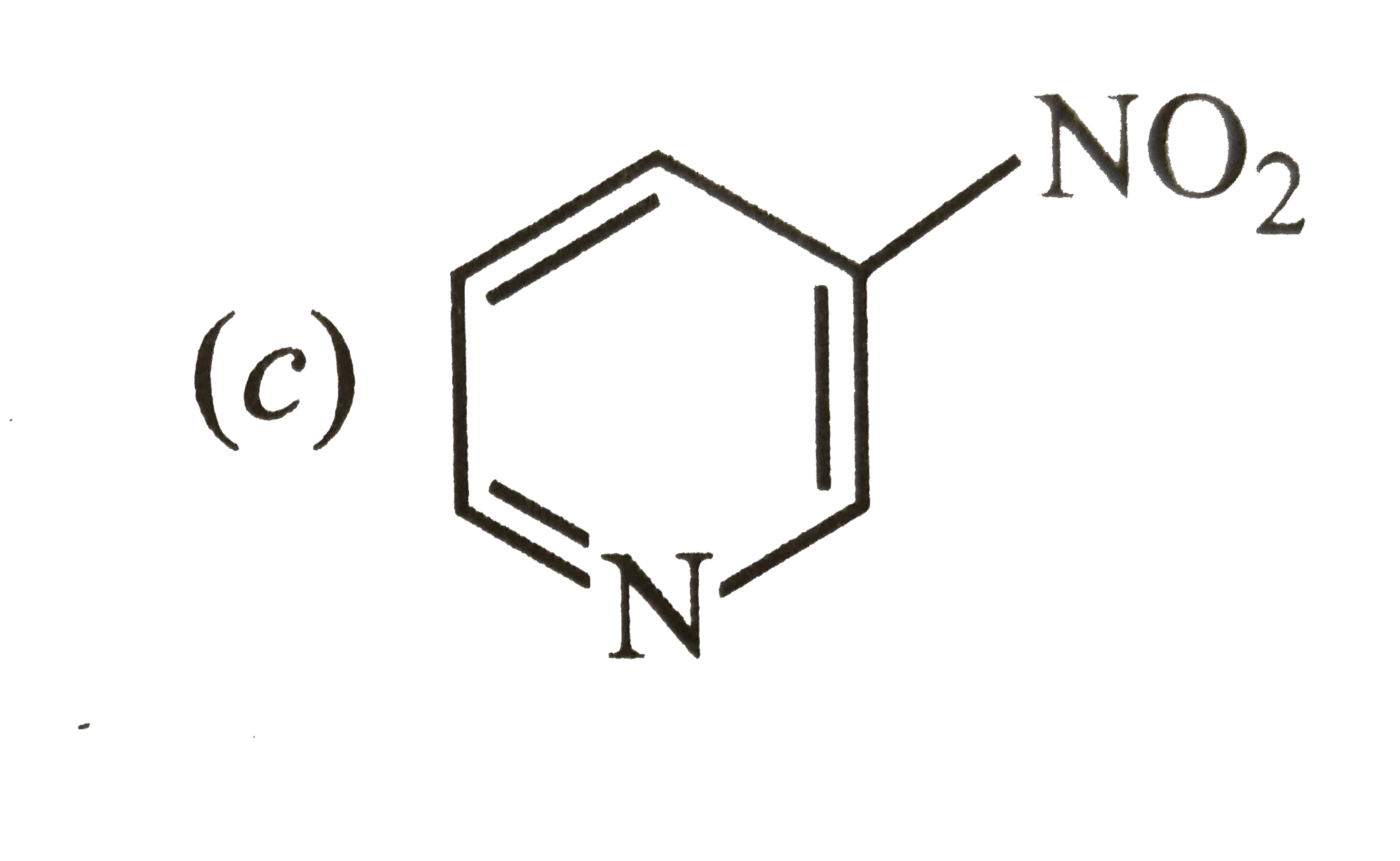Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following generally gets displaced by more electro positive metals in nonmetal displacement reactions. |
|
Answer» `H_(2)` |
|
| 2. |
Which of the following is dimensional formula of intensity of radiation ? |
Answer» SOLUTION :INTENSITYOF radiationisthe HEIGHTOF crestordepthof troughtit isdenoted by a 
|
|
| 3. |
Which of the following acid has the same molecular weight and equivalent weight ? |
|
Answer» `H_(3)PO_(2)` |
|
| 4. |
The temperature at which the volume of a gas is zero is |
| Answer» Answer :B | |
| 5. |
Which compound is obtained on reaction of Diborane with Ammonia at 450 K temperature ? |
|
Answer» `B_3N_2H_6` |
|
| 6. |
Which element of Group 13 is the strongest reducing agent ? |
|
Answer» B |
|
| 7. |
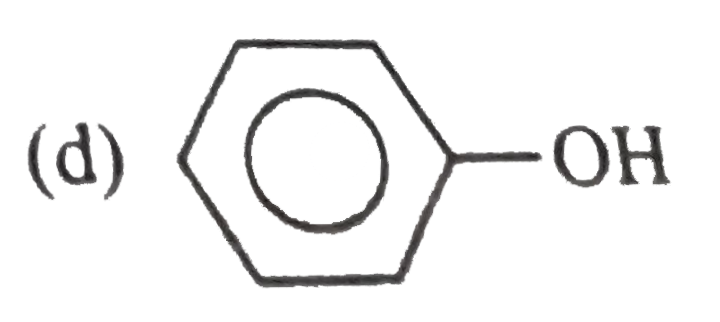
Which of the following pairs are resonance structures of each other ? |
|
Answer»
|
|
| 8. |
What is electrolyte ? Give examples. |
| Answer» SOLUTION :An electrolyte is DEFINED as a compound whose AQUEOUS solution or MELT conducts ELECTRICITY NaCl, `NaNO_3`. | |
| 9. |
Which of the following have no unit? |
|
Answer» Electronegativity |
|
| 10. |
What is the effect of temperatureon viscoisty and why ? |
| Answer» SOLUTION :Viseosity deecreases in the TEMPERATURE because intermolecular FORCE of attraction DECREASES. | |
| 11. |
Two solutions of a substances (non-electrolyte) are mixed in the manner, 480 mL of 1.5 M of first solution with 250 mL of 1.2 M of second solution. |
|
Answer» 1.20 M For solution II,millimoles `=MV=520xx1.2=624` TOTAL millimoles `=720+624=1344` `THEREFORE` Molarity `=(1344)/(480+520)=1.344M` |
|
| 12. |
The salts of which +ve and -ve ions are basic ? |
| Answer» SOLUTION :If the positive ions are `NA^+, K^+ , Ba^(2+)`and negative ions are other than `Cl^(-) , NO_3^(-) , SO_4^(2-)`than the solution is basic. | |
| 13. |
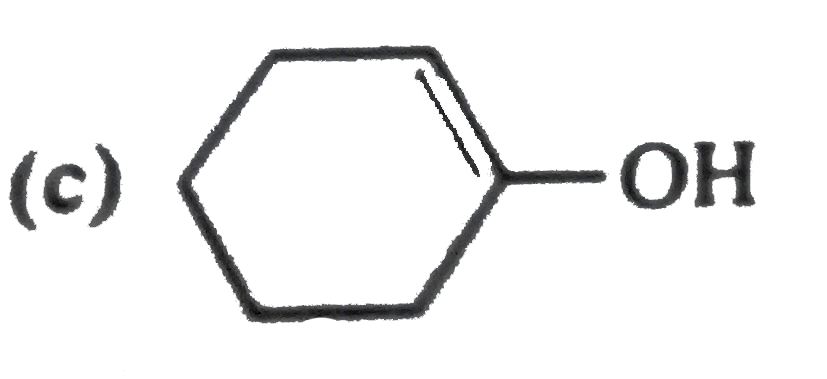
Whichof the followingarrowis usedbetweentwostructurestoindicatethattheseareresonanceforms? |
|
Answer»
|
|
| 14. |
What are the uses of chlorobenzene? |
|
Answer» SOLUTION :(i) Chiorobenzene is USED in the manufacture of pcsticides like DDT. (ii) It is used as high boiling solvent in ORGANIC synthesis. (III)It is used as a fibre swelling agent in TEXTILE processing. |
|
| 15. |
Various successive ionization enthalpies (in kJ mol^(-1)) of an element are given below: The element is |
|
Answer» phosphorus |
|
| 16. |
Which of the following molecules or ions is/are linear? |
|
Answer» `BeCI_(2)` |
|
| 17. |
The temperature of a gas is increased by 1 aC. Then from the following statements pickout the correct one 1) The volume increases by 1/273 of its volume at 0°C at constant pressure 2) The pressure increases by 1/273 of its pressure at 0°C at constant volume 3) The volume decreases by 1/273 of its volume at 0°C 4) The pressure is doubled to its pressure at 0°C |
|
Answer» a,C |
|
| 18. |
what is the nature of the reaction for the following ? DeltaGlt0 |
| Answer» SOLUTION :`DeltaGlt0` : The PROCESS in SPONTANEOUS and FEASIBLE | |
| 19. |
What ar solid fuels ? |
| Answer» SOLUTION :IONIC metal hydrides such as LiH, `LiAlH_(4), NaBH_(4)`) are called solid FUELS because on heating they produce DIHYDROGEN which spontaneously catches fire. | |
| 20. |
What is an intensive property? Give an example. |
| Answer» Solution :A property which is INDEPENDENT on the QUANTITY of matter present in the SYSTEM. Ex.: DENSITY, TEMPERATURE. | |
| 21. |
Write the entropy statement of second law of thermodynamics. |
|
Answer» Solution :WHENEVER a spontaneous process TAKES place, it is accompanied by an increase in the te entropy of the Universe, `DeltaS_"universe" gt DeltaS_"system" +DeltaS_"SURROUNDINGS"` |
|
| 22. |
Which of the following statement is right |
|
Answer» R and S configurational ISOMERS of a COMPOUND are enantiomers B) D and L configurational isomers of a compound are enantiomers c) cis-2-butene and trans-2-butene are a pair of diastereoisomers |
|
| 23. |
What are stoichiometric defects or intrinsic defects in ionic crystals ? |
| Answer» Solution :Stoichiometric defects are those defects in which the ratio of cations to anions REMAINS the same as respesented by the MOLECULAR FORMULA. | |
| 24. |
What is the composition of water gas ? |
| Answer» Solution :Water GAS [ CO + `H_(2)` ] is a mixture of CARBON monoxide and HYDROGEN. | |
| 25. |
What is the pH at the equivalenve point for the titration of 0.20 M aniline (K_(b) = 7.95 xx 10^(-10)) with 0.20 M HCL |
|
Answer» `2.8` `:. pH = 7 - (1)/(2) pK_(b) - 1/2 "LOG"C` |
|
| 26. |
What is meant by open system? Give example. |
|
Answer» SOLUTION :(i) A system which can exchange both matter and energy with its SURROUNDINGS is called an open system. (ii) Hot water CONTAINED in an open beaker is an example for open system (III) In this system, both water vapour and heat is TRANSFERRED to the surroundings through the imaginary boundary (iv) All living things are open systems because they continuously exchange matter and energy with the surroundings. |
|
| 27. |
What is stereoisomerism? Which are its types? |
|
Answer» Solution :The compounds that have the same constitution and sequence of covalent BONDS but differ in relative positions of their atoms or groups in SPACE are called stereoisomers. This special Type of isomerism is called as stereoisomerism and can be classified as GEOMETRICAL and optical isomerism. Stereoisomerism can be classified in two TYPES (i) geometrical (ii) optical isomerism |
|
| 28. |
Which of the following compounds decolourise Br_(2) - H_(2)O and also give positive test with neutral FeCl_(3) : |
|
Answer»
|
|
| 30. |
What are the characteristics of enthalpy? |
|
Answer» Solution :CHARACTERISTICS of enthalpy: (i)Enthalpy is a thermodynamic PROPERTY of a system. Enthalpy H is defined as sum of the internal energy (U) of a system and the product of pressure and volume of the system. That is, H=U + PV (ii)Enthalpy is a state FUNCTION which depends entirely on the state functions T,P and U . (iii)Enthalpy is usually expressed as the change in enthalpy (`DeltaF`) for a process between initial and final states. (iv)At constant pressure, the heat flow (q) for the process is equal to the change in enthalpy which is defined by the equation. `DeltaH=q_P` (v) In an endothermic reaction heat is absorbed by the system from the surroundings that is q > 0 (positive). Therefore, at constant T andP, by the equation above, if q is positive then `DeltaH` is also positive. (vi) In an exothermic reaction heat is EVOLVED by the system to the surroundings that is, q<0 (negative). If q is negative , then `DeltaH` will also be negative (vii)Unit of enthalpy is KJ `mol^(-1)` |
|
| 31. |
The value of ionic product of water increases with increase in |
|
Answer» Acidic nature of SOLUTION |
|
| 33. |
Which of the following is a secondary pollutant ? |
|
Answer» `CO_2` |
|
| 34. |
Which one of the following has inductive, mesomeric and hyperconjugation effect? |
|
Answer» `CH_3CI` |
|
| 35. |
What characteristics do you expect from an electron deficient hydride with respect an electron deficient hydride with respect to its structure and chemical reactions ? |
|
Answer» Solution :Dihydrogen forms molecular compounds with most of the p-block elements. Most familiar examples are `CH_4, NH_3, H_2O` and HF. For convenience hydrogen compounds of non metals have ALSO been considered as hydrides. Being covalent, they are volatile compounds. Molecular hydrides are further CLASSIFIED according to the relative numbers of electrons and bonds in their Lewis structure into : (i) electron-deficient, (ii) electron-precise, (iii) electron - rich hydrides. An electron-deficient hydride, as the name suggests, has too few electrons for writing its conventional Lewis structure. Example : Diborane `(B_2H_6)` In fact all elements of group 13 will FORM electron-deficient compounds. They act as Lewis ACIDS i.e., electron acceptors. Electron-precise compounds have the required number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds (e.g., `CH_4`) which are tetrahedral in geometry. Electron-rich hydrides have excess electrons which are present as lone pairs. Example : Elements of group 15-17 form such compounds. (`NH_3` has 1-lone pair, `H_2O` -2 and HF-3 lone pairs). They will behave as Lewis bases i.e., electron donors. The presence of lone pairs on highly electronegative atoms LIKE N, O and F in hydrides results in hydrogen bond formation between the molecules. This leads to the association of molecules. |
|
| 36. |
What are the anomalous properties of second period elements? |
|
Answer» Solution :(i) In the `1^(st)` GROUP, lithium differs in many aspects from its own family elements. Similarly, in the `2^(nd)` group, beryllium differs in many aspects from its own family. (ii) For EXAMPLE, lithium forms COMPOUNDS with more covalent character. But other alkali metals of this group form only ionic compounds. (iii) Similarly, beryllium forms compounds with more covalent character while the other elements of this family form only ionic compounds. (iv) Lithium and beryllium RESEMBLE more with the elements lying at their right hand side in the `3^(rd)` period than with the other members of their own family. (v) These kinds of anomalies are also observed from `13^(th)` to `17^(th)` groups. (vi) This sort of similarity is commonly referred to as diagonal relationship in the PERIODIC properties. |
|
| 37. |
Which elements are given at the bottom of the periodic table ? |
| Answer» Solution :Elements of 4f and 5f SERIES are given at the BOTTOM of the PERIODIC table. | |
| 38. |
Three students namely A,B,C have done an experiment two time invdividual, for which the correct value is 2.00g. The result are Accurate and precise result is |
|
Answer» C |
|
| 39. |
Which one of the following hvbridisation takes place in the formation of BeCl_(2)? |
| Answer» SOLUTION :sp | |
| 40. |
What are state and path functions? Give two examples. |
|
Answer» Solution :(i) State FUNCTION : A state function is a THERMODYNAMIC property of a system, which has a specific VALUE for a given state and does not depend on the path (or mannery) by which the particular state is reached. Example : Pressure (P), Volume (V), Temperature (T) (ii) Path functions : A path function is a thermodynamic property of the system whose value depends on the path by which the system changes from its initial to final states. Example : Work (w), Heat (Q). |
|
| 41. |
When treated with Pb(CH_(3)COO)_(4) glycol forms |
|
Answer» only acetone |
|
| 42. |
{:('X'("salt solution")+,AgNO_(3)rarr'Y'(ppt.)overset(Na_(3)AsO_(3))rarr'Z'(yellow ppt.)),(,""darr),(,"Soluble in dil"NH_(3)(aq)and Na_(2)S_(2)O_(3)):} 'X' salt solution may be of: |
| Answer» Answer :A | |
| 43. |
Which of the following reactions is/are not feasible : |
|
Answer» `CH_(3)COONa + NCOOH to CH_(3)COOH + NCOONa` |
|
| 44. |
What is the major product of the following reaction ?C_6H_6 + (CH_3)_2 C = CH_2 overset(H_3PO_4)to ? |
|
Answer»

|
|
| 45. |
Which of the following possesses highest resonance energy |
|
Answer»
 has the highest resonance ENERGY, due to delocalization 6 electrons. Further it has AROMATICITY as it SATISFIES 4N +2 rule. has the highest resonance ENERGY, due to delocalization 6 electrons. Further it has AROMATICITY as it SATISFIES 4N +2 rule.
|
|
| 46. |
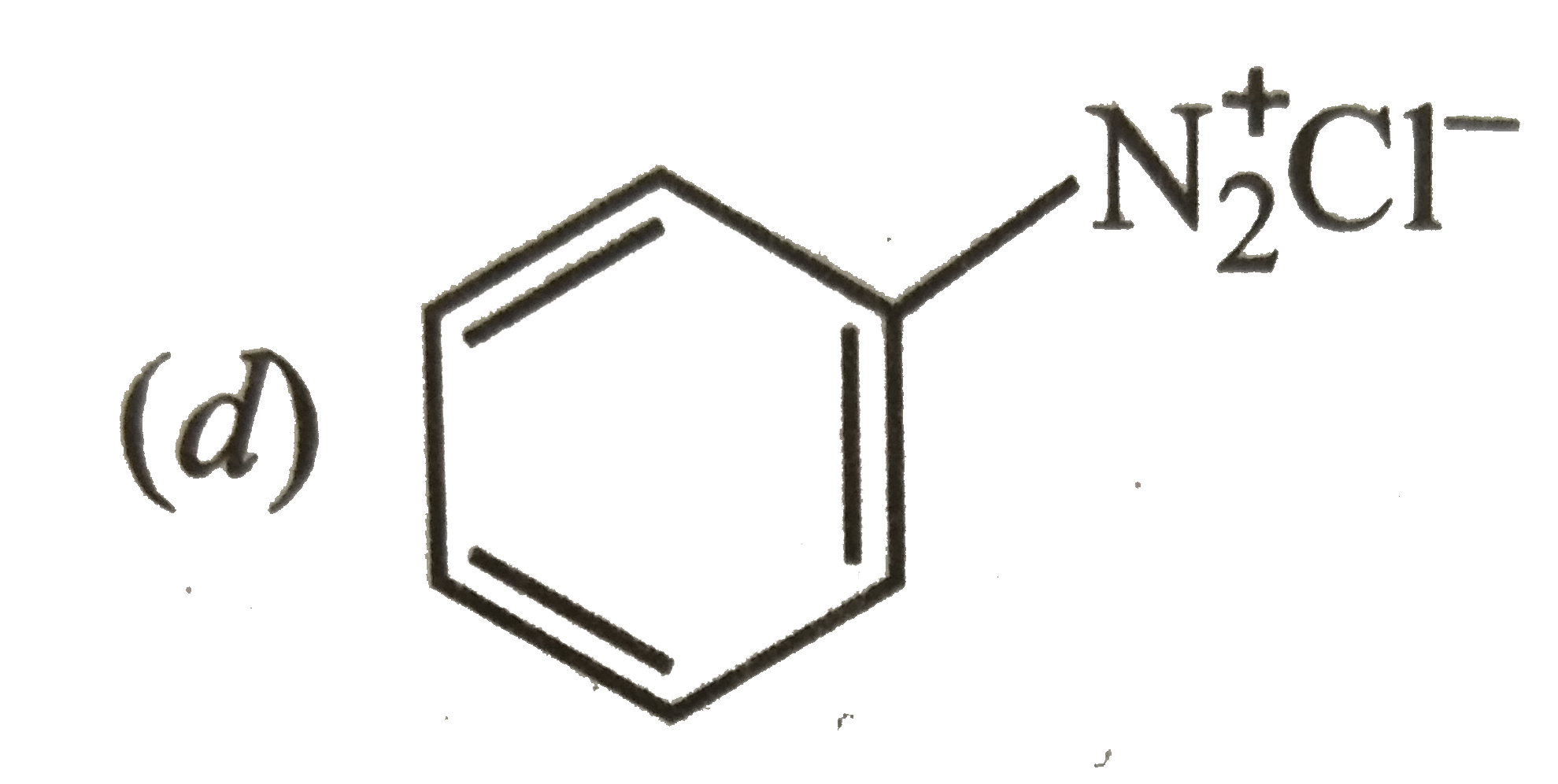
Which of the two: O_(2)NCH_(2)CH_(2)O^(-) or CH_(3)CH_(2)O^(-) is expected to be more stable and why ? |
Answer» Solution : is more stable than is more stable than 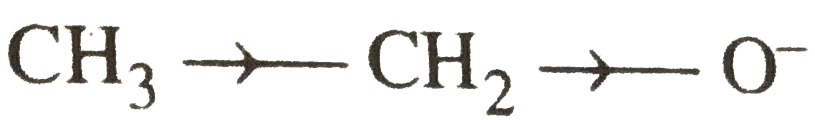 because `NO_(2)` groups has -I-effect and HENCE it tends to disperse the -ve charge on the O-atom. In CONTRAST, `CH_(3)CH_(2)` has +I-effect. It, THEREFORE, tends to INTENSIFY the -ve charge and hence DESTABILIZES it. because `NO_(2)` groups has -I-effect and HENCE it tends to disperse the -ve charge on the O-atom. In CONTRAST, `CH_(3)CH_(2)` has +I-effect. It, THEREFORE, tends to INTENSIFY the -ve charge and hence DESTABILIZES it.
|
|
| 47. |
Which of the following chlorides is covalent ? |
|
Answer» `NACL` |
|
| 48. |
What characteristics do you expect from an electron cleficient hydride with respect to its structure and chemical reactions? |
| Answer» Solution :Due to lack of SUFFICIENT ELECTRONS, they do not form NORMAL covalent bonds but exist in POLYMERIC state (e.g.,`B_2H_6)`. They are Lewis acied capable of REACTING with Lewis bases. | |
| 49. |
Which of the following halides is least stable and has doubtful existence ? |
|
Answer» `Cl_(4)` |
|
| 50. |
Which of the followiing compounds will be suitable for Kjeldahl's method for nitrogen estimation ? |
|
Answer»
|
|