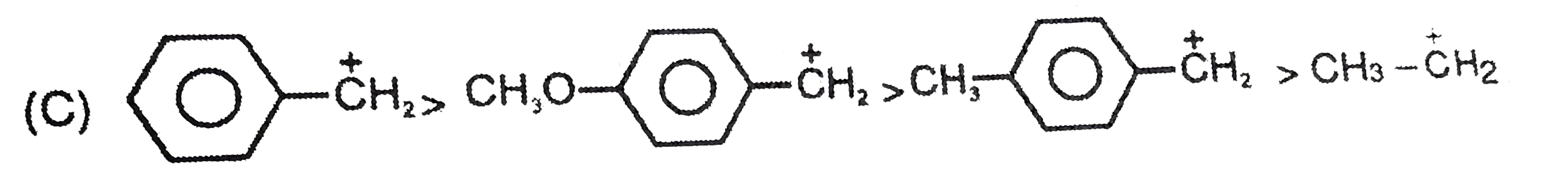Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the effect on solubility of AgCl, when saturated solution of AgCl is added in different solution of 0.1 AgNO_3 and 0.1 NaCl ? |
|
Answer» SOLUBILITY in `AgNO_3 GT` Solubility in NaCl |
|
| 2. |
What are the sources of dissolved oxygen in water ? |
| Answer» SOLUTION :Sources of dissolved OXYGEN in water are (i)photosynthesis (II) natural AERATION and (iii) MECHANICAL aeration | |
| 3. |
We are good at matching things let us try our hand on the following columns : |
|
Answer» |
|
| 5. |
Which one of the following is not a broad spectrum antibiotic ? |
|
Answer» Tetracycline |
|
| 6. |
Which of the following have maximum number of P -H bond ? |
|
Answer» `H_3PO_2` |
|
| 7. |
Type of isomerism not possible with the molecular formula C_(4)H_(10)O is |
|
Answer» chain |
|
| 8. |
What isuncerainty in location of photod of wavelenght 5000Å if wavelenght is known to anaccuracy of 1 pm? |
|
Answer» `7.96xx10^(-14)` `Deltax ge2xx10^(2)m` |
|
| 9. |
What is dry ice and why is it called so ? |
| Answer» SOLUTION :SOLID `CO_2`. It does not WET the SURFACE when it MELTS. | |
| 10. |
What is the use of diborane ? Why BH_(3) exists in the form of diborane ? |
| Answer» Solution :`BH_(3)` EXISTS in the FORM of DIBORANE `(B_(2) H_(6))` because it is electron deficient . Diborane is used as a reducing agent. | |
| 11. |
What isspeedof electromagneticradiationin vacuum ?Whatis itcalled? |
| Answer» Solution :The speedof electromagnaticraditionis`3.0 xx 10^(8) MS^(-1) . `itis knownas speedof rightor velocityof LIGHTAND givethesymboli C | |
| 12. |
Under critical states for one mole of a gas, compressibility factor is |
|
Answer» `3/8` |
|
| 13. |
Which of the following can be used as oxidant and reductant both? |
|
Answer» `HNO_2` |
|
| 14. |
The two sources of Zn, that is, ZnSO_(4) and Zn(CH_(3)COO)_(2)2.H_(2)O, can be purchased at the same price per kilogram of compound. Which is the most economical source of Zn and by how much? |
|
Answer» |
|
| 15. |
Which of the following is not a combustion reaction |
|
Answer» `CO + (1)/(2) O_(2) RARR CO_(2)` |
|
| 16. |
Which of the following statements are not correct- |
|
Answer» the SHAPE of `PCl_(5)` is trigonal pyramidal |
|
| 17. |
Write the structure of the following compounds. BF_(3) |
Answer» SOLUTION : A = B ATOM , B = F ATOMS
|
|
| 18. |
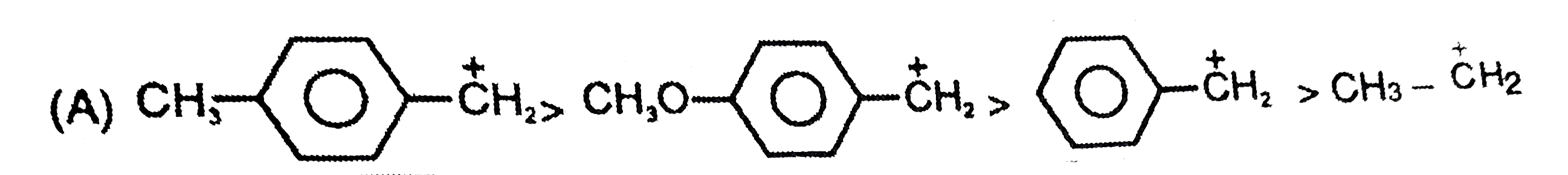
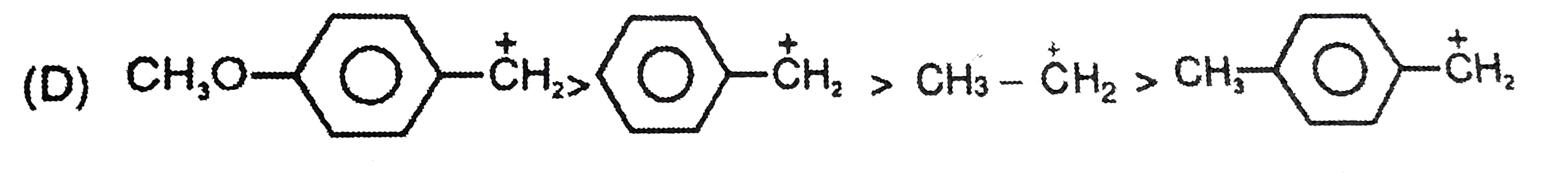
The stability of the carbanions in the following is…… (i) RC -= overset(Θ)C (iii) R_(2)C = overset(Θ)(C )H (iv) R_(3)C - overset(Θ)( C)H_(2) |
|
Answer» `(iv) gt (ii) gt (iii) gt (i)`  `R_(3)-C - underset(SP^(3))overset(Θ)(CH_(2))` |
|
| 19. |
Which of the following metal nitrate on heating does not give its metal Oxide? |
|
Answer» `NaNO_(3)` (C ) `2Ca(NO_(3))_(2(s)) overset(Delta)to 2CaO_((s)) +4NO_(2(g)) +O_(2(g))` (A) `NaNO_(3(s)) overset(Delta)to 2NaNO_(2(s))+O_(2(g))` (D) `4LiNO_(3(s)) overset(Delta)to 2Li_(2)O_((s)) +4NO_(2(g)) +O_(2(g))` `NaNO_(3)` does not form OXIDE but gives `NaNO_(2)` (nitrite) on heating. |
|
| 20. |
Which of the following is correct: |
|
Answer» Due to FORMATION of |
|
| 21. |
In diborane, the two boron atoms are linked together by two ............ bonds. |
| Answer» SOLUTION :HYDROGEN, 3c-2e | |
| 22. |
Which are the atmospheric air pollutants ? |
| Answer» Solution :Gaseous air pollutant contains oxides of carbon, NITROGEN and SULPHUR, HYDROGEN sulphide, HYDROCARBONS, OZONE and other oxidizing agents. | |
| 23. |
Which of the following sequences contain atomic number of only representative elements- |
|
Answer» 3,33,53,87 |
|
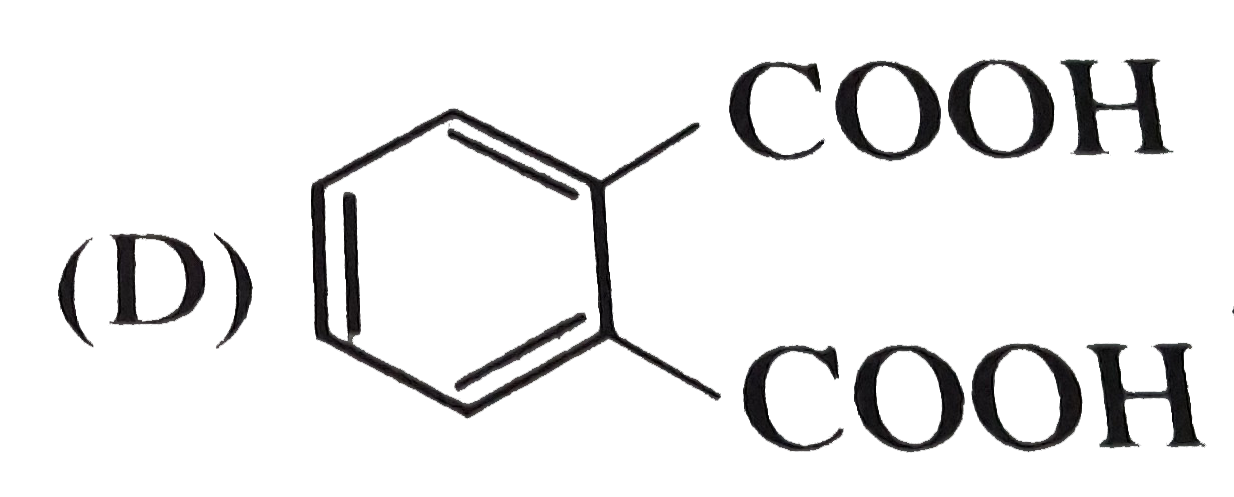
| 24. |
Which of the following reactants on reaction with concentrated NaOH followed by acidification gives the following lactone as the only product |
|
Answer»

|
|
| 25. |
Which of the following is incorrect statement? |
|
Answer» `GeX_4 (X= F, Cl , Br , I)` is more stable than `GeX_2` |
|
| 26. |
The volume of oxygen liberated at STP from 15 mL of 20 volume H_(2)O_(2) is |
|
Answer» 100 ML `THEREFORE ` 15 mL of `H_(2)O_(2)` will liberate `O_(2)` at STP `=15xx20=300 `mL |
|
| 27. |
The value of DeltaH and DeltaS for the reaction C_("(graphite)")+CO_92)(g) rarr 2CO(g0 are170 kJ and 170JK^(-1) respectively. This reaction will be spontaneous at |
|
Answer» 910 K or `TDeltaS gt DeltaH ` or`Tgt ( DeltaH )/( DeltaS) , i.e.,T gt (170000J)/( 170 JK^(-1))`or `T gt 1000K`. Hence, options (b) ISCORRECT. This method can be APPLIED only when `DeltaH` and`DeltaS` both are `+ve`. Alternatively, for equilibrium, `DeltaG =0` `i.e., T DeltaS = DeltaH` or `T= ( DeltaH )/( DeltaS) =( 170000)/( 170) =1000K` For spontaneity , `DeltaG = -ve`which can be so if `T gt 1000 K` ( so that`T DeltaS gt DetalH` in magnitude ) . |
|
| 28. |
Which are the transition elements and inner transition elements ? |
|
Answer» Solution :3d transition element : Sc (Z = 21) to Zn (Z = 30) 4d transition element: Y (Z = 39) to Cd (Z = 48) 5d transition element: La (Z = 57) to HG (Z = 80) 6d transition element: Ac (Z = 89) to Cn (Z = 112) 4F inner transition elements or Lanthanoid series : Ce (Z = 58) to Lu (Z = 71) 5finner transition elements or ACTINOID series: Th (Z = 90) to Lr (Z = 103) |
|
| 29. |
What are ideal solution ? Give example. |
| Answer» Solution :An ideal solution is a solution in which each component i.e. the SOLUTE as WELL as the SOLVENT obeys the Raoult.s LAW over the entire range of CONCENTRATION. | |
| 31. |
The specific test for H_(2)S_(2) is |
|
Answer» It turns STARCH iodide PAPER to blue COLOUR |
|
| 32. |
Write the Lewis structure of the nitrite ion,NO_2^-. |
Answer» SOLUTION :
|
|
| 33. |
why carbon show catendation but silicon does not ? |
| Answer» Solution :As we move down the group 14, the ATOMIC size increasesand HENCETHE STRENGTH of the element -elementbond, i.e, the bonddissociationenthalphy decreasessteadily. Consequentlythe tendencyfor CATENDATION decreasesdown the group with increasingatomic NUMBER. | |
| 34. |
Which element shown less shield effect ? |
|
Answer» Group-12, period-4 |
|
| 35. |
Which of the following alkene is the most stable |
|
Answer» `CH_(3)-CH=CH_(2)` |
|
| 36. |
Which one of the following statements is incorrect in relation to ionization enthalpy? |
|
Answer» Ionization enthalpy increases for each SUCCESSIVE electron. |
|
| 37. |
Which is the product of the following reaction ? |
|
Answer»

|
|
| 38. |
The standard electrode potentials of few metals are give below AI (-1.66 v),cu(+0.34V),Li(-3.05 v),Ag(+0.80 v) and Zn (-0.76) V) Which of these will behave as the strongest oxidising agent and which as the strongest reducing agent ? |
| Answer» Solution :Since `E^(@)` value of LI (-3.05 V) is thelowest while that of AG(+0.80 V) lis the HIGHEST therefore Li is the strongest reducing agent while `Ag^(+)` is the strongest oxidising agent | |
| 39. |
What is the value of the equilibrium constant for the following reaction at 400 K ? 2NOCl(g) hArr 2NO(g) + Cl_(2)(g) Delta H^(@) = 77.5 kJ mol^(-1), R = 8.3124 J mol^(-1) K^(-1), Delta S = 135 J K^(-1) mol^(-1). |
|
Answer» Solution :`Delta G = Delta H - T Delta S` `Delta G = 77.5 XX 1000 J - 400 K + 135 J K^(-1) = 77500 J - 54000 J = 23500 J` `Delta G = -2.303 RT LOG K` `23500J = -2.303 xx 8.314 xx 400 K log K` `log K = (-23500)/(19.147 xx 400 K) = (-235)/(76.588) = -3.068` K = ANTILOG (-3.608) `"Antilog" (0.932 - 4)= "Antilog" (0.93) xx 10^(-4) = 8.55 xx 10^(-4)`. |
|
| 40. |
Which among the following compounds show geometrical isomerismI)1- butene II) 2-butene III) 2-methyl-2-butene IV) 2-pentene |
|
Answer» II,III |
|
| 41. |
The value of carbon-carbon triple bond length is ……………. |
|
Answer» 1.33Å |
|
| 42. |
Which type of attraction force pressure in ideal gas ? |
| Answer» Solution :There is no attraction forces present in IDEAL gas. It is assumed that INTER MOLECULAR forces are not present between the molecular of an ideal gas this ideal gas is HYPOTHETICAL. | |
| 43. |
Which of the following reagent is used to find out carbon-carbon multiple bonds? |
|
Answer» GRIGNARD reagent |
|
| 44. |
Which one of the following is an intensive property? |
| Answer» Answer :A | |
| 45. |
When an alkali metal dissolves in liquid ammonia , the solution can acquire different colours . Explainthe reasons for this type of colour change . |
|
Answer» Solution :The dilute solution of alkali metals in liquid AMMONIA exhibit dark blue colour because ammoniated electrons absorb energy corresponding the red region of the visible light . `M + (x + y) NH_(3) to [M (NH_(3))_(x) ]^(+) + underset("Ammoniated electrons")(e^(-) (NH_(3))_(y))` However , if the concentration INCREASES above 3M, the colour CHANGES to copper-bronze and the solution acquires metallic lustre due to the formationof METAL ion clusters. |
|
| 46. |
What are the salient features of Valence Bond (VB) theory? |
|
Answer» Solution :(i) When half filled orbitals of TWO atoms overlap, a covalent BOND will be formed between them. (ii) The resultant overlapping orbitals are occupied by the two electrons with opposite spins. For example when `H_(2)` is formed, the two 1s electron of two hydrogen atoms get paired up and occupy the overlapped orbitals. (iii) The strength of a covalent bond depends upon the extent of overlap of atomic orbitals. Greater the overlap, larger is the energy RELEASED and stronger will be the bond formed. (iv) Each atomic ORBITAL has a specific direction (except s-orbital which is spherical) and hence orbital overlap takes place in the direction that maximises overlap. (v) Depending upon the nature of overlap, the bonds are classified as `sigma` covalent bond and `pi` covalent bond. (vi) When two atomic orbitals overlap linearly along the axis, the resultant bond is called a sigma `(sigma)` bond. This overlap Is also called "head-on-overlap" or "axial overlap" (vii) When two atomic orbitals overlap Sideways the resultant covalent bond is called a `(pi)` bond. |
|
| 47. |
Which of the following can show geometrical isomerism?(i) Propene (ii) But-1-ene (iii) But-2-ene (iv) Pent-2-ene |
| Answer» SOLUTION :III and IV | |
| 48. |
Which of the following oxides is most acidic in nature ? |
| Answer» Solution :As electropositive character of alkaline earth metals increases down the group , the basic character of their OXIDES increases . Thus BeO is least basic and hence most acidic . Actually , BeO is AMPHOTERIC in nature . | |
| 49. |
Which of the following is correct with respect to bond length of the species ? |
|
Answer» `C_(2) gt C_(2)^(2-)` `Li_(2) = 1 , Li_(2)^(+) = 0.5 , N_(2)= 3 , N_(2)^(+) = 2.5 , O_(2) = 2 ` `O_(2)^(-) = 1.5` . Greater the bond order , SHORTER is the bond length |
|
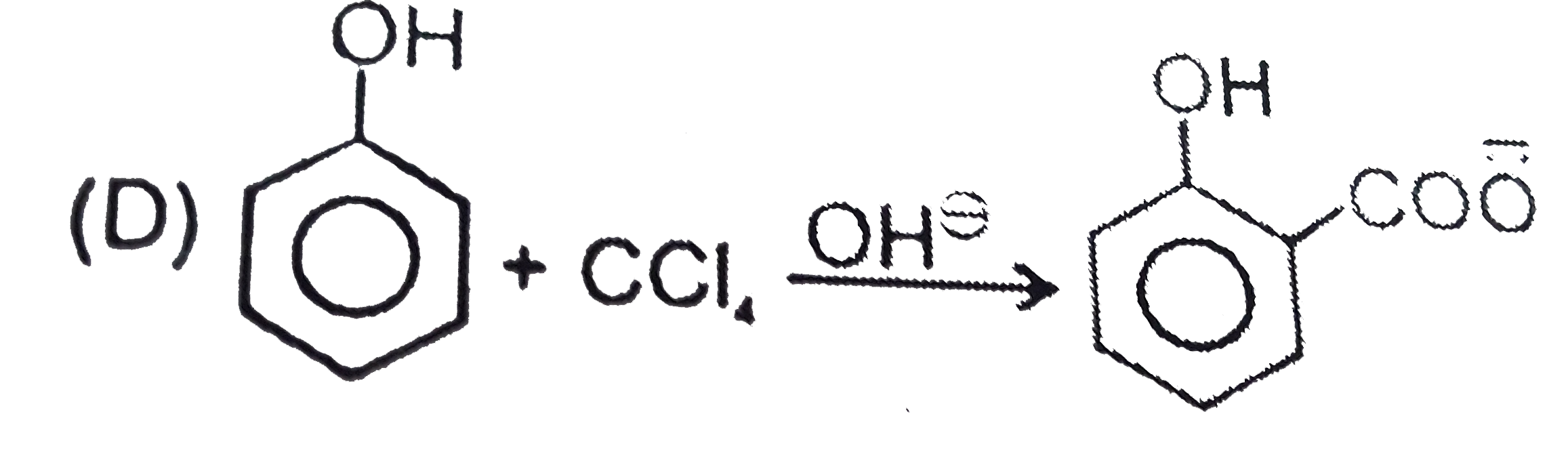
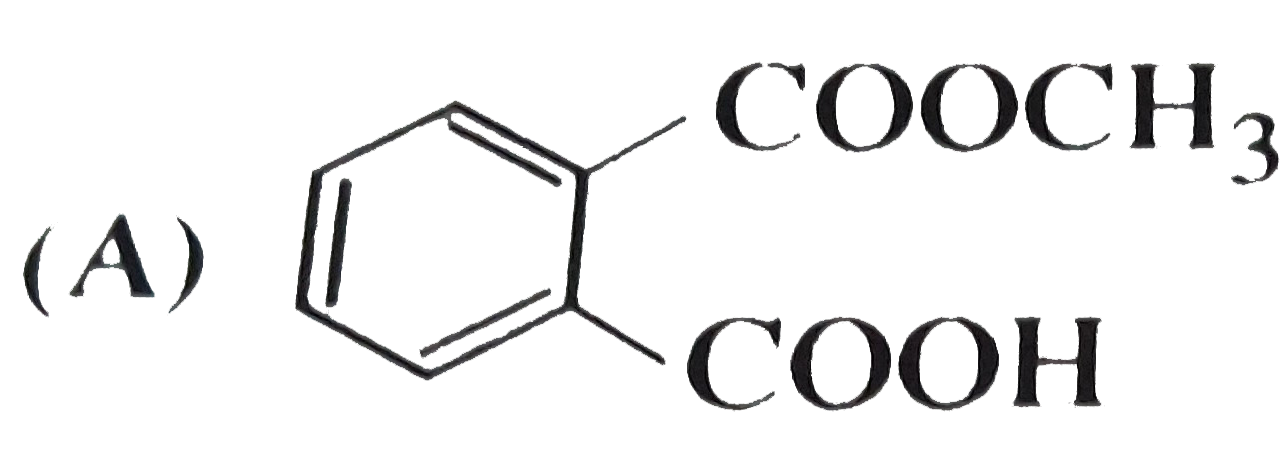
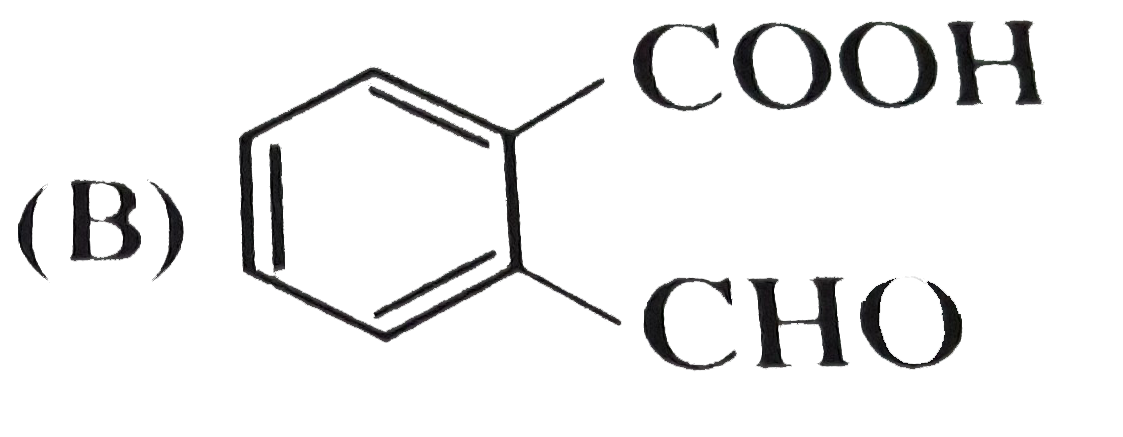
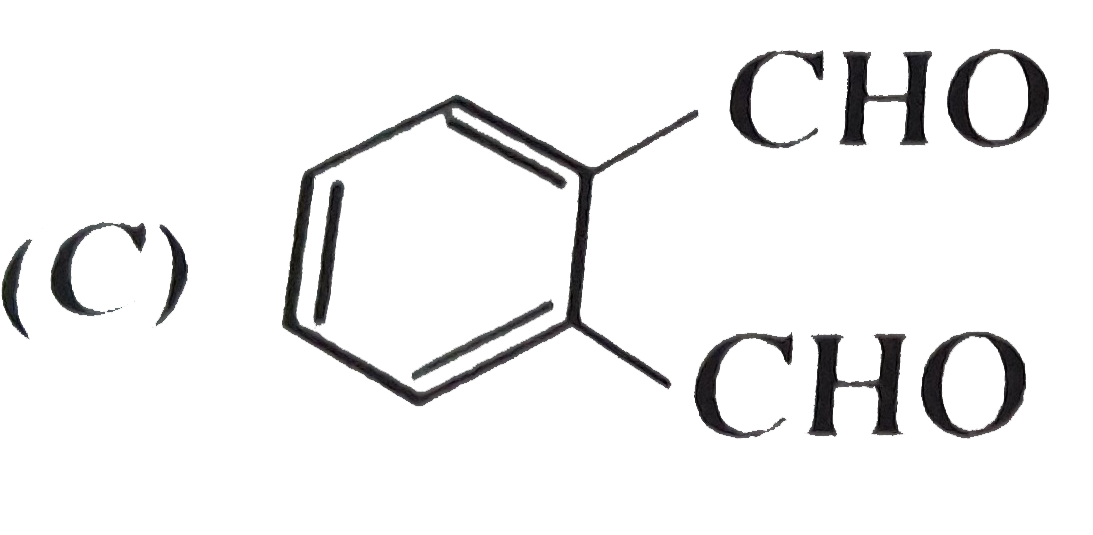
 , Ph-OH does not give Friedel
, Ph-OH does not give Friedel `, `Ph-NH_(2)` give m-nitro aniline, in sufficient amount during nitration. <br><img src=)