Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Van der Waal's radius is used for |
|
Answer» Molecular SUBSTANCES in gaseous STATE only |
|
| 2. |
What happens when H_(3) PO_(3) is heated ? |
|
Answer» Solution :The oxidation state of P in `H_(3)PO_(3)" is "+3`. Since this VALUE is intermediate between the highest (+5) and lowest(-3) oxidation states of P, therefore, when `H_(3)PO_(3)` is HEATED it undergoes DISPROPORTIONATION to form `PH_(3)andH_(3)PO_(4)` with oxidation states of -3 and +5 respectively. `UNDERSET("Orthophosphorous acid")overset(+3)(4H_(3)PO_(3))overset("Heat")tounderset("Phosphine")overset(-3)(PH_(3))+underset("Orthophosphoric acid")overset(+5)(H_(3)PO_(4))` |
|
| 4. |
Which of the following has maximum number of lone pairs associated with Xe? |
| Answer» Solution :`XeF_(2)` | |
| 5. |
Write the atomic number of the element present in the third and seventeenth group of the periodic table. |
|
Answer» Solution :Since it belongs to 3RD period, it will have outermost shell , n = 3. It.s configuration will be `3s^(2)3p^(5)`. `:.{:(He,Ne,Ar),(2,10,18):}` ` :. ` atomic number `(Z = 10 + 2 + 5) = 17` ` :. ` element Cl (Chlorine) |
|
| 6. |
What is screening effect? How is it related to IE? |
|
Answer» |
|
| 7. |
Which of the following does not alter the equilibrium ? |
|
Answer» CATALYST |
|
| 8. |
What is Aromatisation? Give example. |
Answer» Solution :Hexane or higher akanes when heated in presence of vanadium pentoxide `(V_(2)O_(5))`, molybdernium oxide `(MO_(2)O_(3))` or chromium oxide `(Cr_(2) O_(3))` supported on ALUMINA `(Al_(2) O_(3))` at 800 K and 10-20 atm pressure GIVE benzene or its alkyl DERIVATIVES with the liberation of hydrogen. this dehydrogenation process which involves cyclisation of alkanes is known as AROMATIZATION. 
|
|
| 9. |
Xoverset(CoCl_(2))rarrCaCl_(2)+Yuarr, the effective ingredient of X is : |
|
Answer» `OCL^(-)` |
|
| 10. |
What mass of an oxygen gas will occupy 8.21 L of volume at 1 atm pressureand 200 K temperature ? |
| Answer» ANSWER :a | |
| 11. |
The standard value of enthalpy of combustion of benzoic acid is ____ |
| Answer» SOLUTION :`-3237 "KJ MOL"^(-1)` | |
| 13. |
What is difference between STP or NTP and SATP ? |
|
Answer» Solution :STP, 1 ATM `= 1.0132xx10^(5)` Pa SATP, 1 bar `= 1XX10^(5)` Pa TEMPERATURE is same at 273.15 K or `0^(@)C`. |
|
| 14. |
What are isotopes? Write the names of isotopes of hydrogen. |
|
Answer» Solution : (i) Isotopes are atoms of the same element that have the same ATOMIC NUMBER but having DIFFERENT mass numbers (or) Isotopes are atoms with the same number of protons and electrons but differ in number of neutrons. (ii) Hydrogen has THREE naturally occurring isotopes namely PROTIUM `("_(1)H^(1))`, Deuterium`("_(1)H^(2))`,and Tritium `("_(1)H^(3))` 
|
|
| 15. |
The sulphur dioxide obtained by the combustion of 8 gms of sulphur is passed into Bromine water. The solution is then treated with barium chloride solution. The amount of barium sulphare formed is |
|
Answer» 1 mole 8 gm of sulphur is present in `1//4` mole `BaSO_(4)` |
|
| 16. |
The volume of water to be added to 400 mL of N//8 HCl to make it exactly N//12, is : |
|
Answer» 400 mL |
|
| 17. |
What happens when quick lime is heated with silica ? |
| Answer» Solution :`CaO + SiO_2(s) overset(DELTA) to UNDERSET("CAL. Silicate")(CaSiO_3(s))` | |
| 19. |
Which silicon compound is used as lubricant |
| Answer» Solution :Silicone is USED as libricant | |
| 21. |
The value of K_c = 4.24 at 800K for the reaction, CO_((g)) + H_2O_((g)) hArrCO_(2(g)) + H_(2(g))Calculate equilibrium concentrations of CO_(2), H_2, CO and H_2O at 800 K, if only CO and H_2O are present initially at concentrations of 0.10M each. |
|
Answer» Solution :`{:("REACTION :", CO_((g))+, H_2O_((g)) hArr , CO_(2(g)) + , H_(2(g))),("INITIAL conc. :",0.1 M,0.1 M, 0 M, 0 M),("Reaction change :",-xM,-X M,+x M,+x M),("Concentration of equilibrium:",(0.1-x)M,(0.1-x)M,x M, x M):}` where , x= amount of CO consume in reaction = amount of `H_2O` x=made of product = amount of `CO_2` = amount `H_2` `K_c=([CO_2][H_2])/([CO][H_2O])=((x)(x))/((0.1-x)(0.1-x))=x^2/(0.1 -x)^2` `therefore x^2/(0.1-x)^2=4.24` `therefore x^2=4.24(0.01-0.2x - x^2)` `therefore x^2=0.0424-0.848x+4.24x^2` `therefore underseta(3.24x^2)-undersetb(0.848x)+undersetc(0.0424)=0` This is quadratic equation in which , a=3.24 , b=-0.848 and c=0.0424 This equation of quadratic , `ax^2+bx+c` `therefore x=(-bpmsqrt(b^2-4ac))/(2a)` `therefore x=(-(-0.848)pmsqrt((0.848)^2-4(3.24)(0.0424)))/(2(3.24))` `therefore x=(+0.848 pm SQRT(0.7191-0.5495))/6.48` `=(0.848pmsqrt(0.1696))/6.48 =(0.848 pm 0.4118)/6.48` `=1.2598/6.48` `0.4362/6.48` =0.1944 OR 0.0673 The value 0.1994 should be neglected because it will give concentration of the reactant which is more than initial conc. So, x=0.0673 M `therefore` Product of equilibrium `[CO_2]=[H_2]`=0.0673 M Reaction of equili. [CO]=`[H_2O]`= (0.1-x)M =(0.1-0.0673)M = 0.0327 M |
|
| 22. |
Which of the following compound is insoluble in water? |
| Answer» Answer :B | |
| 23. |
Which of the following pair of species can be separated by dil. HCI? |
|
Answer» `Hg(NO_(3))_(2)` and `Hg_(2)(NO_(3))_(2)` |
|
| 24. |
What happens when ethylene is passed through cold dilute alkaline potassium permanganate. |
|
Answer» `(##SUR_CHE_XI_VO2_S_MQP_02_E01_051_A01##)` |
|
| 25. |
Which of the following is most reactive towards nucleophilic substitution reaction? |
|
Answer» `CH_(2)=CH-C L` |
|
| 26. |
What is water gas ? How is it prepared ? |
|
Answer» Solution :An EQUIMOLAR mixture of CO and `H_(2)` is CALLED water GAS. It is prepared by passing steam over red hot coke. `C(s) + H_(2)O(g) overset(1273 K) to CO(g) + H_(2)(g)` |
|
| 27. |
When pressure is expressed in dynes and volume in cm^3the value of R is ………… . |
| Answer» SOLUTION :`8.314xx10^7 " ERGS " K^(-1) MOL^(_1)` | |
| 28. |
Which of the following series contains only electrophiles |
|
Answer» `H_(2)O, SO_(3), H_(3)O^(+)` |
|
| 29. |
The second electron affinity of oxygen is +744 kJ*mol^(-1) then the second electron affinity of sulphur is- |
|
Answer» `-200KJ*mol^(-1)` |
|
| 30. |
Which of the following ions wilt cause hardness in water sample ? |
|
Answer» `Ca^(+2)` |
|
| 31. |
Which of the following compound does not show geometrical isomerism? |
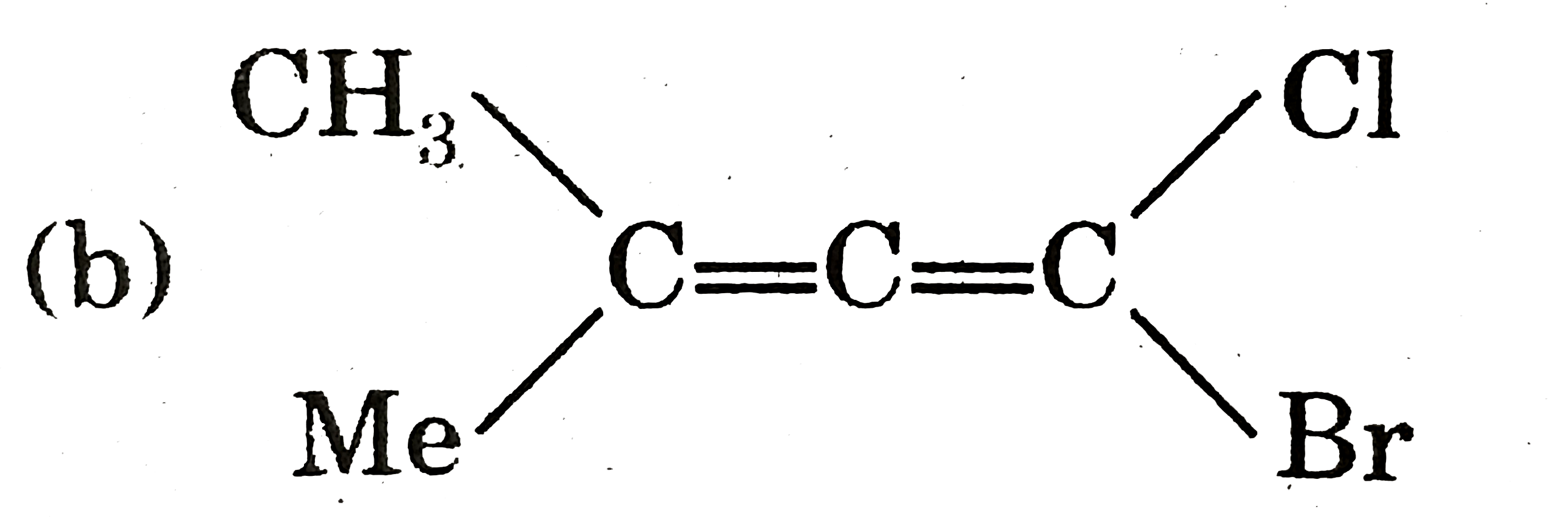
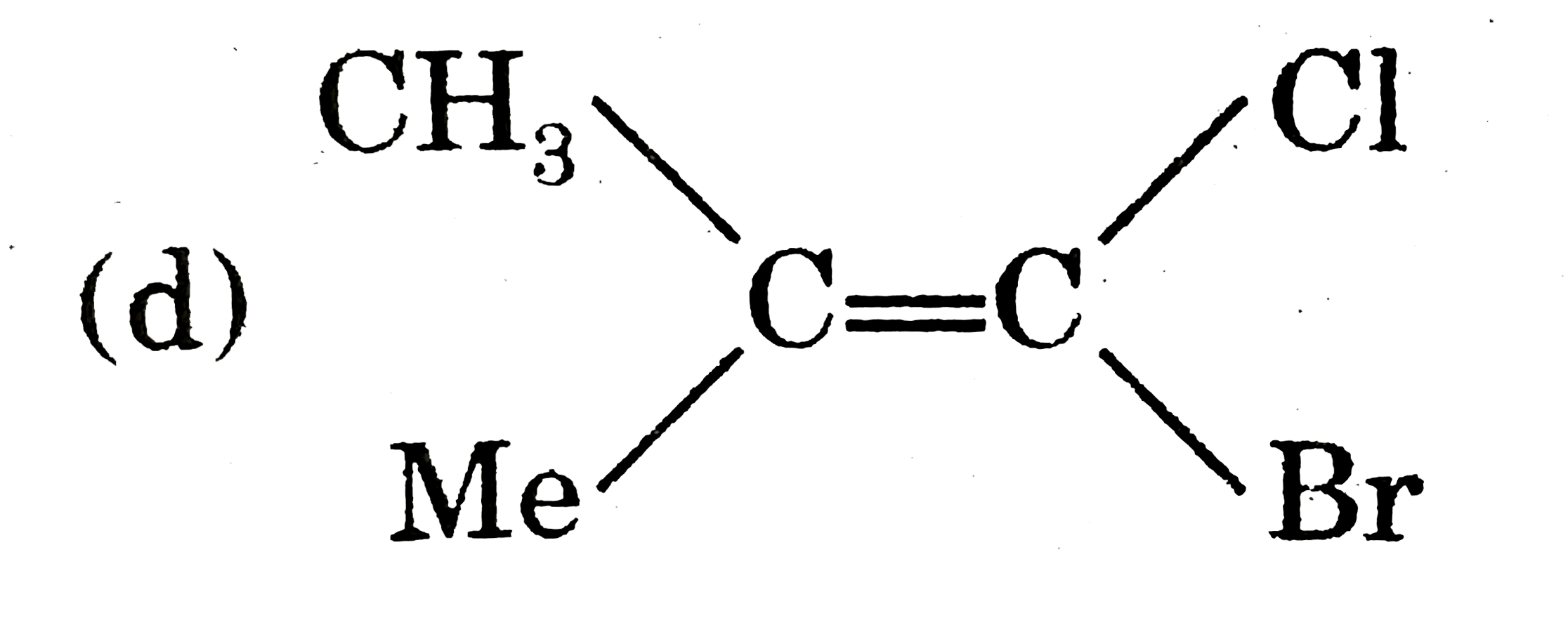
|
Answer»
 `therefore` It cannot show geometrical isomerism |
|
| 32. |
Which of the following can work as a dehydrating agent for alcohols ? |
|
Answer» `H_(2)SO_(4)` |
|
| 33. |
Which of the following compoundsare readily soluble in water ? |
|
Answer» `BeSO_(4)` |
|
| 34. |
What atoms are indicated by the following configurations ? (i) [He] 2s^(1)(ii) [Ne] 3s^(2) 3p^(3)(iii) [Ar] 4r^(2) 3d^(1) |
| Answer» SOLUTION :(a) `._(3)Li` (B) `._(15)P` (C) `._(21)SC` | |
| 35. |
When a sample of baking is strongly ignited in a crucible, it suffered a loss in weight of 3.1 g. The mass of baking soda is |
|
Answer» `16.8g` |
|
| 36. |
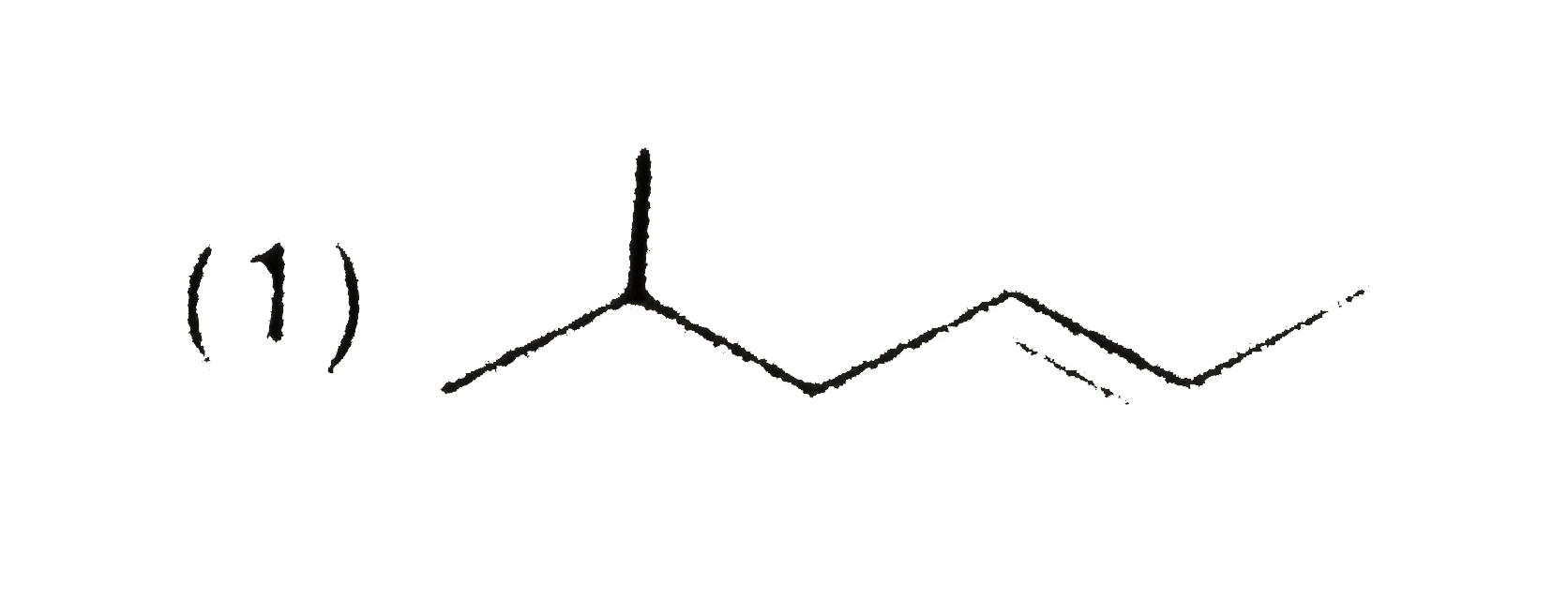
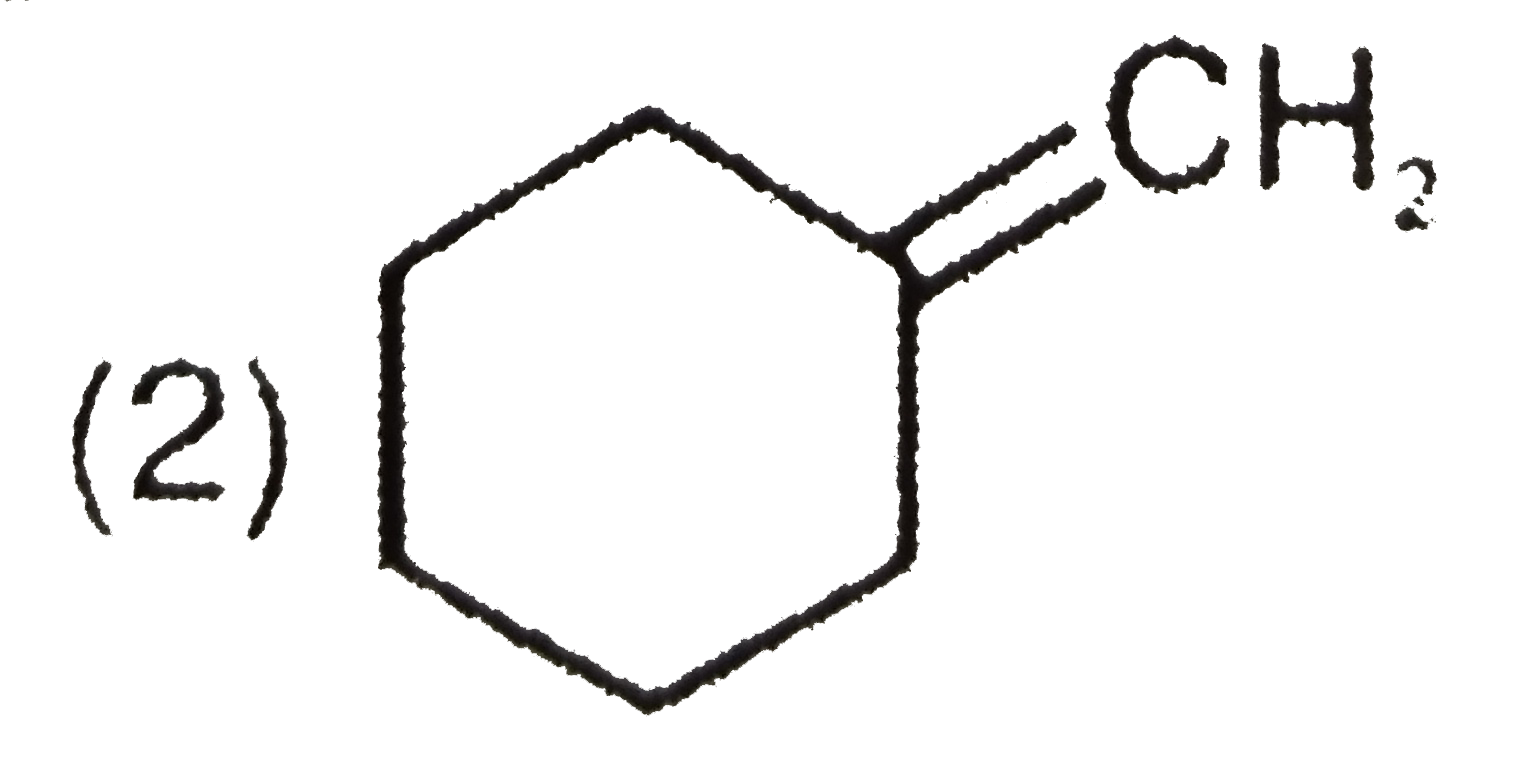
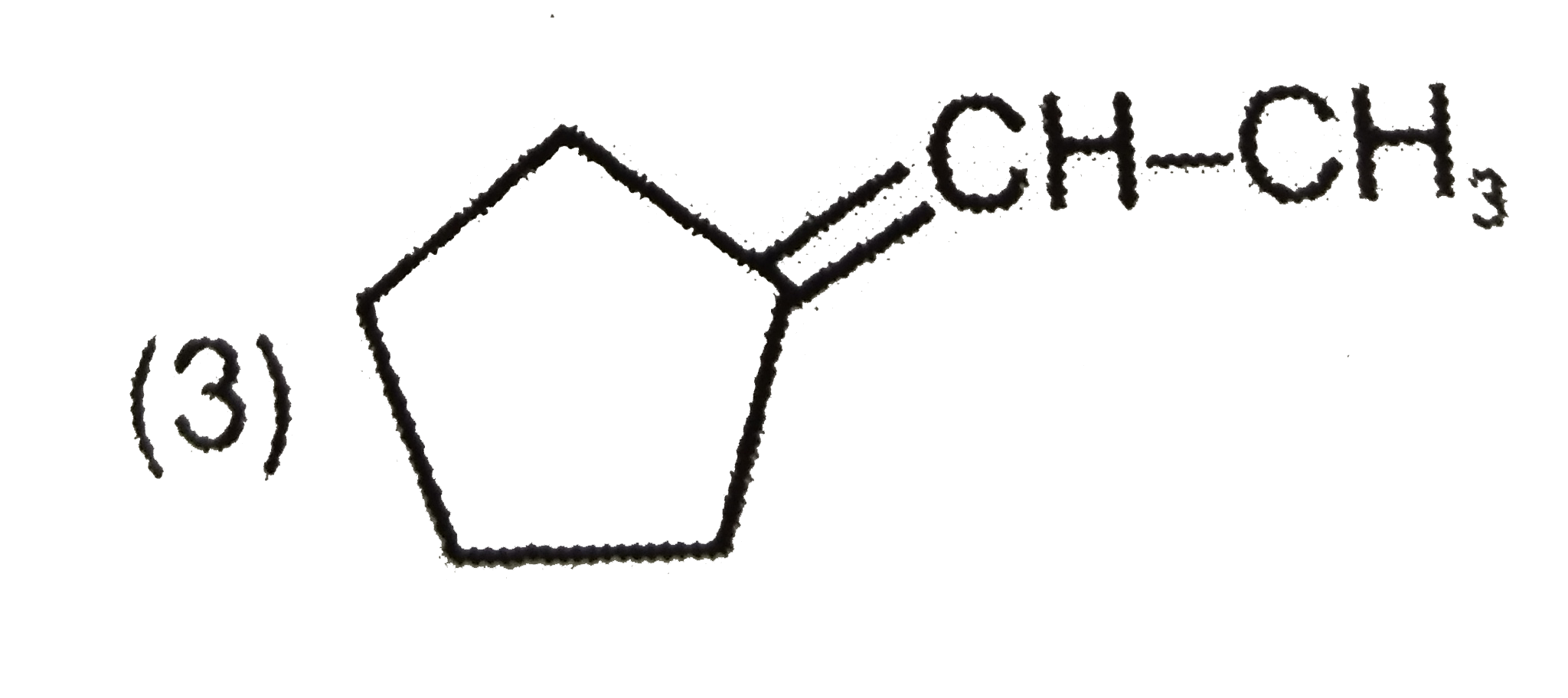
(x) C_(7)H_(12) overset(O_(3))underset(Me_(2)S)toP+Q Compound P responds to Tollen's test and iodoform test but Q does not respond with both the reagents. Structure of compound (x) is : |
|
Answer»
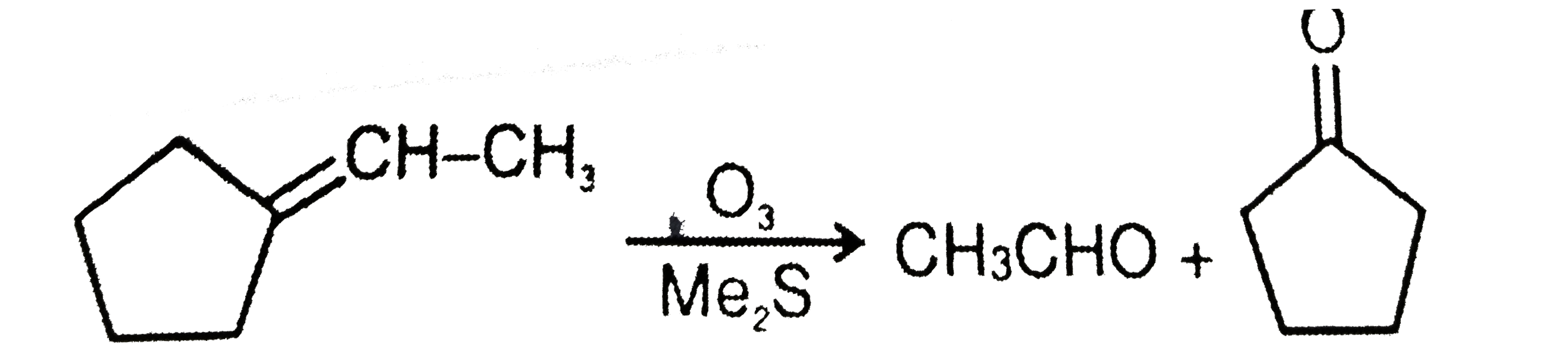
|
|
| 37. |
What is the wavelength (in m) of a particle of mass 6.62 xx 10^(-29)g moving with a velocity of 10^(3)m.s^(-1) ? (h = 6.62 xx 10^(-34) J.s) |
|
Answer» `6.62 xx 10^(-4)` |
|
| 38. |
Which of the following reactions will give alkyne? |
|
Answer»
|
|
| 39. |
The velocity of a gas molecule present in a cubic vessel in a particular direction is 100 cm/sec. The side of that cubic vessel is 10 cm. The time taken for one collision in a given particular direction on one face is |
| Answer» ANSWER :A | |
| 40. |
Which one of these gases has the highest critical temperature ? . |
|
Answer» `W` |
|
| 41. |
Which of the following disproportionates when treated with water ? |
|
Answer» `SO_(3)` |
|
| 42. |
The volume of a gas at 0°C and 700 mm pressure is 760 cc. The number of molecules present in this volume is: |
|
Answer» `1.88 XX 10^(22)` |
|
| 43. |
Which of the following statements is not correct regarding purification of liquids by steam distillation? |
|
Answer» impurities MUST be non-volatile |
|
| 44. |
Water is a .... |
|
Answer» BASIC OXIDE |
|
| 45. |
Which is the IUPAC name of methyl propyl ether? |
|
Answer» Methoxypropane |
|
| 46. |
Which of the following statement is wrong . |
|
Answer» Through SODIUM and potassium are similar chemically , they differ in their behaviour in biological system |
|
| 47. |
In the diagram shown in figure, all pulleys are smooth and massless and strings are light. Match the following (##AKS_TRG_AO_PHY_XI_V01_A_C05_E01_045_Q01.png" width="80%"> |
|
Answer» `K_2 =(K_1+K_3) //2` acid STRENGTH ` ALPHA (1)/( pH)` |
|