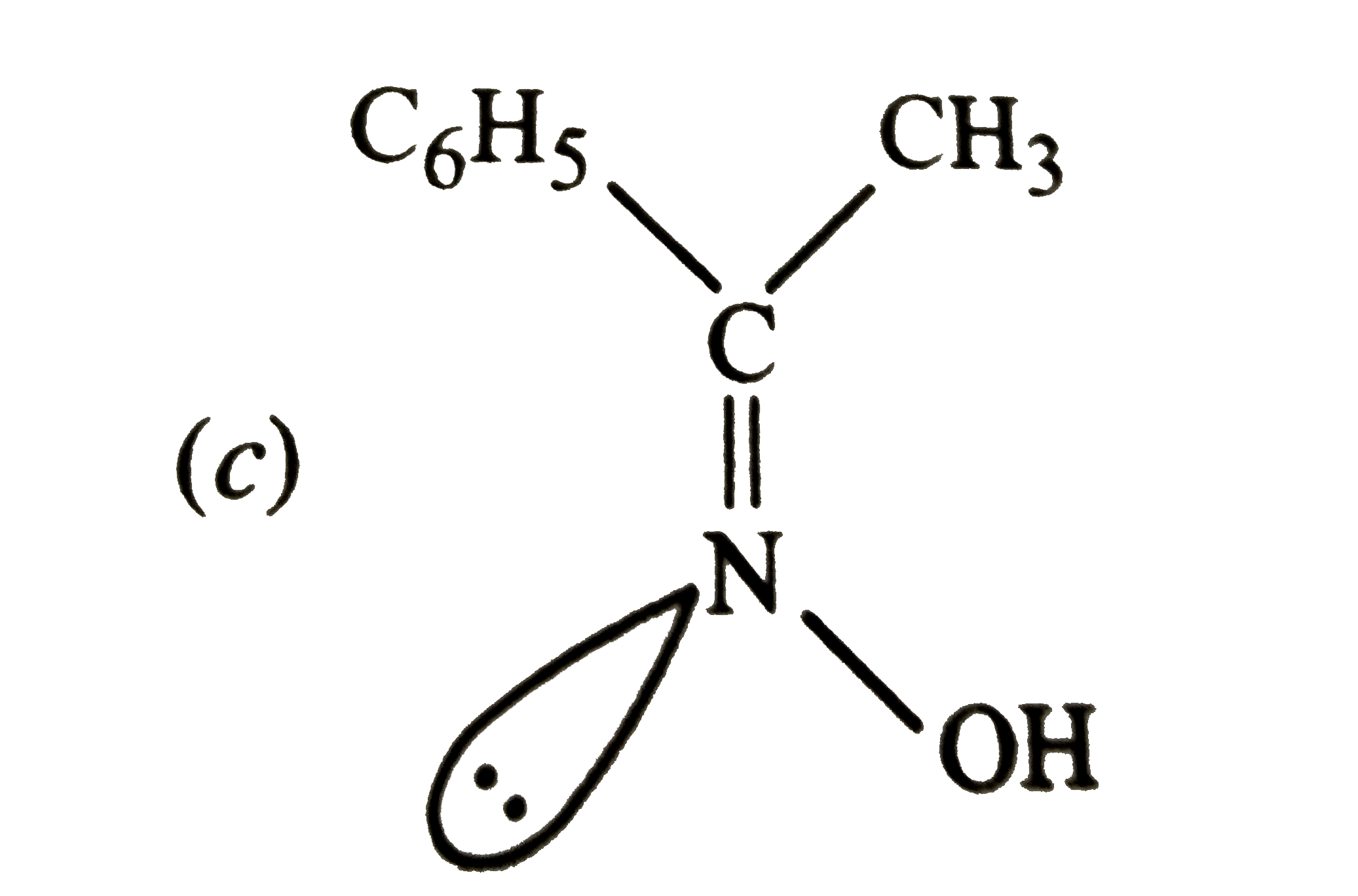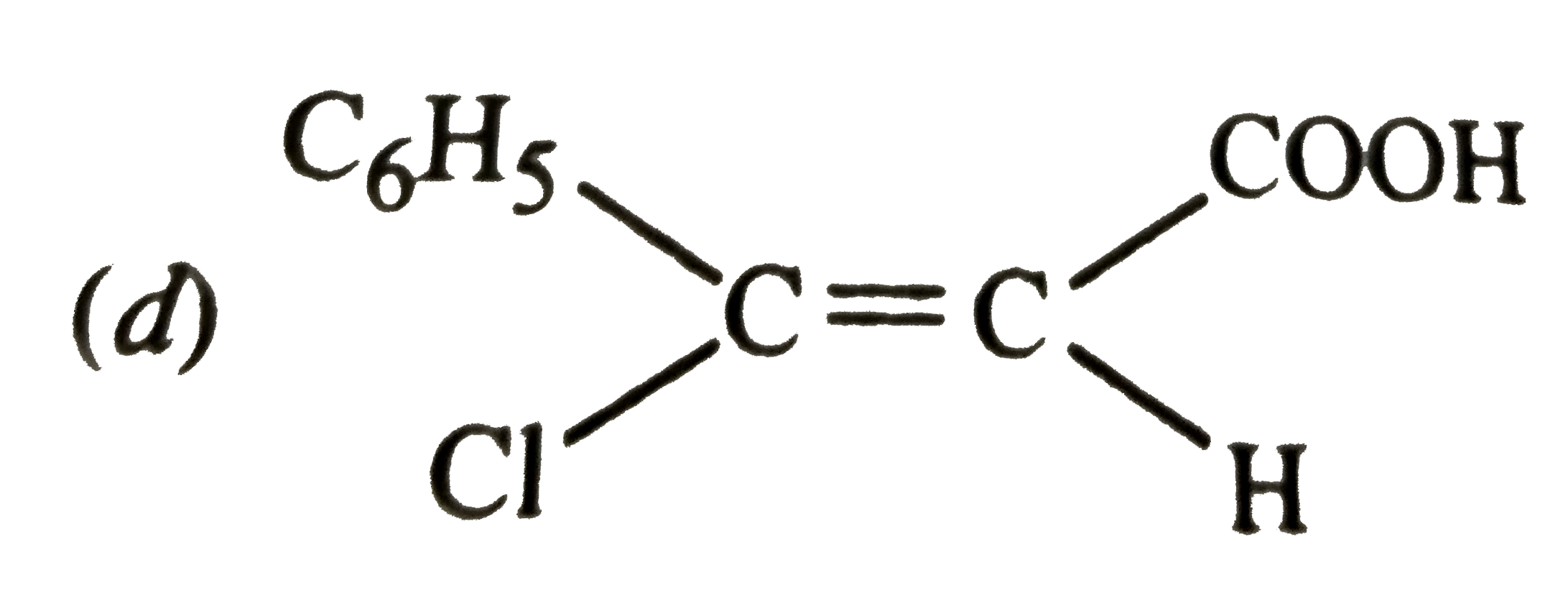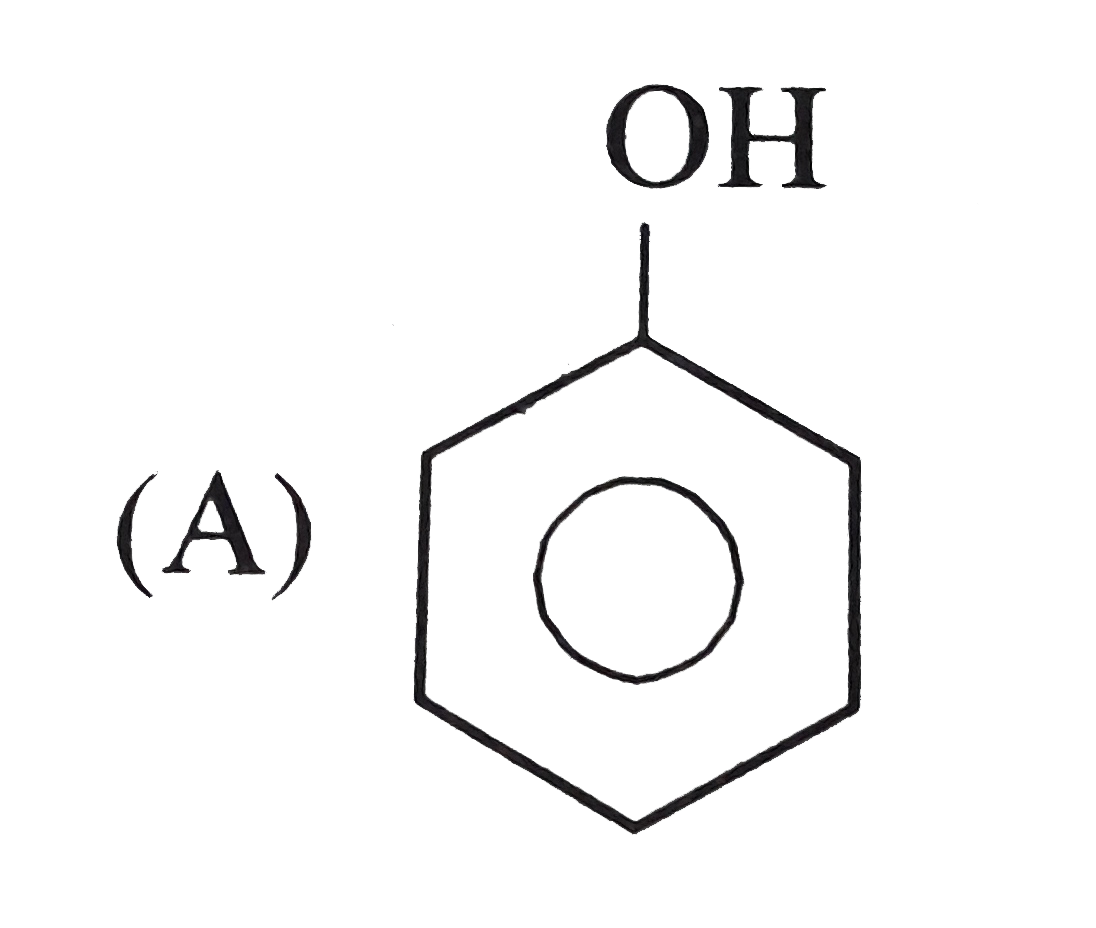Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
V_(1) mL of NaOH of normality x and V_(2) mL of Ba(OH)_(2) of normality y are together sufficient to neutralize exactly 100 mL of 0.1 N HCl. If V_(1) : V_(2) =1 : 4 and if x : y =4 : 1, what fraction of the acid is neutralised by Ba(OH)_(2) ? |
|
Answer» 0.5 |
|
| 2. |
Which substances having intermolecular (van der Waal) forces ? |
|
Answer» Solution :None - polar STATES : e.g., `He, Ne, H_(2), Cl_(2), Br`….. etc. Polar molecules : e.g., HCl, HF, CO, NO Force present between polar and non - polar molecules. |
|
| 3. |
The solubility of lead iodide in water is 0.63 g/litre. Calculate the solubility product of lead iodide. (At mass of Pb = 207 , I = 127 ) |
|
Answer» |
|
| 4. |
What are Clathrate hydrate? Explain it with suitable example. |
|
Answer» SOLUTION :(i) Gas hydrates in which the guest MOLECULES are not bonded CHEMICALLY but retained by the structure of host is called clatharates. (ii) Water FORMS clatharate hydrates, eg methane hydrates `(CH_4.2OH_2O)` which are a type of ice that will burn when a lit match is held to it (iii) The structure of methane hydrate is made of liked polyhedral that contains 20hydrogen bonded water molecules forming a cage in which methane MOLECULE is trapped. (iv) Deposits of methane clatharates occur naturally in deep sea bed. |
|
| 5. |
Whenelectron transistfrom higherenergylevelto lowerenergylevelthen ….. |
|
Answer» energyabsorb |
|
| 7. |
What is called 'a chemical family'? |
| Answer» SOLUTION :The element PRESENT ina group of the PERIODIC table are called CHEMICAL family. This is because the elements possess similar chemical PROPERTIES. | |
| 8. |
Total nubler of mono chlorodervatives obrained by replacement of H by Cl in propene are : |
|
Answer» |
|
| 9. |
The vividh bharati station of All India Radio, Delhi broadcasts on a frequency of 1.368 kHz. Calculate the wave length of the electromagnetic radiation emitted by the transmitter. Which part of the electromagnetic spectrum does it belong ? |
|
Answer» Solution :GIVEN `V=1.368kHz=1.368xx10^(3)s^(-1)` `"Velocity of light, c "= 3xx10^(8) ms^(-1)` Wavelength, `lambda=?` `lambda=(c)/(v)=(3xx10^(8)ms^(-1))/(1.368xx10^(3)s^(-1))=219.3m` The wavelength of electromagnetic RADIATION emitted by TRANSMITTER is 219.3 m. This is a characteristic radio wave wavelength. |
|
| 11. |
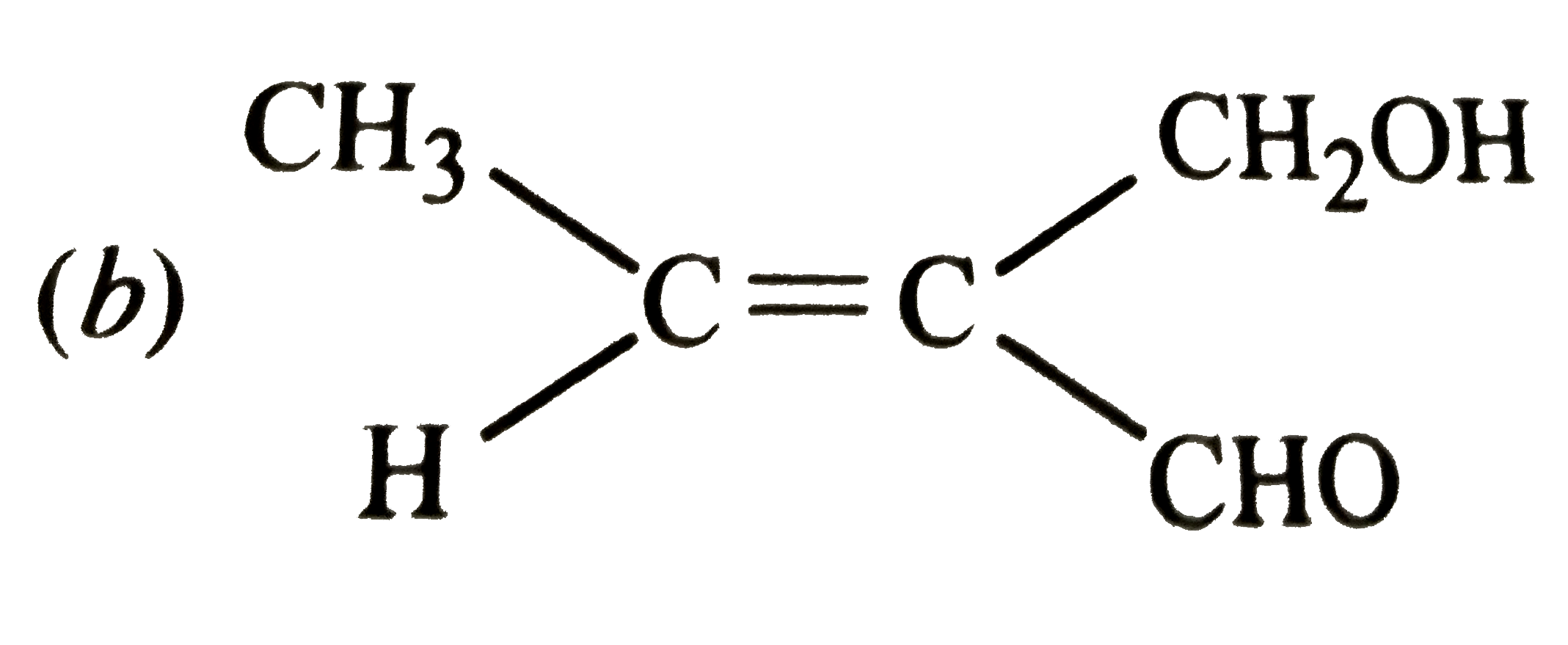
Which of the following is paramagnetic |
|
Answer» `Na_(2)O` |
|
| 12. |
What would be the work done on// by the system if the internal energy of the system falls by the 100 joules evenwhen 200 joules of heat is supplied to it ? |
|
Answer» |
|
| 13. |
Which of the following isotopes of hydrogen is most reactive for Cl_(2)? |
|
Answer» H |
|
| 14. |
Which of the following is ferromagnetic ? |
| Answer» Answer :B | |
| 15. |
Yellow phosphrous on reaction with Ca(OH)_(2) gives |
|
Answer» `Ca(H_(2)PO_(4))_(2)` |
|
| 16. |
Use the following data tocalculate Delta_("lattice") H^(c-) for NaBr. Delta_("sub") H^(c-) for sodium metal =108.4 kJ mol^(-1) Ioniztion enthalpy ofsodium = 496kJ mol^(-1) Electron gain enthalpy of bromine = - 325 kJ mol^(-1) Bond dissociation enthalpy of bromine =192kJ mol^(-1) Delta_(f)H^(c-)for NaBr(s) = - 360.1 kJ mol^(-1) |
|
Answer» Solution :Proceedas in CALCULATION of LATTICE enthalpy for NaCl on page . We GET `Delta_("lattice") H^(@) ( NaBr)= - 360 .1 kJ MOL^(-1)` |
|
| 17. |
which of the following can be used in an electrode ? |
|
Answer» a NAIL |
|
| 18. |
Wavelengthassociatedwith electronmotion …. |
|
Answer» increasewithincreasein speedof ELECTRON. |
|
| 19. |
Which of the following is used as nitrogeneous fertilizer in agriculture ? |
|
Answer» `NaNO_(3)` |
|
| 20. |
Which functions are needed to understand the second law of thermodynamics ? |
| Answer» SOLUTION :ENTROPY and FREE ENERGY. | |
| 21. |
Which of the following is not a Bravais lattice ? |
|
Answer» FACE CENTRED orthorhombic |
|
| 22. |
Which of thefollowing is not a Bravais lattice ? |
|
Answer» END CENTRED cubic |
|
| 23. |
The total no. of isomers for the compounds of the formula C_(4)H_(8)O are….. |
|
Answer» 3 |
|
| 24. |
What is reaction quotient (Q). |
| Answer» Solution :Reaction QUOTIENT is the ratio of PRODUCT of MOLAR concentrations (or) partial pressures of the PRODUCTS to that of the reactants at GIVEN time. | |
| 25. |
underset(P_(1), V_(1), T_(1))("State") A overset("Rev")rarr underset(P_(2), V_(2), T_(2))("State B"): If P_(1) = P_(2) then |
|
Answer» `underset("UNIVERSE")(Delta S) = 0` `:. Delta underset("universe")(S) = 0` |
|
| 26. |
When we move from HF to HCl , the boiling point sharply buton moving further to HBr and HI the boiling point increases. Why ? Or Out of HF, HCl , HBr and HI which has boiling point and why? |
|
Answer» Solution :In H-F , there is H-boiling point is high . There is no H-bonding in HCl . So the boiling point is less . On moving further to HBr and HI, the size of the molecule increases and so the VAN der WAALS forces increase and so does the boiling point . Hence , HCl has the LOWEST boiling point. |
|
| 27. |
Water is the working fluid used in Kota thermal power plant for generating electricity. Coal is combusted for generating heat as per reaction, C + O_(2) rarr CO_(2). 0.01 % of the released CO_(2) gas is absorbed in water and gets coverted to weak acid, H_(2)SO_(3) which dissociated to give H^(+) as H_(2)CO_(3) rarr 2H^(+) + CO_(3)^(2-). The percentage dissociattion of acid is 5 %. Assume no ionisation of water. From this information answer the questions. if in a certain application [H^(+)] concentration can maximum be 10^(-5)M, then, (P) Calculate maximum moles of H^(+)(x) and CO_(3)^(2-)(y) in the water water if 10^(9) litres of H_(2)O is used (Q) Calculate maximum moles of carbon (z) which can be burnt so that water remains fit to be used Hence, write the value of (xy^(2)//z) |
|
Answer» |
|
| 28. |
Which of the following reactions increases production of dihydrogen from synthesis gas ? |
|
Answer» `CH_(4(G)) + H_2O_((g))UNDERSET"Ni"overset"1270 K" to CO_((g)) + 3H_(2(g))` `CO_((g)) + H_(2)O_((g)) overset"673 K"to CO_(2(g)) + H_(2(g))` |
|
| 29. |
What are the types of air pollutants? Give examples. |
|
Answer» SOLUTION :(i) Air pollutants EXISTS in two major forms namely: (a) aseous air polutants (b) Particulates. (ii) Gaeous air pollutats : Oxides of sulphur. Oxids of nitrogen, oxides of carbon and hydrocarbons are the GASEOUS air pollutants. (iii) PARTICULATE pollutants : Particulate pollutants are small solid particles and liquid droplets suspended in air . Example - dust, POLLEN, smoke , soot, aerosols. |
|
| 30. |
Two elements x (at mass = 75) and y(at mas=16) combine to give a compound having 75.8% x. The formula of the compound is |
|
Answer» xy Mole ratio of `y =(24.2)/(16)=1.5` Simple ratio `=x:y = 1:1.5=2:3=x_(2)y_(3)` |
|
| 31. |
What is the product obtained from the HCl, HBr and Hi in presence of peroxide ? |
| Answer» SOLUTION :`C OVERSET(.)(l), Boverset(.)(R), overset(.)(I)` : Respectively free radicals are formed. | |
| 32. |
The value of K_(w) is 9.55xx10^(-14) at a certain temperature. Calculate the pH of water at this temperature . |
|
Answer» Solution :Here, we are given : `K_(w)= 9.55xx10^(-14)` Now, as for WATER, `[H_(3)O^(+)]=[OH^(-)] :. K_(w)=[H_(3)O^(+)][OH^(-)]=[H_(3)O^(+)][H_(3)O^(+)]=[H_(3)O^(+)]^(2)`, i.e., `[H_(3)O^(+)]^(2) = 9.55xx10^(-14) or [H_(3)O^(+)]=sqrt(9.55xx10^(-14))=3.09xx10^(-7)M` `:. PH = - log [H_(3)O^(+)]=-log (3.09xx10^(-7))=-[log 3.09+ log 10^(-7)]=- [ 0.49-7]=6.51`. |
|
| 33. |
Which of the following process is important in food industry? |
|
Answer» Dehydration |
|
| 34. |
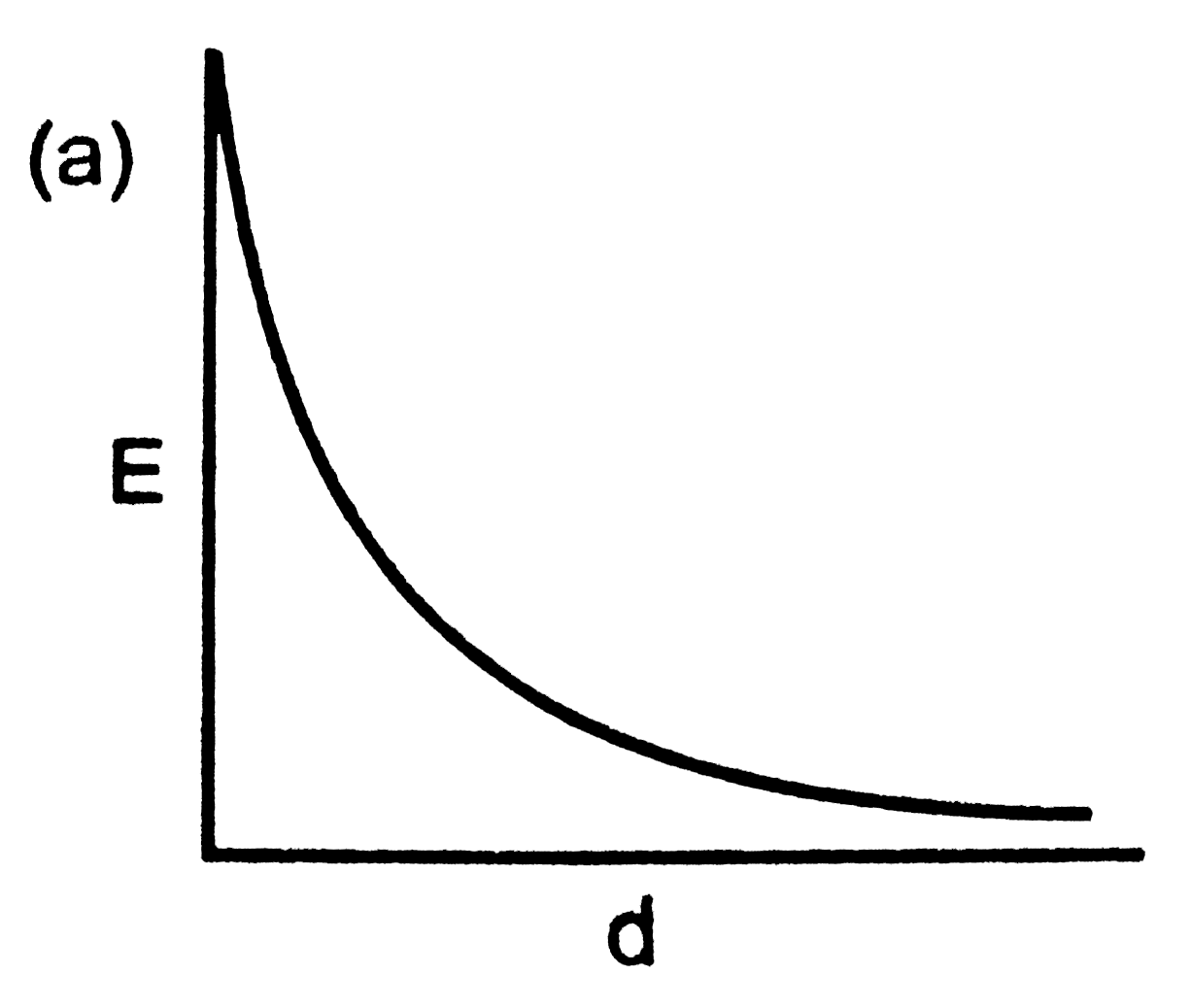
When two hydrogen atoms approach each other toformH_(2) molecule, the potential energy diagram obtained is : (d = interatomic distance ) |
|
Answer»
|
|
| 35. |
The weight of one molecule of compound C_(60)H_(22) is : |
|
Answer» `1.2 xx 10^(-20) g` ` (C_(60)H_(122))=60xx12+122=842g` Mass of ONE molecular `= (842)/(6.022)xx10^(-23)g` `=1.4 xx 10^(-21)g`. |
|
| 36. |
Which of the following has the highest boiling point ? (i)2-methylpentane, (ii)2,3-dimethylbutane , (iii)2,2-dimethylbutane. |
| Answer» Solution :As the branching INCREASES , surface area decreases. As a result, magnitude of van der WAALS forces of attraction decreases and hence the boiling point decreases. Now 2-methylpentane has the largest surface area and hence has the highest boiling point. Further because of the presence of two BRANCHES on the same carbon, 2,2-dimethylbutane has LOWER surface area and hence has lower boiling point than that of 2,3-dimethylbutane. Thus, the overall order of decreasing boiling points is : 2-methylpentane (333 K) gt 2,3- dimethylbutane (331 K) gt 2,2-dimethylbutane (323 K) | |
| 37. |
The wavelength of a spectral line emitted by hydrogen atom in the Lyman series is 16/15R cm. The value of n_(2) if R is Rydberg constant |
|
Answer» 1 |
|
| 38. |
What is the effect of resonance? Write about its types ? |
|
Answer» Solution :The resonance effect is defined as the polarity produced in the molecule by the interaction of two `pi`-bonds or between a `pi`-bond and lone pair of electrons present on an adjacent atom. The effect is transmitted through the chain. These are two types as resonance or mesomeric effect designed of R or M effect. (a) Positive resonance (+R) or mesomeric (+M) effect: (i) Definition: When the transfer of electron is AWAY from an atom or substituent group attached to the conjugated system. This electron displacement makes CERTAIN POSITIONS in the molecule of high electron densities. So this is called positive resonance (+R) effect. (ii) e.g. In aniline `-OH` group containing (+R) and (+M) effect and the non-bonding electron pair of N is transfer in BENZENE ring and aniline becomes polar. Due to this effect the electron density is increases at some position.  Its structure (II, III) and (IV) are polar and lone pair electron of `overset(..)(N)` at definite position and negative charge and more electron density. (iii) The other examples of +R or +M effect containing groups are as under. `-X, -OH, -OR, -OCOR, -NH_(2), -NHR, -NR_(2)- NHCOR` (b) Negative resonance effect (-R) or negative mesomeric effect (-M): (i) Definition: When the transfer of electron is towards the atom or substituent group attached to the conjugated system then these groups are called (-R) or (-M) effect. (ii) e.g. : In nitrobenzene, `-NO_(2)` group containing (-R) means (-M) effect and the electron pair of `pi`-bond with N of out side of ring. Therefore nitrobenzene molecule become polar and at certain position the electron density decrease in ring and become positive  Structure (II), (III), (IV) are polar. The electron pair of ring transfer at out side in O-N bond and (+) charge is at specificposition and electron density is less (iii) (-R) means (-M) effect containing groups are `-COOH, -CHO, gt C= O, -CN, -NO_(2)` |
|
| 39. |
Which of the following compounds does not evolve CO_2 gas, on oxidative ozonolysis? |
|
Answer»

|
|
| 40. |
Which is true for an element present in group 13 of the periodic table? |
|
Answer» It is a gas at room TEMPERATURE. |
|
| 41. |
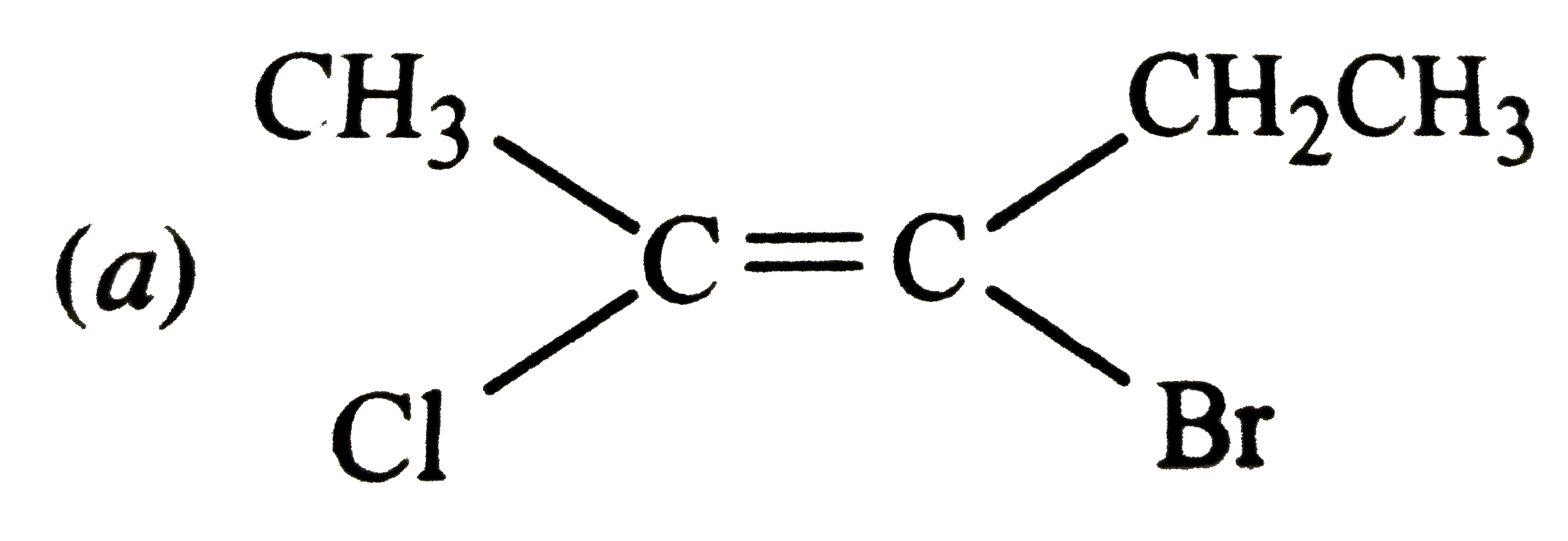
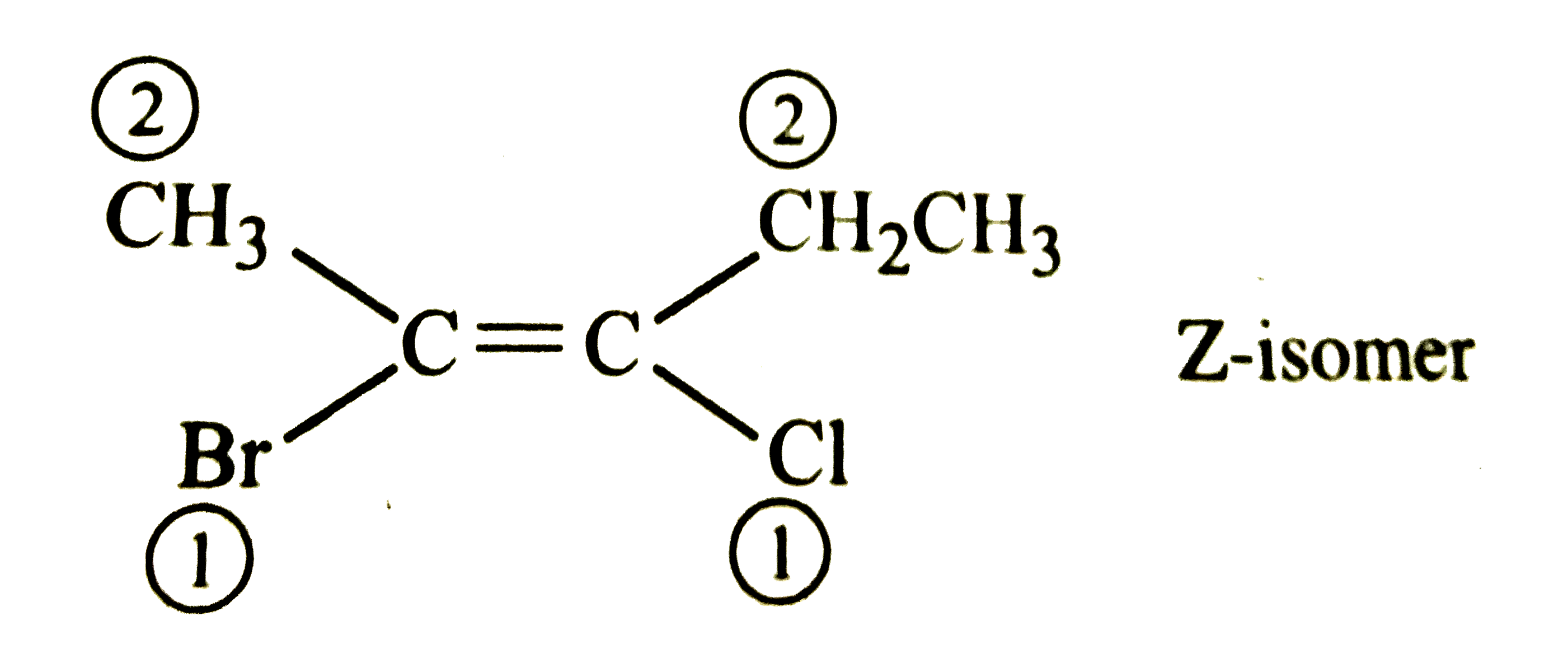
The Z-isomer among the following is |
|
Answer»
|
|
| 42. |
What is called for natural spray of liquid produced by condensation of vapours present in air ? |
| Answer» SOLUTION :mist | |
| 43. |
Which product is formed when the compoundistreated with concentrated aqueous KOH solution |
|
Answer»
|
|
| 44. |
Which experiment is responsible for finding out the charge on an electron? |
|
Answer» Millikam's oil DROP EXPERIMENT |
|
| 45. |
What is temporary hardness of water? |
| Answer» SOLUTION :Water containing BICARBONATES of CALCIUM and magnesium. | |
| 46. |
Which of the following has the lowest boiling point ? |
|
Answer» `CH_(4)` |
|
| 47. |
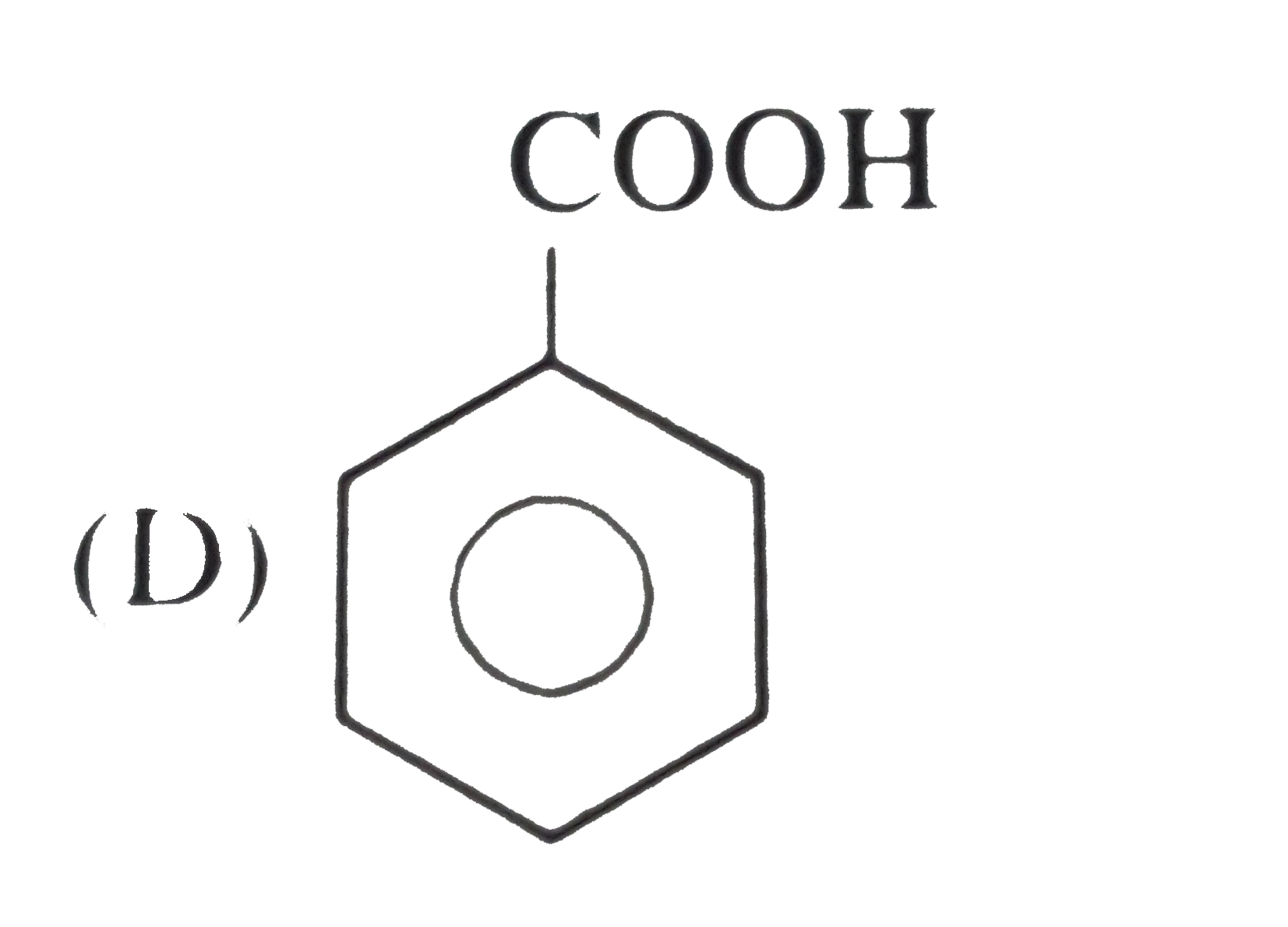
Which of the following is the weakest acid ? |
|
Answer»
|
|
| 48. |
Which of the following terms are unitless ? |
|
Answer» Molality Mole fraction `=("No. of moles of SOLUTE")/("No. of moles of solution")= ("moles")/("moles")` `=("No. of moles of SOLVENT ")/("No. of moles of solution") = ("moles")/("moles")` Mass per CENT `=("Mass of solute in gram")/("Mass of solution in gram") xx 100` |
|
| 49. |
Which of the following series of compounds have same mass percentage of carbon? |
|
Answer» `CO_(2), CO` |
|