Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What happens when an electron hits a zinc sulphide screen and what does it prove ? |
| Answer» SOLUTION :A SCINTILLATION is produced which in turn PROVES that ELECTRON has particle CHARACTER. | |
| 2. |
Which of th e following represents the correct set of four quantum number of 3d orbital electron ? |
|
Answer» `n=3,L=3,m =2,s=+1/2` n=3, l = ,m =(-2) to (+2) `S=+1/2 ro -1/2` |
|
| 3. |
When N and S both are present in an organic compound, the sodiun fusion extract with FeCl_3gives |
| Answer» Answer :D | |
| 4. |
Discuss VSEPR model applied for linear, trigonal planar, tetrahedral and octahedral geometries of molecules. |
|
Answer» `O_(3)` |
|
| 5. |
Which of the following reaction does not take place |
|
Answer» `CuSO_(4)+ZnrarrZnSO_(4)+Cu` |
|
| 6. |
What is the shape of a methane molecule ? |
| Answer» SOLUTION :TETRAHEDRAL | |
| 7. |
Which one between p and sp-orbital has more directional characteristics and why? |
| Answer» Solution :Directional nature of sp-orbital is greater than that of p-orbital. Becausethe two LOBES of p-orbitals are SIMILAR in SIZE and possess same electron density while ONE of the two lobes of sp-orbitals is larger in size and have higher electron density. | |
| 8. |
The RMS speed of hydrogen is sqrt7 times the RMS speed of nitrogen. Ifis the temperature of the gas. Then |
|
Answer» `T_(H_(2))=T_(N_(2))` Hence,`(T(H_(2)))/(T(N_(2)))=7(M(H_(2)))/(M(N_(2)))=l((2)/(28))=(1)/(2)` `T_(h_(2))ltT_(N_(2))` |
|
| 9. |
The substance not likely to contain CaCO_(3) is ….. |
|
Answer» dolomite |
|
| 10. |
Which of the following is relatively the weakest acid |
|
Answer» `HA (2xx10 ^(-4) ) ` |
|
| 11. |
What should be the tolerable limit of fluoride ions in drinking water ? What happens if it is higher than 10 ppm ? |
| Answer» Solution : 1 PPM or 1 MG `DM^(-3)` Higher concentration is harmful to bones and teeth | |
| 12. |
Which one of the following elements is unable to form MF_6^(2-) ion? |
|
Answer» GA |
|
| 13. |
Which of the following statements about HCO_(3)^(-) are correct ? 1. It is a Bronsted acid 2. It can ionize in water to form CO_(3)^(2-) (aq) 3. It does not exist in aqueous solution 4. It is a Bronsted base. Select the correct answer using the codes given below |
|
Answer» 1, 2 and 3 It can ionize in water to `CO_(3)^(2-)` ion. It can exist in AQUEOUS solution. Hence, only 3 is wrong |
|
| 14. |
Which of the following is not an aromatic |
|
Answer»
|
|
| 15. |
What is the pH of 1M HCI ? |
| Answer» Solution :Mathematically its ZERO, ACTUALLY pH scale is for only SOLUTE solution and it is for the solution having concentration less than 1 M. | |
| 17. |
Water become useless for drinking purpose if the fluoride concentration exceeds |
| Answer» Answer :C | |
| 18. |
Which of the following compounds undergoes sublimation? |
|
Answer» NAPHTHALENE (B) Camphor (C ) `HgCl_(2)` (D) `NH_(4)Cl` |
|
| 19. |
The simplest way to check whether a system is a colloid is by |
|
Answer» Tyndall EFFECT |
|
| 20. |
Which of the following elements does not show disproportionation tendency? |
|
Answer» CI |
|
| 21. |
Which of the following is CORRECT regarding reductant ? |
|
Answer» It causes oxidation of the other CHEMICAL SPECIES involved in the reaction |
|
| 22. |
Total volume of atoms present in a face centred cubic unit cell of a metal is (r=atomic radius) |
|
Answer» `20/3pir^(3)` If fcc number of atoms per unit cell =4 `THEREFORE` volume of FOUR atoms=`4times4/3pir^(3)=16/3pir^(3)` |
|
| 23. |
Write the Balance chemical equation for theKc= ([CaO_(s)] [CO_(2(g))])/([CaCO_(s)]) |
| Answer» SOLUTION :`CaCO_(3)(S)hArr CaO(s)+CO_(2)(g)` | |
| 24. |
Which one of the following is prepared in industry by the auto oxidation of 2-alkyl anthraquinol? |
|
Answer» HEAVY WATER |
|
| 25. |
Which one of the following is the formula of sodium nitroprusside? |
|
Answer» A)`Na_(4)[Fe(CN)_(5)NO_(5)` |
|
| 26. |
The treatment of benzene with isobutene in the presence of sulphuric acid gives |
|
Answer» isobutylbenzene 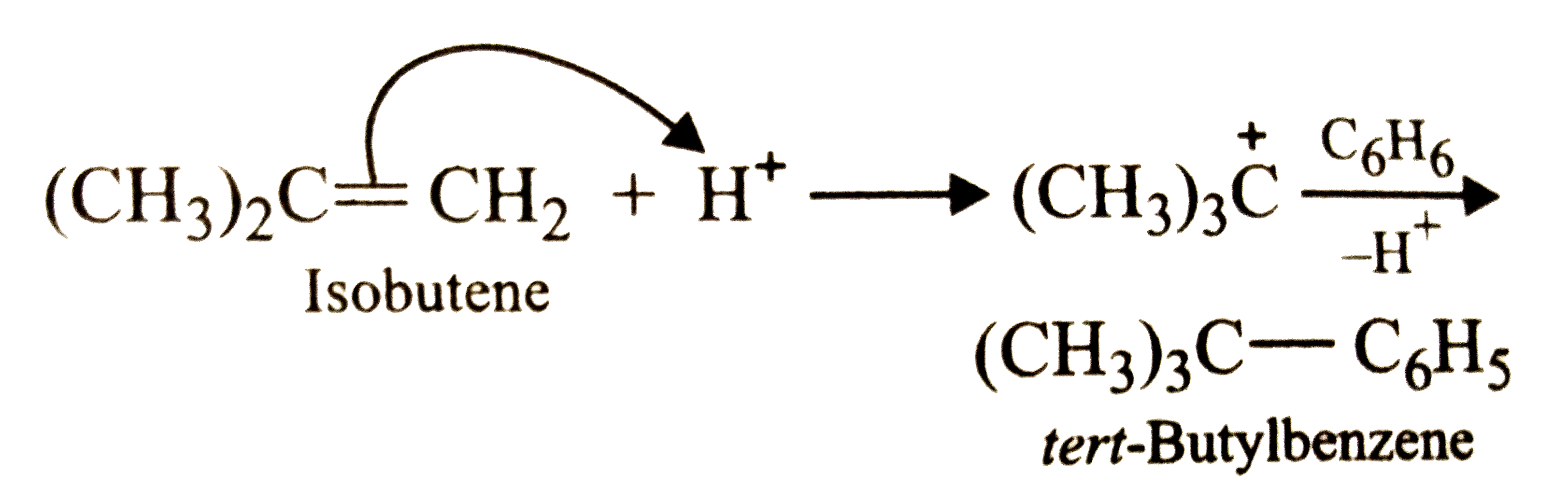
|
|
| 27. |
What happens when benzene is added to hydrogen. |
Answer» Solution :Benzene reacts with hydrogen n PRESENCE of Pd nickel as a catalyst at 475-500 K GIVES CYCLOHEXANE. 
|
|
| 29. |
Which of the following subtances will absorb more hert energy? |
|
Answer» CONC. `H_(2)SO_(4)` |
|
| 30. |
Which of the following order for ionization energy is correct? |
|
Answer» `Be gt B gt C gt N gt O` |
|
| 31. |
The structure ofIF_(7)is |
|
Answer» Trigonal bipyramid |
|
| 32. |
Which alkali metal ion forms largest hydrate ion in aqueous solution and why? |
| Answer» Solution :`Li^(+)` forms largest hydrated cations because it has highest HYDRATION ENERGY. It has SMALLEST SIZE therefore, it is most hydrated. | |
| 33. |
The vapour density of N_(2) O_(4) at certain temperature is 30 . What is the percentage dissociation of N_(2) O_(4) at that temperature ? [Hint : alpha = (D - d) // d ( n - 1)] |
|
Answer» `53.3` % |
|
| 34. |
Which statement is/are true about resonance ? |
|
Answer» It decreases the energy of system. |
|
| 35. |
Which of the following halide of group 14 does not exist?a) CF_4b) Cl_4c) SiF_4d) PbI_4 |
|
Answer» `CF_4` |
|
| 36. |
What is the maximum number of electrons that can be present in an atom in which the highest principal quantum number is 4 ? |
|
Answer» SOLUTION :Keeping in VIEW aufbau principle, the electronic configuration of atom having highest value of n = 4 will be `1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) 4s^(2) 3d^(10) 4P^(6)` (After 4p, filling of 5s starts) Hence, maximum number of electrons present = 36 (KR) |
|
| 37. |
Which of the following is an example of super octet molecules? |
| Answer» Answer :D | |
| 38. |
What is the hybridisation state of benzyl carbonium ion? |
|
Answer» `SP^(2)` |
|
| 39. |
Three gases A, B and C were taken at partial pressures of 1 bar each along with excess of liquide D so thet following equation was established. A(g)+B(g)hArrC(g)+D(g) Calculate partial pressure of C (in pascal ) when equalibrium gets established in the container at 300 k. Given:DeltaG^(@)f A (g) =- 200 kcal/mole DeltaG^(@)f D(l)=-49.58 kcal/mole DeltaG^(@)f B (g0 =-100 Kcal/mole DeltaG^(@)f D (g)=-49.58 kcal/mole DeltaG^(@)f C (g) =- 250 kcal/mole R=2 cal/mol K, In 2=0.7 sqrt5= 2.24 all data are given at 300 K. Divede your answer by 10 and fill the OMR. |
|
Answer» |
|
| 40. |
Which of the following statements are correct about the reaction intermediate ? |
|
Answer» Intermediate (III) is unstable because in this carbon is attached to 5 atoms. |
|
| 41. |
Which of the following is a pair of functional isomers? |
|
Answer» `CH_(3)COCH_(3).CH_(3)CHO` |
|
| 43. |
Which of the following have the same number of unpaired electrons in 'd' orbitals |
|
Answer» Cr `._24Cr=3d^5 4s^1 =5` `._25Mn=3d^5 4s^2 =5` `._26Fe^(3+)=3d^5 4s^0=5` |
|
| 44. |
Which of the following metals is not used as a catalyst in addition reaction of alkyne ? |
|
Answer» Ni |
|
| 45. |
{:('X'("salt solution")+,AgNO_(3)rarr'Y'(ppt.)overset(Na_(3)AsO_(3))rarr'Z'(yellow ppt.)),(,""darr),(,"Soluble in dil"NH_(3)(aq)and Na_(2)S_(2)O_(3)):} The correct statement about above experiment may be: |
|
Answer» `'Y' = AGCI` and insoluble in dil. `HNO_(3)` |
|
| 46. |
The solubility product of AgCl is 2.8xx10^(-10) at 298 K. Calculate the solubility of AgCl in (i) pure water (ii) 0.1M AgNO_(3) solution, and (iii) 0.1M HCl solution. |
|
Answer» Solution :(i) `K_(sp)" of "AGCL=underset(s)([Ag^(+)])underset(s)([Cl^(-)])` `therefore` Solubility `S=sqrt(K_(sp))=sqrt(2.8xx10^(-10))=1.67xx10^(-5)"mol "DM^(-3)` (ii) `AgCl_((s))hArrAg^(+)(aq)+Cl^(-)(aq)` `underset(0.1M)(AgNO_(3))tounderset(0.1M)(Ag^(+))+underset(0.1M)(NO_(3)^(-))` `therefore` Solubility of AgCl in the pressure of 0.1M `AgNO_(3)=K_(sp)/c=(2.8xx10^(-10))/0.1` `=2.8xx10^(-9)"mol "dm^(-3)` (iii) `AgCl_((s))hArrAg^(+)(aq)+Cl^(-)(aq)` `underset(0.1M)(HCL)toH^(+)+underset(0.1M)(Cl^(-))` `therefore` Solubility of AgCl in the pressure of 0.1M HCl = `K_(sp)/c` = `(2.8xx10^(-10))/0.1` `=2.8xx10^(-9)" mol "dm^(-3)` |
|
| 47. |
Which of the following are non-polar molecules ? I. NCl_(3) II SO_(3) III. PCl_(5) |
|
Answer» Ionly 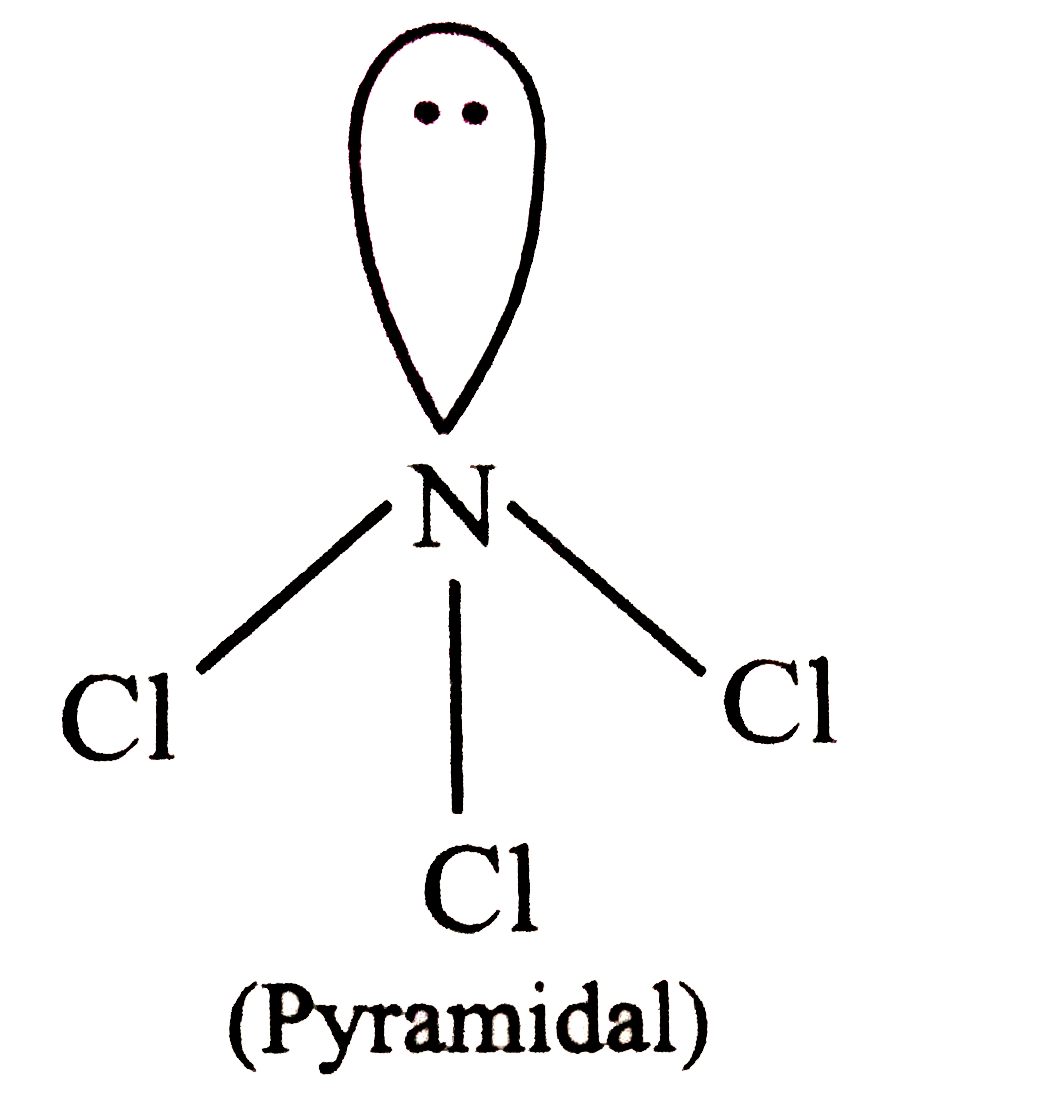 Electronegativity of N and Cl are EQUAL (=3.0). Hence, all the N-Cl BONDS are non polar. 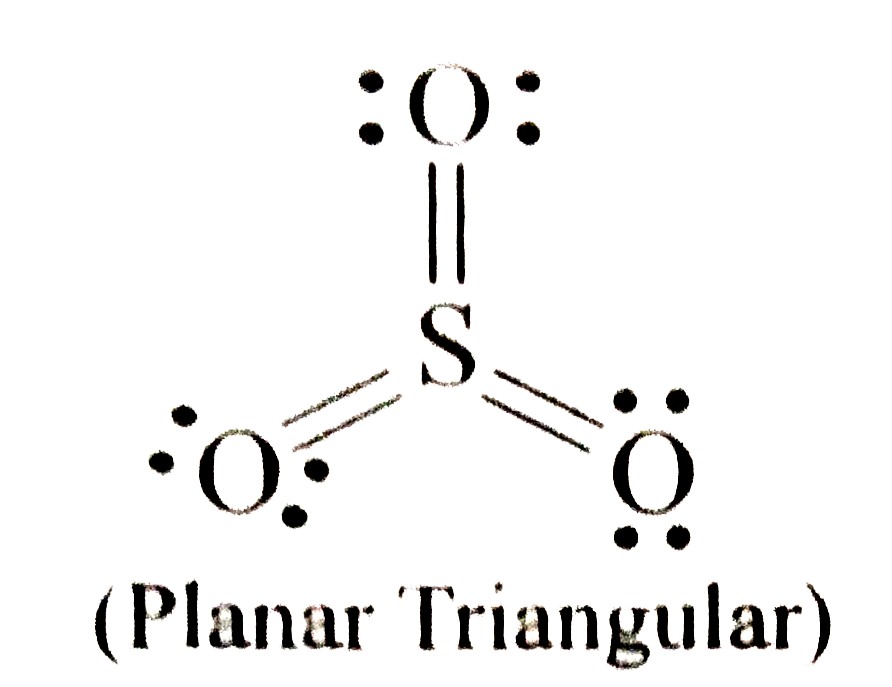 It is SYMMETRICAL . Hence, its DIPOLE moment is ZERO, i.e., it is non-polar . In `PCl_(5)` . bond moments do not cancel out. Hence, is is polar |
|
| 48. |
What are the techniques to reduce particulate pollutants? |
| Answer» Solution :The particulates from AIR can be removed by USING lectrostatic PRECIPITATORS. gravity setling chambers, wet scrubbers or by cyclone collectors. These techniques are base on washing away or settling of the PARTICULATE matter. | |
| 49. |
Which of the following substance on heating liberates two different gases which can be attracted into the applied magnetic field |
|
Answer» `LiNO_(3)` |
|







