Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the pH of a buffer solution prepared by dissolving 0.1 mole of sodium acetate and 0.2 mole of acetic acid in enough water to make a dm^(3) of solution? (K_(a) of acetic acid = 1.8xx10^(-5)). |
|
Answer» Solution :`pK_(a)=-logK_(a)` = `-log1.8xx10^(-5)` = -(5.2553) = 4.7447 `pH=pK_(a)+"LOG"(["salt"])/(["ACID"])` = `4.74+"log"0.1/0.2` = `474+log0.5` = 4.74 - 0.3010 = 4.44 |
|
| 2. |
Under whatcondition DeltaHbecomesequal to DeltaU? |
| Answer» Solution :`DeltaH = DeltaU` during a process which is carried out in a CLOSED vessel `( DeltaU = 0 )` or number of moles of gaseous products `=` number of moles of gaseous REACTANTS or the reaction does not involve any gaseous or PRODUCT. | |
| 3. |
What is the reason behind the blue colouration of alkali metals with liquid ammonia? M+(x+y)NH_(3)to[M(NH_(3))_(x)]^(+)+[e(NH_(3))_(y)]^(-) |
| Answer» Solution :The blue COLOUR of the solution is due to the ammoniated ELECTRON which absorbs energy in the visible REGION of light and thus imparts blue colour to the solution. The solutions are paramagnetic and on standing slowly liberate hydrogen RESULTING in the formation of amide. In concentrated solution, the blue colour changes to bronze colour and become diamagnetic. | |
| 4. |
The shape of CH_4, SO_4^(2-), PO_4^(-3) is |
|
Answer» Trigonal PLANAR |
|
| 5. |
Which of the following is not an intensive property? |
| Answer» ANSWER :D | |
| 6. |
Which of the following IUPAC name of the molecule is not correct? |
|
Answer» `UNDERSET("4-methylpent-2-ene")(CH_(2)=CH-CH_(2)-underset(underset(CH_(3))(|))(CH)-CH_(3))` |
|
| 7. |
Vapour Pressure is depend upon ? |
| Answer» SOLUTION :(i) TEMPERATURE(II) LIQUID | |
| 9. |
Which zone is called ozonosphere ? |
| Answer» SOLUTION :STRATOSPHERE | |
| 10. |
The uncertainity in momentum of an electron is 1 xx 10^(-5) kg.m//s. The uncertainity in its position will be (h = 6.62 xx 10^(-34)kg.m//s) |
|
Answer» `1.05xx10^(-28)m` |
|
| 11. |
The stability order of alkene in following compounds is : |
|
Answer» `IltIIltIIIltIV` |
|
| 12. |
Which is expected to be more stable, O_2 NCH_2CH_2O or CH_3CH_2O and why? |
|
Answer» |
|
| 13. |
Which of the following is not the consequence of global warming? |
|
Answer» INCREASE in AVERAGE TEMPERATURE of Earth. |
|
| 14. |
The reagent which gives the same reduction product with propionaldehyde and acetone is |
|
Answer» `LiAIH_(4)` |
|
| 15. |
Which of the following are examples of Birch reduction ? |
|
Answer»
|
|
| 16. |
When 3, 3-dimethylbutan-2-ol is heated with conc.H_(2)SO_(4), then main product formed is |
|
Answer» 3, 3-Dimethylbutene-1 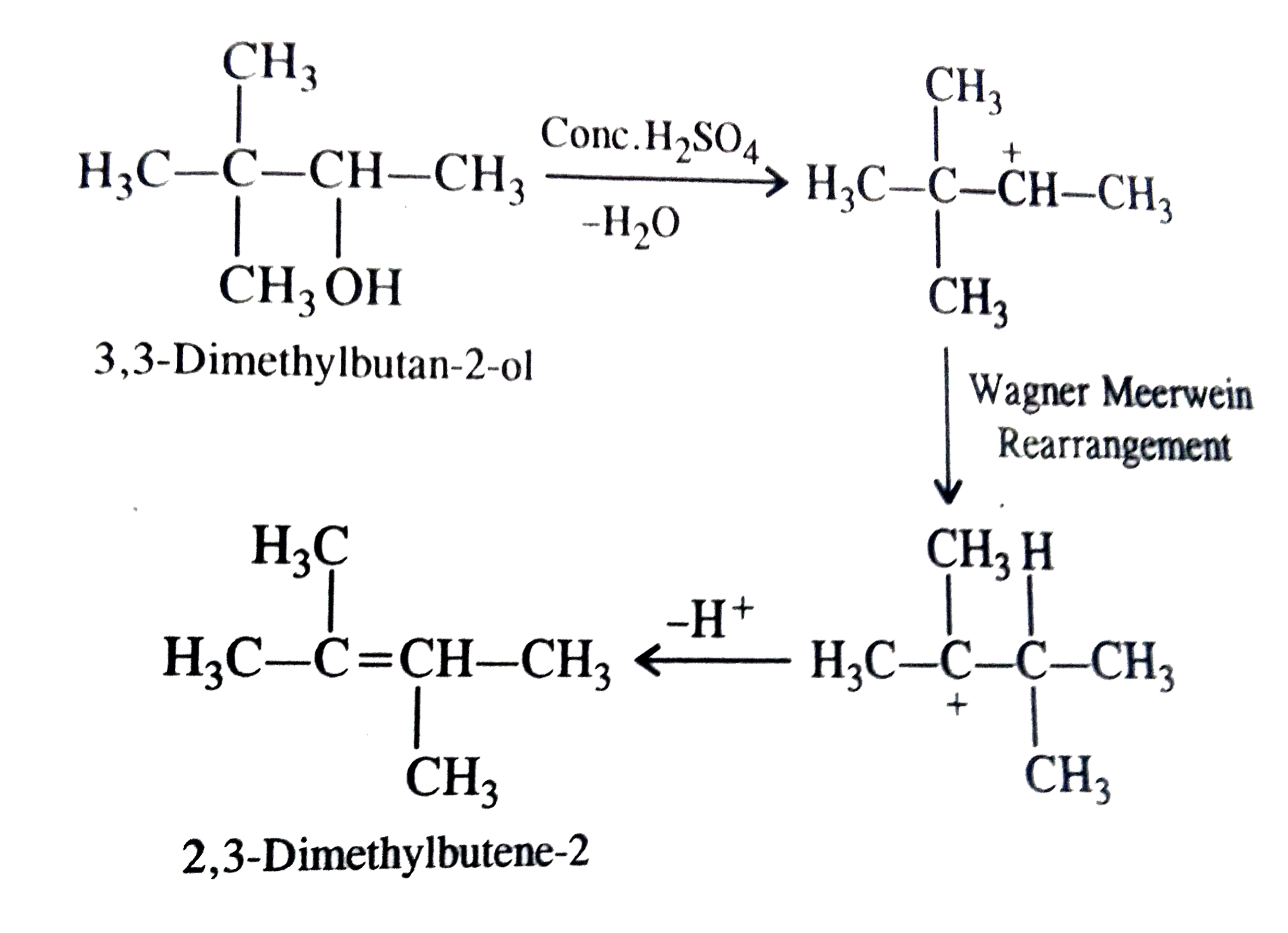
|
|
| 17. |
What are the values of n, l, m and s for 3p_(x) electron ? |
|
Answer» <P>3,2,1,0 for `p_(x)` orbital, `L = 1, m = -1, s = ½` |
|
| 19. |
Which of the following relativelymore strong acid in aqueous solutions? |
|
Answer» `HClO_4` |
|
| 20. |
Which of the following is not the use of alkali metals . |
|
Answer» White metal , an ALLOY of lithium and lead is used in manufacture of bearing for motore engines . |
|
| 21. |
Which among the following is a redox reaction ? |
|
Answer» `Ca(OH)_(2) + 2HClrarr CaCl_(2) + 2H_(2)O` |
|
| 22. |
Which of the following is a set of reducing agents ? |
|
Answer» `HNO_(3),Fe^(2+),f_(2)` |
|
| 23. |
What is the IUPAC name of |
|
Answer» 2, 3-dimethyl, 7-bromooctane<BR>2-bromo-5, 6-dimethyloctane |
|
| 24. |
The threshold energy for photoelectric emission of electrons from a metal is 3.056 xx 10^(-15) joule. If light of 4000 Å wavelength is used, will the electrons be efected or not ? (h = 6.63 xx 10^(-34) Joule sec) |
|
Answer» Energy of the incident light `= hv = h (C)/(lamda) = (6.63 xx 10^(-34) xx 3 xx 10^(8))/(4000 xx 10^(-10)) J = 4.97 xx 10^(-19) J` Thus is less than threshold energy |
|
| 25. |
What is meant by point defect in crystals ? |
| Answer» Solution :The defect caused by MISSING or DISPLACEMENT of ATOMS or ions in a CRYSTAL is called point defect. | |
| 26. |
The screening effect of d-electrons is |
|
Answer» <P>EQUAL to p - ELECTRONS |
|
| 27. |
X ml of H_(2) gas effuses through a hole in a container in 5 seconds. The time taken for the effusion of the same volume of the gas specified below under identical conditions is |
|
Answer» 10 seconds : He or `(t)/(5)=sqrt((M_(X))/(2))` The equaloty HOLDS GOOD only for (b). |
|
| 28. |
The temperature of the system, decreases in an_________. |
|
Answer» ISOTHERMAL EXPANSION |
|
| 29. |
Which solute has the greatest solubility (in mol/L) in water at 25^(@)C and 1 atm? |
|
Answer» `CH_(4)` |
|
| 30. |
Which orbitals are involved in bond overlapping of each carbon in benzene ? |
|
Answer» Solution :(i) C-C sigma BOND is formed by overlapping of `SP^(2)-sp^(2)` hybridized orbital of two CARBON atoms. (ii) The sigma bond is formed between C-H by overlapping of `sp^(2)` hybridized orbital of C and 1S orbital of H and form a bond between C and H. (iii) `pi`-bond is formed by overlapping of 2p-2p ORBITALS of each two carbon. |
|
| 31. |
What is the distance between Na^+ and Cl^- in a NaCl crystal if its density is 2.165 "g cm"^(-3) ?NaCl crystallises in the fcc lattice |
|
Answer» |
|
| 32. |
Valence electrons in the atom of elements A is 4 and in the element B is 2. Most probale compund from A and B is |
|
Answer» `AB_(3)` |
|
| 33. |
Which contain more strong H-bond in the followi!)g pair ? (i) H_(2)O and H_(2)S (ii) NH_(3) and PH_(3) (iii) HF and HCl. |
| Answer» SOLUTION :(i) `H_(2)O` (ii) `NH_(3) ` (iii) HF | |
| 34. |
Which one of the following is correct order? (A) T_(2) gt D_(2) gt H_(2) (order of BP) (B) T_(2) gt D_(2) gt H_(2) (order of BE) (C) T_(2)=D_(2)=H_(2) (order of BL) (D) T_(2) lt D_(2) lt H_(2) (order of reactivity with Cl_(2)) |
|
Answer» A, B & C |
|
| 35. |
The stability of hydrophobic sol is due to |
|
Answer» SOLVATION of COLLOIDAL particles |
|
| 36. |
Write reaction for autoprotolysis of water. How is ionic product of water related to ionization constant of water ? Derive the relationship. |
|
Answer» SOLUTION :Autoprotolysis of `H_(2)O` TAKES place as : `{:(H_(2)O+H_(2)O,HARR,H_(3)O^(+)+OH^(-)),(""H_(2)O,hArr,H^(+)+OH^(-)):}` For IONIZATION of `H_(2)O`, `K_(i)=([H^(+)][OH^(-)])/([H_(2)O])=(K_(w))/([H_(2)O])=(K_(w))/(55.55 "mol" L^(-1)) or K_(w)=K_(i)xx55.55`. |
|
| 37. |
The sulphate of an element contains 42.2% element. The equivalent mass would be |
|
Answer» 17 |
|
| 38. |
Which of the following statement about solution of alkali metals in liquid ammonia is correct |
|
Answer» The solution have strong OXIDIZING properties |
|
| 39. |
Which of the following is strong reducing agent ? |
|
Answer» `Al_(2)O_(3)` |
|
| 40. |
What is the ratio of number of molecules, if the mass ratio of N_(2) and O_(2) is 4:1? |
|
Answer» SOLUTION :Ratio of molecules of `N_(2) and O_(2)=` Ratio of NUMBER MOLES of `N_(2) and O_(2)=(4)/(28):1/(32)` `=1/7 : 1/(32)=32:7` |
|
| 41. |
Which of the following particle having same kinetic energy, would have the maximum de-Broglie wave length |
|
Answer» `ALPHA` - particle |
|
| 42. |
Write the following as net ionic equations and designate the nucleophile, substrate, and leaving group in each base. |
Answer» SOLUTION :the net ionic equations are as FOLLOWS for (a) and (B) and there is no abbreviated equation possible for (c ). Nucleohiles, substrates and leaving groups are labeled ACCORDINGLY. 
|
|
| 43. |
Which of the following groups shows negative inductive effect? |
| Answer» Solution :More ELECTRONEGATIVE GROUP attracts ELECTRONS. | |
| 44. |
What is meant by isoelectronic species ? |
| Answer» Solution :Isoelectronic SPECIES are atoms, INOS or molecules having same number of ELECTRONS. | |
| 45. |
What is (X) and (Y) in the above reaction? |
|
Answer» `RCH_(2)CH_(2)CHO,RCH_(2)CH_(2)CH_(2)OH` |
|
| 46. |
Which of the following groups cannot participate in resosnace with benzene ring ? |
|
Answer» `-COCL` 
|
|
| 47. |
Which of thefollowingspecieshas thelargestand thesmallest size ? Mg^(2+) ,Al,Al^(3+) |
|
Answer» Solution :(i) Mgand A1belongto thethirdperiod.Acrossa periodatomicradiidecreasedue toincreases nuclear charge . Thereforeatomicsizeof ALIS smaller thanthat of Mg. (ii)Further cations are smaller thantheir parentatoms. Therefore `Mg^(2+)` si smallerthan Mg and `Al^(3+)` issmallerthan Al. (III) `Mg^(2+)` and `Al^(3+)` are isoelectronicions. AMONG isoelectronicions, higher the + vechargesmallerthesize. Thereforeionicradiusof `Al^(3+)` is smallerthan thatof `Mg^(2+)` Fromthe abovediscussionsit followsthat MGHAS thelargestwhile`Al^(3+)` has thesmallestsize. |
|
| 48. |
Which one of the following is not a crystalline hydrate? |
|
Answer» `CH_(4).20H_(2)O` |
|
| 49. |
Which of the following statement is false? |
|
Answer» `CA^(2+)` ions are not important in maintaining the regular beating of the heart |
|
| 50. |
Which of the following alkaline earth metal is cofactor of enzyme to utilise ATP in phosphate transfer ? |
|
Answer» Ca |
|


