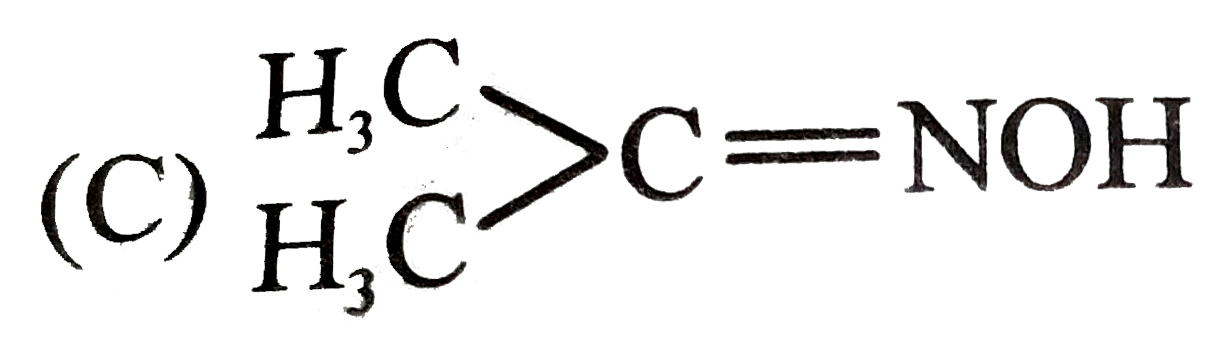Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following orbitalsare possible 1p , 2s, 2p , 3f |
|
Answer» SOLUTION :2S and2pare ORBITALS But1p and3forbital are notpossible |
|
| 2. |
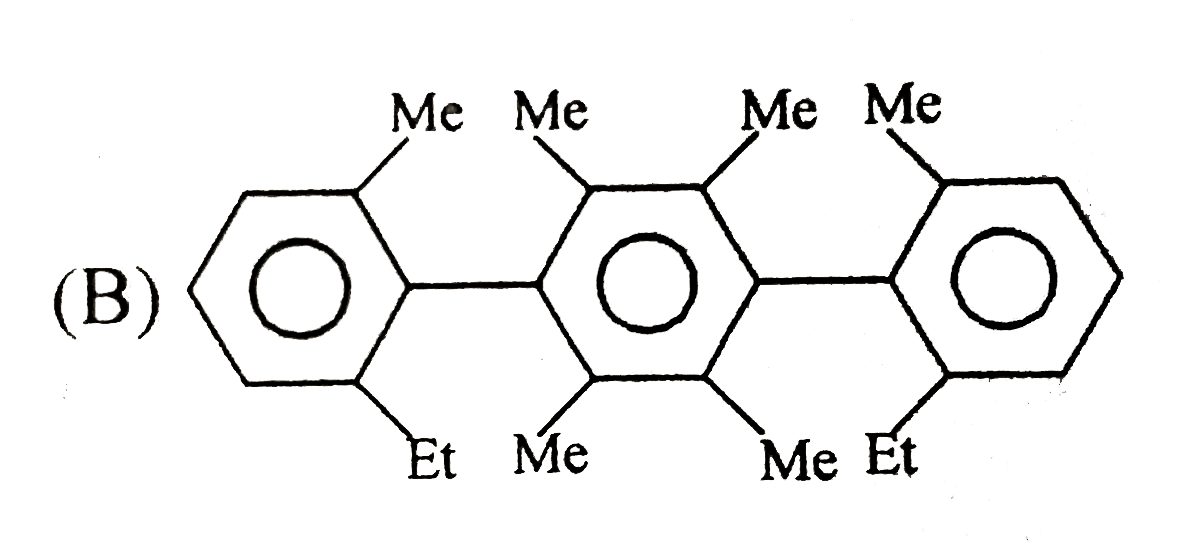
Which will show geometrical isomerism ? |
|
Answer» `CH_(3)CH=NOH`  
|
|
| 3. |
Which of the following statements is correct regarding the slag obtained during the extraction of a metal like copper or iron ? |
|
Answer» The SLAG is lighter and has LOWER metling point than the METAL. |
|
| 4. |
Which amongthe followingare statefunctions ? |
|
Answer» Internal ENERGY |
|
| 5. |
Write the expression for the equilibrium constant, K_c for each of the following reactions (1)2NOCl_((g)) hArr 2NO_((g)) + Cl_(2(g)) (2) 2Cu(NO_3)_(2(s))hArr 2CuO_((s)) + 4NO_(2(g)) + O_(2(g)) (3)CH_3COOC_2H_(5(aq)) + H_2O_((l)) hArr CH_3COOH_((aq)) + C_2H_5OH_((aq)) (4) Fe_((aq))^(3+) + 3OH_((aq))^(-) hArr Fe(OH)_(3(s)) (5)I_(2(s)) + 5F_(2(s)) hArr 2IF_(5(g)) |
|
Answer» Solution :(1)`K_c=([NO_((g))]^2[Cl_(2(g))])/([NOCl_((g))]^2)` (2)`K_c=([CuO_((s))]^2[NO_(2(g))]^4 [O_(2(g))])/([Cu(NO_3)_(2(s))]^2)` In this equilibrium CUO and `Cu(NO_3)_2` are solid. So, at equilibrium its CONCENTRATION are not changed and constants. So, ignore in expression, `K_c=[NO_(2(g))]^4 [O_(2(g))]` (3)`K_c=([CH_3COOH_((aq))][C_2H_5OH_((aq))])/([CH_3COOC_2H_(5_(aq))][H_2O_((l))])` Here , `H_2O` is liquid its concentration is constant during REACTION, so ignore it. `K_c=([CH_3COOH_((aq))][C_2H_5OH_((aq))])/([CH_3COOC_2H_(5(aq))])` (4)`K_c=([FE(OH)_(3(s))])/([Fe_((aq))^(3+) ][OH_((aq))^(-)]^3)` In this equilibrium `Fe(OH)_3` is solid and amount of solid i.e. its concentration is equal to density. Density is constant at temperature, so its not affect the equilibrium constant. `(Fe(OH)_(3(s)))` is ignore `therefore K_c=1/([Fe^(3+)][OH^-]^3)` (5)`K_c` for the product `IF_5` of the reaction of `I_(2(s))` and `F_(2(g))` is , `K_c=([IF_(5(g))]^2)/([I_(2(s))] [F_(2(g))]^5)` `I_(2(s))` is solid so its concentration remain constant so it is ignore in expression. `therefore K_c=([IF_(5(g))]^2)/([F_(2(g))]^5)` |
|
| 6. |
What are called earths ? |
| Answer» Solution :Long time BACK,chemicals areclassifiedinto SPRITS and EARTHS. Sprits are generallyvolatile SUBSTANCES .Warths are those chemicals which are ALMOST non- voltatile ,thermally very stable and were present on the earth , where they were placed . | |
| 7. |
Which one of the following can be recycled ? |
|
Answer» Garbage |
|
| 8. |
What is the formula of cryolite ? |
|
Answer» `Na_3 AlF_6` |
|
| 9. |
What is the value of K_p for decomposition of NH_4HS ? P = Total pressure |
|
Answer» `K_p=P` |
|
| 10. |
Which among the following is used as cathode in Castner Kellner cell ? |
| Answer» ANSWER :A | |
| 11. |
Which of the following carbides is used for welding ? |
|
Answer» `Be_4C` |
|
| 12. |
What is the weight of one molecule of CO_(2) ? (M.wt of CO_(2)=44g/mole) |
|
Answer» `3.37xx10^(-23)g` `6.022xx10^(23)` molecule `= 44 g CO_(2)` `:.` 1 molecule of `CO_(2)=(?)` `= (44)/(6.022xx10^(23))=7.0307 XX 10^(-23)` `= 7.31xx10^(-23)g` |
|
| 13. |
The structure of 4-methyl pent-2-ene is |
|
Answer» `(CH_(3))_(2)CH-CH_(2)CH=CH_(2)` |
|
| 14. |
The solubilities of carbonates decreases down ther magnesium group due to decrease in |
|
Answer» INTER IONIC attraction |
|
| 15. |
Which of thefollowingstatements is incorrectin relation to ionizationenthalpy ? (a)ionizationenthalpyincreasesfor eachsuccessiveelectron . Thegreatestincrease in ionizationenthalpyis experienced on removal ofelectrons from corenoblegasconfiguration. End ofvalence electron fromorbitalsbearing lowern value is easier thanfromorbitalhavinghigher n value. |
| Answer» SOLUTION :Statement (d ) isincorrect . Thecorrectstatementis : Removalof ELECTRON from orbitals bearinglower nvalue isdifficult than from orbital havinghigher n value . Allother STATEMENTS arecorrect. | |
| 16. |
Which of the following will racemize in presence of acid or base traces in aqueous medium? |
|
Answer» `Me-UNDERSET(O)underset(||)C-underset(CH_(3))underset(|)OVERSET(CH_(3))overset(|)C-Ph` |
|
| 17. |
What is Bio-chemical oxygen demand ? |
| Answer» SOLUTION :The AMOUNT of oxygen required by bacteria to break down the organic matter present in a certain volume of a SAMPLE of WATER, is called BIOCHEMICAL Oxygen Demand (BOD). | |
| 18. |
Which of the following compound possess geometrical isomerism ? |
|
Answer» Alkane |
|
| 19. |
What is the state of hybirdization of carbon in (a) CO_(3)^(2-) (b) diamond (c ) graphite ? |
| Answer» SOLUTION :(a) `SP^(2)` , (b) `sp^(3)`, (c ) `sp^(2)` | |
| 20. |
What volume of 0.1 M H_(2)SO_(4) is required in litres to neutralize completely 1 litre of a solution containing 20 g of NaOH ? |
|
Answer» `5.0` Reaction : `H_(2)O+2NaOH to Na_(2)SO_(4)+2H_(2)O` `(M_(1)V_(1))/(1)=(M_(2)V_(2))/(2)` `0.1xxV_(1)=(0.5xx1)/(2)` `V_(1)=2.5 L` |
|
| 21. |
What is the reaction to increase the intensity of red colour by adding HgCl_2 positive in equilibrium of red colour by adding HgCl_2 positive in equilibrium of Fe_((aq))^(3+) + SCN_((aq))^(-) hArr [Fe(SCN)]_((aq))^(2+) |
| Answer» Solution :`HgCl_2` is added in solution So, `Hg^(2+)` IONS are increases and combine with `SCN^-` and form `(Hg(SCN)_4)^(2-)`. `[SCN]^(2-)` DECREASES. To MINIMIZE this EFFECT `[FE(SCN)]^(2-)` form by forward reaction. The solution becomes more red. | |
| 22. |
The value of carbon-carbon single bond length is …………………… |
|
Answer» 1.43Å |
|
| 23. |
Which of the following in not a charateristic of a crystalline solid ? |
|
Answer» Define and characteristic heat of FUSION. |
|
| 24. |
When an alkali metal dissolves in liquid ammonia the solution can acquire different colours. Explain the reasons for this type of colour change |
| Answer» Solution :A dilute solution of alkali metal in `NH_3` has deep blue colour due to the formation of ammoniated ELECTRONS which ABSORB in the RED REGION of VISIBLE light. | |
| 25. |
Write the oxdation number of the underlined atoms in each of the following: (i)KMnO_(4) (ii) H_(4)^(+) (iii) S_(2)O_(3)^(2-) (iv) Na_(3)PO_(4) |
|
Answer» |
|
| 27. |
Which of the following is true about the value of refrective of quartz glass ? |
|
Answer» <P>POSITIVE |
|
| 28. |
What are the values of n_(1) and n_(2) respectively for H_(beta) line in the Lyman series of hydrogen atomic spectrum? |
| Answer» Solution :`alpha implies2 to 1, BETA implies3 to 1 ` | |
| 29. |
Under the name auricome, H_2O_2 is used |
|
Answer» As an antiseptic |
|
| 30. |
Which of the following compounds has hydrogen bonding? |
|
Answer» `HCL` |
|
| 32. |
The sharp melting point of crystalline solids is due to ______ |
|
Answer» a REGULAR arrangement of CONSTITUENT particles observed over a short distance in the crystal lattice. |
|
| 33. |
What are activating and deactivatikn groups |
|
Answer» Solution :(i) Whenmonosubstiutedhenzeneundergoesand electrophilicsubstitution reaction thefunctionalgroupalreadyattached to it. (ii) Somegroupincreasesthe reactivityof BENZENERING ANDARE knownas ACTIVATING groupgroups |
|
| 34. |
Which principle states that "A given compound always contains exacly the same proportion of elements by weight" ? |
|
Answer» Law of conservation of mass |
|
| 35. |
Which is the strongest acid in the following |
|
Answer» `HCIO_(4)` |
|
| 36. |
Write the half eaction for fthe following redox reac tiojns (a)2Fe^(3+)(Aq)+2I^(-)(aq) rarr 2 Fe^(2+)(aq)+I_(2)(aq) (b)Zn(s)++2 H^(+)(aq)rarr Zn (c )AI(s)+3Ag^(+)(aQ)rarrAI^(3)rarr AI^(3+)(aq)+3Ag (s) |
| Answer» SOLUTION :`{:((a) 2I^(-)(aq) to I_(2)(aq) +2E^(-)),(ul(FE^(3+)(aq)+e^(-) to Fe^(2+)(aq)"]"xx2)),(2Fe^(3+) (aq)+2I^(-)(aq)to2Fe^(2+)(aq)+I_(2)(aq)),((b)ZN(s)toZn^(2+)(aq)+2e^(-)),(ul(2H^(+)(q)+2e^(-)toH_(2)(g))),(Zn(s)+2H^(+)(aq)toZn^(2+)(aq)+H_(2)(g)),((C)Al(s)to Al^(3+)(aq)+3e^(-)),(ul(Ag^(+)(aq)+e^(-) to Ag(s)"]"xx3)),(Al(s)+3Ag^(+)(aq) to Al^(3+)(aq)+3Ag(s)):}` | |
| 37. |
Which of the following metal sulphate is more soluble in water |
|
Answer» `BasO_(4)` `BeSO_(4) gt MgSO_(4) gt CaSO_(4) gt SrSO_(4) gt BaSO_(4)` |
|
| 38. |
Which of the following statements regarding heat of hydrogenation of alkenes is true |
|
Answer» Cis-2-butene has HIGHER HEAT of HYDROGENATION than trans-2-butene |
|
| 39. |
What is "Calgon" ? Give its molecular formula. |
| Answer» SOLUTION :Sodium hexametaphosphate commercialy CALLED .calgon. Its MOLECULAR FORMULA is `Na_6P_6O_18`. | |
| 41. |
Under which situations can an amorphous substances change to crystalline form ? |
| Answer» Solution :As a result of slow heating and cooling over a long PERIOD , an amorphous SOLID acquires some CRYSTALLINE character , e.g., window GLASS of old BUILDINGS . | |
| 42. |
Write the half cell reaction and the overall cells reaction for the electrochemical cell : Zn|Zn^(2+)(1.0M)||Pb^(2+)(1.0M)|Pb Calculate the standard e.m.f for the cell if standard electrode potentials (reduction ) for Pb^(2+)|Pb and Zn^(2+)|Zn electrodes are -0.126 V and -0.763 V respectively. |
|
Answer» Solution :Zn ELECTRODE acts as anode while Pb electrode acts as cathode and , therefore , oxidation OCCURS at zinc electrode and REDUCTION occurs at lead electrode . The half cell reactions are : Oxidation half reactions : `Zn(s) rarr Zn^(2+)(aq)+2e^(-)` Reduction half reaction : `Pb^(2+)(aq) +2e^(-) rarr Pb(s)` Overall cell reaction `Zn(s) +Pb^(2+)(aq) rarrZn^(2+) (aq) +Pb(s)` E.M.F = `E_R^@ - E_L^@` `=-0.126-(-0.763)` = 0.637 V. Note . The EMF of the cell can also simply be calculated as : `E_("cell")^@=E_R^@-E_L^@` If the value of `E_("cell")^@ ` is positive , then the cell is correctly REPRESENTED , i.e. , oxidation occurs at left electrode (anode) and reduction occurs at the right electrode (cathode). On the other hand , if the value of `E_("cell")^@` comes out to be negative , then the cells is wrongly represented . Its order is reversed to get positive value for `E_("cell")^@`. |
|
| 43. |
The volume of perhydrol required to get one liter of oxygen at STP is |
|
Answer» 20ml |
|
| 44. |
Writethe electronic configurationof the followingions : (a )H^(-)( b) Na^(+)(c ) O^(2-)(d ) F^(-) |
|
Answer» Solution :(a ) `H^(-)(2E ) ` So ,`1s^(2)` ( b) `Na^(+) (11-1)=10^(2) ` So `1s^(2)2S^(2) 2p^(6)3S^(0)` (c ) `O^(2) (8+2)=10^(2) ` So `1s^(2) 2s^(2) 2p^(6)` (d ) `F^(-)(9+1) = 10^(2) `So`1s^(2) 2s^(2) 2p^(6)` |
|
| 45. |
What will be the correct order of vapour pressure of water, acetone and ether at 30^(@)C. Given that among these compounds, water has maximum boiling point and ether has minimum boiling point ? |
|
Answer» Water `LT` ether`lt ` acetone |
|
| 46. |
Which of the following statement is not correct in respect of hydrophilic sol. ? |
|
Answer» The particles are hydrated |
|
| 47. |
Which of the following compound is known as inorganic benzene ? |
|
Answer» BORON |
|
| 48. |
The species in which the N atom is in a state of sp hybridisation is |
|
Answer» `NO_(2)^(+)` 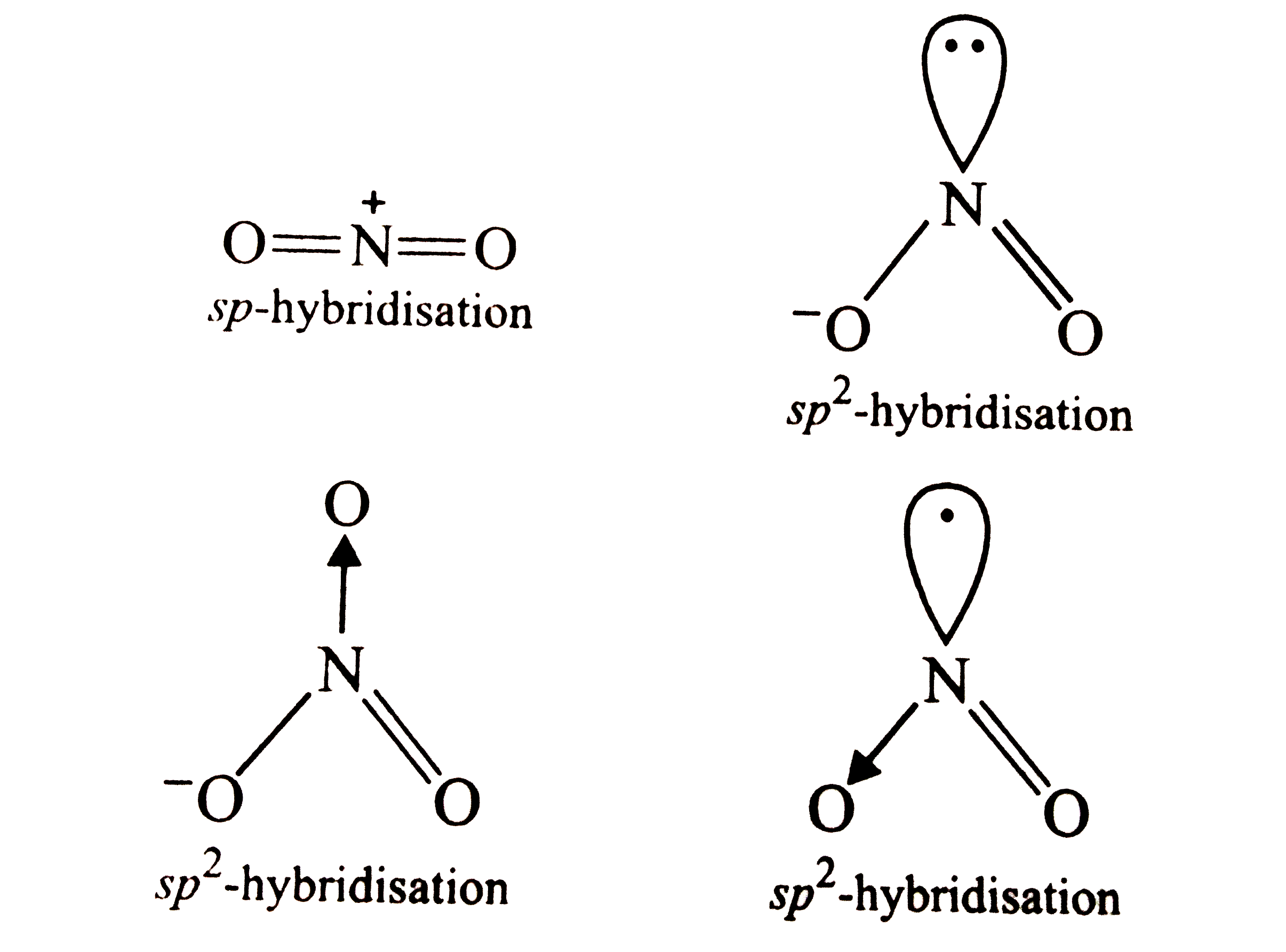
|
|
| 49. |
What volume of 6M HCl and 2M HCl should be mixed to get one litre of 3M HCl? |
|
Answer» Solution :Suppose the volume of 6M HCl required to OBTAIN 1L of 3M HCl = XL Volume of 2M HCl required = (1-x)L Applying the molarity equation `M_1 V_1 + M_2V_2 = M_3V_3 ` `6 MHCL + 2MHCl = 3MHCl` `6x+2(1-x) = 3 XX 1` `6x + 2 -2x = 3 ` ` 4X =1 ` `x =0.25 L` HENCE, volume of 6 M HCl rquired = 0.25 L Volume of 2M HCl required = 0.75 L |
|
| 50. |
Which of the following reactions give meso product ? |
|
Answer»
B) Cis ALKENE `UNDERSET(OH^(-)) overset("cold" KMnO_4)to` cis HYDROGENATION `to 1 ` cis addition is anti additionracemic mixture (CAR) cis syn addition-meso C) C) Trans alkene- epoxide formation with peroxidesepoxide on hydrolysis gives trans diols so anti addition Trans-anti addition (ATM) gives meso D) Cyclo hexen is cis alkene anti addition racemic so (D) is not the correct ANSWER |
|