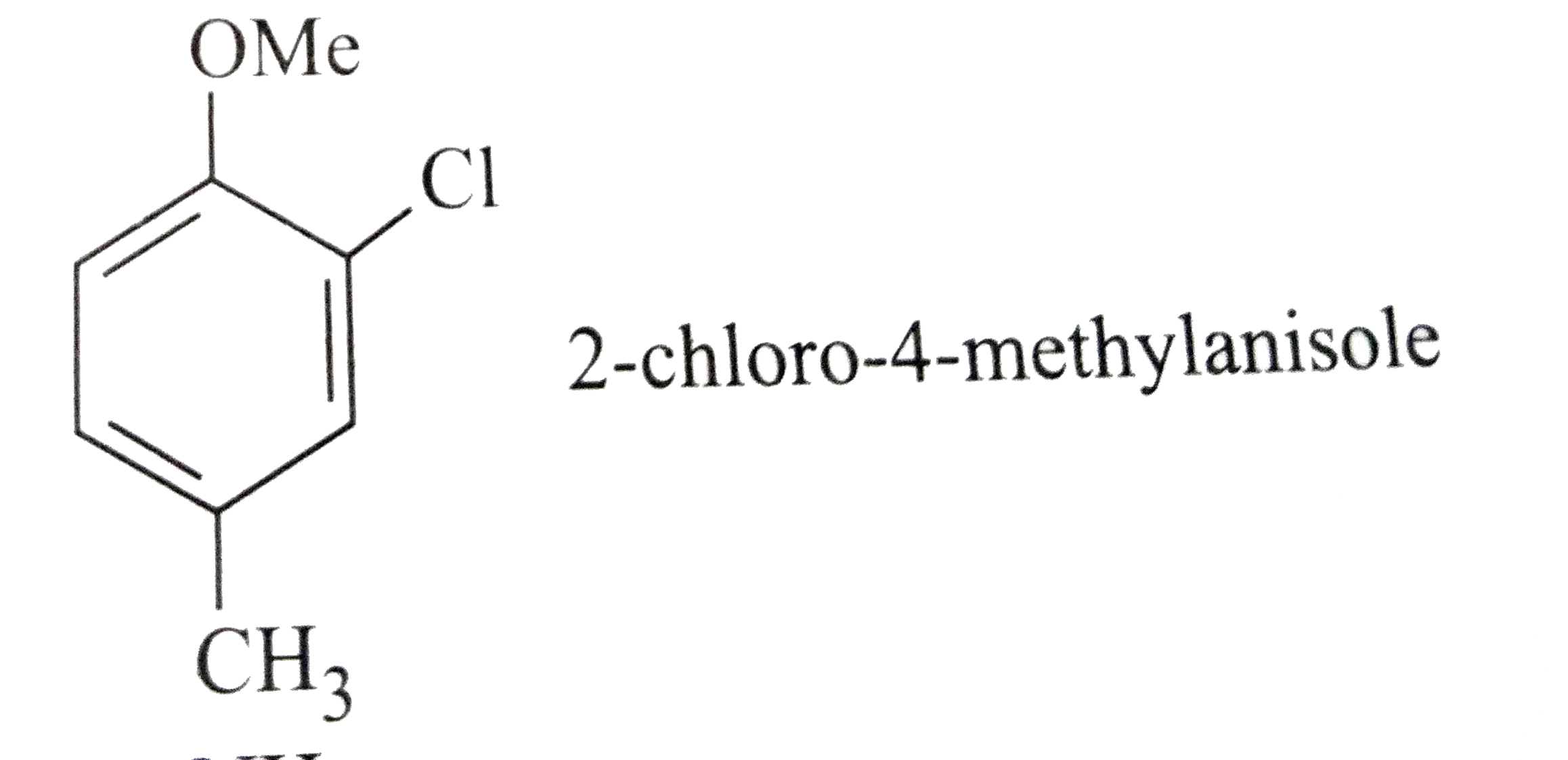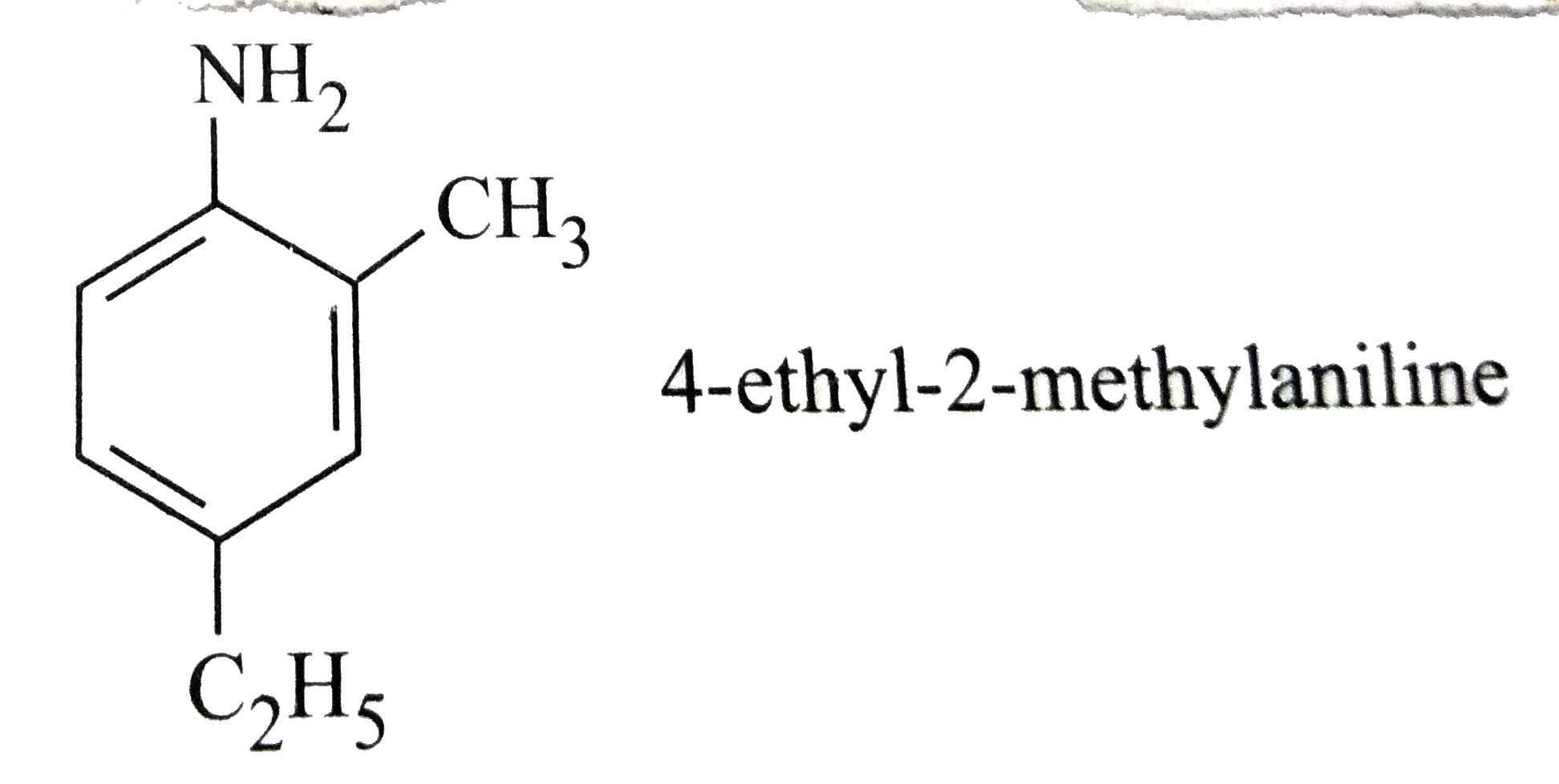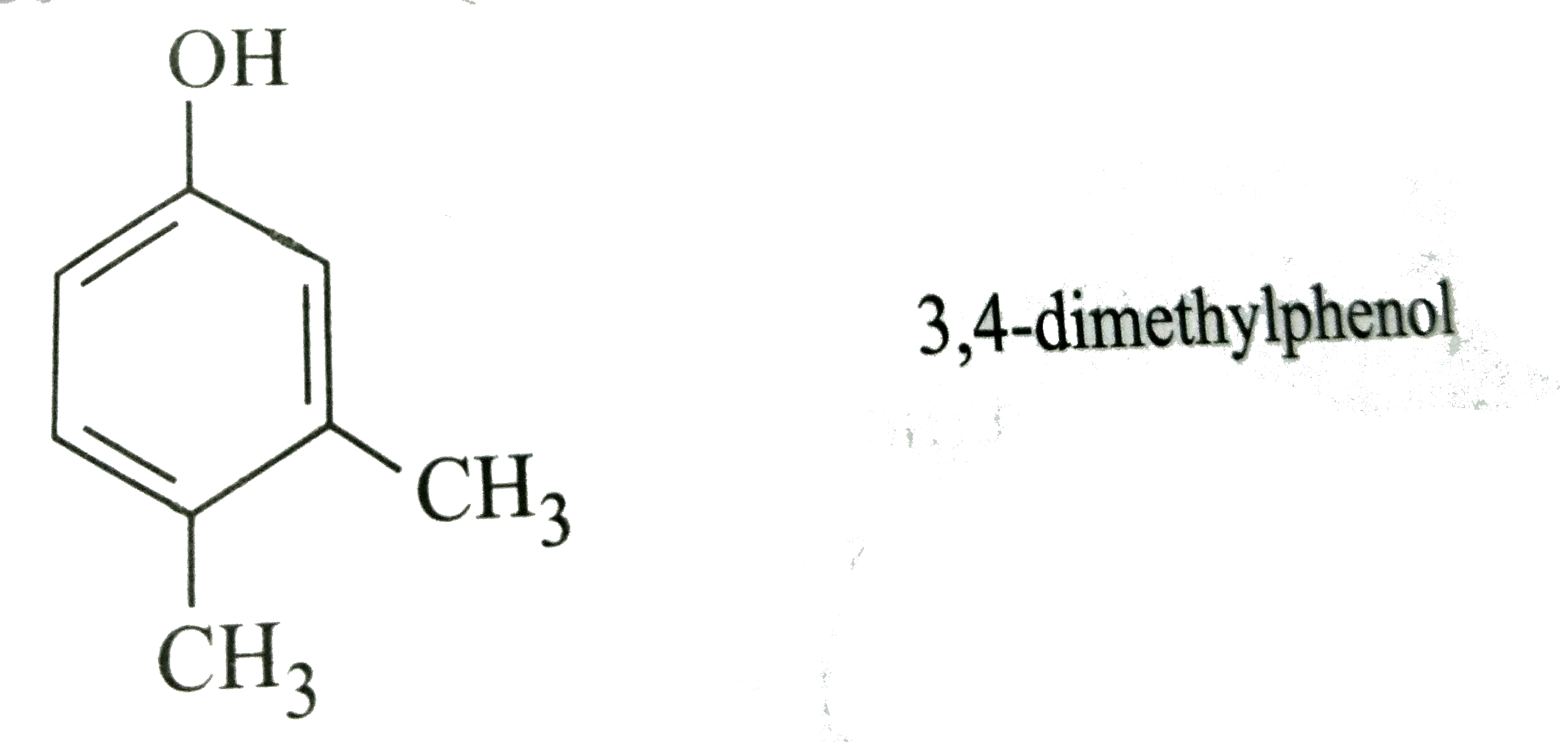Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following will occur if a 0.1 M solution of a weak acid is diluted to 0.01 M at constant temperature ? |
|
Answer» `[H^(+)]` will decrease to 0.01 M |
|
| 2. |
The units of Vander Waals constant 'b' and 'a' respectively |
|
Answer» `molL^(-1)andL"ATM"^(2)mol^(-1)` |
|
| 3. |
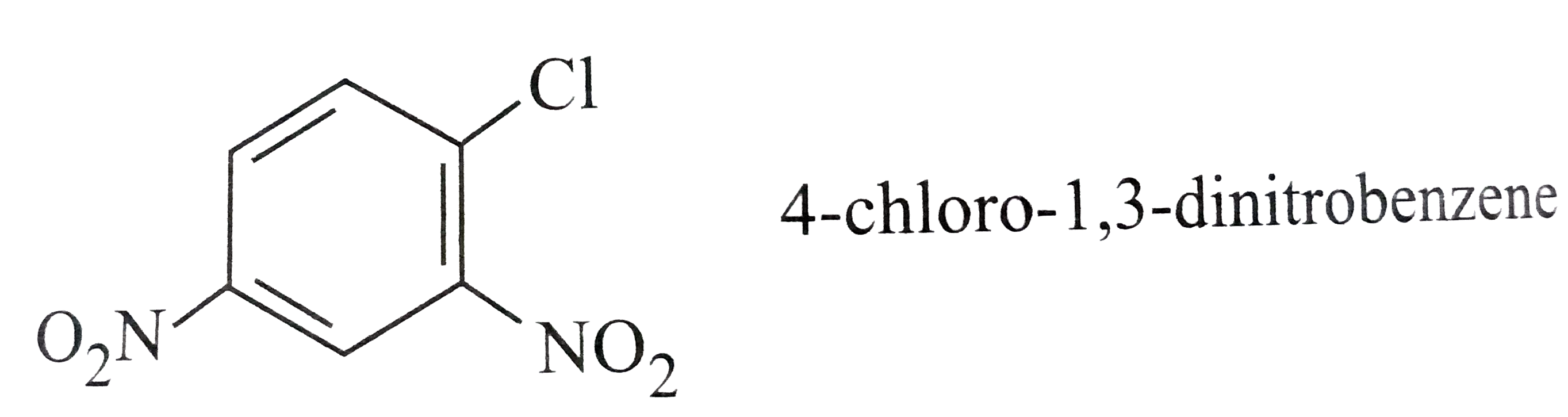
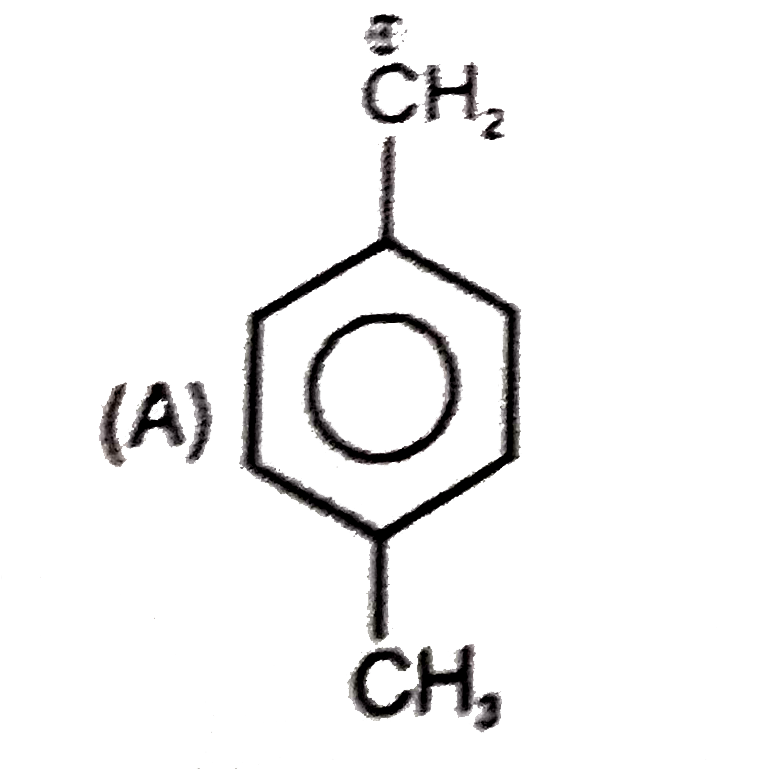
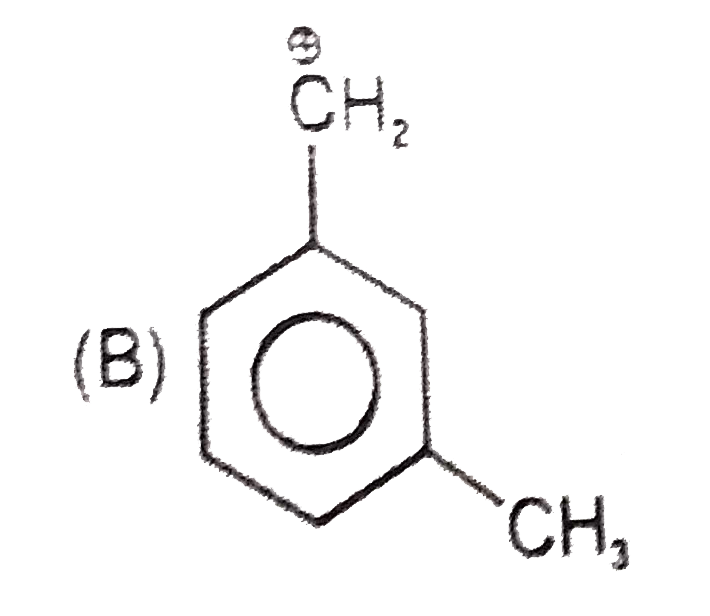
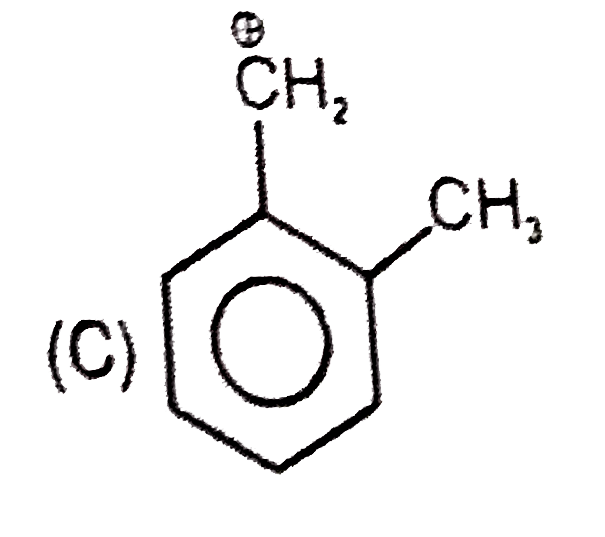
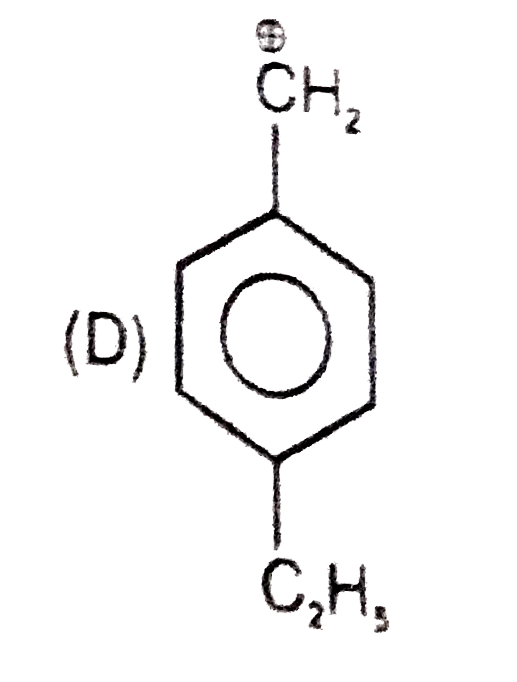
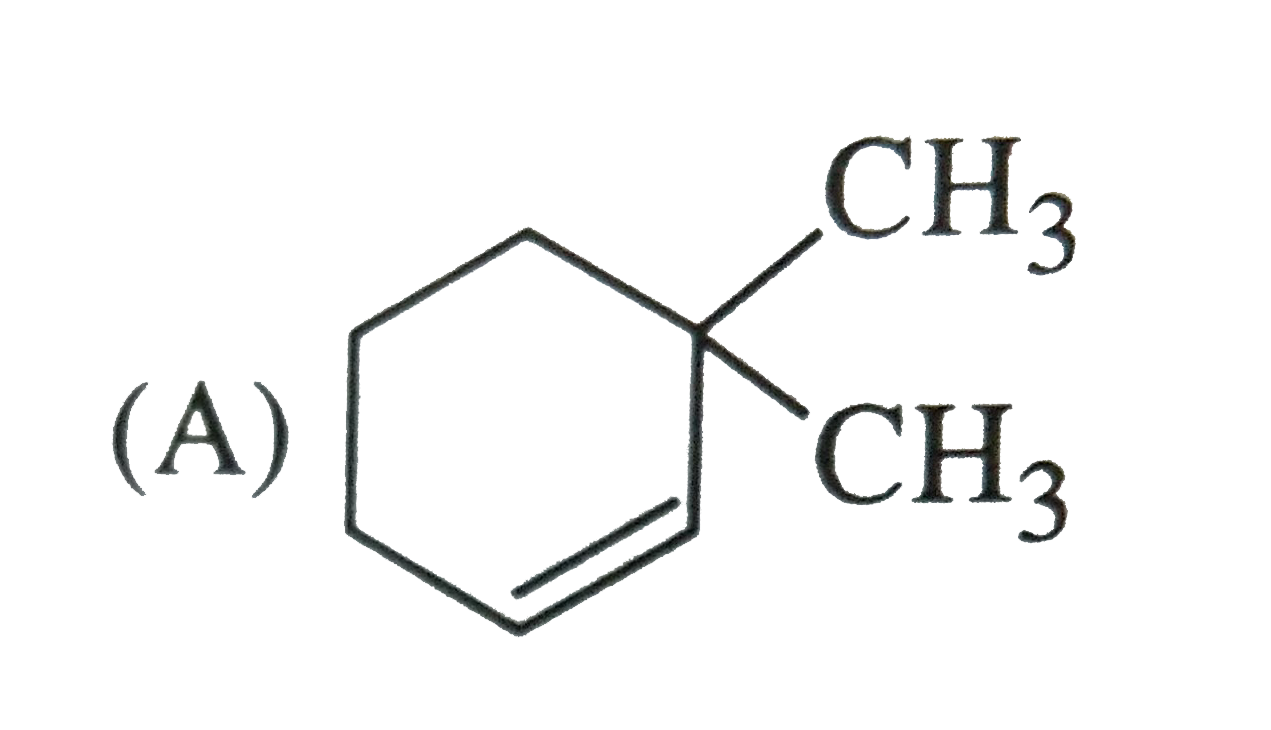
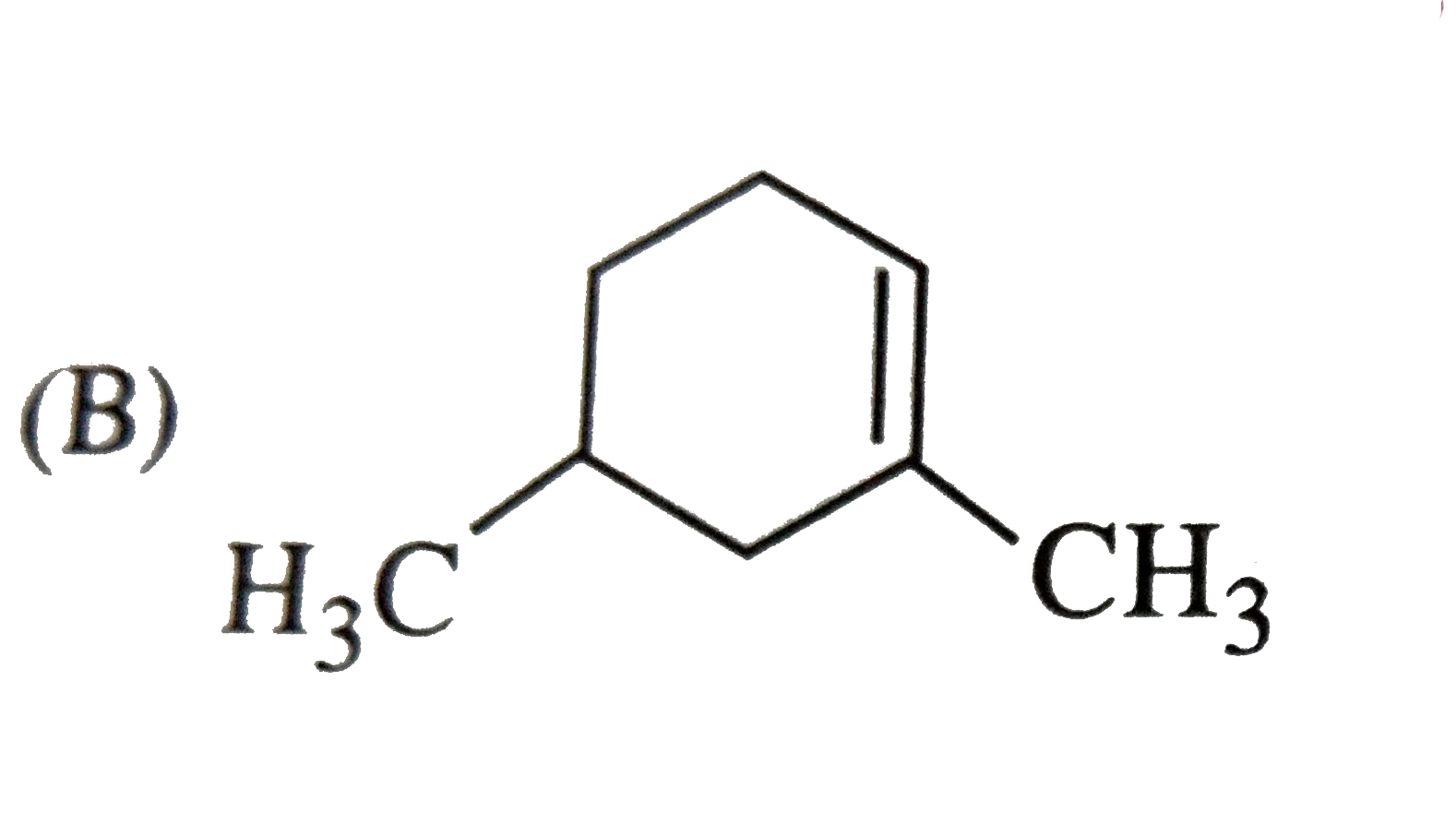
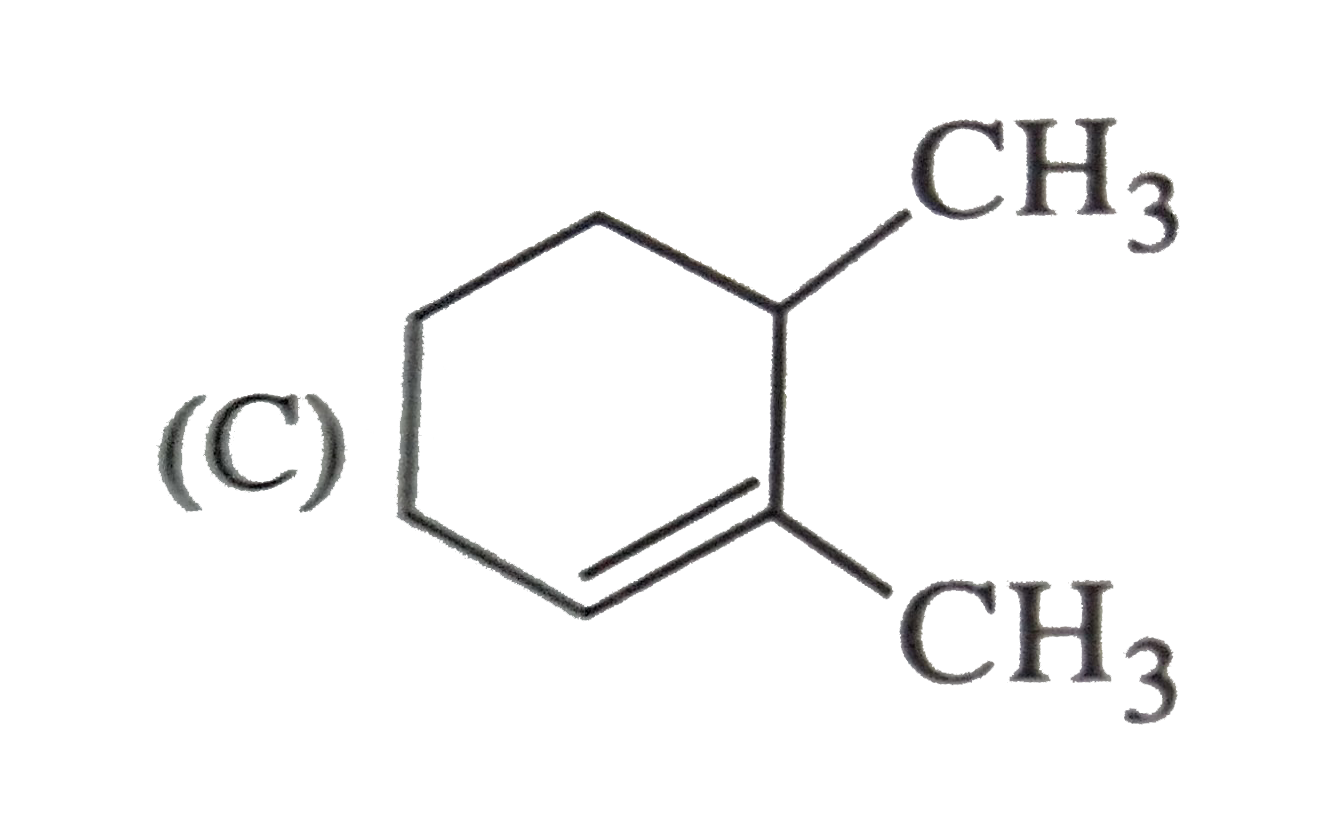
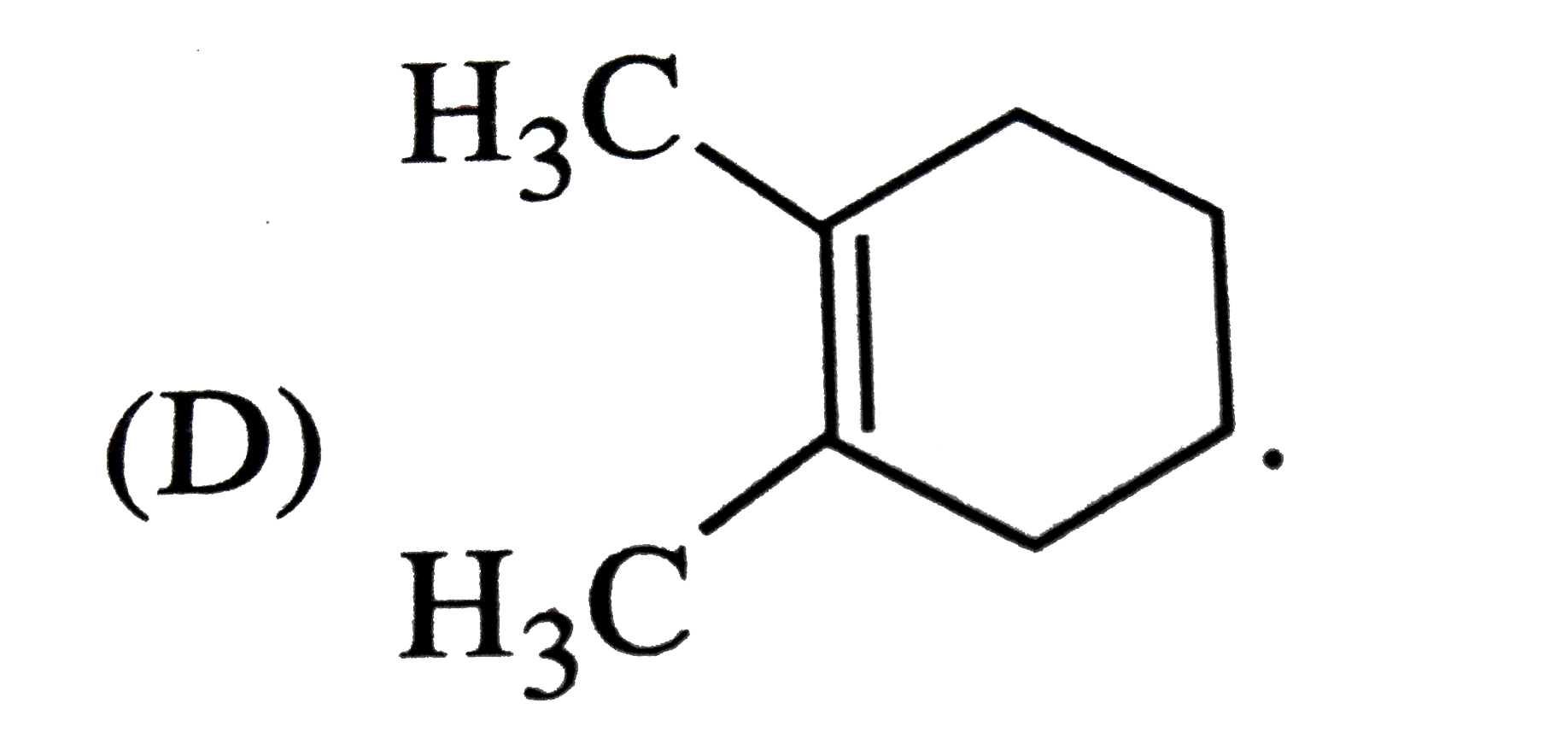
Which of the following names of substituted benzene compounds is not correct? |
|
Answer»
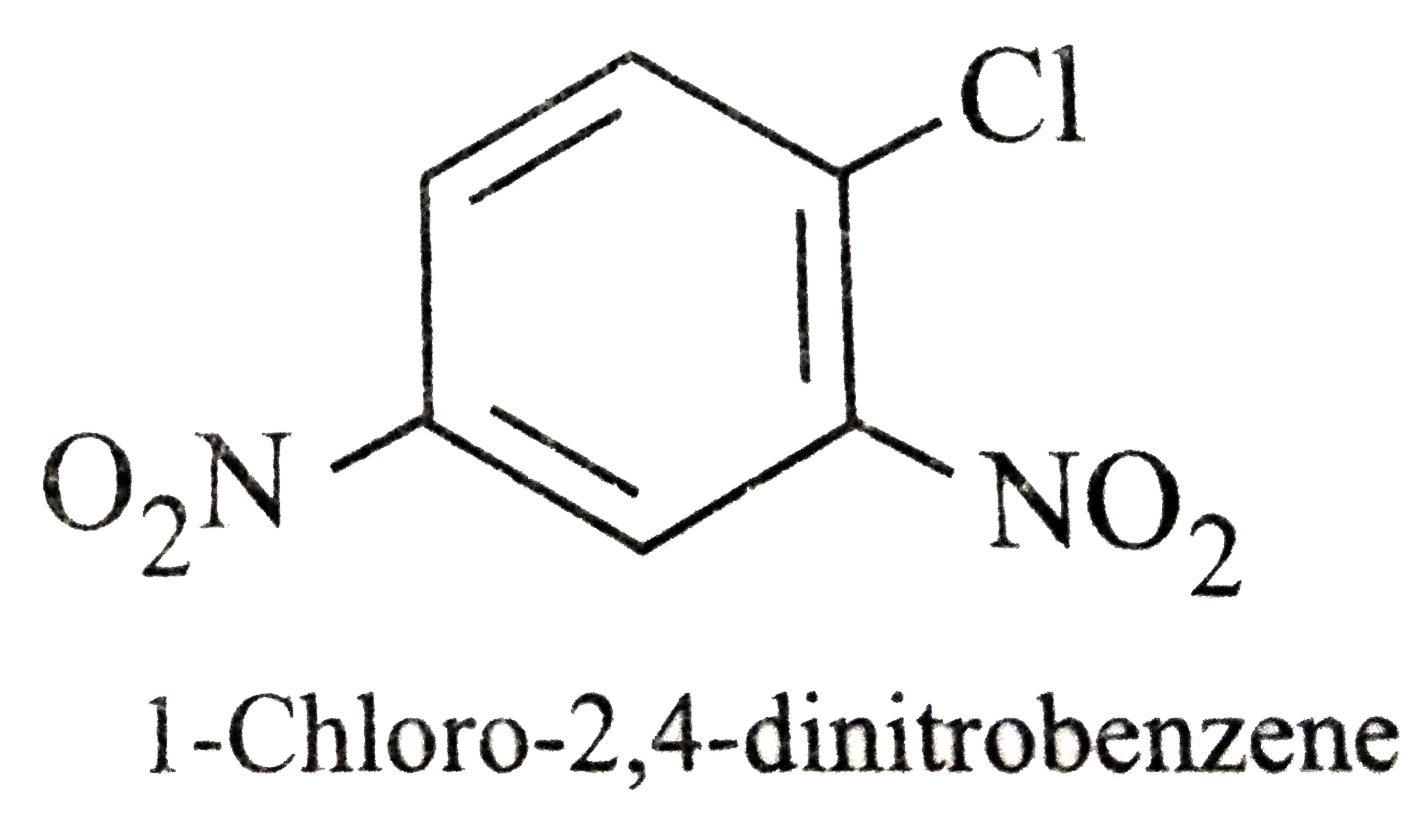
|
|
| 4. |
Write the requirements for aromatic character in compound. |
|
Answer» Solution :Benzene was considered as parent .aromatic. compound. Now the name is applied to all the ring system whether or not having benzene ring, possessing following characteristics. (a) Requirements for aromatic character in compound : (i) Compound must havr cyclic (ring) STRUCTURE. (ii) Cyclic compound must have all atoms in one planer structure. (iii) Complete delocalisation of the `pi` electronsin the ring. (iv) Presence of `(4n+2)pi` electrons in the rinf where .n. is an inetger (n=0, 1, 2,...). This is often referred to as Huckel Rule. (b) Huckel.s rule for aromaticity : If the number of electrons in planar cyclic compound equals to (4n+2), then these compounds possess aromatic properties, (aromaticity) where n = number of cyclic structures, (4n+2) = total delocalized electron of atoms present in aromatic ring). (c) Aromatic hydrocarbon compounds : (i) Benzene is aromatic in nature. Number of rings in benzene = n =1 NUmber of double bonds in benzene = 3 so, number of `PI2(p)` electrons = 6 `therefore` According to Huckel rule, if (4n+2) `pi` electrons are present in structure then it is considered as aromatic. So for benzene , n=1 and (4n+2) = 4(1) + 2 = 6, so benzen obeys Huckel rules and benzene is aromatic in nature and it possess aromatic characteristics.  (ii) Cyclopentadienyl anion aromatic in nature. According to Huckel rule, n = 1 and hence (4n+2) = 6 In cyclopentadienail anion TWO double bond have `2 xx 2 = 4pi` + two lone `pi` electrons, so it is aromatic in nature.  (iii) Cycloheptatrienyl cation is aromatic in nature. Number of ring = n = 1 `therefore (4n+2) = 6`  In this compound three double bonds are present, so no. of `pi` electrons `= (3 xx 2) = 6`. So it follows Huckel rule, so cycloheptatriene, n = 1(4n+2)=6, cation is aromatic in nature. It possess positive charge `sp^(2)` carbon so it also possess PLANARITY. (iv) Nepthalene is aromatic in nature.  Number of ring = n= 2 It having five double bonds so no. of `pi` electrons `= (5 xx 2) = 10` electrons Napthalene follows Huckel rules and so it is aromatic innature. (v) Anthrecene and Phenanathrene are aromatic compounds. 
|
|
| 5. |
What do you understand by the term oxidation number? |
| Answer» Solution :Oxidation number refers to the number of charges an atom WOULD have in a molecule or an IONIC compound, if ELECTRONS were transferred completely. The oxidation NUMBERS reflect the number of electrons transferred. | |
| 6. |
The r.m.s. velocity of hydrogen is sqrt(7) times the r.m.s. velocity 1.0 g cm^(-3) and that of wate vapours is 0.0006 g cm^(-3), then the volume of water molecules in 1 L of steam at this temperature is |
|
Answer» `T(H_(2)) = T(N_(2))` or `(3RT_((H_(2))))/(2) = 7 xx (3RT_((N_(2))))/(28)` or `T_((N_(2))) = 2T_((H_(2)))` `:. T_((N_(2))) gt T_((H_(2)))` |
|
| 7. |
Which of the following compound will gives positive test with NaHCO_(3)? CH_(3)CO OH,PhSO_(3),PhOH |
|
Answer» |
|
| 8. |
Write general and empirical forumula of silicon. |
| Answer» Solution :General formula of silicon is `(R_2SiO)_n` . Where, R = METHYL or PHENYL group. Empirical formula of silicon is `R_2 SiO`. (Which is very SIMILAR to KETONS and hence they are known as silicons) | |
| 9. |
Write the MO electronic configuration of a diatomic molecule having a bond order of three . |
| Answer» SOLUTION :`N_(2) = KK[SIGMA (2s)]^(2) [sigma ^(**)(2s)]^(2)[ PI(2p_(x))]^(2) [pi^(**)(2p_(y))]^(2) [pi(2p_(z))]^(2)` | |
| 10. |
The relative number of mass of elements, 'X' and 'Y' in a compound is 0.25 and 0.5. The empirical formula of compound is |
|
Answer» XY Mole ratio of 'X' = 0.25/0.25=1 Mole ratio of 'Y' 0.5/0.25=2 EMPIRICAL FORMULA `=XY_2` |
|
| 11. |
Which of the following are valid iodometric titractions? |
|
Answer» `HClO+2I^(-)+H^(+)rarrCl^(-)+I_(2)+H_(2)O` |
|
| 12. |
Which of the following equation represents standard heat of formation of ethanol? |
|
Answer» `CH_(3)CHO_((l)) + (1)/(2)H_(2(g)) OVERSET(Ni)rarr C_(2)H_(5)OH_((l))` |
|
| 13. |
Which one of the following does not give alkane? |
|
Answer» Reaction of `CH_3I` with NA in ether |
|
| 14. |
What volume of water is to be added to 100 cm^3 of 0.5M NaOH solution to make it 0.1 M solution? |
|
Answer» `200cm^3` `0.5xx100=0.1xxV_2 rArr V_2=500cm^3` Volume of WATER to be added to 100 `cm^3` of solution = `500-100=400cm^3` |
|
| 15. |
Which of the following ion is stable in water ? |
| Answer» Answer :D | |
| 16. |
Write short note on Ionic or Saline Hydrides. |
|
Answer» Solution :These are stoichiometric compounds of dihydrogen formed with most of the s-block elements which are highly electropositive in character. Such as LiH, `BeH_2` and `MgH_2`. In fact `BeH_2` and `MgH_2` are polymeric in structure. The ionic hydrides are crystalline, non-volatile and non-conducting in solid state. However, their melts conduct electricity and on electrolysis liberate dihydrogen gas at anode, which confirms the existence of `H^-` ion. `2H^(-)` (melt) `overset"anode"to H_(2(g)) + 2e^(-)` Saline hydrides react violently with water producing dihydrogen gas. `NaH_((s)) + H_2O_((AQ)) to NaOH_((aq)) + H_(2(g))` Lithium HYDRIDE is RATHER unreactive at moderate temperatures with `O_2` or `Cl_2`. It is, THEREFORE, used in the SYNTHESIS of other useful hydrides, e.g., `8LiH+Al_2Cl_6 to 2LiAlH_4 + 6LiCl` `2LiH + B_2H_6 to 2LiBH_4` |
|
| 17. |
Whichof the followingproperties isassociatedwithwholenumberforan atom ? |
|
Answer» ATOMICRADIUS |
|
| 18. |
What happens when an alkaline solution of potassium ferricyanide is reacted with H_(2)O_(2)? |
|
Answer» Potassium ferricyanide is oxidised to potassium FERROCYANIDE becomes colourless and `H_(2)O_(2)` is oxidised. |
|
| 19. |
What is a 'semiconductor' ? Describe the two main types of semiconductors and contrast their cnduction mechanisms . |
|
Answer» SOLUTION :Substances whose conductance LIES inbetween that of metals (CONDUCTORS) and insulators are called semiconductors. Two MAIN TYPES of semiconductors are n-type and p-type. |
|
| 20. |
The second ionization enthalpy of alkali metals are very high. Give reason |
| Answer» Solution :The removal of one ELECTRON from the alkali metals causes the formation of monovalente having very STABLE ELECTRONIC configuration. Therefore it becomes very difficult to remo the SECOND electron from the stable noble gas configuration, GIVING rise to very high second ionization energy values. | |
| 21. |
What is the wave length of H, line in Balmer series of hydrogen spectrum? (R = Rydberg constant) |
|
Answer» `36//5R` |
|
| 22. |
What properties of water make it useful as a solvent ? What types of compound can it (i) dissolve and (ii) hydrolyse ? |
|
Answer» Solution :Water is a colourless and tasteless liquid. The unusual properties of water in the condensed phase (liquid and solid states) are due to the PRESENCE of extensive hydrogen BONDING between water molecules. This leads to high freezing point, high boiling point, high heat of vaporisations and high heat of fusion in comparison to `H_2 S` and `H_2Se`. In comparison to other liquids, water has a higher specific heat, THERMAL conductivity, surface tension, dipole moment and dielectric CONSTANT, etc. These properties allow water to play a key role in the biosphere. It is an excellent solvent for transportation of ions and molecules required for plant and animal metabolism. Due to hydrogen bonding with polar molecules, even covalent compounds LIKE alcohol and carbohydrates dissolve in water. Physical properties of `H_2O` and `D_2O`  
|
|
| 23. |
Which of the following two reactions does HNO_(3) not acts as an oxidising agent ? Give an example 2HNO_(3)+P_(2)O_(5)to2HPO_(3)+N_(2)O_(5) |
|
Answer» <P> SOLUTION :`2HNO_(3)+P_(2)O_(5)to2HPO_(3)+N_(2)O_(5)` |
|
| 24. |
What is the criteriafor spontaneityin terms of free energy change ? |
| Answer» Solution :If`DeltaG`is NEGATIVE, PROCESS is SPONTANEOUS , if `DeltaG` is POSITIVE , direct process is non-spontaneousand if`DeltaG=0`, the processis in equilibrium | |
| 25. |
Which stoichometric defect in crystals increases the density of a solid ? |
| Answer» SOLUTION :INTERSTITIAL DEFECT | |
| 26. |
Which of the following expressions represents the spectrum of Balmer series (If n is the principal quantum number of higher energy level) in Hydrogen atom? |
|
Answer» `BAR(UPSILON) = (R(n-1) (n+1))/(n^2)` |
|
| 27. |
Which of the following metal does not produces dihydrogen gas with dilute hydrochloric acid? |
| Answer» Solution :Metals which are below .H. in electro chemical series not react with MINERAL acids.s | |
| 28. |
What is the basic principle of a television picture tube or fluorescent light tubes ? |
| Answer» Solution :The television picture TUBE is a cathode ray tube in which a picture is PRODUCED due to fluorescence on the television screen coated with SUITABLE material. SIMILARLY, fluorescent light tubes are also cathode ray tubes coated inside with suitable material which produced visible light on being HIT with cathode rays. | |
| 29. |
Which statements is//are correct? |
|
Answer» `0.1M NH_(3)` solution will precipitate `Fe(OH)_(2)` from a `0.1M` solution `Fe^(2+)`. |
|
| 30. |
When 22.4 litres of H_(2) (g) is mixed with 11.2 litres of Cl_(2)(g), each at 273 K at 1 atm the moles of HCl (g). formed is equal to |
|
Answer» a) 2 moles of HCl gas 
|
|
| 32. |
Which alkyne yields butanoic acid (CH_3-CH_2 - CH_2 - CO_2 -CO_2H). As the only organic product on treatment with ozone followed by the hydrolysis ? |
|
Answer» 1-butyne |
|
| 33. |
What factor make the separation of Lanthanides a formidable task |
|
Answer» SIMILARITY in ionic size |
|
| 34. |
When CI_(2) gas reacts with hot and concentrated sodium hydroxide solution the oxidation number of chlorine changes from |
|
Answer» zero to +1 and zero to -5 Here O.N of CI increases from +1 CIO to +5 in `CIO_(3)^(-)` while that of CI decrease form +1 in `CIO_(3)^(-)` while that of CI DECRESES form +1 in `CIO^(-)` to -1 `CI^(-)` THEREFORE it is a disproportionation REACTION |
|
| 35. |
Which of the following radius of orbits in hydrogen atom are possible ? |
|
Answer» 52.9 pm For FIRST SHELL , r= 52.9 pm For second shell , r =` 52.9xx4 = 211.6 `pm |
|
| 36. |
Which of the follwoings is the least stable form of the mentioned compound? |
|
Answer» Eclipsed |
|
| 37. |
The stabilization of a halffille d-orbitals is more pronouced than that of p-orbital why? |
|
Answer» SOLUTION :They exactly HALF filled orbitals have GREATER stability.the reason for their stability are (i)symmetry(ii)EXCHANGE ENERGY. (i)Symmetry:The half filled orbitals are more symmetrical than partially filled orbitals and this symmetry leads to greater stability . (ii) |
|
| 38. |
Which one are correct about the reaction? HgS+HCI+HNO_(3) rarrH_(2)HgCl_(4)+NO +S+H_(2)O |
|
Answer» HG is reduced |
|
| 39. |
Which of the product of the following reaction ? |
|
Answer»
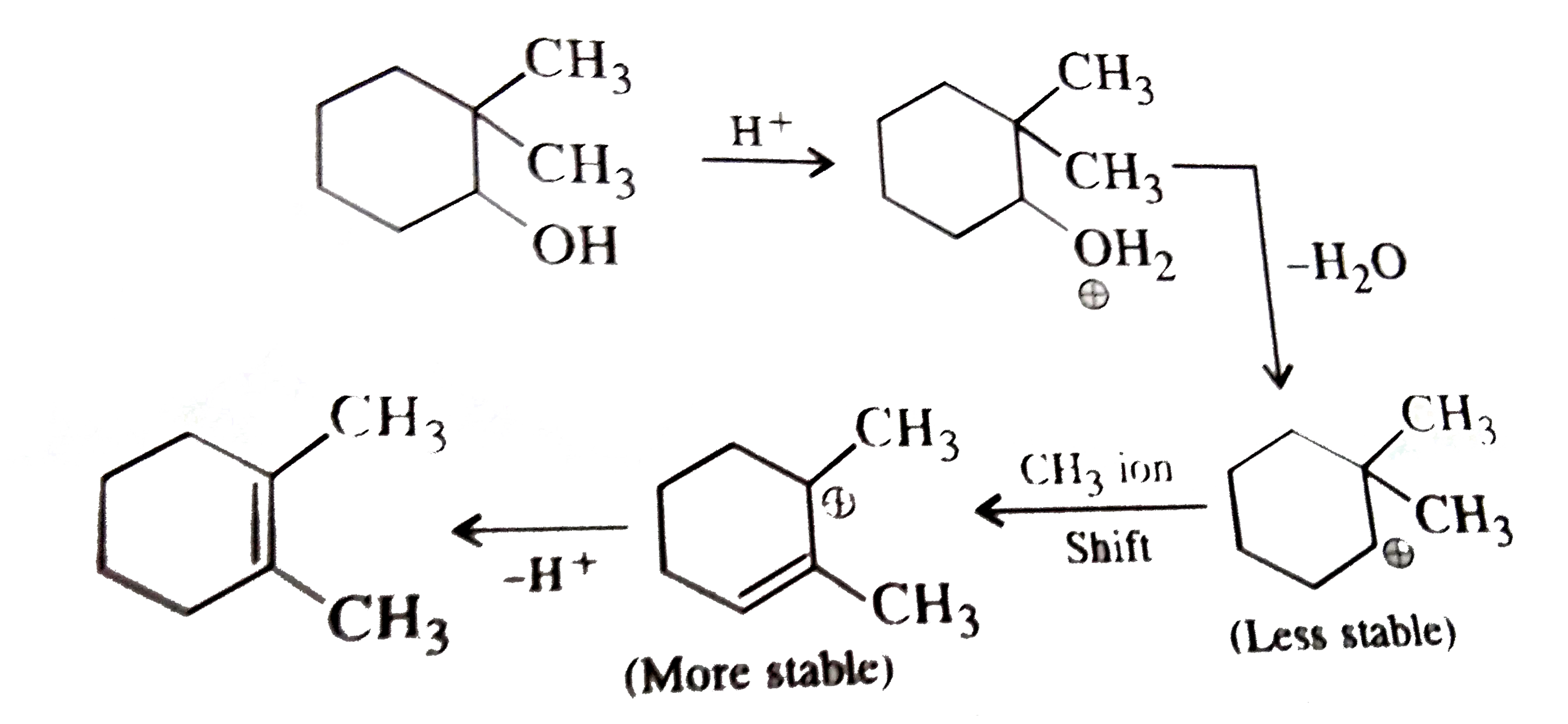
|
|
| 40. |
When iron reacts with Brion in acid solution Br_2is liberated. The equivalent mass of KBrO_3in this reaction is |
|
Answer» M/8 |
|
| 41. |
Which one of the following reagents is useful for separating aniline from nitrobenzene? |
|
Answer» AQ. `NaHCO_(3)` |
|
| 42. |
There is no importance of Dalton's law with reference to ........... |
|
Answer» ISOTOPES |
|
| 43. |
Which of thefollowing has maximum numberofunpairedelectrons ? |
|
Answer» `FE^(3+)` |
|
| 44. |
Which of the following molecules have the highest mean speed at the same temperature |
|
Answer» `CO_2` |
|
| 45. |
Which of the following is the most abundant alkali metal (in combined state) in nature ? |
|
Answer» K |
|
| 46. |
The standard enthalpies of formation of H_(2)O_(2(l)) and H_(2)O_((l)) " are " -187.8 kJ "mole"^(-1) and -285.8 kJ "mole"^(-1) respectively. The Delta H^(0) for the decomposition of one mole of H_(2)O_(2(l)) " to " H_(2)O_((l)) and O_(2(g)) is |
|
Answer» `-473.6 kJ. "mole"^(-1)` `Delta H= Sigma` Heat of formation of PRODUCTS `-Sigma`Heat of formation of reactants `=(-285.8) + (0) - (-187.8) = -98` |
|
| 47. |
What are functional group inter conversions? |
|
Answer» Solution :Organic SYNTHESIS involves functional group inter conversions.A PARTICULAR functional group can be CONVERTED into other functional group by REACTING it with suitable reagents. For example, the carboxylic acid group` (-COOH)` PRESENT in organic sacids can be transformed into `-COCI` by treating the acid with `SOCI_(2)` reagent. |
|
| 48. |
What happens to the solubility of BaSO_(4) when a few drops of BaCl_(2) solution is added to its saturated solution? Give reason. |
|
Answer» |
|
| 49. |
Which of the following is the most likely Lewis structure of nitroslyl chloride NOCl |
|
Answer»
|
|
| 50. |
underset("Salt of iron")((X)) overset(Na_(2)S_(2)O_(3))rarr underset("Violet complex")((Y)) rarr underset("Green solution")((Z)) Write oxidation number of Fe in (X) as first digit, (Y) as second digit, (Z) as third digit in OMR. |
|
Answer» `[Fe(S_(2)O_(3))_(2)]^(-)+Fe^(3+) overset("Boil")rarr 2Fe^(2+)+S_(4)O_(6)^(2-)` `ulbar(2Fe^(3+)+S_(2)O_(3)^(2-)rarr 2Fe^(2+)("green")+S_(4)O_(6)^(2-))` `X rarr 3, Y rarr 3, Z rarr 2` |
|