Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is paramagnetic- |
|
Answer» `CN^(-)` `K K(sigma_(2S))^(2)(sigma_(2s)^(**))^(2)(sigma_(2p_(z)))^(2)(pi_(2p_(x)))^(2)(pi2_(p_(y)))^(2)(pi_(2p_(x))^(**))^(2)(pi_(2p_(y))^(**))^(1)` OWING to the presence of one UNPAIRED electron, it is paramagnetic in nature. |
|
| 2. |
Which of the following has maximum neutralization enthalpy ? |
|
Answer» `NH_(4) and CH_(3) COOH` |
|
| 3. |
Write Lewis dot symbols for the following elements : Mg, Na, B, O, N, Br |
| Answer» Solution :`overset(.)MG,""overset(.)NA,""overset(.)underset(.)B.,"".overset(..)underset(.)O:,"".overset(..)underset(.)N.,"".underset(..)overset(..)Br:` | |
| 4. |
The size of isoelectronic species-F^(-), Ne and Na^(+) is affected by ... |
|
Answer» nuclear charge (Z) |
|
| 5. |
Which element is the end product of every natural radioactive series? |
|
Answer» Pb |
|
| 6. |
Which is the structural formula of 4-methyl-hex-5-yne-2-one? |
|
Answer» `CH_(3)-CO-underset(underset(CH_(3))(|))(CH)-CH_(2)-C -=CH` |
|
| 7. |
What is meant by 'demineralished water '? |
| Answer» SOLUTION :Water free from all caption and anions is called demineralised water. Hard water is passed through caption EXCHANGE resin FOLLOWED by anion eexchange resin to FORM demeneralised water. | |
| 8. |
What do you understand by the following statements ? enthalpy of combustion of carbon disulphide is = -1065.2 kJ. |
| Answer» SOLUTION :When 1 mole of `CS_(2)(1)` is COMPLETELY burnt in `O_(2)`, 1065.2 kJ of heat is liberated. | |
| 9. |
Which of the following is least acidic ? |
|
Answer» `C_(2)H_(5)OH` `C_(2)H_(5)OH lt C_(6)H_(5)OH lt CH_(3)COOH lt ClCH_(2)COOH` |
|
| 10. |
Which one of the following sets of ions represents a collection of isoelectronic species ? |
|
Answer» `N^(3-),O^(2-),F^(-),S^(2-)` |
|
| 11. |
Write the structure of isobutylene ? |
| Answer» SOLUTION :`CH_(3)-UNDERSET(CH_(3))underset(|)(C)=CH_(2)` | |
| 12. |
What is environmental chemistry ? |
| Answer» SOLUTION :It is the STUDY of CHEMICALS and CHEMICAL PROCESS. | |
| 13. |
What is hydrogen bonding? |
| Answer» SOLUTION :Hydrogen BONDING is because of dipole-dipole interactions between the molecule in which hydrogen atom is covalently BONDED to a HIGHLY electronegative atom | |
| 14. |
Whereis the nodal planein P_(x ) ,P_(y) and P_(z) orbital ? |
|
Answer» SOLUTION :Thenodalplanein `p_(X )p_(y) p_(z)`prespectively in YZ. zx andxy. Note: The PLANE from twoaxesexceptpxorbital |
|
| 15. |
Use of sodium carbonate is.... |
|
Answer» in preparation of borex. |
|
| 16. |
Which of the following is not common property of Be and Al ? |
|
Answer» Their OXIDES are amphoteric |
|
| 17. |
Which one of the following decomposes easily on heating ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 18. |
Write the relationship between K_(sp) and S of an A_(2)B type of salt. |
|
Answer» Solution :Consider an electrolyte `A_(2)B`, WHOSE SOLUBILITY is S MOL `DM^(-3)`. In a saturated solution, `A_(2)B_((s))hArr2A^(+)+B^(2-)` `therefore[A^(+)]=2S" mol "dm^(-3)and(B^(2-))=S" mol "dm^(-3)` `K_(sp)=[A^(+)]^(2)[B^(2-)]=(2S)^(2).S=4S^(3)` `thereforeK_(sp)=4S^(3)` |
|
| 19. |
What is the molarity of HCl solution that has a density of 1.17 g/mL at 25^(@)C? |
|
Answer» 36.5 M 1.17 G HCl dissolve in 1 mL `= 0.001` L solution molarity `= (1.17)/(36.5xx0.001)=32.054M` |
|
| 20. |
Which of the following is not correct ? |
|
Answer» Iodine oxidises sodium THIOSULPHATE to sodium tetrathionate `I_(2)+2Na_(2)S_(2)O_(3)rarrNa_(2)S_(4)O_(6)+2NaI` In this REACTION `I_(2)` acts as an oxidising agent. |
|
| 21. |
The substances that cause depletion of ozone or make it thinner are abbreviated as |
| Answer» SOLUTION :ODS | |
| 23. |
Which pair among the following is enantiomers |
|
Answer»

|
|
| 24. |
Which of the following statements regarding Friedel-Crafts reactions is wrong? |
|
Answer» ALKYLATION of benzene with an alkyl chloride requires only a CATALYTIC amount of a Lewis acidsuch as aluminum chloride |
|
| 25. |
What is chlorosis? |
| Answer» SOLUTION :Slowing down the formation of chlorophyll in plants DUE to presence of `SO_2` as POLLUTANT is called chlorosis. | |
| 26. |
When 20 ml of methane and 20 ml of oxygen are exploded together and the reaction mixture is cooled to laboratory temperature. The resulting volume of the mixture is |
|
Answer» Solution :`CH_(4)^((g))+2O_(2)^((g))rarrCO_(2)^((g))+2H_(2)O(l)` `{:("Initial","20 ml","20 ml",-,-),("After reaction","10 ml",-,"10 ml",-):}` |
|
| 27. |
Which of the following statements is /are incorrect about two organic compounds which are stereoiso iners? |
|
Answer» They MUST be enantiomers |
|
| 28. |
Which of the following is not an example of disproportionation reaction? |
|
Answer» `Cl_(2)+2NaOH to NaCl+NaOCl+H_(2)O` |
|
| 30. |
The volume occupied by 8.8 gm of CO_2 at 31.1^@C and 1 bar pressure (R = 0.083 "bar. It K"^(-1)mol^(-1)) is |
| Answer» ANSWER :C | |
| 31. |
Which setsofionsis isoelectronic ? |
|
Answer» `NO^(+) , C_(2)^(2) CN^(-)N_(2)` |
|
| 32. |
Which one of the following does not contain neutron? |
|
Answer» ORDINARY hydrogen |
|
| 33. |
The treatment of acetone with excess of RMgX gives: |
| Answer» SOLUTION :`R - UNDERSET(OH)underset(|)OVERSET(R )overset(|)C-R` | |
| 34. |
The second ionization enthalpy is |
|
Answer» smaller than the FIRST ionization enthalpy |
|
| 35. |
The volume of CO_(2) formed when a mixture of 2 mol NaHCO_(3) and 1 mol Na_(2)CO_(3) at STP, is- |
|
Answer» 2 EQUIVALENT `H_(2)` gas |
|
| 36. |
What is dry ice ? Why is it called so ? How to prepare a pure sample of CO ( Carbon monoxide ) ? |
|
Answer» Solution :Solid `CO_(2)` is called DRY ice.It is called dry ice because it directly changes into GASEOUS state without becoming liquid. `"HCOOH" underset (373 K) OVERSET ("Conc" H_(2)SO_(4)) (rarr) CO+H_(2) O.` |
|
| 37. |
Which of the following has the maximum number of unpaired electrons ? |
| Answer» Answer :D | |
| 38. |
Which of the following has the highest tendency to give the reaction M_((g))^+overset("Aqueous")underset("Medium")toM_((aq))^+ |
|
Answer» Na |
|
| 39. |
which of the following molecules would have a carbon -halogen bond most suscptible to nucleophilic subsition ? |
|
Answer» 2- fluourbutane  Decreasing order of RATE of `S_(N^(2))` REATION .` |
|
| 40. |
What are the conclusions of Rutherford's a-rays scattering expermiment? |
|
Answer» SOLUTION :(i)Rutherford bombarded a thin gold foil with a strea, of FAST moving a-particles. (ii)It was observed that most of the a-particles passed through the foil. (iii)Some of them were deflected through a small angle. (iv)Very few a-particles were reflected back by `180^(@)` (V)Based on these observations,he proposed that in an atom,there is a tiny positively changed NUCLEUS and the electrons are ,moving around the nucleus with high speed. |
|
| 41. |
Write the resonance structures of ozone. |
Answer» SOLUTION :`)
|
|
| 42. |
Which of the following is heterolytic compound ? |
|
Answer» PYRROLE |
|
| 43. |
When two molecules of an ideal gas collide |
|
Answer» heat is LIBERATED |
|
| 44. |
Two moles of an ideal gas is expanded spontaneously into a vacuum. The work done is |
| Answer» ANSWER :A | |
| 45. |
Whichis thecorrectset of all fourquantumnumberforvalanceelectronof Na ? |
|
Answer» `1,-0,0, + (1)/(2)` |
|
| 46. |
Whichone iscorrectfor hydrogen |
|
Answer» The energyof 3dorbital is LESSTHAN 3p |
|
| 47. |
Why BeH_2 molecule has zero dipolemoment although the Be-H bonds are polar ? |
|
Answer» Solution :`BeH_(2)` is a linear molecule (H-Be-H) with bond angle equal to `180^(@)`. ALTHOUGH Be-H bonds are polar on account of ELECTRONEGIVITY difference between Be (1.5) and H(2.1) atoms. The bond polarities cancel each other. The molecule has resultant dipole moment of zero. |
|
| 48. |
Which is the electrophile of the following ? |
|
Answer» `OVERSET(+)(N)O_(2)` |
|
| 49. |
The % yield of Ammonia as a function of time in the reaction N_(2)(g) + 3H_(2)(g) hArr2NH_(3)(g), Delta H lt O " at " (P, T)is given below: If this reaction is conducted at T_(2) gt T_(1), then plot the % yield of NH_(3)as a function of time on same graph |
Answer» SOLUTION :Initially on increasing temperature the rate of REACTION increases, however since the reaction is exothermic therefore % yield of `NH_(30`GET decreased overall after a certain interval of TIME. 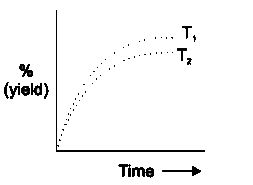
|
|
| 50. |
Which of the following compound(s) reduces chromates and dichromates? |
| Answer» Answer :A::B::C::D | |


