer» |
|
| 38. |
The number of silver atoms present in a 90% pure silver wire weighing 10 g is (Ag = 108) |
|
Answer» `5.57xx10^(22)` 108 g of Ag CONTAINS `6XX10^(23)` ATOMS of Ag 9 gm of Ag contains `(6xx10^(23)xx9)/(108)` `=0.5xx10^(23)=5xx10^(22)` atoms of Ag |
|
| 39. |
The number of significant figures in it are |
|
Answer» One |
|
| 40. |
The number of significant figures in electronic charge 1.602 xx 10^(@)C |
|
Answer» 1 |
|
| 41. |
The number of significant figures in 3.040 dm is |
|
Answer» infinite |
|
| 42. |
The number of significant figures in each of these given numbers respectively are (i) 506.20 (ii) 0.003402 |
| Answer» Solution :`506.20=5,0.003402=4` | |
| 43. |
The number of significant figures in 6.0023 are |
|
Answer» 5 |
|
| 44. |
The number of significant figures in 10500 |
|
Answer» Three |
|
| 45. |
The number of significant figures in 0.0045 are |
|
Answer» Two |
|
| 46. |
The number of sigma of pi-bonds in butene 3-yne are |
|
Answer» 5 SIGMA and 5 pi |
|
| 47. |
The number of sigma (sigma) bonds in 1-butene is ……… |
|
Answer» 8 |
|
| 48. |
The number of sigma (sigma) and pi (pi) bonds in pent-2-en-4-yne is …… |
|
Answer» `13 sigma`-BONDS and no `PI`-bonds `{:(,,sigma,pi),(sigma-"bonds = 10",-,1,0),(pi-"bonds=3",=,1,1),(,-=,1,2):}` |
|
| 49. |
The number of sigma and pi bonds present in ethene is |
|
Answer» `6sigma and no PI` |
|
