Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Usingthe periodictable, predictthe formulaof compoundformedbetweenelementxgroup13and anotherelementY ofgroup16. |
|
Answer» SOLUTION :The valency of X (group 13) = 3 The valency of Y (group 16) = 2 The COMPUND has 2 atoms of X and 3 of Y. Hence, the formula `= X_2Y_3` |
|
| 2. |
The number of sigma and pi (TT) bonds present in benzene respectively are |
| Answer» Solution :`SIGMA = 12 , PI =3 ` | |
| 3. |
When two substances A and B are powdered together in a pestle and mortar, a larger amount of heat is given out and a substance C is formed. The properties of C are found to be different from those of A and B. It is substance C an element, compound or mixture ? |
|
Answer» |
|
| 4. |
Water is bounded in liquid state because of .. ......... bond of water in liquid state. |
|
Answer» van-der-Walls ATTRACTION force |
|
| 5. |
What do you understand by the terms acidity and basicity? |
|
Answer» Solution :ACIDITY: The number of hydroxyl IONS present in one mole of a BASE is known as the acidity of the base. BASICITY: The number of replaceable hydrogen ATOMS present in a molecule of the acid is referred to as its basicity. |
|
| 6. |
What is chemical bonding ? How it is formed and give its types.OR Explain the formation of a chemical bond. |
|
Answer» Solution :The ATTRACTIVE force which holds various consti- tuents (atoms, ions, etc.) together in different chemical species is called a chemical bond. Every system tends to be more stable and bonding is nature.s way of lowering the ENERGY of the system to attain stability. The types of chemical bonding are as under. (l) Kossel - Lewis approach (2) The valence shell ELECTRON pair repulsion (VSEPR) theory (3) Valence bond theory (VB) (4) Molecular orbital theory (MO) |
|
| 7. |
The table given below lists the bound dissociation enegrgy (E_(diss)) for single covelent bonds formed between C and atom A,B,D,E. What of the atoms has smallest size? |
| Answer» Solution :The smallest the size of the ATOM, more strongly it is bonded. Because SMALL and comparable size of atom have strong force of attraction between then and are strongly bonded to each other. Therefore, a very high enegry is required to break or dissociate that bond. | |
| 8. |
Writer four characteristic properties of p-block elements. |
|
Answer» a) p-Block elements include both metals and non-metals but the number of non-metals is much higher than that of metals. Further, the metabolic character increases from TOP to bottom within a group and non-metallic character inicreases from LEFT to right along a period in this block. C) They mostly form covalent compounds. Some of them show more than one (variable) oxidation states in their compounds. Their oxidising character increases form left to right in a period and reducing character increases from top to bottom in a group. |
|
| 9. |
Which zeolite catalyst is used to convert alcohols directly into gasoline ? |
|
Answer» ZSM - 5 |
|
| 10. |
Which are true statements among the following ? |
|
Answer» `,13` |
|
| 11. |
Which of the following is a polar molecule ? |
|
Answer» `BF_(3)` |
|
| 12. |
Which product is obtained by the reaction of chlorine with excess amount of ammonia ? |
|
Answer» `NH_4Cl` |
|
| 14. |
Which of the following reactions involve oxidation reduction? |
|
Answer» `2Rb +2H_(2) O rarr 2RbOH+H_2` |
|
| 15. |
What is the action of hydrogen halide on alkene? |
|
Answer» Solution :ALKENE reacts with hydrogen HALIDE to form alkyl halide. The addition is governedby MARKOWNIKOFF's rule. ` R - CH = CH_(2) + HK rarr R - UNDERSET(X)(|)(CH) - CH_(3)` |
|
| 16. |
When H_(2)S is passed through nitric acid, the product is |
|
Answer» Rhombic(S) |
|
| 17. |
The temperature of 2 moles ofan ideal gas is raisedfrom 27^(@)Cto 77^(@)C. What is the value forDeltaH - DeltaU for the process ? ( R= 8.3 J K^(-1)mol^(-1)) |
|
Answer» 415J `:. H_(2)-H_(1) = ( U_(2)+nRT_(2)) - ( U_(1)+nRT_(1)) ` `= ( U_(2)-U_(1))+nRT(T_(2)-T_(1))` or `DeltaH = DeltaU + nR DELTAT` or `DeltaH- DeltaU = nRT = 2 XX 8.3 xx 50 = 830 J` |
|
| 18. |
Which of the following acids has the smallest dissociation constant ? |
|
Answer» `CH_(3)CHFCOH` |
|
| 19. |
What is the positive and negative resonance effect ? Explain with examples |
|
Answer» Solution :The resonance effect is defined as the polarity produced in the molecule by the interaction of two `pi`-bonds or between a `pi`-BOND and lone pair of electrons present on an adjacent atom. The effect is transmitted through the chain. These are two types as resonance or mesomeric effect designed of R or M effect. (a) Positive resonance (+R) or mesomeric (+M) effect: (i) Definition: When the transfer of electron is away from an atom or substituent GROUP attached to the conjugated SYSTEM. This electron displacement makes certain positions in the molecule of high electron densities. So this is called positive resonance (+R) effect. (ii) e.g. In aniline `-OH` group containing (+R) and (+M) effect and the non-bonding electron pair of N is transfer in benzene ring and aniline becomes polar. Due to this effect the electron density is increases at some position.  Its structure (II, III) and (IV) are polar and lone pair electron of `overset(..)(N)` at definite position and negative charge and more electron density. (iii) The other examples of +R or +M effect containing groups are as under. `-X, -OH, -OR, -OCOR, -NH_(2), -NHR, -NR_(2)- NHCOR` (b) Negative resonance effect (-R) or negative mesomeric effect (-M): (i) Definition: When the transfer of electron is towards the atom or substituent group attached to the conjugated system then these groups are called (-R) or (-M) effect. (ii) e.g. : In nitrobenzene, `-NO_(2)` group containing (-R) means (-M) effect and the electron pair of `pi`-bond with N of out side of ring. Therefore nitrobenzene molecule become polar and at certain position the electron density DECREASE in ring and become positive  Structure (II), (III), (IV) are polar. The electron pair of ring transfer at out side in O-N bond and (+) charge is at specificposition and electron density is less (iii) (-R) means (-M) effect containing groups are `-COOH, -CHO, gt C= O, -CN, -NO_(2)` |
|
| 20. |
Which of the following compound is amphoteric in nature ? |
|
Answer» `CH_(3)COCl` |
|
| 21. |
Which of the following neutral molecule is not lewis bese? |
|
Answer» `NH_(3)` |
|
| 22. |
What is the difference between the notations l and L? |
| Answer» Solution :L' represents azimuthal quantum number which can have value 0, 1, 2 etc. L represents second BOHR orbit for which n = 2 always | |
| 23. |
What is compressibility factor (Z). |
|
Answer» Solution :In case of real gases, the DEVIATION from ideal BEHAVIOUR can be meausred in terms of compressibility FACTOR (2). Z is mathematically defined as `Z=(PV)/(nRT)` |
|
| 24. |
Which of the elements Na, Mg, Si and P wouldhave thegreaterdifferencebetweenthe firstand thesecondionizationenthalpies . Brieflyexplainyour answer. |
|
Answer» SOLUTION :AmongNa, Mg ,Si and P , Na isanalkalimetal. Ithas onlyone electronin thevalenceshell thereforeits `Delta_(i)H_(2)`is verylow . However afterremoval of theone electron . It acquiresneongas configurationi.e.,`Na^(+)(1S^(2)2s^(2)2p^(6))` . Thereforeits `Delta_(i) H_(2)` is expectedto be veryhigh. CONSEQUENTLY thedifference in firstand secondionizationenthalpieswouldbe greaterin case of Na. Howeverit maybe notedhere THATIN caseof Mg ,Si and Palthough their `Delta_(i) H_(1)`will be muchhigherthanthat of abut their`Delta_(i) H_(2)`will be muchlower than thatof Na . As aresultthe difference intheirrespective`Delta_(i) H_(1)` and `Delta_(i) H_(2)` would bemuch lowerthan thatof Na. |
|
| 25. |
Which of the following compound gives white precipitate with silver nitrate solution? |
|
Answer» `NaCI` |
|
| 26. |
Which of the followingcan be used as the halide component for friedal crafts reaction |
|
Answer» CHLOROBENZENE |
|
| 27. |
The solution of (CH_3COOH + CH_3COONa) will be acidic, basic or neutral ? Give the equation to calculate its pH. |
|
Answer» Solution :This solution is a APPROXIMATELY 4.76 PH CONTAINING acidic buffer. Its pH is calculate by following EQUATION `pH=pK_a "-log" (["Conjugate base," CH_3COO^-])/(["ACID," CH_3COOH])` |
|
| 28. |
Which is correct in case of p-orbitals ? |
|
Answer» They are spherically symmetrical `*` p-orbitals have strong directional character . `*` They are three fold degenerate orbitals. |
|
| 29. |
The standard heat of formation of NO_(2)(g) and N_(2)O_(4)(g) are 8.0 and 2.0kcal mol^(-1) respectively . The heat of dimerization ofNO_(2) in kcal is |
|
Answer» `10.0` `N_(2)+ 2O_(2) rarr N_(2)O_(4), DeltaH=2kcal` ....(II) Aim `: 2NO_(2) rarrN_(2)O_(4) , DeltaH=?` Eqn. (ii) `- 2 XX `Eqn. (i) gives `0= N_92)O_94) - 2NO_(2)`or `2NO_(2) rarr N_(2)O_(4), DeltaH= 2-2(8)= -14.0 kcal` |
|
| 30. |
The substances which contain species with unpaired electrons in their orbitals behave as paramagnetic substances. Such substances are weakly attracted by the magnetic field. The paramagnetism is expressed in terms of magnetic moment. The magnetic moment is related to the number of unpairedelectrons according to the following relation : Magnetic moment, mu = sqrt(n(n+2)) B.M. Where .n. = number of unpaired electrons. B.M. stands for Bohr magneton, a unit of magnetic moment. An ion of a d-block elements has magnetic moment 5.92 B.M. Select the ion among the following. |
|
Answer» `Zn^(+2)` |
|
| 31. |
Write the formation of sigma covalent bond diagrammatically. |
|
Answer» Solution :It is a covalent bond formed by axial overlapping of half filled atomic orbitals ALONG the internuclear AXIS. Axial overlapping is ALSO called end to end or HEAD to head overlapping. Example : 1. S-S overlapping : It involves overlap of two half filled s-atomic orbital. This occurs during the formation of H2 molecule. The bond is s-s s bond. 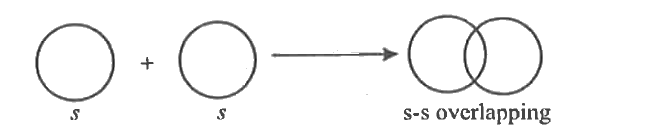 2. s-p overlapping : It involves overlap of half filled s orbital and half filled p-orbitals. This occurs during the formation of hydrogen halide molecule (HCL, HBr, Hl) 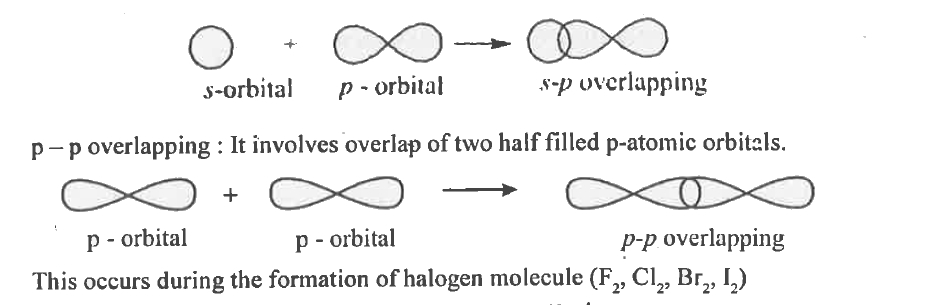
|
|
| 32. |
Which of the following is an analgesic ? |
|
Answer» Streptomycin |
|
| 33. |
What is the value of [H_3O^+] and [OH^-] of pure water at 298 K temp. ? |
| Answer» SOLUTION :`[H_3O^+]=[OH^-]=1.0xx10^(-7)` | |
| 34. |
Write reactions to justify the amphoteric nature of Al. |
|
Answer» `2Al+3H_2SO_4 rarr Al_2(SO_4)_3+3H_2` `2Al+2NaOH+6H_2O rarr 2Na^+(Al(OH)_4)^-+3H_2` |
|
| 35. |
Which of the following molecular orbitals has two nodal planes ? |
|
Answer» `sigma _(2s)` |
|
| 36. |
Which of the following is contaminant responsible for Bhopal gas tragedy |
|
Answer» `CO_(2)` |
|
| 37. |
Which of the statement given below are correct ? (i) Pure dihydrogen 99.5 % is obtained by heating hydrocarbons at 1270 K temperature using Fe catalyst. (ii) In acidic medium H_2O_2decolourises potassium permanganate solution. (iii) Ca^(2+) and Mg^(2+) ions can be precipitated by calgon to soften the hard water (iv) The strength of 15 volume H_2O_2 at STP is 45.54 gram. |
|
Answer» (i),(II) |
|
| 38. |
Which of the following aredisproportionation redox changes? |
|
Answer» `(NH_(4))_(2)Cr_(2)O_(7) rarrN_(2) +Cr_(2)O_(3) + 4H_(2)O` `Cl^(+)rarrCl^(5+)+4E" "Cu^(+)+erarrCu` |
|
| 39. |
Which of the following are not true about hexagonal close packing ? |
|
Answer» It has a coordination number of 6. |
|
| 40. |
What is the pressure exerted by 10^(23) gas molecules, each molecule of mass 10^(-23)g, present in a container of volume one L and RMS speed of 1000 m s^(-1)? |
|
Answer» Solution :KINETIC GAS equation `PV = (1)/(3) mnC^(2)` Pressure of the gas (P) ` = (mnC^(2))/(3V) = (10^(-23) xx 10^(23) xx 10^(10))/(3 xx 1000) = 3.33 xx 10^(8)` dyne `cm^(-2)` |
|
| 41. |
Which of the following is the correct order of increasing enthalpy of vaporisation ? |
|
Answer» `NH_(3) lt PH_(3) lt AsH_(3)` |
|
| 42. |
The repeating structural unit is silicone is |
|
Answer» `SiO_(2)` |
|
| 43. |
The TLV values of four pollutants A, B, C and Dare 9ppm, 10ppm, 100ppm and 500 ppm respectively. The most toxic amount them is |
|
Answer» A |
|
| 44. |
Which of the following are correct- |
|
Answer» Only lyman SERIES is OBSERVED in both emission and absorption spectrum |
|
| 45. |
Write the favourable factors for the formatJon of ionic bond. |
|
Answer» Solution :The easy of formation of the POSITIVE and negative ions from the respective neutral atoms. The arrangement of the positive and negative ions in the solid, that is, the lattice of the crystalline compound. `M_((g)) RARR M_((g))^(+) + e^(-)` .... Ionization enthalpy`(Delta_(i) H)` `X_((g)) + e^(-) rarr X_((g))`.... electron gain enthalpy `(Delta_(eg)H)` Obviously Ionic bonds will be formed more easily between elements with comparatively low ionization enthalpies and elements with comparatively high negative value of electron gain enthalpy. And there will be for are lattice energy (enthalpy) `(Delta_(L)H)` of crystal.`M_((g))^(+) + X_((g))^(-) rarr MX_((s))` The ionization enthalpy `(Delta_(i) H)` is always endothermic. Electron gain enthalpy `(Delta_(eg))` process may be exothermic or endothermic. And lattice enthalpy `(Delta_(L)H)` always endothermic. Ionic compounds in the crystalline state consist of orderly three - dimensional arrangements of cations and anions held together by coulombic interaction energies. These compounds crystallise in different crystal STRUCTURES determined by the size of the ions, their packing arrangements and other factors. In ionic SOLIDS, the sum of the electron gain enthalpy and the ionization enthalpy may be positive but still the crystal structure gets stabilized due to the energy released in the formation of the crystal lattice. |
|
| 46. |
The substances which contain species with unpaired electrons in their orbitals behave as paramagnetic substances. Such substances are weakly attracted by the magnetic field. The paramagnetism is expressed in terms of magneticmoment The magnetic moment is related to the number of unpaired electrons according to the following relation : Magnetic moment, mu = sqrt(n(n+2)) B.M. Where ‘n’ = number of unpaired electrons. B.M stands for Bohr magneton, a unit of magnetic moment. Which of the following has the highest magnetic moment? |
|
Answer» `FE^(+2)` |
|
| 47. |
Using stock notation represetn the following comoounds : HauCI_(2),TI_(2),FeO,F_(2),CuI,CuO,MnO and MnO_(2) |
|
Answer» Solution :By applying the various rules for determining the oxidation number of diffeeent atoms in a compund the oxidation number of each metallic element in the given compound is a s follows In `HaucI_(4) Au` ha +3 in `TI_(2)O` TI has +1 , in `FeO,Fe` has +2 in `Fe_(2)O_(3) Fe` has +3 in CuI Cu has +1 in CuCOCu +2 in MnO MN hs +2 while in `MnO_(2)` Mn has +4 oxidation state THEREFORE these compounds may be repersented as `Hau(III) CI_(4),TI_(2)O,Fe_(2)(III)O_(3),Cu(I)I,,Cu(II)O` and `Mn(iv)O_(2)` |
|
| 48. |
Which of the following has see saw shape? |
|
Answer» `PCl_(5)` |
|
| 49. |
Three mole of glyoxal are obtained by the ozonolysis, followed by hydrolysis (in presence of Zn) of |
| Answer» Solution :Benzene | |
| 50. |
Write the structures of the compounds from the following data. |
Answer» SOLUTION :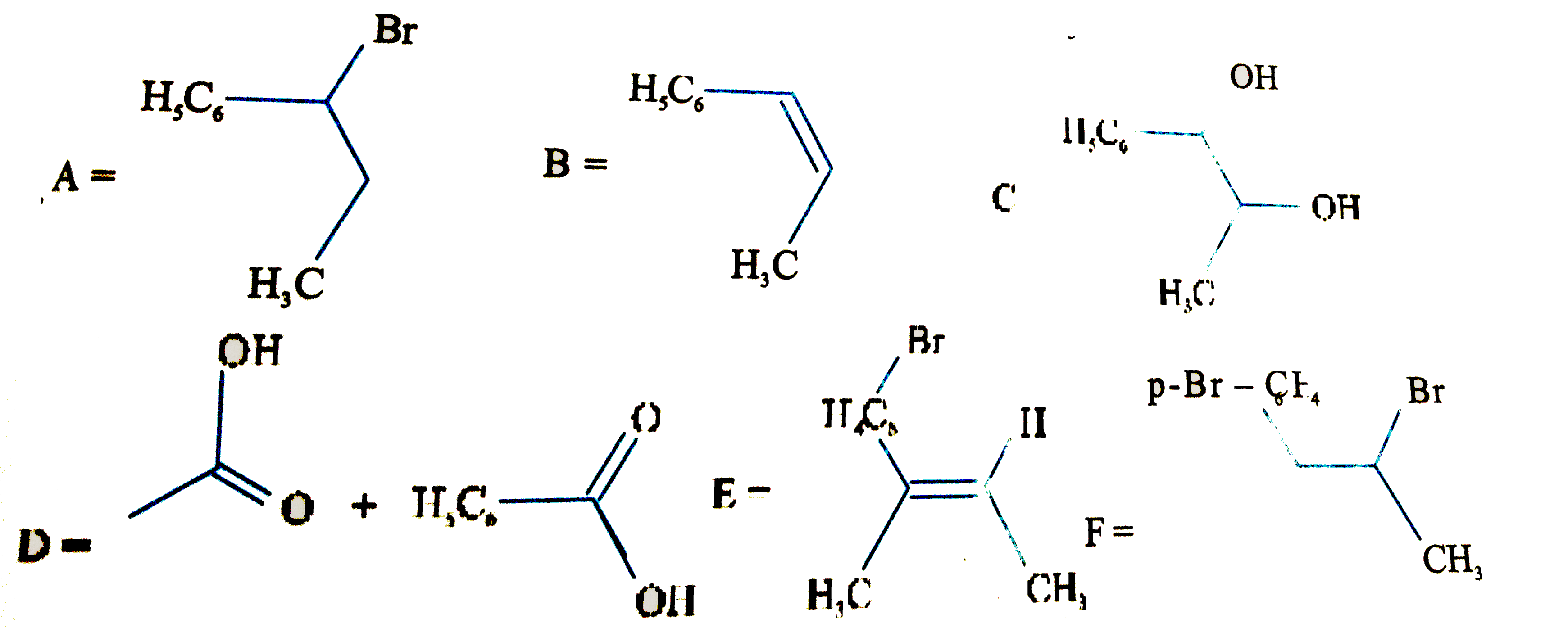
|
|