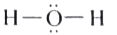Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which combination cannot be used for the preparation of hydrogen gas in the laboratory? Zn/conc.H_(2)SO_(4)"II) " Zn//dil.HNO_(3) III) pure Zn/dil. H_(2)SO_(4) IV) granulated Zn/dil. HCl |
|
Answer» I and II |
|
| 2. |
Which chain is taken as parent chain ? Why |
| Answer» SOLUTION :A is taken as LONG parent chain as A or B has same `-C_(6)H_(13)` in such case the chain which has more subsitute is taken as parent chain | |
| 3. |
Which of the following reactions is/are correct ? |
|
Answer» `C_xH_y + (x + y/2) O_2 to xCO_2 + y/2 H_2O` |
|
| 4. |
Using the Periodic Table, predict the formulae of compounds which might be formed by the following pairs of elements, aluminium and sulphur. |
| Answer» Solution :Aluminium BELONGS to GROUP 13 with a VALENCY of 3, Sulphur belongs to group 16 with a valency of 2. Hence the formula of the compound formed would be `Al_2S_3` | |
| 5. |
Which of the following solids is not an electrical conductor ?{:("(A) Mg(s)", " (B) TiO (S)"),("(C)"I_(2)"(s)","(D)"H_(2)O "(S)"):} |
|
Answer» (A) only |
|
| 6. |
Using the Periodic Table, predict the formulae of compounds which might be formed by the following pairs of elements, silicon and bromine |
| Answer» Solution : Silicon is group 14 element with a VALENCY of 4, bromine belongs to the HALOGEN FAMILY with a valency of 1. HENCE the formula of the compound formed would be `SiBr_(4)`. | |
| 8. |
Which of the following reactions are endothermicreactions ? |
|
Answer» COMBUSTION of methane |
|
| 9. |
What is the boiling point of water in degree farenheit ? |
|
Answer» 210 `=9/5xx 100 + 32 = 180 + 32 = 212` |
|
| 10. |
Which of the following is//are the hazardous pollutant(s) present in automobile exhaust gases ?(i) N_(2) (ii) CO (iii) CH_(4 ) (iv) Oxides of nitrogen |
|
Answer» II and iii |
|
| 11. |
The total pressure of a mixture of 8g of oxygen and 14g of nitrogen contained in a 11.2L vessel at 0°C is. |
| Answer» ANSWER :C | |
| 12. |
Three samples of water A,B and C have the D.O. values 1mg/l , 3mg/l , and 5 mg/l respectively . The more polluted sample of water is |
|
Answer» A |
|
| 13. |
What is meant by 100-volume hydrogen peroxide? |
| Answer» Solution :A 30% solution is marketed as 100 VOLUME hydrogen peroxide indicating that at STP, 100 volumes of OXYGEN are liberated per MILLIMETER of the solution | |
| 14. |
Which one of the following has longest covalent bond distance ? |
| Answer» Answer :D | |
| 15. |
Versene, a chelating agenthaving chemical formula C_(2)H_(4)N_(2)(C_(2)H_(4)O_(2)Na)_4. If each mole of this compound could bind 1 mole fo Ca^(2+), then the rating of pure versene expressed as mg of CaCO_(3) bound per g of chelating agent is |
|
Answer» 100 mg RATING = mg of `CaCO_(3)` needed per g chelating agent mol. Wt` =380 = (100 xx 10^(3))/(380) = 263 mg` |
|
| 16. |
Write the structural formula of isobutyl alcohol. |
| Answer» SOLUTION :`CH_(3)-underset(CH_(3))underset(|)(CH)-CH_(2)-OH` | |
| 17. |
Which of the following statements is not true for lithium ? |
|
Answer» It is the hardest alkali metal |
|
| 18. |
Which of the following elements can show covalency greater than 4 ? |
|
Answer» Be They can have 8 ELECTRONS maximum in the valence shell. In other words, we can SAY that it can have a maximum covalency of 8/2 = 4. Elements P and S have vacant d- orbitals in their respective valence shells and hence it can accommodate more than 8 electrons in their valence shell RESPECTIVELY. In other words, we can say that it can SHOW a covalency of more than 4. |
|
| 19. |
Which of the following oxides is formed on burning sodium in oxygen ? |
|
Answer» `Na_(2)O` |
|
| 20. |
Which of the following reactions will result in the formation of a chiral centre in the product? |
|
Answer» `CH_3CH =CH_2 + HBR to `<BR>`CH_2 = CH_2 + HOBr to ` |
|
| 21. |
Which of the following can be used as the halide component for Friedel-Craft's reaction ? |
|
Answer» Chloroethane  But in Chlorobenzene, Bromobenzene, Chloroethene LONE PAIR of halogen are delocalised with `pi` bonds, so attain double BOND character. |
|
| 22. |
Which of the following molecules can be optically active |
|
Answer» `CH_2 = C= CH_2`  Allens are optically active even though they do not have any chiral centre but acts as chiral MOLECULE due to the PRESENCE of chiral axis.Hence ALLENES with different substituted ATOMS are optically active. |
|
| 23. |
What is orbital (atom orbital)? |
| Answer» SOLUTION :ORBITAL is a three DIMENSIONAL REGION in space around the nucleus in which the probability of FINDING an electron is maximum. | |
| 24. |
The standard pH of drinking water is................. |
| Answer» SOLUTION :6.5 to 8.5 | |
| 25. |
Write the postulates of kinetic theory of gases. |
|
Answer» SOLUTION :1. Gases are made up of large number of the minute particles. 2. Pressure is exerted by a gas. 3. There is no LOSS of kinetic theory. 4. Molecules of gas attract on on e ANOTHER 5. Kinetic energy of the molecules in directly proportional to absolute TEMPERATURE. 6. Actual volume of the gaseous molecule very SMALL. 7. Gaseous molecules are the at always in motion. 8. There is more influence of gravity on movement of gaseous molecule. |
|
| 26. |
The wave length of H_(delta) line of Balmer series of a hydrogen atom is nearly (R = 1.08 xx 10^(7) m^(-1)) |
|
Answer» 4090 Å |
|
| 27. |
Write the expressions of equilibrium constants in terms of partial pressure and active masses for 2BrCl_((g))hArrBr_(2(g))+Cl_(2(g)) |
| Answer» SOLUTION :`K_(P)=((P_(Br_(2)))(P_(Cl_(2))))/((P_(BrCl))^(2))," "K_(c)=([Br_(2)][Cl_(2)])/([BrCl]^(2))` | |
| 28. |
What is the proportion of oxygen in H_(2)O and H_(2)O_(2) ? |
|
Answer» `1:2` |
|
| 29. |
The values of critical pressure, critical volume and critical temperature for hydrogen gas are 12.97bar, 0.005 Lmol^(-1) and 33.2K. Calculate the compressibility factor. |
|
Answer» SOLUTION :Compressibility FACTOR (z) for one mole of gas is GIVEN interms of critical pressure `(P_C)`, critical VOLUME`(V_C)` and critical temperature `(T_C)` as `z=(P_C V_C)/(RT_C)` Substituting values , `z = (12.97 xx 0.065)/(0.032 xx 33.2) = 0.306`. |
|
| 30. |
What is the maximum number of moles of CO_(2) that could be obtained from the carbon in 4 moles of Ru_(2)(CO_(3))_(3) ? |
|
Answer» |
|
| 31. |
Which colour will appear in the Lassaigne's test if the compound contains both nitrogen and sulphur ? |
|
Answer» |
|
| 32. |
What is metamerism? Give example |
|
Answer» Solution :It arises due to different alkyl chains on either side of the functional GROUP in the molecule is CALLED metamerism e.g `C_(4)H_(10)O` ETHER represents two metameres. (i) methoxypropane `(CH_(3)OC_(3)H_(7))` (ii) ethoxypropane `(C_(2)H_(5)OC_(2)H_(5))` |
|
| 33. |
Write IUPAC names of the products by the ozonolysis of the following compounds : (i) Pent-2-ene (ii) 3,4-Dimethylhept-3-ene (iii) 2-Ethylbut-1-ene (iv) 1-Phenylbut-1-ene |
Answer» Solution :  : Selection of REACTIVE OZONOLYSIS product : In intermediate of unstable ozonolysis, if we do not draw line in between it is not COMPULSARY. If there is ONE DOUBLE bond in between, put .O. on that carbon. Therefoe C=O is formed and their is two different structure. Do not change the structure of double bonded carbon having result or yield as 2 product. (iii) 
|
|
| 34. |
What kind of a bond is formed when the orbitals of two atoms A and B undergo (i) s-s overlap (ii) s-p overlap? |
| Answer» SOLUTION :In both (i) and (ii) only `SIGMA`- bonds are FORMED. Because s-orbitals are non directional in nature, always overlap ALONG the internuclear axis only. So s-orbitals form `sigma`- bonds only. The `sigma`- bonds in (i) and (ii) are denoted as `sigma_(s-s)` and `sigma_(s-p)` respectively. | |
| 35. |
Which substance is called as a calgon for business purpose ? |
|
Answer» SODIUM pyrometa PHOSPHATE |
|
| 36. |
What is the other name for classical smog ? |
|
Answer» REDUCING SMOG |
|
| 37. |
What is order of thermal energy of liquid, solid and gas. |
| Answer» SOLUTION :Order of THERMAL ENERGY : Solid `to` Liquid `to` GAS | |
| 38. |
The tendensy for catenationdecreases down thegroup 14 ? |
| Answer» SOLUTION :As we MOVE down the group 14, the ATOMIC size increasesand hencethe strength of the element -elementbond, i.e, the bonddissociationenthalphy decreasessteadily. Consequentlythe tendencyfor catendation decreasesdown the group with increasingatomic NUMBER. | |
| 39. |
Which of the following is Dobereiner 's triad ? |
|
Answer» `NE,Ca,NA` |
|
| 40. |
The solubility of a salt of weak acid (AB) at pH 3 is Y xx 10^(-3) mol L^(-1). Thevalue of Y is ............ (Given that the value of solubility product of aB (K_(sp))=2xx10^(-10) and the value of ionization constant of HB (K_(a))=1xx10^(-8)) |
|
Answer» `B^(-)(aq)+ H^(+) (aq) hArrHB (aq)` ...(ii) Adding eqns (i) and (ii), we get `{:(,AB(s),+,H^(+)(aq),hArr,A^(+)(aq),+,HB(aq)),("INITIAL conc",x,,10^(-3),,0,,0),("Final conc.",x-S,,10^(-3),,S,,S),(,,,,,,,):}`(S = solubility when pH = 3) From eqn. (i), `K_(SP)=[A^(+)][B^(-)]` Fromeqn. (ii), `K_(a)=([HB])/([B^(-)][H^(+)])` `:. (K_(sp))/(K_(a))=([A^(+)][HB])/([H^(+)])` `(2xx10^(-10))/(10^(-8))=(SxxS)/(10^(-3))` or `S^(2) = 2 xx10^(-5) = 20 XX 10^(-6) or S = 4.47 xx 10^(-3)` Hence , y = 4.47. |
|
| 41. |
Which of the following statements are false? |
|
Answer» `BeCl_(2)` is a linear molecule in the VAPOUR STATE but it is POLYMERIC FORM in the solid state |
|
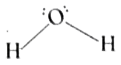
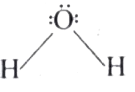
| 42. |
Draw the molecular structure of water. |
|
Answer»
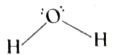
|
|
| 43. |
Which electron is remove from Be and B during ionisation enthalpy? |
| Answer» SOLUTION :During IONISATION ENTHALPY ELECTRONS removed from `2s^(2) `and 2p ORBITAL in Be and B elements respectively. | |
| 44. |
Those substaces can be separated by steam distillation which are |
|
Answer» steam volatile and INSOLUBLE in water |
|
| 45. |
Which of the following is optically active ? |
|
Answer» 3 - Chloropentane |
|
| 46. |
What is the orbital angularmomentum forelectronin 2sorbital ? |
|
Answer» `(1)/(2) .(H)/(2PI)` for`2s l=0= sqrt(0(0+1)) .(h)/(2pi)=0` (for2 s orbital l=0) |
|
| 47. |
Three acyclic alkenes (x,y,z) on catalytic hyrogenation give same alkane. On reaction with HCl (x,y,z) form same major tertiary halide product.Reductive ozonolysis of mixture of (x,y,z) gives a mixture of two moles of CH_2=O one moles of CH_3CH=O one mole of acetone, one mole of butanone and one mole of 2-methyl propanal, x,y and z do not have any stereoisomers. x,y,z are |
|
Answer» CHAIN isomers |
|
| 48. |
Unlike phosphorus, nitrogen shows little tendency for catenation. Or Phosphorus shows greater tendency for catenation than nitrogen. |
| Answer» Solution :Due to smaller size, the lone pairs of electronc on the two N-atoms repel the bond pair of N-N bond. In CONTRAST, because of COMPARATIVELY larger size of P, the lone pairs of electrons on the two P atoms do not repel the bond pair of the P-P bond to the same extent. As a result, N-N single bond is WEAKER `(159kJmol^(-1))` than P-P single bond `(213kJmol^(-1))`. Consequently, NITROGEN shows little tendency for catenation or PHOSPHORUS has greater tendency for catenation. | |
| 49. |
Three acyclic alkenes (x,y,z) on catalytic hyrogenation give same alkane. On reaction with HCl (x,y,z) form same major tertiary halide product.Reductive ozonolysis of mixture of (x,y,z) gives a mixture of two moles of CH_2=O one moles of CH_3CH=O one mole of acetone, one mole of butanone and one mole of 2-methyl propanal, x,y and z do not have any stereoisomers. (x,y,z)overset(H_3O^(**))to addition product . The correct statement is |
|
Answer» All THREE ALKENES will GIVE 3 different major hydration PRODUCTS |
|
| 50. |
Three acyclic alkenes (x,y,z) on catalytic hyrogenation give same alkane. On reaction with HCl (x,y,z) form same major tertiary halide product.Reductive ozonolysis of mixture of (x,y,z) gives a mixture of two moles of CH_2=O one moles of CH_3CH=O one mole of acetone, one mole of butanone and one mole of 2-methyl propanal, x,y and z do not have any stereoisomers. What is true about x,y,z |
|
Answer» These have molecules formula `C_3H_6` |
|