Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following pairs of ions are isoelectronic and isostructural ? |
|
Answer» `CO_(3)^(2-), SO_(3)^(2-)` `"" ClO_(3)^(-) "" SO_(3)^(2-)` Number of `e^(-)` :4242 Hybridization : `" " sp^(3) "" sp^(3)` both are having ONE lone pair on central ATOM hence they are pyramidal. |
|
| 2. |
Write ideal gas equation and explain its terms. |
|
Answer» SOLUTION :`PV=nRT` P-Pressure, n-No. of MOLES, V-volume, R-Gas CONSTANT, T-Temperature. |
|
| 3. |
Which of the following ion has the highest value of ionic radius? |
|
Answer» `Li^(+)`  `O^(2-) " and " F^(-)` have larger size than `Li^(+)" and " B^(+)` `O^(2-)" and " F^(-) ` are isoelectronic, but `O^(2-)` has smaller nuclear CHARGE (8) than `F^(-)` (g) ,As an effect of less EFFECTIVE nuclear charge `O^(2-)` has higher ionic radius. |
|
| 4. |
Which of the following metals is required as cofactor byh all enzymes utilisation ATP in phosphate transfer? |
|
Answer» K |
|
| 5. |
Which is more stable PCl_(5)orPCl_(3) ? Or What happens when PCl_(5) is heated ? |
|
Answer» Solution :`PCl_(3)` is more STABLE than `PCl_(5)` because on heating `PCl_(5)` decomposes to form `PCl_(3)andCl_(2)` `PCl_(5)overset(Delta)toPCl_(3)+Cl_(2)` For reason, REFER to FIG. 11.35, page 11//134. Since two axial bonds being weaker than three equatorial bonds, therefore, `PCl_(5)` decomposes on heatin to form `PCl_(3)andCl_(2)`. |
|
| 6. |
Using MO theory predict whcihof the following species has the shortest bond length? |
|
Answer» `O_(2)^(2-)` |
|
| 7. |
Which of the following is not an isomer of 3-methylbut-1-yne ? |
|
Answer» Pent-1-yne |
|
| 9. |
Which of the following compounds will exhibit cis-trans (geometrical) isomerism? |
|
Answer» But-2-ene |
|
| 10. |
When an excess and a very dilute aqueous solution of KI is added to a very dilute aqueous solutions of silver nitrate, the colloidal particles of silver iodide which are associated with the Helmholtz double layer:- |
|
Answer» `AgIvdotsAg^(+)vdotsI^(-)` |
|
| 11. |
Which of the following is not obtained when propyl chloride and methyl chloride react with sodium in dry ether? |
| Answer» Answer :C | |
| 12. |
Which one is more reactive between alkane and alkene ? Why? |
| Answer» SOLUTION :Alkene is more REACTIVE than the alkane, because in alkene weak `pi`-bond is present between two carbon atoms , which attract the electrophilic reactant towards themselves by their ELECTRON sources. This `pi`-bonds easily is broken down in presence of electrophilic GROUP and FORM two new single bond. | |
| 13. |
Xenon crystabllises in the face- centred cubic lattic and the edge of the unit cell is 620 pm. What is the nearest neighbour distance and what is the radius of xenon atom ? |
|
Answer» Solution : Herea = 620 PM, d = ?, r = ? For the face - CENTRED cubic ` = d = a/sqrt2 = 620/1.414 = 438 .5 ` pm ,` r = d/2 = (438.5)/2 = 219. 25 ` pm |
|
| 14. |
Which block elements form the interstitial hydrides compound ? |
| Answer» SOLUTION :d-block and f-block ELEMENTS FORMED interstitial hydride COMPOUND. | |
| 15. |
What do you understand by (i) Electron-deficient (ii) Electron-precise (iii) Electron-rich compounds of hydrogen? Provide justification with suitable examples. |
|
Answer» Solution :(1) Electron deficient hydrides: Compounds in which central ATOM has incomplete octet, are called electron deficient hydrides. For example, `BeH_(2)`, `BH_(3)`, are electron deficient hydrides. (ii) Electron precise hydrides: Those compounds in which exact NUMBER of electrons are present in central atom or the central atom contains complete octet are called precise hydrides e.g., `CH_(4)`, `SiH_(4)`, `GeH_(4)`, etc. are precise hydrides. (iii) Electron rich hydrides: Those compounds in which central atom has one or more lone pair of excess electrons are called electron rich hydrides. e.g., `NH_(3)`, `H_(2)O`. |
|
| 16. |
The treatment of alkyl chlorides with aqueous KOH solution leads to the formation of alcohols but in the presence of alcoholic KOH solution, alkenes are the major product. Explain. |
| Answer» Solution :In aqueous solution, KOH is almost completely ionised to give `OH^(-)` IONS which being a strong nucleophile brings about a substitution reaction of alkyl halides to form alcohols. In aqueous solution, `OH^(-)` ions are highly hydrated. This solvation reduces the basic character of `OH^(-)` ions which therefore, abstract fails to abstract a HYDROGEN atom from the `beta`-carbon of the alkyl chloride to form an alkene. In CONTRAST, an ALCOHOLIC solution of KOH contains alkoxide (`RO^(-)`) ions which being a much stronger base than `OH^(-)` ions preferentially eliminates a molecule of HCI from an alkyl chloride to form ALKENES. | |
| 17. |
Which compound has cis and trans Isomers? |
|
Answer» `CH_(3)-CHCl-CHO` |
|
| 18. |
Which group-2 elements are found abundantly? |
|
Answer» CA and Mg |
|
| 19. |
Which of the following is true for a spontaneous process? |
|
Answer» `DELTA G gt 0` |
|
| 20. |
When bromoethane is subjected to Wurtz reaction, the hydrocarbon inixture so obtained consists of |
|
Answer» butane only <BR>butane and ethane `2C_2H_5Br + 2Na underset(Delta)overset("Dry ETHER ")to underset("Butane")(C_2H_5-C_2H_5 ) + 2NaBr ` `C_2H_5 Br + NA to C_2H_5^(-) Na^(+)` `C_2H_5 Na^(+) + H-CH_2 - CH_2 - Br to underset("Ethane")(C_2H_6) + CH_2 = underset("Ethene")(CH_2+ NaBr)` |
|
| 21. |
which out of K, Mg, Ca and AI from amphoteric oxide? |
| Answer» SOLUTION :AI FORMS amphoteirc oixde, i.e., ACIDIC as well as basic in nature. | |
| 22. |
x gm of water is mixed with 69 gm of ethanol . The mole fraction of ethanol in the resulting solution is 0.6 . What is the value of x in gm |
|
Answer» 54 |
|
| 23. |
Which one is correct Lewis structure of N_(2)? |
|
Answer»
|
|
| 24. |
What is the concertration of copper sulphate (in mol L^(-1)) if 80 of it is dissolved in enough water to make a final volume of 3 L? |
|
Answer» 0.0167 MOLES of `CuSO_4=(80)/(159.5)=0.50` Volume of solution = 3L MOLARITY `=("Moles of SOLUTE")/("Volume of solution in L")=(0.50)/(3)` `=0.167mol L^(-1)` |
|
| 25. |
Which one of the following dissolves more rapidlyin bolld haemoglobin than oxygen? |
|
Answer» OZONE Nitrous OXIDE |
|
| 26. |
The shape of orbital for which l = 1 is |
|
Answer» Spherical |
|
| 27. |
Van der waals constant, b of a gas is 4.42 centrilitre*mol^(-1). How near can centres of 2 molecules approach each other - |
|
Answer» 127.2 pm |
|
| 28. |
Which of the following species are stabilised by addition of electron- |
| Answer» Answer :A::B | |
| 29. |
Why are the P-Cl bonds in PC l_(5) not of same length? |
| Answer» Solution :In `PCl_(5)`, two axial P-Cl bonds and three equatorial P-Cl bonds are present. An axial BOND pair is repelled by three equatorial bond PAIRS at `90^(@)` and one axial bond pair at `180^(@)`. Similarly, an equatorial bond pair is repelled by two axial bond pairs at `90^@` and two equatorail bond pairs at `120^(@)`. thus, an axial bond pair is repelled by three electron pairs while an equatorial bond pair is repelled by two electron pairs. thus, axial bond pair is repelled by two electron pairs. thus, axial bond pair SUFFERS greater repulsion & hence slightly longer than equatorial bonds. | |
| 30. |
Write Lewis strcutrue of the following compounds and show formal charge on each atom : HNO_(3), NO_(2), H_(2) SO_(4) |
Answer» Solution :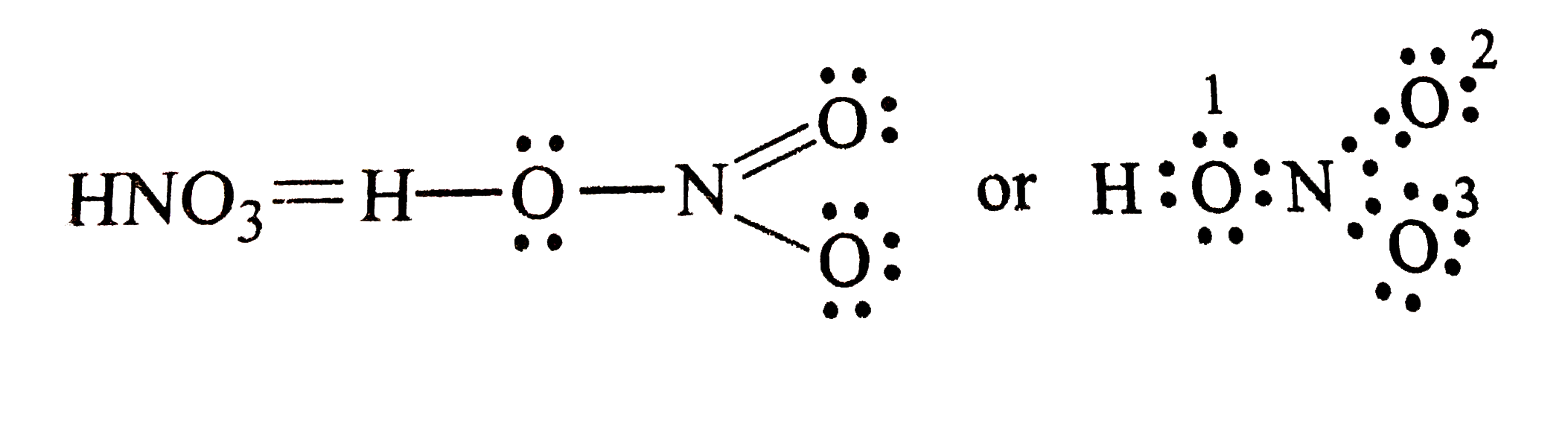 Formal charge = `V - L - (1)/(2) S` Formal chargeon H = ` 1 - 0 - (1)/(2) XX 2 = 0 ` Formal charge on `O (1) = 6 - 4 - (1)/(2)(4 ) = 0 ` formal chargeon (1) `N = 5 - 0 - (1)/(2) (8) = 1` Formal charge on ` O(2) = 6 - 4 - (1)/(2) (4) = 0 ` Formal chargeon `O (3) = 6 - 6 - (1)/(2) (2) = - ` Hence , we write the structure of `HNO_(3)` with write structrue of `HNO_(3)` formal CHARGES as 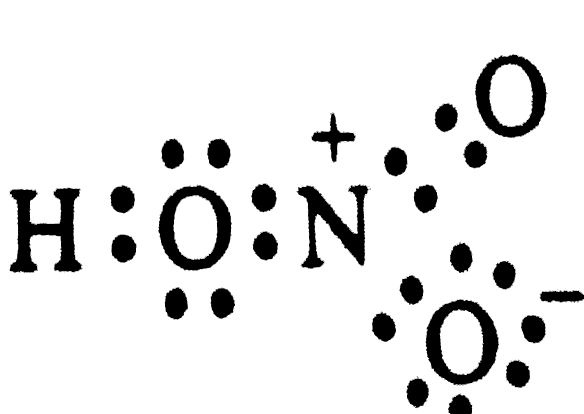 Similary, we have 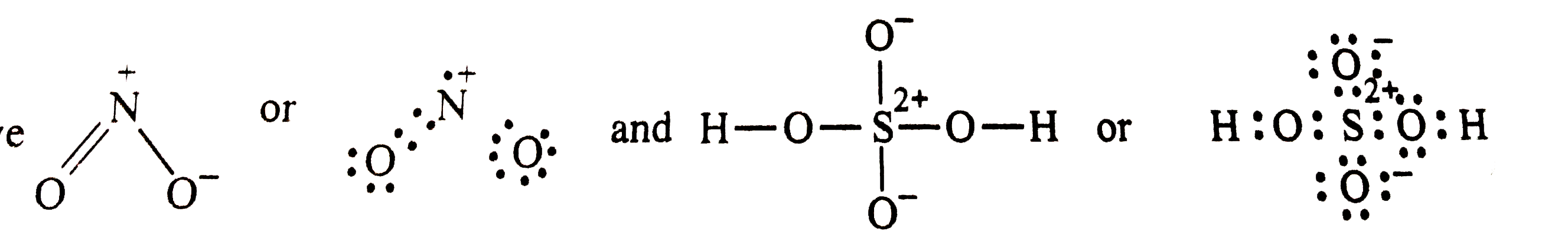
|
|
| 31. |
Whodiscovered e//mfor electron? Andhow ? |
|
Answer» SOLUTION :J.J. Thomsonmeasuredthe RATIOOF electricalcharge (e ) tothemass ofelectron`(m_(e ) )` by usingcathoderay tubeand APPLYING electricaland magneticfieldperpendicularto eachotheraswellas tothe pathof elements R.a. Milikan (1868 - 1953)deviseda methodknownas oildropexperiment (1906 -1914)todeteminethe chargeon theelectron. |
|
| 32. |
Which of the following group possess m-directional effect ? -OH, -O CH_(3), -Cl, -NO_(2), -COOH, -NHCOCH_(3), -SO_(3)H, -Br, -COCH_(3) |
| Answer» Solution :m-directional groups : - `NO_(2), -COOH, -SO_(3)H, COCH_(3)` (rest groups SHOWS ortho and PARA directional EFFECT). | |
| 33. |
Two samples of gases 'a' and 'b' are at the same temperature. The molecules of 'a' are travelling 4 times faster than molecules of 'b'. The ratio of M_(a)//M_(b)will be |
|
Answer» `1//4` |
|
| 34. |
When two ice-cubes are pressed each other, they unite to form one cube. Which of the following forces is responsible to hold them together ? |
|
Answer» DIPOLE forces |
|
| 35. |
Difine critical temperature (Tc) |
| Answer» SOLUTION :It is the TEMPERATURE above which the gas cannot be liquified by application of EXTERNAL pressure. | |
| 36. |
Which one of the following is most reactive towards electrophilic attack :- |
|
Answer»
|
|
| 37. |
Write the IUPAC names of the compound: CH_2 = CH - CH -= C - CH_3 |
Answer» SOLUTION :
|
|
| 38. |
Which of the following contains three pairs of electrons? |
|
Answer» Carbanion |
|
| 40. |
The temperature at which the average speed of oxygen molecules is double that of the same molecules at 0^@C is |
| Answer» Answer :B | |
| 41. |
When propyne reacts with aqueous H_(2)SO_(4) in the presence of HgSO_(4), the major product is |
|
Answer» Propanal `CH_(3)-underset(OH)underset(|)(C)=CH_(2) overset("Rearrangement")rarr underset("Acetone")(CH_(3)-underset(O)underset(||)(C)-CH_(3))` |
|
| 42. |
Which of the following species acts as a Lewis acid and also as a Lewis base ? |
|
Answer» `SO_2` |
|
| 43. |
Which of the following factors will favour the backward reaction? Cl_(2) (g) + 3F_(2(g)) hArr 2Cl F_(3(g)), Delta H= - ve |
|
Answer» ADDITION of inert gas at constant pressure |
|
| 44. |
Which of the following statement is true when ethanol dissolves in water. |
|
Answer» the mixture is a MAXIMUM boiling point azeotrope `Delta V_(mix) gt 0`. `Delta H_(MAX) gt 0`. |
|
| 45. |
Which one is the most stable carbanion? |
|
Answer» `(CH_(3))_(2)CH^(-)` |
|
| 47. |
The size of Cl^(-)=1.81ÅandCl=0.99Å. Explain |
|
Answer» Solution :1 `Cl-1s^(2)2s^(2)2p^(6)3s^(2)3p^(5),Cl^(-)-1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)` 2. Generally anions are LARGER than their parent atom 3. `Cl^(-)` had 18 electrons and Cl has 17 electrons. The later EXPERIENCES an greater effective inward pull by the nucleus. 4. This DECREASES the size of Cl atom when compared to `Cl^(-)` ion. |
|
| 48. |
What do you understand by 'triple point' of a substance ? |
| Answer» SOLUTION :Triple point of a substance represents the CONDITIONS at which all the three phases of the substance, i.e., SOLID, liquid and GAS exist together, e.g., triple point of water is `0.01^(@)C` at 4.58 mm pressure where ice, liquid water and water VAPOUR coexist. | |
| 49. |
Zinc blende has ________ arrangement of S^(2-) ions whereas wurtzite has _____ arrangement of S^(2-) ions. |
|
Answer» |
|







