Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statement (s) for crystal having schottky defect is/are correct |
|
Answer» Schottky defect arises due to ABSENCE of CATION & anion from position which they are expected to occupy |
|
| 2. |
Write the tautomer of acetaldehyde and give its IUPAC name. Which of the two is more stable and why ? |
Answer» SOLUTION : The tautomer, eth-1-en-1ol (i.e., vinyl ALCOHOL) is less stable than acetaldehyde because C = O `pi`-BOND `(364 KJ mol^(-1))` is MUCH stronger than C=C `pi`-bond `(254 kJ mol^(-1))`. |
|
| 3. |
Which of the following is purifiedby vacuum distillation |
|
Answer» ETHANOL |
|
| 5. |
Which of the following is not attacked by hot sodium hydroxide solution ? |
|
Answer» Silicon |
|
| 6. |
Which of the following does give violet colour with neutral ferric chloride ? |
|
Answer» ACETIC acid |
|
| 7. |
What is the difference between 13-15 and 12-16 compounds ? |
| Answer» Solution :Group 13-15 compounds such as AIP, GaAs, etc. have large covalent CHARACTER but Group 12-16 compounds such as ZnS, CDS, HgTe, etc.do not have perfect covalent bonds but POSSES SUFFICIENT ionic character. Howver, all of them are semiconductors. | |
| 8. |
What do you understand by 'auto-protolysis' of water? What is significance? |
|
Answer» Solution :AUTO-pyotolysis of water means 'self-ionisation' which PROCEEDS as FOLLOWS: `"" underset("Acid-I")(H_(2)O(l))+underset("Base-II")(H_(2)O(l)) Leftrightarrow underset("Acid-II")(H_(3)O^(+)(aq))+underset("Base-I")(O^(-)(aq))` Because of auto protolysis, water behaves both as a Bronsted acid and Bronsted base i.e. it is amphoteric in nautre `"" underset("Acid-I")(H_(2)O(l))+underset("Base-II")(NH_(3)(l)) Leftrightarrow underset("Acid-II")(NH_(4)^(+)(aq))+underset("Base-I")(OH^(-)(aq))` |
|
| 9. |
Why boron forms electron deicient compounds ? |
| Answer» Solution :BORON has only THREE ELECTRONSIN the OUTER mostshell which itcan share with other atoms.Hence,in itscompounds, there areonly 6 electronspresent aroundB-atomi.e. ,i.e,octet is not complete. | |
| 10. |
The time taken for diffusion of CO_(2(g)) is twice than that of unknown gas of same volume under identical condition. Calculate the molecular weight of unknown gas. (C = 12, O = 16). |
|
Answer» 11 gm/mol `(r_(1))/(r_(2))=(V_(1)t_(2))/(V_(2)t_(1))=SQRT((M_(2))/(M_(1)))` `V_(1)=V_(2) "" t_(2)=2t_(1)` `(2t_(1))/(t_(1))=sqrt((44)/(M_(1)))` `therefore 2=sqrt((44)/(M_(1)))` `therefore 4=(44)/(M_(1)) "" therefore M_(1)=11" gm mole"^(-1)` |
|
| 11. |
Which compound with react with Zn to form 2-butene ? |
|
Answer» 2,3-DIBROMOBUTANE |
|
| 12. |
Which of the following is paramagnetic with bond order 0.5 |
|
Answer» `O_(2)` |
|
| 13. |
Write the names of isotopes of hydrogen . What is the mass of these isotopes ? |
| Answer» SOLUTION :Protium `""_(1)^(1)H`, Deterium `""_(1)^(2)H` or D, TRITIUM `""_(1)^(3)H` , or T. The mass ratio of protium, : deutrium : tritium =1:2:3 | |
| 14. |
What is STP ? |
| Answer» Solution :STANDARD TEMPERATURE and pressure means `273.15 (0^(@)C)`temperature and 1 BAR (i.e. exactly `10^(5)` pascal)pressure. These values approximate greezing temperature of water and ATMOSPHERIC pressure at sea level. | |
| 15. |
Write the structures of the following : (i) Acetone (ii) Ethylene glycol (iii) Sec-butyl alcohol. |
|
Answer» Solution :(i) `CH_(3)-OVERSET(O)overset(||)(C)-CH_(3)` (ii) `HO-CH_(2)-CH_(2)-OH` (iii) `CH_(3)-UNDERSET(OH)underset(|)(CH)-C_(2)H_(5)` |
|
| 16. |
The silver salt of a fatty acid on refluxing with an alkyl halides gives an |
|
Answer» acid |
|
| 17. |
Which among the following is E-Isomer ? |
|
Answer»
|
|
| 18. |
Which oneof the followingelementsposeda challenge to Newlands 'slaw of octaves ? Li, Be , B , C , N , O ,F , Ne , Na |
| Answer» SOLUTION :Newland'slaw ofoctavesstates thatwhen ELEMENTSARE arranged order of increasingatomicweights then theeveryeighth elementresemblesthe firstone . Butwhen innergas (NE wasdiscovered. Then it WASTHE ninthwhichresembles the firstone . Thereforethe elementinert gas Ne POSED a challange to Newlands's law ofoctaves. | |
| 20. |
Which is correct of the following in structure of ? (i) 2-methyl-6-ethylnonance (ii) 6-ethyl-2-methylnonane (iii) 4-ethyl-8-methylnonane |
| Answer» SOLUTION :(II) Because, the no. of substituents are written ACCORDING to ALPHABETICAL order | |
| 21. |
What is molar molecular volume at STP, if compressibility factor Z lt 1 ? |
|
Answer» Solution :Molar volume of Ideal gas decreases. EX. `V_(m)=22.4` Lit at STP When `Z lt 1`, then `V_(m)lt 22.4 L` |
|
| 22. |
Which kind of defets are intorduced by doping ? |
|
Answer» Disolocation defect |
|
| 23. |
When zeolite is treated with hard water, the sodium ions are exchanged with ...... |
| Answer» SOLUTION :`CA^(2+)` IONS and `MG^(2+)` ions | |
| 24. |
What is the dominant intermolecular force or bond that must be overcome in converting liquid CH_(3)OH to a gas ? |
|
Answer» Dipole-dipole interaction DUE to intermolecular hydrogen bonding in methanol, it ex.1st as ASSOCIATED molecule. |
|
| 25. |
Which of the following compounds has the smallest bond angle in its molecule |
|
Answer» `NH_3` |
|
| 26. |
What is the percent composition of a mixture of (S)-(+)-2- butanol, [+-](25)/(D)=+13.52^(0), and (R)-(-)-2- butanol, [+-](25)/(D)=-13.52^(0), with a specific rotation [+-](25)/(D)=+6.76^(0)? |
|
Answer» 75%(R) 25%(S) |
|
| 27. |
The unit of surface tension is |
|
Answer» `"DYNES CM"^2` |
|
| 28. |
Which of the following representation of wave functions of molecular orbitals and atomic orbitals are not correct ? |
|
Answer» `Psi_(MO)=Psi_(A) PM Psi_(B)` |
|
| 29. |
Which of the following reactions will evolve CO_(2)(g) as product ? |
|
Answer» Charcoal `overset("stream(excess)")(rarr)` d) `MgC_(2) O_(4) overset(Delta)(rarr) MgCO_(3) + CO`, `CO + (1)/(2) O_(2) rarr O_(2) , MgCO_(3) rarr Mgo + CO_(2)` |
|
| 30. |
Which of the following reaction has maximum Delta S value? |
|
Answer» `Ca_((s)) + (1)/(2) O_(2(g)) to CaO_((s))` |
|
| 31. |
Which one of the following options does not represent ground state electronic configuration of an atom ? |
|
Answer» `1s^(2) 2s^(2) 2P^(2) 3S^(2) 2p^(6) 3d^(8) 4s^(2)` |
|
| 32. |
Which of the following specles does not exert a reasonance effect ? |
|
Answer» `C_(6)H_(5)NH_(2)` |
|
| 34. |
Water is polar in nature. |
| Answer» Solution :EVEN though `SP^(3)` HYBRIDIZATION in there is `H_(2)O` but due to LONE pair - lone pair repulsion exists. | |
| 36. |
Two moles of an ideal gas at 2 atmand 27^(@)Care compressed isothermally to halfthe volume by an externalpressure of4 atm. Calculate w, q and DeltaU. |
|
Answer» `:. w = -P_(EXT) DELTAV=- 4 atm xx ( 12.71 -25.42) L= 4 xx 12.71 L atm= 50.84 Latm=50.84 xx 101.3 J= 5150 J` |
|
| 37. |
Ionic ompounds like AgCl, CaF_2, and BaSO_4 are insoluble in water. This is because, |
|
Answer» ionic compounds are insoluble in water |
|
| 38. |
Which of the following compounds is paramagnetic |
|
Answer» `KO_2` |
|
| 39. |
Which of the following is used as adsorbent? |
|
Answer» SILICA GEL and alumina |
|
| 40. |
Third period contain 8 elements. Fourth period contain 18 elements. Seven period contain 32 elements. |
|
Answer» Solution :For sixth PERIOD , `n = 6` So, `LAMBDA = n - 1, lambda = 6 - 1 = 5 `  Total sub orbitals ` = 1 + 3 + 5 + 7 = 16` Number of electronns = 32 ` :. 32` elements present in 6th period. ENERGY of orbital : `6s lt 4f lt 5d lt 6p`. |
|
| 41. |
Which of the following reagents is used for decarboxylation of carboxylic acid ? |
|
Answer» `NaHCO_(3) + KCl` |
|
| 42. |
Which of the following is green house gas and also causes depletion of ozone layer |
|
Answer» CFCS |
|
| 43. |
The repeating unit of silicones |
|
Answer» Rsio |
|
| 44. |
What is Mayer's relation? |
|
Answer» <P> Solution :The difference in MOLAR heat capacity values for a gas ut constant pressure `(C_(p))` and at constant volume`(C_(v))` is called Mayer.s RELATION`C_(p)-C_(v)=R=` Molar gas consant. For a monoatomic gas LIKE helium,`c_(p)=5R/2" and "C_(v)=3R/2` |
|
| 45. |
Which of the following is an electrophilic reagent? |
| Answer» Answer :C | |
| 46. |
Which of the following reaction is catalysed by enzyme maltase |
|
Answer» STARCH`to`maltose |
|
| 47. |
Which of the following has highest electron attraction capacity? |
| Answer» ANSWER :D | |
| 48. |
Which of the following sets of quantum number is/are not possible ? |
|
Answer» `{:(n, l , m_l , m_s),(4,4,-2,+1//2):}` In option (c) `m_l` cannot be - 3 for l = 2 because `m_1 =- 1` _____ 0 ______ `+ 1` |
|
| 49. |
X underset(P_(2)O_(5))overset("Heat")rarr Malonic acid underset(432K)overset(Delta)rarr YX and Y in the above reaction sequence are |
|
Answer» `C_(3)O_(2), CH_(3)COOH` `underset("Malonic acid")(CH_(2) (COOH)_(2)) overset(P_(2)O_(5))rarr underset((X))(O = C = C) = O + 2H_(2)O` |
|
| 50. |
Which of the following is more stronger acid than Phenol ? |
|
Answer» Ethanol 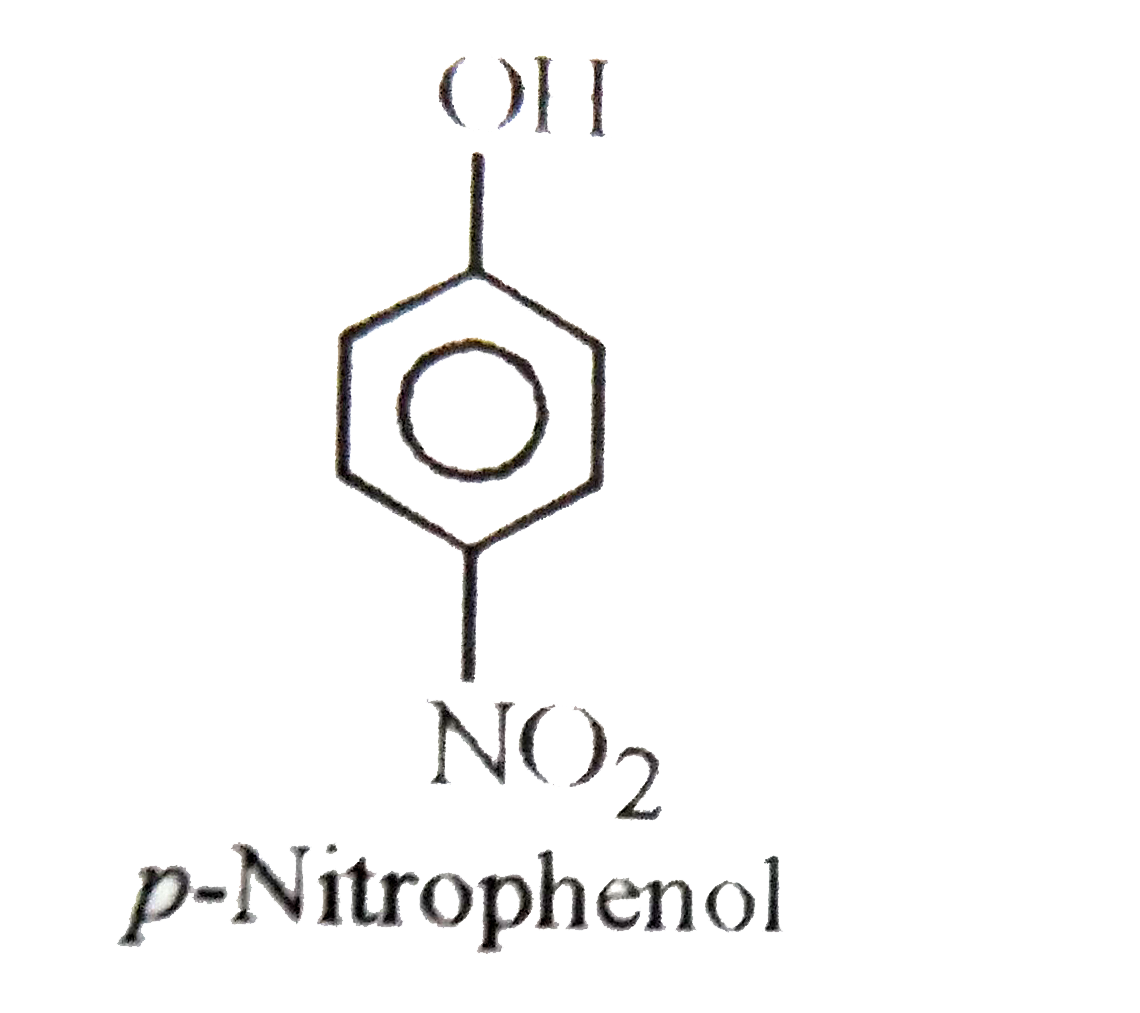
|
|



