Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When ethyl bromide is added to potassium t-butoxide, the product is ethyl t-butyl ether. underset("ethyl bromide")(CH_3CH_2-Br)+underset("ptassium t-butoxide")((CH_3)_3C -OK)tounderset("ethyl t-butyl")((CH_3)_3C-O)-underset("ether")(CH_2CH_3) Which of the following statements is/are correct ? |
|
Answer» when the concentration of ETHYL bromide is DOUBLED rate is also doubled. |
|
| 2. |
Which one is the more acidic in aqueous solution ? |
|
Answer» `NiCl_2` |
|
| 3. |
Which are only Lewis acid but not Bronsted acid of CO_2, BCl_3, NH_3, CH_3NH_2, NO_2^+ and C_6H_5NH_3^+ ? |
| Answer» SOLUTION :`CO_2 , BCl_3 , NO_2^+` | |
| 4. |
What are the four prominent lines in Balmer series of hydrogen spectrum? |
|
Answer» |
|
| 5. |
Whichorbitalsare equinian orbitalsfrom the given ? |
|
Answer» SOLUTION :(i) `3d_(XY), 3d_(Z),3d_(xz)`areequivalent . (ii) `4d_(xy) ,4d_(yz),4d_(z^(2))` areequivalent |
|
| 6. |
Which statement is correct for H_3PO_3 & H_3PO_4 ? |
|
Answer» `H_3PO_3` is a MONO basic and reducing AGENT. |
|
| 8. |
Write the common names of the following halides (a) CH_(3)-CH-CH_(2)Br b) CH_(3)CH_(2)CHCl_(2) c) CH_(2)-overset(I)overset(|)underset(I)underset(|)C-CH_(3) (d) CHBr_(3) e) C Cl_(4) |
|
Answer» Solution :a. Isobutyl bromide b. Propylidene chloride C. Propylidene IODIDE d. Bromoform E. CARBON tetra chloride. |
|
| 9. |
Which one of the following is incorrect statement ? |
|
Answer» for a system at equilibrium, Q is always less than the equilibrium constant. |
|
| 10. |
What type of atomic orbitals can oversap to form molecular orbitals ? |
| Answer» Solution :Atomic ORBITALS with COMPARABLE ENERGIES and proper orientaiton. | |
| 11. |
Which element do you think would have been named by Lawrence Berkeley laboratory. |
| Answer» SOLUTION :LW (LAWRENCIUM). BK (BERKELIUM) | |
| 12. |
What are the conditions under which real gases approach ideal behaviour? |
| Answer» SOLUTION :Only at HIGH TEMPERATURE and LOW PRESSURE. | |
| 13. |
Who modified Bohr's theory by introducing elliptical orbits for electrons path? |
|
Answer» RUTHERFORD |
|
| 14. |
The sub-shells filled one by one for 4th period elements are |
|
Answer» 3D, 4S and 4P |
|
| 15. |
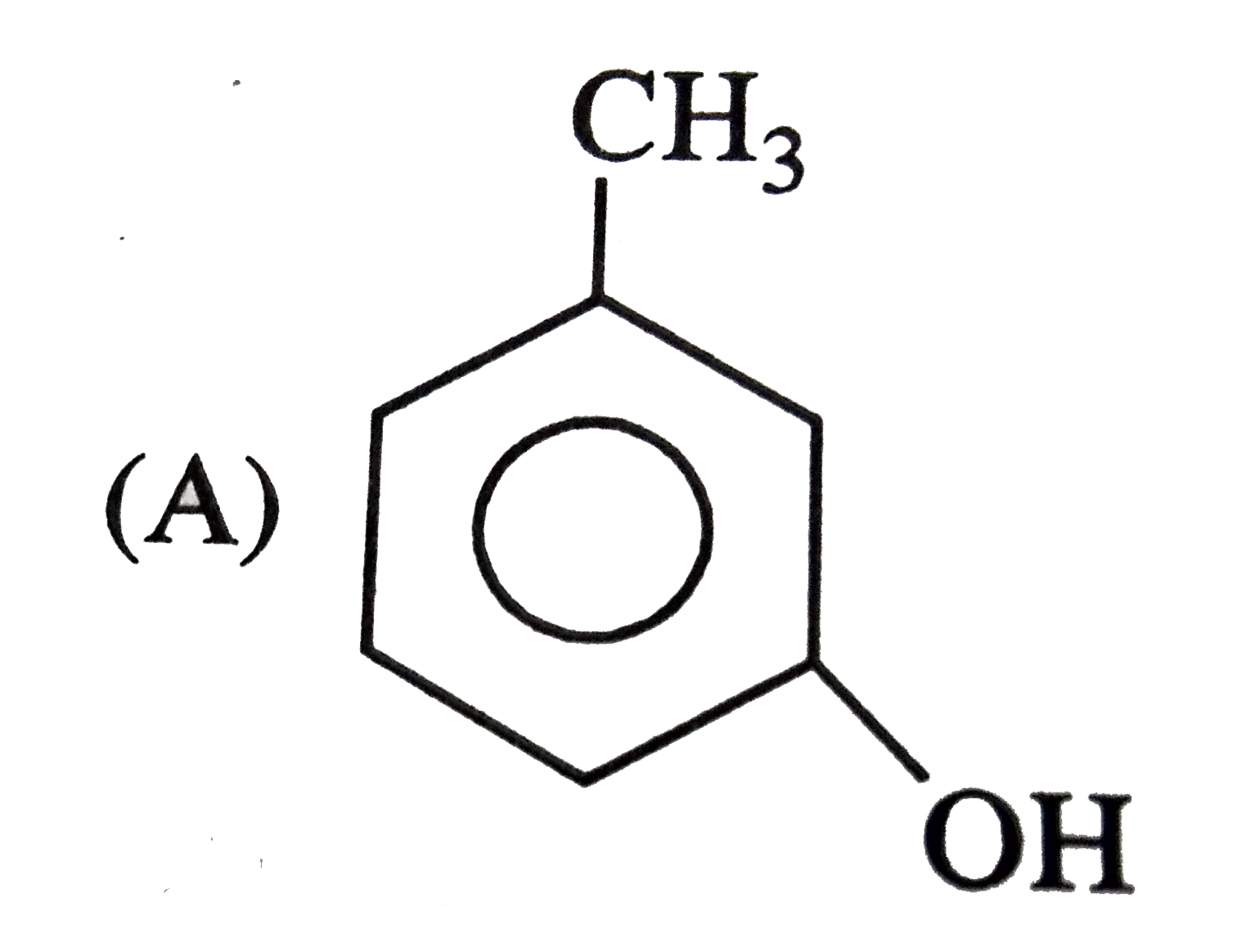
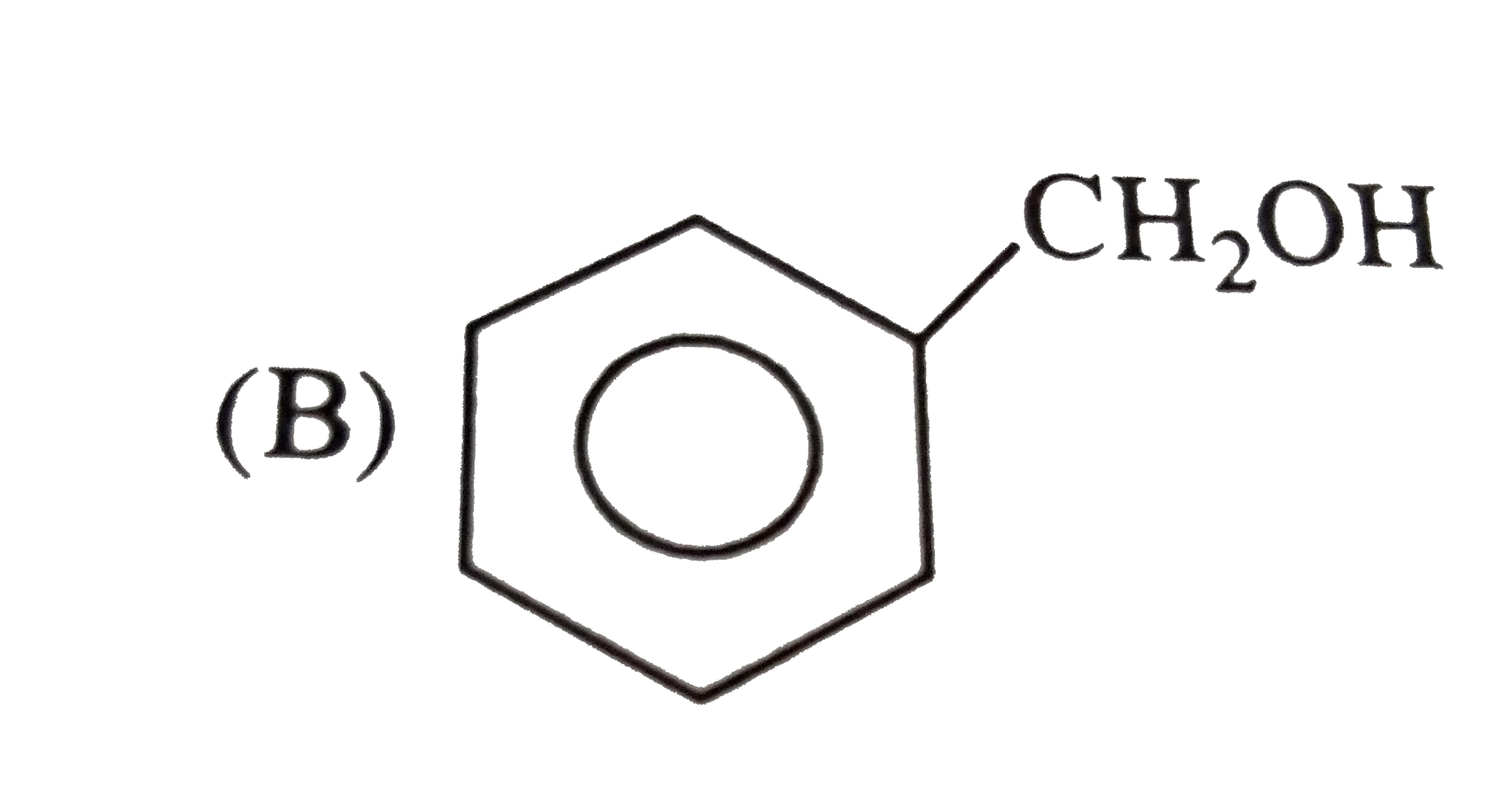
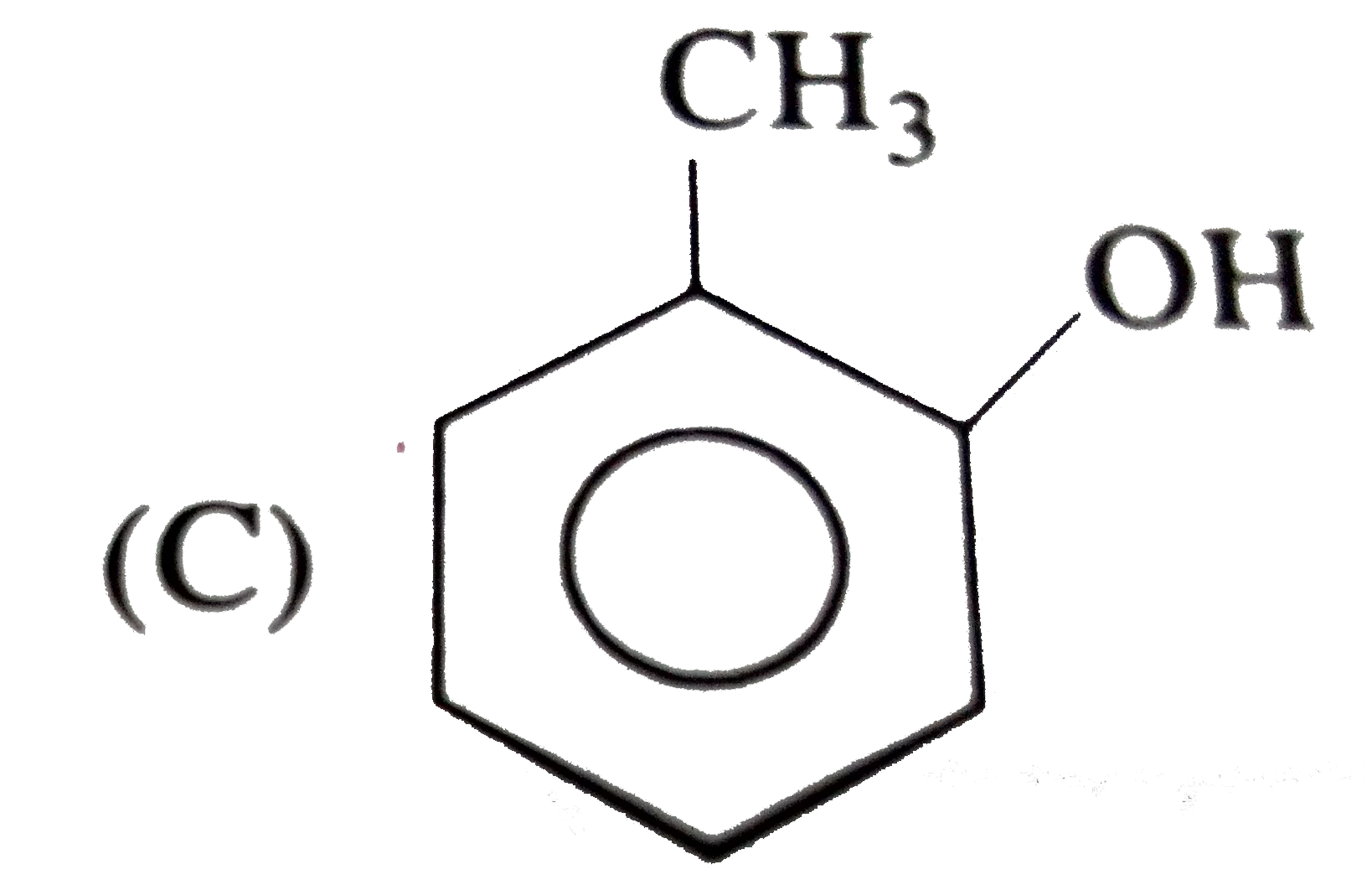
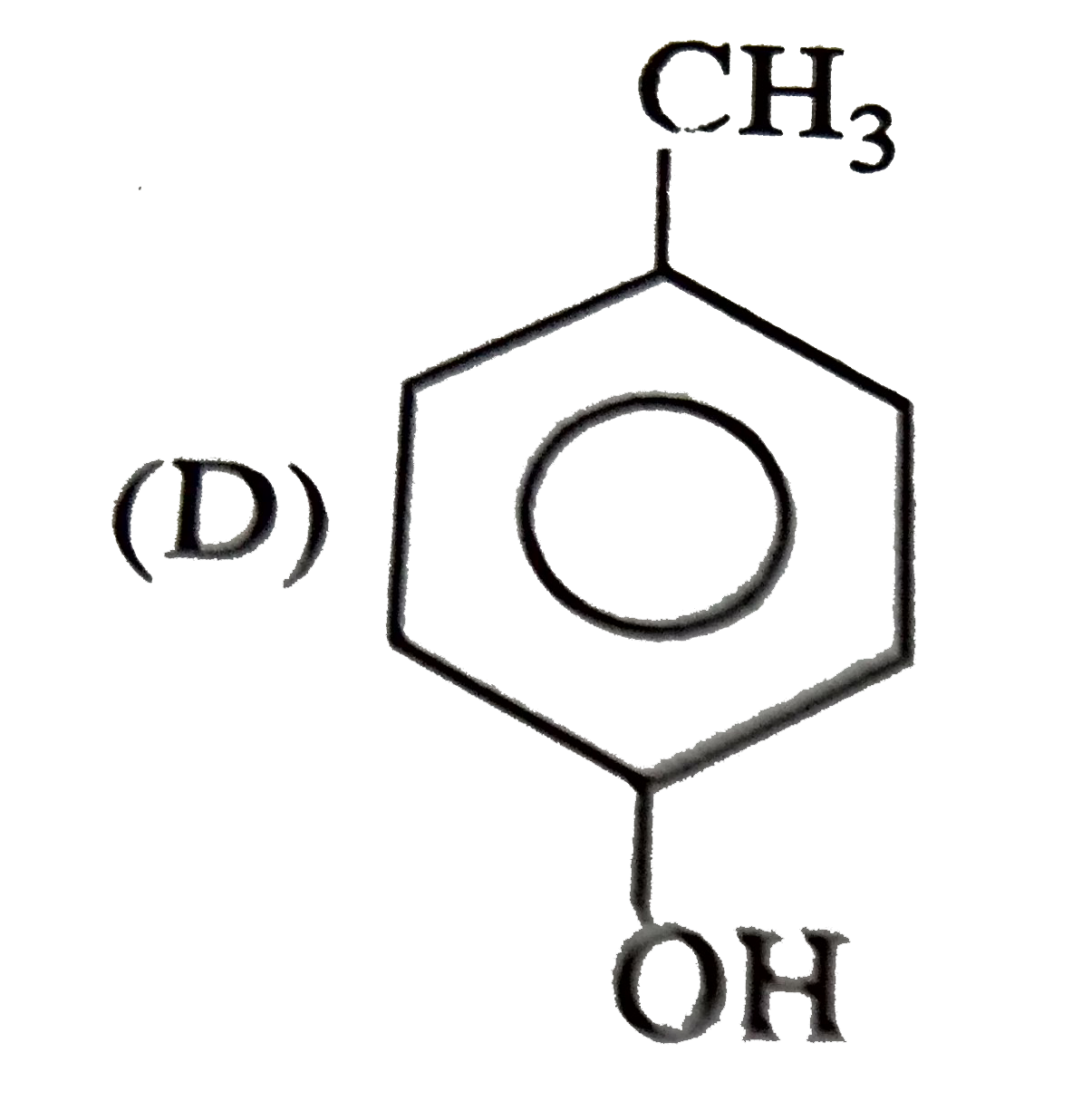
The structure of the compound that gives a trobromo derivative on treatment with bromine water is |
|
Answer»
|
|
| 16. |
What is a functional group ? Write the functional groups of the following : (i) Thioalcohol (ii) Isothiocyanate (iii) Thiocyanate and (iv) Sulphonic acid (v) Sulphones (vi) Sulphoxides. |
Answer» 
|
|
| 17. |
What is haemolysis? Why intravenous fluid are isotonic to blood ? |
|
Answer» Solution :(i) The osmotic pressure of the blood cells is approximately equal to 7 atm at `37^(@)C.` (ii) The intarvenous injcctions should have same osmotic pressure as that of the blood (ISOTONIC with blood). (iii) If the intervenous SOLUTIONS are too dilute that is hypotonic, the solvent from OUTSIDE of the cells FLOW into the cell to normalise the osmotic pressure and this process is called haemolysis causes the cells to burst. (iv) On the other. hand, if the solution is too concentrated, that is hypertonic, the solvent molecules will flow out of the cells, which causes the cells to shrink and DIE. (v) For this reason, the intervenous fluids are prepared such that they ar istonic to blood (`0,9%` mass/volume sodium chloride solution). |
|
| 18. |
What is oxidation number of .C. in C_(3)O_(2) ? |
| Answer» SOLUTION :The OXIDATION NUMBER of .C. in `C_(3)O_(2)" is "+4/3`. | |
| 19. |
Which of the following is incorrect about SI units ? |
|
Answer» Density in `KGM^(-3)` |
|
| 20. |
The uncertainity principle and the concept of wave nature of matter were proposed by----- and ---- respectively |
|
Answer» PAULI, Hund |
|
| 21. |
Which one of the following will have greatest average speed of its molecules ? |
|
Answer» 0.5 mol of `O_(2)` at 500 K `O_(2)=sqrt((500)/(2))=3.95,CO_(2)=sqrt((400)/(44))=3.0` `He=sqrt((200)/(4))=7.0, NH_(3)=sqrt((300)/(17))=4.2` Thus, He at 200 K will have highest average speed. |
|
| 22. |
What happened when more solution of CdCl_2 is added in saturated solution of CdS and starred? |
|
Answer» No EFFECT. |
|
| 23. |
What is molar volume of gas ? What is its value. |
|
Answer» Solution : ..Volume of 1 mole IDEAL gas at STP is known as molar volume of ideal gas... At STP (273.15 K temperature and 1 BAR PRESSURE) the volume of 1 mole ideal gas is 22.710981 L `mol^(-1)` it is molar volume. |
|
| 24. |
Which one of the following is a standard solution ? |
|
Answer» It contains one GRAM EQUIVALENT mass of the substance in one LITRE solution |
|
| 25. |
Which of the following statements is correct |
|
Answer» oxidation number of Fe in `[Fe(H_(2)O)_(5)NO]SO_(4)` is +1 `therefore =+1` Thus the O.N of Fe is +1 and the correct structure of `[Fe(H_(2)O)_(5)overset(+)NO]SO_(4)^(2-)` |
|
| 26. |
Which of the following is not lyophilic |
|
Answer» Gelatin sol (a) is not CORRECT |
|
| 27. |
What is the steps involve in the calculation of molecular formula from empirical formula ? |
|
Answer» SOLUTION :MOLECULAR mass and empirical formula are used to deduce molecular formula of the compound . Steps to calculate molecular formula: 1. Empirical formula is found out from the percentage composition of elements 2. Empirical formula mass can be found from the empirical formula 3. Molecular mass is found out from the GIVEN data 4. Molecular formula = `("Empirical formula")_(N)` 5. where, n = `("Molecular mass")/("Empirical formula mass")` |
|
| 28. |
Which of the following compounds will show cis-trans isomerism? |
|
Answer» `(CH_(3))_(2)C=CHC_(2)H_(5)` 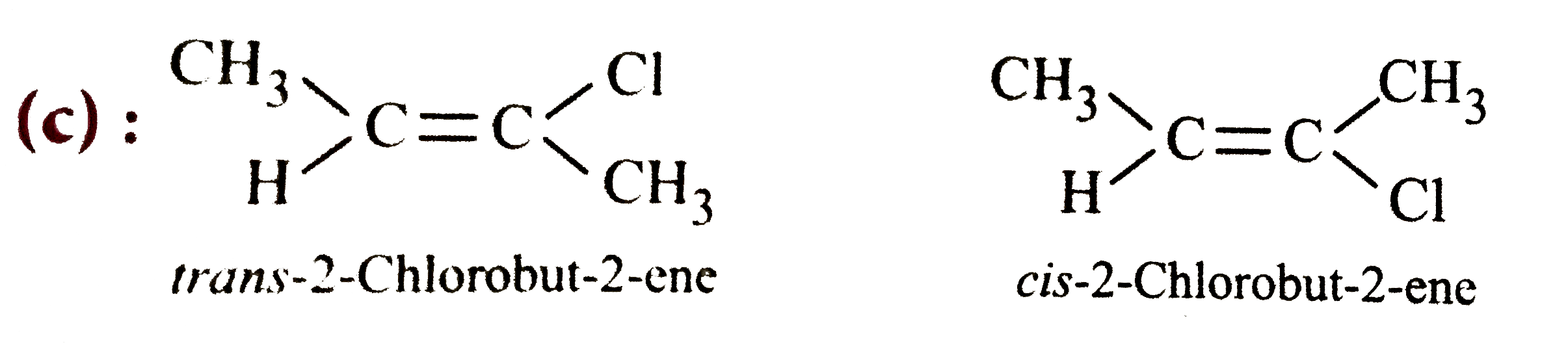
|
|
| 29. |
Two moles of ammonia occupy a volume of 5 L at 27^@C and 9.32 atm pressure. Calculate the compressibility factor of the gas. |
|
Answer» Solution :In the PRESENT case, `P= 9.32" atm, " "" V=5L, ""T = 27 + 273 = 300 K` `n = 2, " and " "" R=0.0821 L " atm " K^(-1) mol^(-1)` The COMPRESSIBILITY factor Z is given by `Z=(PV)/(nRT)` `:. "" Z= (9.32xx5)/(2xx0.0821xx300) =0.946` Hence, the compressibility factor for NH3 under the given conditions is 0.946. |
|
| 30. |
The sizesof followingspeciesincrease in theorder : |
|
Answer» `MG^(2+) LT Na^(+)lt F^(-)lt A1` |
|
| 31. |
Which of the following set of raw materials are used in the manufacture of Na_(2)CO_(3) by Solvay process? |
|
Answer» `Ca(OH)_(2), NH_(3).CO_(2)` |
|
| 32. |
What is obnormal molar mass? |
| Answer» Solution :The dissociation or ASSOCIATION of SOLUTE molecules would ALTER the total number of particles present in the solution and hence affect the RESULTS of measured colligative PROPERTIES.In such solution determined using colligative properties would be different from the actual molar mass and it is called abnormal molar mass | |
| 33. |
What is meant by 'autoprotolysis' of water? What is its significance? |
| Answer» Solution :SELF IONISATION of water is CALLED autoprotolysis of water`H_2O+iffH_3O^+ +OH-`This makes water CAPABLE of reacting with both acids and bases(AMPHOTERIC). | |
| 34. |
The systematic name of (CH_(3))_(2)CH- COOH |
|
Answer» 2-Propanoic acid |
|
| 35. |
What are electrophiles and nucleophiles ? Explain with examples |
Answer» Solution : When the BONDING interaction take place between ELECTROPHILIC REAGENT and nucleophilic reagent, then electrophilic reagent accept ELECTRON pair |
|
| 36. |
What is present in troposphere ? |
| Answer» SOLUTION :TROPOSPHERE contains substances like dioxygen, dinitrogen, CARBON dioxide, water vapour, ETC. | |
| 37. |
Write equilibrium constant (equilibrium law) for the following reaction. (i)4NH_(3(g)) + 5O_(2(g)) hArr 4NO_((g)) + 6H_2O_((g)) (ii)H_(2(g)) + I_(2(g)) hArr 2HI_((g)) (iii)N_(2(g)) + 3H_(2(g)) hArr 2NH_(3(g)) |
|
Answer» Solution :(i)`K_c=([NO]^4 [H_2O]^6)/([NH_3]^4 [O_2]^5)`…..(EQ.-i) (ii)`K_c="[HI]"^2/([H_2][I_2])`…(Eq. -ii) (iii) `K_c=([NH_3]^2)/([N_2][H_2]^3)`….(Eq. -iii) |
|
| 38. |
What will be the mass of oxygen liberated by decomposition of 200 mL hydrogen peroxide solution with a strength of 34 g per litre ? |
|
Answer» `25.5 g` 1 L of the solution contains 34 g of `H_(2)O_(2)` 200 mL of the solution contains `(34)/(1000)xx200=6.8 g` of `H_(2)O_(2)` `underset(2xx34 g)(2H_(2)O_(2))to 2H_(2)O+underset(32 g)(O_(2))` 68 g of `H_(2)O_(2)` GIVES 32 g of `O_(2)` 6.8 g of `H_(2)O_(2)` gives `= (32)/(68)xx6.8=3.2g` of `O_(2)` |
|
| 39. |
Zeolites are used as 1) ion exchangers 2) molecular sieves 3) water softener The correct uses are |
|
Answer» 1,2 only |
|
| 40. |
Which of the following elements does not show allotropy ? |
|
Answer» NITROGEN |
|
| 41. |
Which of the following has pentagonal bipyramidal shape? |
|
Answer» `PCl_(5)` |
|
| 42. |
What is adsorption chromatography? Write its type |
|
Answer» Solution :This method is based upon the differential adsorption of the various components of a mixture on a suitable adsorbent. Since same compounds are more STRONGLY desorbed than the other, they will travel though the column at different rates and thus get separated. Following are two main types of chromatographic technique based on the principle of different adsorption. (i) Column chromatography and (II) THIN LAYER chromatography |
|
| 43. |
Which of the following has least bond energy? |
|
Answer» `F_2` |
|
| 44. |
Which of the following statement is not true about amorphous solids ? |
|
Answer» On heating they MAY BECOME crystalline at certain temperature |
|
| 45. |
Which of the following thermodynamic relation is correct ? |
|
Answer» `dG =V d p -SdT` `:. G= U + PV - TS` Differentiating it completely ,we GET `dG=dU +PdV+ VdP - TdS - SdT`…(i) But `dU =DQ+dw` and `w= -PdV` `:. dU = d-PdV` or `dq = dU+ PdV`…(ii) Further, for averversible process, `dS= ( dq)/(T)` or `dq=TdS`....(iii) From (ii)and (iii), `dU +PdV=T DeltaS` Substituting this in (i),weget`dG =VdP - SdT` |
|
| 46. |
Which of the following statements is false? |
|
Answer» Work is a STATE function |
|
| 47. |
What [NH_4^(+) ] in a solution that contains 0.02M NH_3(K_b =1.8 xx 10^(-5) ) and 0.01 M KOH ? |
|
Answer» `9 xx 10^(-6)` ` [NH_4^(+)] =3.6 xx 10 ^(_5) ` |
|
| 48. |
Tile 4f-subshell is successively filled for |
|
Answer» RARE earths |
|
| 49. |
Which of the following is not a thermodynamic function? |
|
Answer» INTERNAL energy |
|