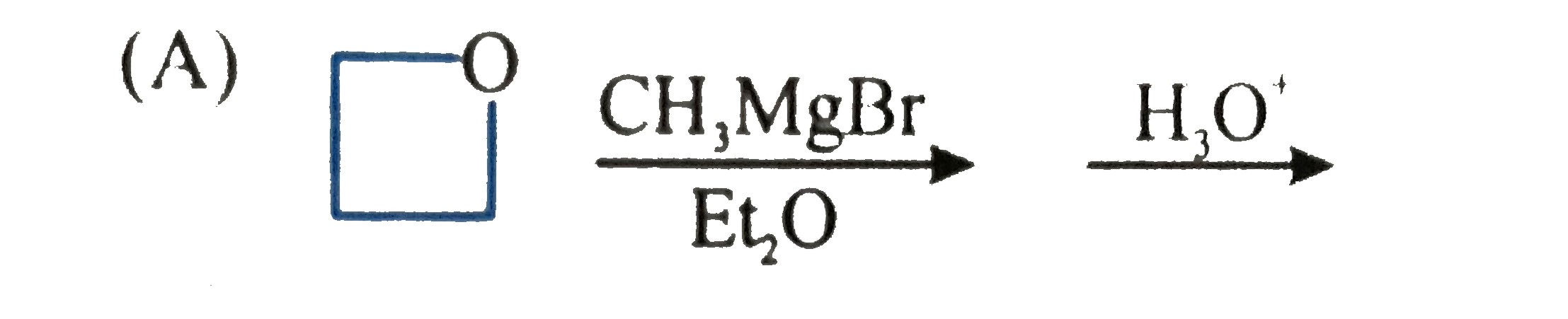Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is a false statement? |
|
Answer» Free radicals, carbonium ions or carbanions are REACTION intermediates. |
|
| 2. |
The stronest acid among the following is |
|
Answer» BUTYRIC acid |
|
| 3. |
When an iden gas undergoes unrestrained expansion, no cooling occurs because the molecules |
|
Answer» are above inversion temperature |
|
| 5. |
Which relationship does hold good for van der Waals' gas under all conditions where all terms have their usual meaning? |
|
Answer» `PV=NRT` |
|
| 6. |
The solubility of CaF_(2) is 2xx10^(-4) mole /litre . Its solubility product is |
|
Answer» `2.0 xx 10^(-4)` ` K_(Sp) = 4S^(3) =4 xx (2xx 10 ^(-4))^(3) =32 xx 10 ^(-12) ` |
|
| 7. |
What is acid rain ? Explain the formation of acid rain by atmospheric pollutant. |
|
Answer» Solution :Rain water has a pH of 5.6. Because rain water reacts with `CO_2` present in atmosphere and formed `H^+` ions. `H_2 O_((l)) + CO_(2(g)) hArr H_2 CO_(3(aq))` `H_2 CO_(3(aq)) hArr H_(("aq"))^+ + HCO_(3(aq))^(-)` When the pH of the rain water drops below 5.6, it is called acid rain. Acid rain refers to the ways in which acid from the atmosphere is deposited on the earth.s surface. Oxides of nitrogen and sulphur which are acidic in nature can be blown by wind ALONG with solid PARTICLES in the atmosphere and finally settle down either on the ground as dry DEPOSITION or in water, fog and snow as wet deposition.  Oxides of sulphur and nitrogen formed and mix in the atmosphere. Which is responsible for the acid rain. Burning of fossil fuels which contain sulphur and nitrogenous matter such as coal and oil in power stations and furnaces or petrol and diesel in motor engines produce sulphur dioxide and nitrogen oxides. `SO_2 and NO_2` after OXIDATION and reaction with water are major contributors to acid rain, because polluted air usually contains particulate matter that catalyse the oxidation. `2SO_(2(g)) + O_(2(g)) + 2H_2 O_((l)) to 2H_2 SO_(4(aq))` `4NO_(2(g)) + O_(2(g)) + 2H_2 O((l)) to 4HNO_(3(aq))` Thus, ammonium salts are also formed and can be seen as an atmospheric haze aerosol of fine particles. Aerosol particles of oxides or ammonium salts in rain drops result in wet deposition. `SO_2` is also absorbed directly on both solid and liquid ground surfaces and is thus deposited as drydeposition |
|
| 8. |
Which of the fallowing has highest chlorine content? |
| Answer» SOLUTION :Pyrene | |
| 9. |
Write phosphorous reacts with aqueous caustic soda to give hypophosphite and phosphine. |
|
Answer» Solution :a) The ionic skeleton equation is `P_(4)overset(OH^(-))rarrPH_(3)+H_(2)PO_(2)^(-)` b) Writing oxidation numbers `overset(0)(P_(4))rarroverset(-3)(P)overset(+1)(H_(3))+overset(+1)(H_(2))overset(+1)(H_(2))overset(+1)(P)overset(-2)(O_(2)^(-))` c) Locating atoms undergoing change in oxidation numbers `overset(0)(P_(4))rarroverset(-3)(P)H_(3)+H_(2)overset(+1)(P)O_(2)^(-)` d) Dividing the reaction into two two halves and balancing in acidic medium, separately Oxidation half - reaction : `P_(4)rarrH_(2)PO_(2)^(-)` Step 1 : Balance phosphorous atoms `P_(4)rarr4H_(2)PO_(2)^(-)` Step 2 : Balance oxygen atoms `P_(4)+8H_(2)Orarr4H_(2)PO_(2)^(-)` Step 3 : Balance hydrogen atoms `P_(4)+8H_(2)O+8OH^(-)rarr4H_(2)PO_(2)^(-)+8H_(2)O` Step 4 : Balance CHARGE `P_(4)+8OH^(-)rarr4H_(2)PO_(2)^(-)+4e^(-)".....(a)"` Reduction half - reaction : `P_(4)rarrPH_(3)` Step 1 : Balance phosphorous atoms `P_(4)rarr4PH_(3)` Stpe 2 : Balance hydrogen atoms `P_(4)+12H_(2)Orarr4PH_(3)+12OH^(-)` Step 3 : Balance charge `P_(4)+12H_(2)O+12E^(-)rarr4PH_(3)+12OH^(-)` e) Equalising the electrons and adding the two halves `"eq (a)"xx3+"eq (b)"xx"1, we get"` `{:(3P_(4)+24H_(2)Orarr12H_(2)PO_(2)^(-)+12e^(-)),(P_(4)+12H_(2)O+12e^(-)rarr4PH_(3)+12OH^(-)),(bar(4P_(4)+12OH^(-)+12H_(2)Orarr"")),(""4PH_(3)+12H_(2)PO_(2)^(-)),(I_(4)+3OH^(-)+3H_(2)OrarrPH_(3)+3H_(2)PO_(2)^(-)):}` This is the balanced equation. |
|
| 10. |
When CH_(2) = CH - COOH is reduced with LiAlH_(4), the compound obtained will be |
|
Answer» `CH_(3) - CH_(2) - COOH` |
|
| 11. |
what are the biological importance of sodium and potassium. |
| Answer» SOLUTION :The biological importance of SODIUM and potassiumk are : (a) Both Na and K are present in body 0.15% and 0.35% (b) Na is found OUTSIDE the cells in the blood PLASMA as well as in the intercellular fluids. K ions are present inside cells. | |
| 12. |
What is polar covalent bond? Explain with example. |
|
Answer» Solution :(i) Ifa covalent bnd is formed between atoms having different electronegativities, the atom with higher electronegativity will have greater tendency to attract the shared pair of ELECTRONS towards itself than the other atom. As a result, the cloud of shared electron pair gets distorted and polar covalent bond is formed. (ii) Example: HF- Hydrogen fluoride: The electronegativities of hydrogen and fluorine on Pauling.s SCALE are 2.l and 4 respectively. It MEANS that fluorine attract the shared pair of electrons approximately twice as much as hydrogen which leads to partial NEGATIVE charge OuoThe atom and partial positive charge on hydrogen atom. Hence , the H-F bond is said to be a polar covalent bond. |
|
| 13. |
Which of the following parameters correctly represent conditions for a spontaneous process with no non P-V work involved? (P) (dH)_(S,P)lt0 (Q) (dU)_(S,P)lt0 (R ) (dG)_(T,P)lt0 (S) (dH)_(P,T)gt0 (T) (d,S)_(universe)gt0 (U) (dS)_(U,V)gt0 |
|
Answer» Options (Q),(R ) and (T) are CORRECT |
|
| 14. |
Which of the following unit is derived unit ? |
|
Answer» TEMPERATURE |
|
| 15. |
Write IUPAC name of following |
Answer» SOLUTION :
|
|
| 16. |
The standardmolar entropy of H_(2)O(l)is 70 JK^(-1) mol^(-1). Will the standard molar entropy of H_(2)O(s) be more, or less than 70 JK^(-1) mol^(-1) ? |
| Answer» Solution :In ice, moleculesof`H_(2)O` are LESS RANDOM than in LIQUID water. HENCE , molar entropy of ice, `H_(2)O(s)`, will be less thanthat of liquid water, `H_(2)O(l)`. | |
| 17. |
Which of thefollowingspecieshas thehighestelectronaffinity ? |
|
Answer» `F^(-)` |
|
| 18. |
What are crystalline hydrates? Explain it with example. |
|
Answer» Solution : (i) In these, hydrogen bonding is very important. OFTEN the water molecules serve to fill in the interstices and bind together structure. (ii) A specific example is `CuSO_(4)5H_(2)O`. (iii) ALTHOUGH there are five water molecules for every divalent copper ion, only four are coordinated to the cation, it.s six-coordination being completed from sulphate ANIONS. The fifth water molecule is held in place of hydrogen bonds, O- H O, between it and TWO coordinated water molecules and then coordinate sulphate anion. (iv) Water forms hydrated salts during crystallization. Examples, `Na_(2)CO_(3)10H_(2)O`, `FeSO_(4).7H_(2)O`. (v) The water present in the hydrates is called as water of hydration |
|
| 19. |
Which one of the compound is achiral : |
|
Answer»

|
|
| 20. |
What will be the change in boiling point of alkene compound on addition of each -CH_(2) group ? |
| Answer» Solution :On addition of each `-CH_(2)` BOILING point increases approximately by 20-30 K. | |
| 21. |
The solubility of metal halides depends on their nature , lattice enthalpy and hydration enthalpy of the individual ions . Amongst fluorides of alkali metals , the lowest solubility of LiF in water is due |
|
Answer» ionic nature of lithium FLUROIDE |
|
| 22. |
Which of the following statements is(are) correct |
|
Answer» In SEMI conductor , electrical resistivity DECREASES with a rise in temperature |
|
| 23. |
The surface tension of water at 20°C is 72.75 dyne cm^(-1) . Its value in SI system is |
|
Answer» `2.275 N m^(-1)` |
|
| 24. |
The state of a gas can be described by quotingthe relationship between "…................". |
|
Answer» pressure, VOLUME, TEMPERATURE |
|
| 26. |
Which of the following properties increases across a period ? |
|
Answer» REDUCING PROPERTY |
|
| 27. |
What happens when calcium carbide is treated with water? |
|
Answer» Ethane is formed |
|
| 28. |
Which of the following solution has the highest boiling point ? |
|
Answer» 1) `5.85%` solution of NaCl |
|
| 29. |
What change would be observed when covalent bond is form ? |
| Answer» SOLUTION :OCTET STRUCTURE is FORM. | |
| 30. |
What are ternary Hydrides ? Give examples. |
| Answer» Solution :Ternary hydrides are COMPOUNDS in which the MOLECULAR is constituted by HYDROGEN and TWO types of element , e.g., `LiB_(4)` or `LiAlH_(4)`. | |
| 31. |
Which of the following relation is correct for an ideal regarding its pressure (p) and translational kinetic energy per unit volume (E ) ? |
|
Answer» `p=(3)/(2)E` `P=(2)/(3)E` |
|
| 32. |
What do you meanby Entropyof vaporisation or Entropy offusion or Entropy of sublimation . |
| Answer» SOLUTION :REFER to the TEXT. | |
| 33. |
What volume of H_(2) at NTP is required to convert 2.8g of N_(2) in to NH_(3)? |
|
Answer» 2240ml |
|
| 34. |
What are non-ideal solution ? Give example. |
|
Answer» Solution :(i) The solution which do not obey Raoult.s law over the ENTIRE range of concentration are CALLED non-ideal solutions. The devintion of the non-ideal solution from the Raoult.s law may be POSITIVE (or) negative. (II) Example. Ethyl alcohol and cyclohexane. |
|
| 35. |
Which of the following is associated with Torsional strain ? |
|
Answer» Repulsion between BOND pair of electrons |
|
| 36. |
The uncertainity in the momentum of a particle is 3.31 xx 10^(-2) kg ms^(-1). The uncertainity in its position is (in metres) |
|
Answer» `1.59xx10^(-33)` |
|
| 37. |
When 2-methyl butyl bromide is treated with sodium ethoxide I ethanol, what will be the major product? |
|
Answer» 2-methyl but-2-ene |
|
| 38. |
What is mechanism of the reaction? |
| Answer» Solution :In the ORGANIC reactions, that series of SIMPLE STEPS which COLLECTIVELY represent the chemical change, from substrate to product, this is CALLED as the mechanism of the reaction. The sloweststep in the mechanism determines the overall rate of the reaction. | |
| 39. |
When PbO_(2) reacts with concentrated HNO_(3), the gas evolved is |
|
Answer» `NO_(2)` |
|
| 40. |
The value of amgnetic moment is zero in the case of antiferromagnetic substances because the domians ……. |
|
Answer» get oriented in the direction of the APPLIED MAGNETIC field. |
|
| 41. |
Which point defect in crystals of a solid decreases the density of the solid ? |
| Answer» SOLUTION :SCHOTTKY DEFECT. | |
| 42. |
What is photoelectric effect ? State the result of photoelectric effect experiment that could not be explained on the basis of laws of classical physics. Explain this effect on the basis of quantum theory of electromagnetic radiations. |
Answer» Solution :Photoelectric effect : When radiation with certain minimum frequency (p0) strike the surface of a metal, the electrons are ejected from the metal surface. This phenomenon is called photoelectric effect. The electrons emitted are called photoelectron  Equipment for studying the photoelectric effect. Light of a particular frequency strikes a clean metal surface inside a vacuum chamber. Electrons are ejected from the metal and are counted by a detector that measures their kinetic energy. The result observed in this experiment were (i) The electrons are ejected from the metal surface as soon as the beam of light strikes the surface, i.e., there is no time lag between the striking of light beam and the ejection of electrons from the metal surface. (ii) The num ber of electrons ejected is proportional to the intensity or brightness of light. (iii) For each metal, there is a characteristic minimum frequency, VQ (also known as threshold frequency) below w hich photoelectric effect is not observed. At a frequency , the ejected electrons come out with certain kinetic energy. The kinetic energies of these electrons increases with the increase of frequency of the light used. The above observation cannot be explained by the electromagnetic wave theory. According to this theory, since radiations were continuous, therefore, it should be possible to accumulate energy on the surface of the metal, irrespective of its frequency and thus, radiations of all frequencies should be able to eject electrons. Similarly, according to this theory, the energy of the electrons ejected should depend upon the intensity of the INCIDENT radiation. Particle Nature of Electromagnetic Radiation : To explain the phenom ena of ‘black body radiation’ and ‘photoelectric effect’, Max Planck in 1900, put forward a theory known after his NAME as Planck.s quantum theory. This theory was further extended by Einstein in 1905. The important points of this theory are as FOLLOWS : (i) The radiant energy is emitted or absorbed not continuously but discontinuously in the form of small discrete packets of energy.Each such packet of energy is called a ‘quantum’. In case of light, the quantum of energy is called a ‘photon’. (ii) The energy of each quantum is directly proportional to the frequency of the radiation, i.e., E v or E = hv where, h is a proportionality constant, called Planck.s constant, Its value is approximately equal to `6.626 xx 127 erg s or 6.626 xx 10^(34)` JS. (iii) The total amount of energy emitted or absorbed by a body will be some whole number quanta. Hence, E = nhv (where, n is any INTEGER), |
|
| 43. |
Which bond angelthetawould result in the maximum dipole moment for the triatomic molecule XY_2 shown below ? |
|
Answer» ` THETA = 90^@` `mu` is MAXIMUM . |
|
| 44. |
What is the significance of the term isolated gaseous atom while defining the ionization enthalpy. |
|
Answer» |
|
| 45. |
Writethe namesand atomic numbers of the followingelements : (i) The fourthalkali metal (ii) The thirdalkaline earthmetal (iii) Thefifthelementof the firsttransition series (iv) Thefirstinnertransition element and (v) Thesixthnoblegas. |
| Answer» Solution :(i) Rb (Z= 37), (ii) CA (Z = 20 ), (ii) Mn(Z=25), (IV) CE(Z= 58), (v) Rn (Z=86) | |
| 46. |
Which of the following reactions will produce the 2-pentyne in good yield? CH_(3)-CH_(2)-C-=C-CH_(3) |
|
Answer»
|
|
| 47. |
Which of the following equation is a balanced equation ? |
|
Answer» `FeS_(2) + 4O_(2) rarr 8 SO_(2) + 2Fe_(2) O_(3)` |
|
| 48. |
Write the chemical reactions to show the amphoteric nature of water. |
|
Answer» Solution :WATER is AMPHOTERIC in NATURE and it behaves both as an acid as well as base. With acids STRONGER than itself (eg. `H_(2)S`) it behaves as a base and with bases stronger than itself (eg, `NH_(3)`) it acts as an acid. (i) As a base: `H_(2)O_((L))+H_(2)S_((aq))toH_(3)O_((aq))+HS^(-)` (ii) As an acid: `H_(2)O_((l))+NH_(3(aq))toOH_((aq))^(-)+NH_(4(aq))^(+)` |
|
| 49. |
Which of the following molecule does not obey the octet rule and also has Ione pair on the central atom? |
|
Answer» `"CC"l_(4)` |
|
| 50. |
Which elements of group shows difficult tendency of value electron gain enthalpy? |
|
Answer» Solution :OXYGEN and sulphur shows different tendency of electron gain ENTHALPY because electron gain enthalpy of oxygen is LESS NEGATIVE than sulphur. Electron gain enthalpy of fluorine is less negative than chlorine. |
|