Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What do you mean by isoelectronic species ? |
| Answer» SOLUTION :`F- (10 ELECTRONS),N^(3-), O^(2-), Ne ,Al^(3+),MG^(2+),Na^+` | |
| 2. |
Which of the following compounds will not evolve H_2 gas on reaction with alkali metals ? |
|
Answer» ethanoic acid |
|
| 3. |
What is the circumference of the second orbit of hydrogen atom? |
|
Answer» SOLUTION :`"CIRCUMFERENCE "= 2pir` `"But R"=N^(2)xx0.529Å=2^(2)xx0.529Å=2.116Å` `"Circumference "=2xx3.14xx2.116Å=13.3Å` |
|
| 4. |
van der Walls forces are maximum in the following substance |
|
Answer» HBr |
|
| 5. |
Whichp , d , forbitalsare notpossible? Why ? |
| Answer» SOLUTION :1p, 1D, 2D, 1F, 3f arenot possiblein shortdistance(r )suchorbitalsare notpossible . | |
| 6. |
Which of the following substances of lithium is most stable ? |
| Answer» ANSWER :A | |
| 7. |
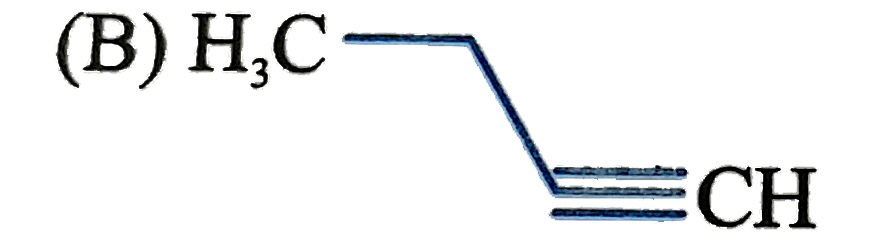
What will be the end peoduct (B) ? |
|
Answer» 1,2-Dibromobenzene |
|
| 8. |
Transitional kinetic energy of a gas molecule________ |
|
Answer» is constant at constant TEMPERATURE |
|
| 9. |
What are periodic properties ? Give example. |
|
Answer» Solution :The term PERIODICITY of properties indicates that the ELEMENTS with similar properties reappear at certain regular intervals of atomic number in the PERIODIC table. Ex 1. Atomic radii,2. Ionisation energy 3. ELECTRON AFFINITY 4. Electron negativity. |
|
| 10. |
What is the role of sodium carbonate in salt analysis |
| Answer» Solution :Water and acid insoluble SALTS are FUSED with sodium CARBONATE. Sodium carbonate EXTRACT of the salt is then tested and qualitatively ANALYSED. | |
| 11. |
The value of e/m for an electron is |
|
Answer» `1.78xx10^(8)c//g` |
|
| 12. |
Which of the following pair of orbitals possess two nodal planes |
|
Answer» `p_"xy", d_(x^2-y^2)` |
|
| 14. |
Which of the followingstatements regarding the inter molecular collisions in a gas is not correct |
|
Answer» DUE to INTER molecular collisions , directions of motion of molecules CHANGES |
|
| 15. |
Writethe electronicconfigurationof element withatomicnumbers89and 90and predictwhetherthey ared- orf- blockelements . |
|
Answer» Solution :Electronicconfigurationof Rn (Z =86) is`[Xe]^(54)4f^(14)5d^(10)6s^(2)6p^(6)` Afterfilling6p- subshellfilling 7S- subshellstartsat Fr (Z= 87)= `[Rn]^(86)7s^(1)` and iscompleteat Ra `(z=88)= [Rn]^(86)7s^(2). ` Thereforethe fillingof 5f- subshellshould beginin ACCORDANCEWITH aufbauprinciple. But DUETO (i) stillsmallerenergydifference between5f- and 6d- orbitalsthan between4f- and5d- orbitalsand (ii)greaterstabilityof theradon core thenexttwo electrons(i.e.,89 thand 90 th)afterthefilling of 7s- orbitalsprefer to enter 6d- orbitalsbeforefillingthe 5f- ORBITALS. Thusthe electronicconfigurationare : `Ac (Z=89)= [ Rn]^(86)7s^(2)6d^(1)` andThe `(Z= 90)= [Rn ] ^(86)7s^(2)6d^(2)` Hereagain theyshould becalledd- blockelementsbut arecalledf-blockelements becausethe INNER 5f- orbitalsare stillempty. |
|
| 16. |
Which of the following reactions is classified as an ene reaction? |
|
Answer»
|
|
| 17. |
when NaCI solution is added to Fe(OH)_3sol then : |
|
Answer» `[Fe(OH)_3]Fe^(3+)` is FORMED |
|
| 18. |
Which of the following statements is right |
|
Answer» All caboxylic acids exhibits functional isomers as esters |
|
| 19. |
What is the Kelvin temperature or Absolute temperature ? |
| Answer» SOLUTION :ABSOLUTE temperature is `0^(@)C` and KELVIN temperature is 273.15 K. | |
| 20. |
Which product is obtained in reaction of methanal in the presence of concentrated NaOH? |
|
Answer» `CO + H_(2)` |
|
| 21. |
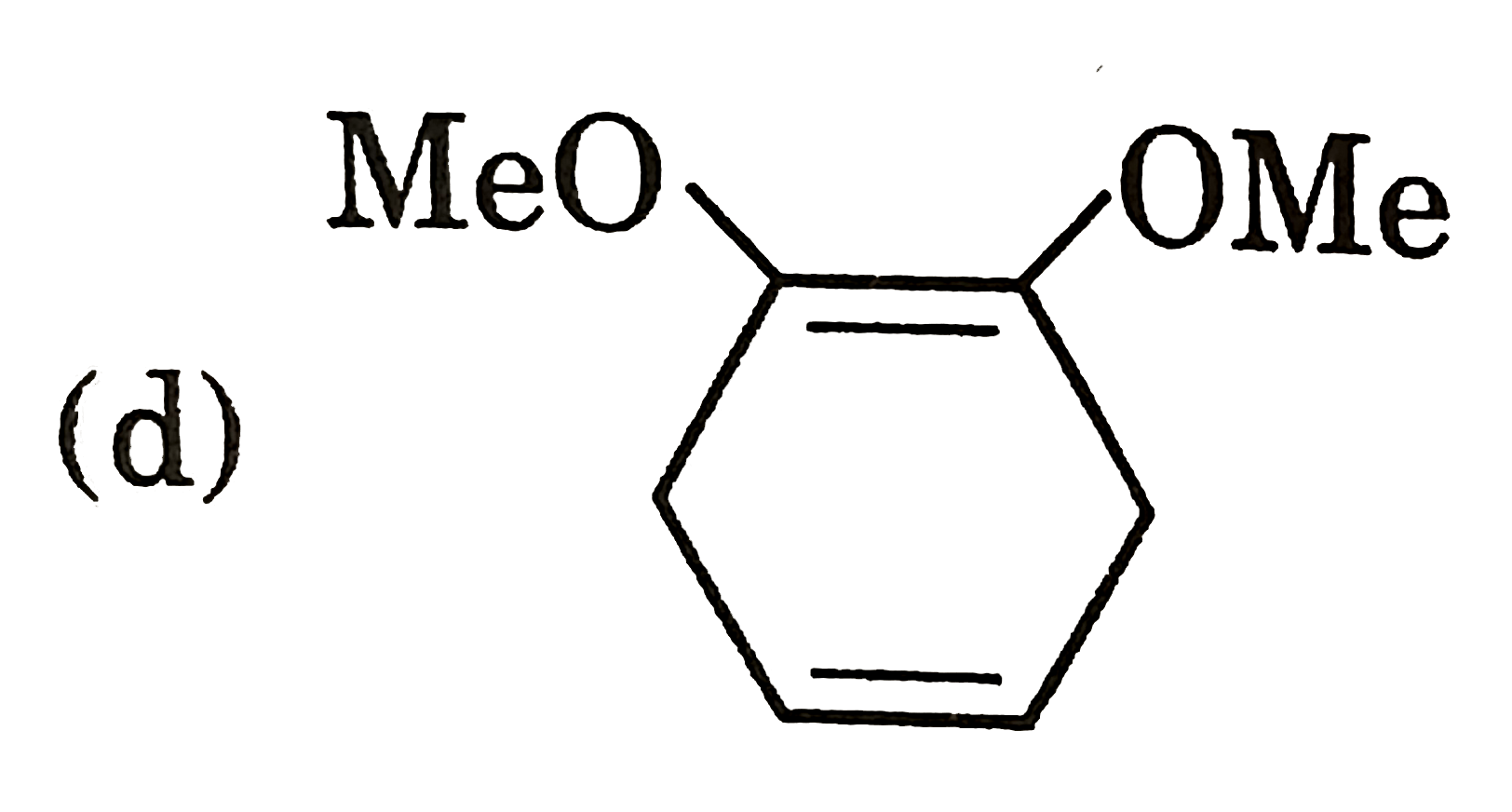
Draw energy level diagram for He-2 molecule.Calculate its bond order. |
Answer» Solution :ATOMIC number of HELIUM is `2:1s^(2)` 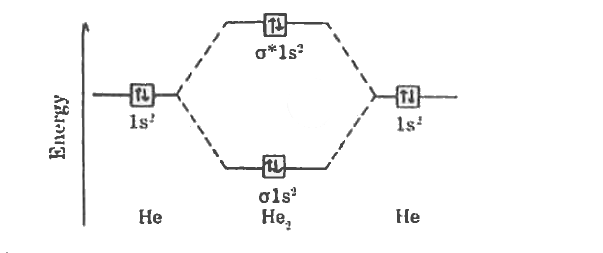 (a) Electronic CONFIGURATION of `He_(2)` MOLECULE is `s1s^(2)s^(**)s1s^(2)` (2) Bond ORDER `=(2-2)/(2)=0` © Magnetic property = diamagnetic. |
|
| 22. |
Which of the following is not a surfactant ? |
|
Answer» `CH_(3)-(CH_(2))_(15)-underset(CH_(3))underset(|) overset(CH_(3))overset(|)(N^(+))-CH_(3)Br^(-)` |
|
| 23. |
Which of the following statements is correct for CsBr_(3) ? |
|
Answer» It is a COVALENT compound <BR>It CONTAINS `CS^(3+)` and `Br^(-)` ions |
|
| 24. |
Using stock notation, represent the following compounds: a. HAuBr_(4) ,b. Tl_(2)O ,c. FeO , d. Fe_(2)O_(3), e. CuBr, f. CuO g. MnO, h. MnO_(2) |
Answer» SOLUTION :
|
|
| 25. |
Which of the following statement(s) is/are correct? |
|
Answer» the system of constant entropy and constant volume will attain the equilibrium in a STATE of minimum energy |
|
| 26. |
Which type of electronic effects seen in structure of molecule? |
| Answer» SOLUTION :(i) INDUCTIVE effect (ii) RESONANCE effect (iii) hyperconjugation effect. (iv) electromeric effect: In presence of REAGENT | |
| 27. |
What is difference between vapour and gas ? |
| Answer» Solution :When temperature of GAS DECREASES than its `T_(C )` then its CONVERTED in to vapour. If we apply pressure liquification occurs. | |
| 28. |
What would be the molality of 20% (mass/mass) aqueous solution of Kl? (molar mass of Kl = 166 g mol^(-1)) |
|
Answer» 1.51 |
|
| 29. |
What is the oxidation number of the underlined atoms in each of the following molecules / ions ? (a)ulClO_3^(-) (b) ulBrF_3 (c) ulCH_4 (d) ulC_6H_(12)O_6 (e) Na_2ulB_4O_7 (f) Na_4|__Fe(CN)_6] (g) ulN_2H_4. |
|
Answer» Solution :Suppose x be the oxidation number of the UNDERLINED atom : `{:((a),ClO_3^(-),x+(-2xx3)=-1),(,,x-6=-1" or "x=+5),((b),BrF_3,x+(-1xx3)=0),(,,x-3=0" or "x=+3),((c),CH_4,x+1(1xx4)=0),(,,x+4=0" or "x =-4),((d),C_6H_(12)O_6,6xxx+1xx12+(-2xx6)=0),(,,6x+12-12=0" or "x=0),((e),Na_4[FE(CN)_6],(+1xx2)+(xxx4)+(-2xx7)=0),(,,+2+4x-14=0 " or "x=+3),((f),Na_4[Fe(CN)_6],(+1xx4)+x+1(-1xx6)=0),(,,+4+x-6="or "x=+2),((G),N_2H_4,2x+4=0" or "x=-2):}` |
|
| 30. |
Write Schrodinger wave equation in the briefest possible form |
| Answer» Solution :`HAT(H) PSI = E Psi " where " hat(H)` is CALLED Hamiltonian OPERATOR. | |
| 31. |
Write the Lewis dot symbols and predict the valencies you expect for the following elements : Nitrogen , Fluorine and Neon |
Answer» SOLUTION :
|
|
| 32. |
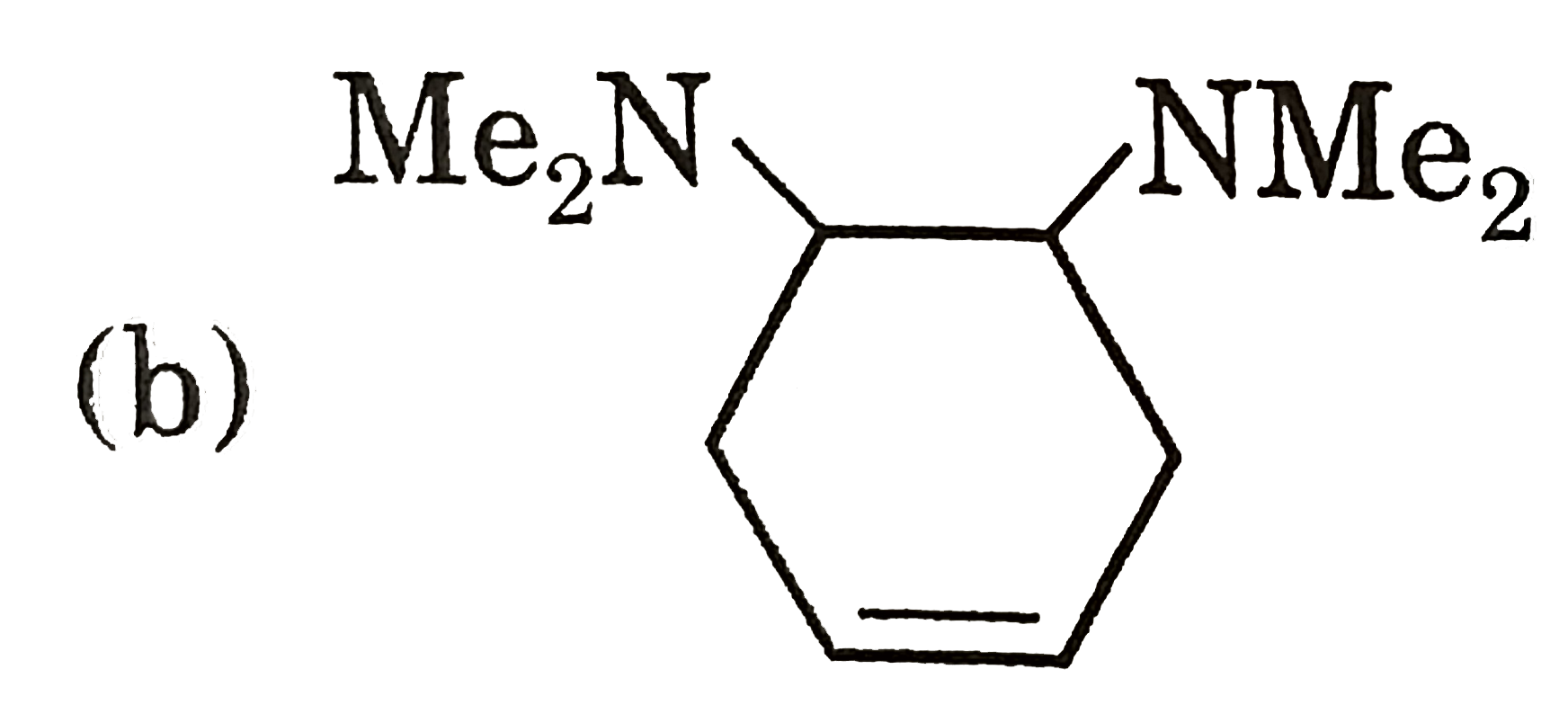
Which of the following order is/are correct for the rate of E2 reaction ? |
|
Answer» 5-Bromocycloheptene GT 4-Bromocycloheptene |
|
| 33. |
The work function for caesium atom is 1.9 eV. Calculate (a) the threshold wavelength and (b) the threshold frequency of the radiation. If the caesium element is irradiated with a wavelength 500 nm, calculate the kinetic energy and the velocity of the ejected photoelectron. |
|
Answer» Solution :(a) WORK function `(W_(0)) = h v_(0)` `:. v_(0) = (W_(0))/(h) = (1.9 xx 1.602 xx 10^(-19)J)/(6.626 xx 10^(-34) Js) = 4.59 xx 10^(14) s^(-1) (1 EV = 1.602 xx 10^(-19)J)` (B) `lamda_(0) = (c)/(v_(0)) = (3.0 xx 10^(8) ms^(-1))/(4.59 xx 10^(14) s^(-1)) = 6.54 xx 10^(-7) m = 654 xx 10^(-9) m = 654nm` (c) K.E. of ejected electron `= h (v - v_(0)) = hc ((1)/(lamda) - (1)/(lamda_(0)))` `= (6.626 xx 10^(-34) Js) (3.0 xx 10^(8) ms^(-1)) ((1)/(500 xx 10^(-9) m) - (1)/(654 xx 10^(-9) m))` `= (6.626 xx 3.0 xx 10^(-26))/(10^(-9)) ((154)/(500 xx 654)) J = 9.36 xx 10^(-20)J` K.E. `= (1)/(2) mv^(2) =9.36 xx 10^(-20) J or kg m^(2) s^(-2)` `:. (1)/(2) xx (9.11 xx 10^(-31) kg) v^(2) = 9.36 xx 10^(-20) kg m^(2) s^(-2)` or `v^(2) = 2.055 xx 10^(11) m^(2) s^(-2) = 20.55 xx 10^(10) m^(2) s^(-2) or v = 4.53 xx 10^(5) ms^(-1)` |
|
| 35. |
What happens whensodium peroxide dissolves in water? |
| Answer» SOLUTION :`Na_2O_2+2H_2O RARR 2NaOH+H_2O_2` | |
| 36. |
Which law of thermodynamics deals with equivalence of different forms of energies? |
| Answer» SOLUTION :FIRST LAW of THERMODYNAMICS. | |
| 37. |
The root mean square velocity of an ideal gas at constant pressure varies with density d as |
| Answer» ANSWER :D | |
| 38. |
Which carbide liberates methane gas on hydrolysis |
|
Answer» `Be_(2)C` |
|
| 39. |
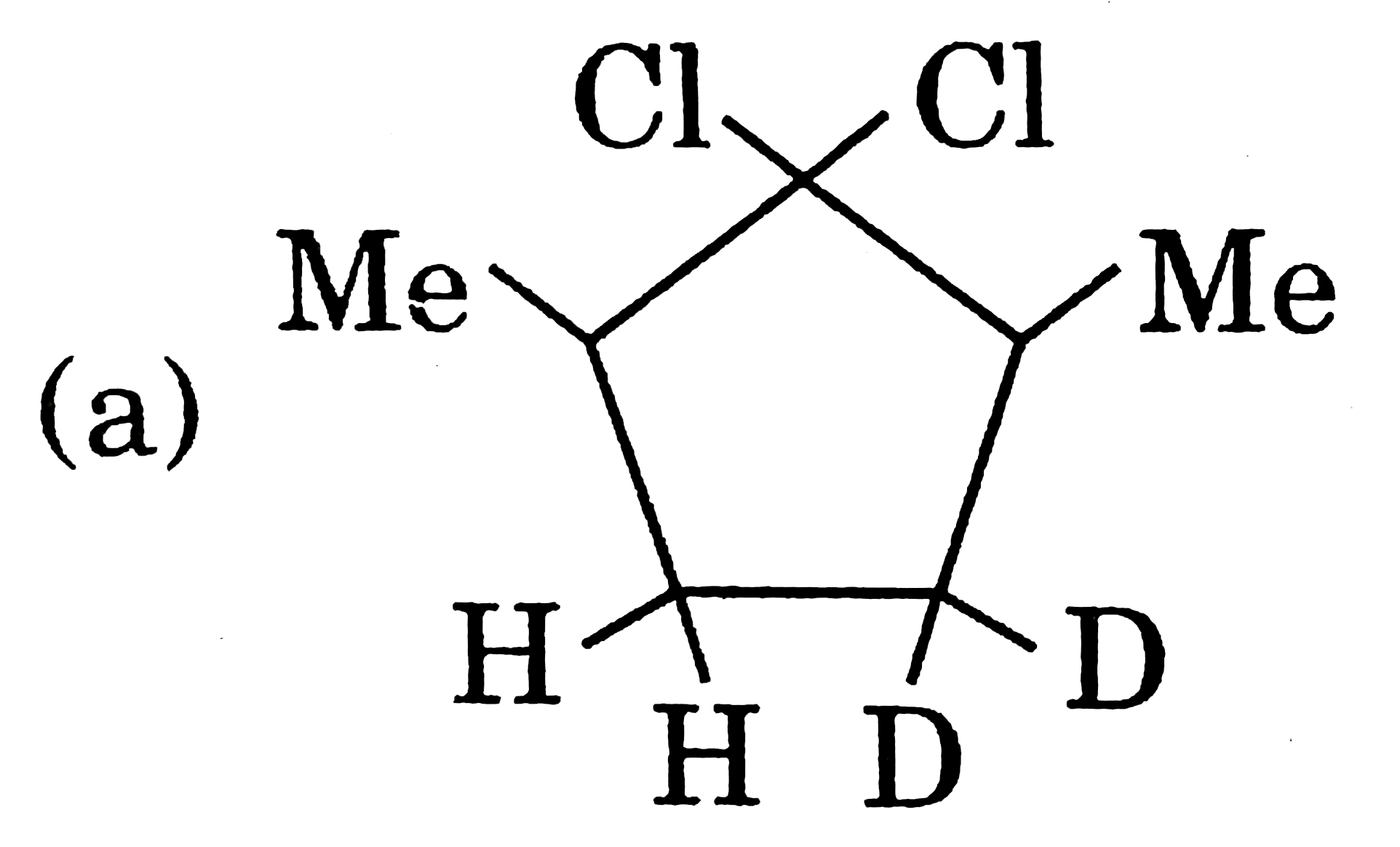
Which of the following structure is least stable? |
|
Answer»
|
|
| 40. |
The solution in water sulphates down theBe group is : Be gt Mg gt Ca gt Sr gt Ba . This is due to |
|
Answer» INCREASE in MELTING GROUP |
|
| 41. |
Which of the following compounds will produce butanone when treated with H_(2)SO_(4) , HgSO_(4) and water? |
|
Answer» `CH_(3)-CH_(2)-CH=CH_(2)` |
|
| 42. |
Which of the following expression for equilibrium constant of the reaction 2N_2O_((g)) + O_(2(g)) hArr 4NO_((g)) ? |
|
Answer» `([N_2][O_2])/[NO]^4` |
|
| 43. |
What will be the pressure of 10 gram of a gas kept under atmospheric pressure, if its temperature is changed from 546 K to 273 K ? |
|
Answer» `(1)/(2)` BAR Initial pressure = ATMOSPHERIC pressure `= P_(1)=1` bar Final temp. `= T_(2)=273` Kelvin. Final pressure `= P_(2)=` According to Gay Lucca.s Law : `P prop T` (n.V constant) So, the temperature will be 273 K from 546 K. Means if we half it, them the pressure will also be halved, it will be `(1)/(2)` bar from 1 bar. OR `(P_(1))/(T_(1))=(P_(2))/(T_(2))` (n.V constant) `therefore ("1 bar")/("546 kelvin")=(P_(2))/("273 kelvin")` `therefore P_(2)=("273 kelving" xx"1 bar")/("546 kelvin")=(1)/(2)` bar |
|
| 44. |
what is the distance betweenNa^(+) and Cl^(-)in a NaCl crystal if its density is 2.165"g cm"^(-3)? NaCl crystallises in the fcc lattice. |
|
Answer» |
|
| 45. |
To a solution of excess of KI in dilute H_(2)SO_(4). 25 mL of an unknown solution of KMnO_(4) were added. The liberated iodine was exactly reduced by 42.5 mL of N//10 Na_(2)S_(2)O_(3) solution. Calculate the concentration of KMnO_(4) solution. |
|
Answer» |
|
| 46. |
Which will show geometrical isomerism? |
|
Answer»
|
|
| 47. |
Which one of the following molecules has the smallest bond angle ? |
|
Answer» `NH_(3)` |
|
| 48. |
Which of the following is not a redox reaction? |
|
Answer» `H_(2) + F_(2) to 2HF` |
|
| 49. |
What is theelectronicconfigurationof the elementwith (i)Z= 57 . Is ita d- orf- blockelement ? |
|
Answer» Solution :As explainedin the foot thenote onpage`3//14`the electronicconfigurationof ` ( La = 57 ) " is"1 s^(2)2S^(2)2p^(6) 3S^(2)3p^(6)3d^(10)4s^(2)4p^(6)4d^(10)5s^(2)5s^(6) 6S^(2)5d^(1)` instead of the EXPECTED `1s^(2)2s^(2) 2p^(6) 3s^(2) 3p^(6)3d^(10) 4s^(2) 4p^(6) 4d^(10) 5s^(2) 6s^(2) 4f^(1)` Sincethe lastelectronenters the 5d- orbitalit is calledd- blockelementand not thef-blockelement . |
|
| 50. |
The well known antipyretic 2-Acetoxy benzoic acid is obtained when |
|
Answer» Salicylic acid is treated with phenol in the PRESENCE of `POCl_(3)` 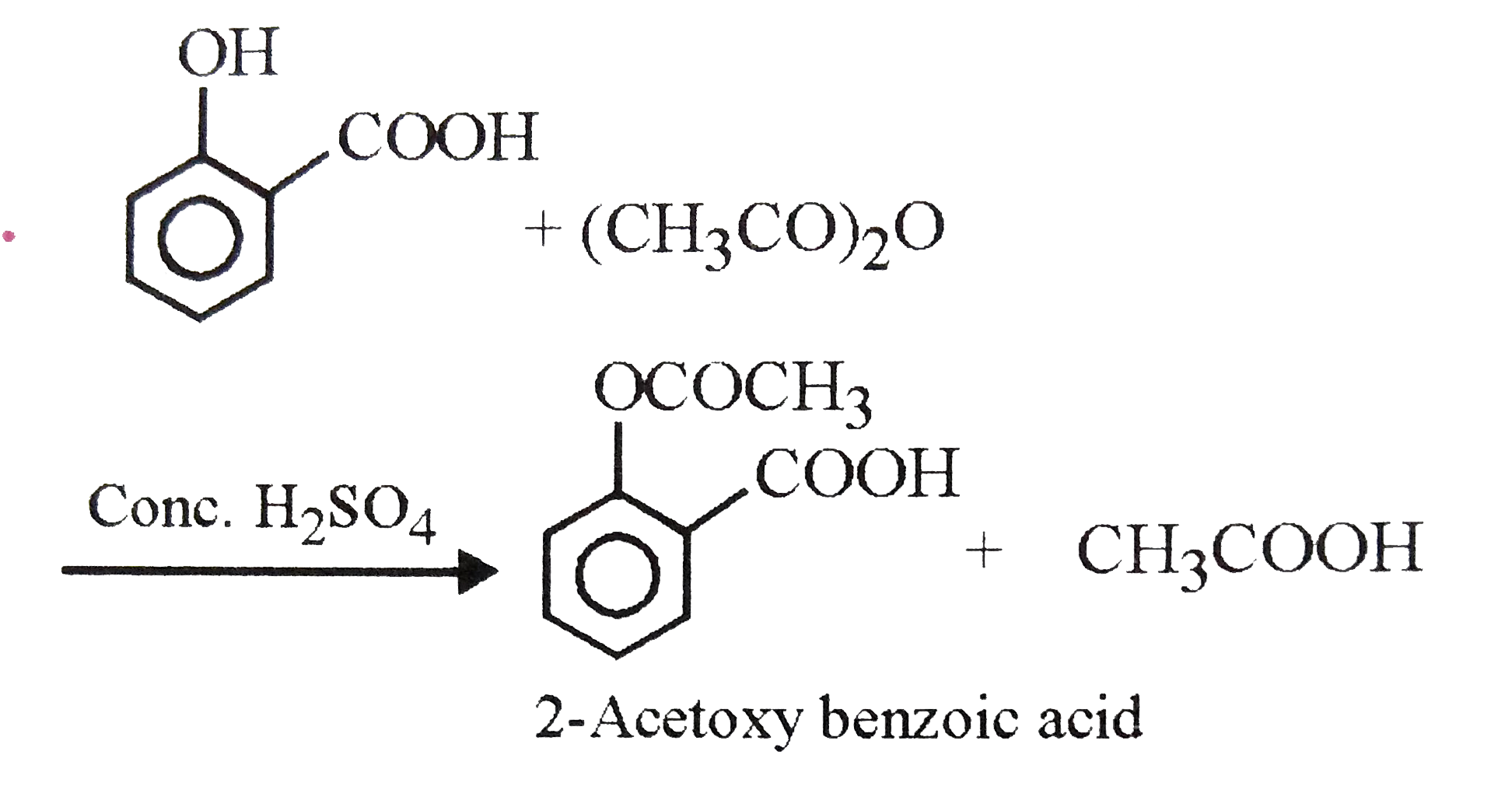
|
|