Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Using Slater's rule calculate the effective nuclear charge on a 3p electron in aluminium and chlorine .Explain how these results relate to the atomic radii of the two atoms. |
Answer» Solution :`ubrace(Al^(13) 1s^(2))_((N-2)) ubrace(2s^(2) 2P^(6))_((n-1))ubrace(3s^(2) 3p^(1))_((n))` 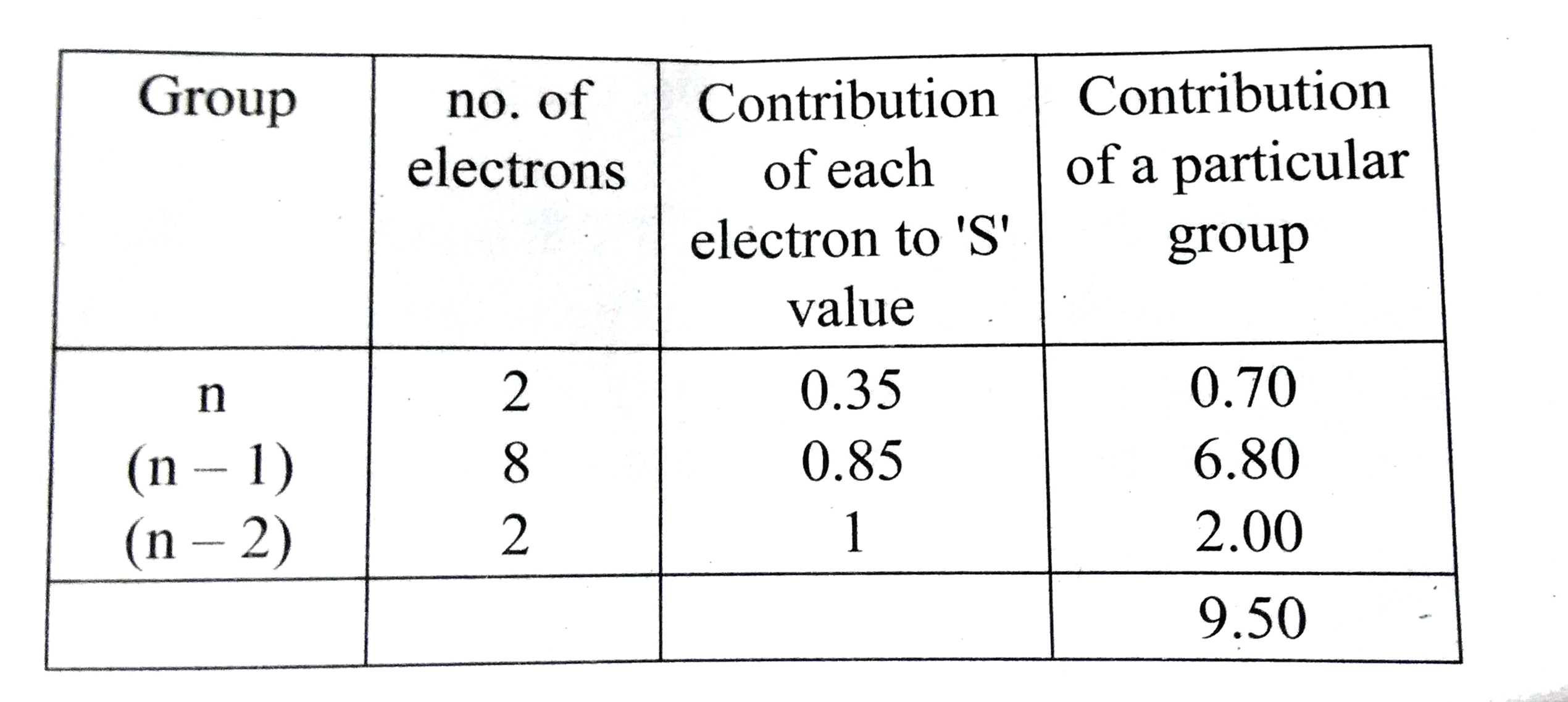 `:.` EFFECTIVE nuclear change =Z-S=13-9.5 `(Z_(eff))_Al=3.5` Electronic CONFIGURATION of CHLORINE `ubrace(1s^(2))_((n-2)) ubrace(2s^(2) 2p^(6))_((n-1)) ubrace(3s^(2) 3p^(5))_((n))` 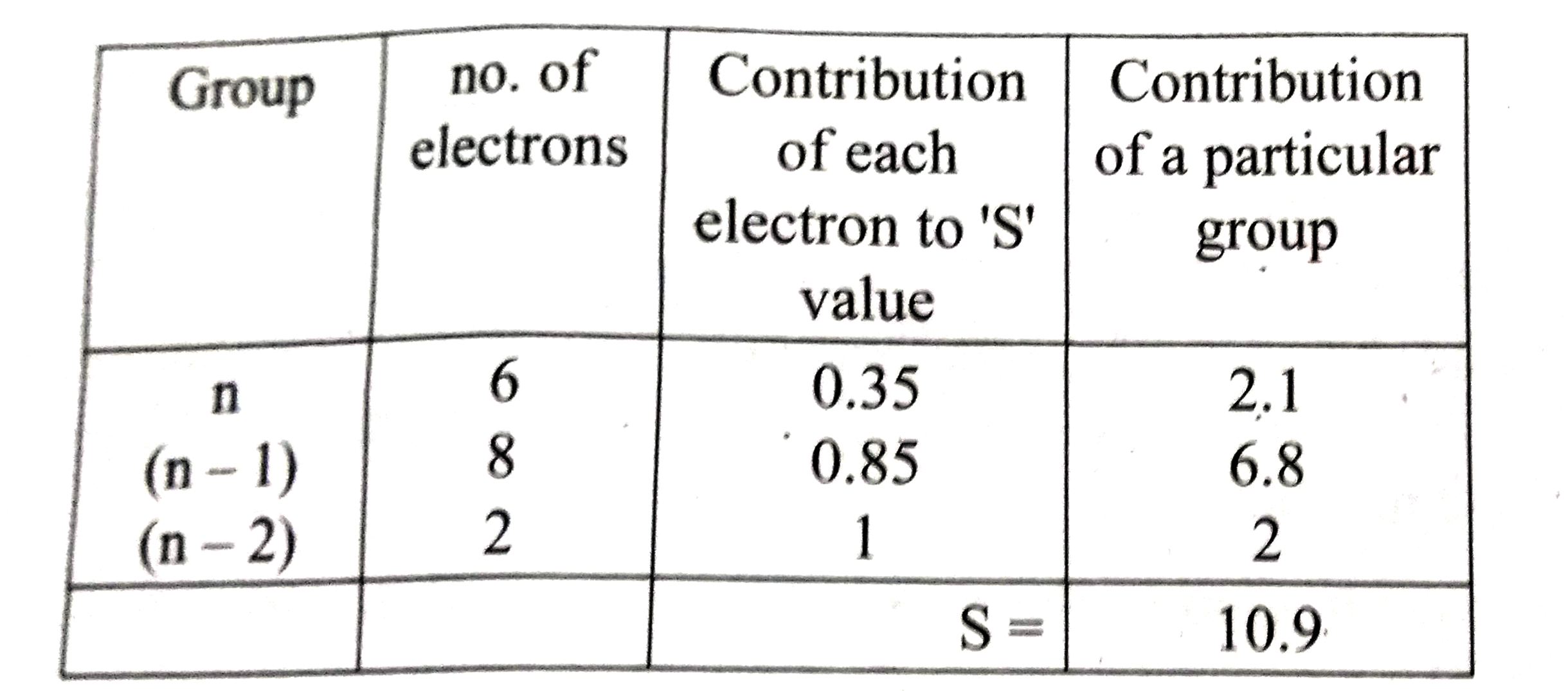 `:.` Effective nuclear charge =Z-S=17=10.9 `(Z_(eff))_Cl=6.1` `(Z_(eff))_Clgt(Z_(eff))_Al` and hence `r_(Cl) ltr_(Al)` |
|
| 2. |
Which of the following reacts with ammonical cuprous chloride? |
|
Answer» `CH_(4)` |
|
| 3. |
Which of the following reactions does not show the acidic nature of ethyne? |
|
Answer» Acetylene reacts with SODAMIDE to form sodium acetylides. |
|
| 4. |
what is Ideal gas equation ? Derive equation of ideal gas and give characteristic of R and its value. |
|
Answer» Solution :Ideal gas equation : The equation get through the COMBINATION of Boyle.s, Charle.s and Avogadro.s law is called ..Ideal Gas Equation... Ideal gas equation : pV = nRT …..(Eq. -i) Ideal gas equation is a relation between four variables (p, V, n, R). It describe the state of any gas, so it is called equation of state. Derivation the equation of ideal gas : (i) Boyle.s law : `V prop (1)/(p)` ....(constant T and n) (II) Charles. Law : `V prop T` ....(constant p and n) (iii) ASvogadro Law : `V prop n` ....(constant p and T) Thus, `V prop (nT)/(p)""`....(Eq. -i) Id standard constant = R, `therefore V =R((nT)/(p)) ""` ....(Eq. - ii) Thus, [pV = nRT] This (Eq. -i) is know as ideal gas equation. Characteristic of gas constant and value R : R is known as gas constant. Value of R is same for each gases. So it is known as UNIVERSAL gas constant. Value of R depends upon the measurements unit p, V and T. Calculation of R value :Volume of one mole of an ideal gas under STP conditions (273.15 K temperature and 1 bar pressure) is 22.710981 L `mol^(-1)`. Value of R for 1 mole of an ideal gas can be calculated under these conditions as follows : where, p = 1 bar `= 10^(5)` PA V = mole volume `= 22.71 mol^(-1) = 22.71xx10^(-3)m^(3)` n = 1 mol,T = 273.15 K pV = nRT.....(according to Eq. -5.24) `therefore R=(pV)/(nT) ""`...(Eq. -5.26) `therefore R=((10^(5)Pa)(22.71xx10^(-3)m^(3)))/((1mol)(273.15 K))` `= 8.3141 Pa m^(3)mol^(1)K^(-1)` `therefore R = 8.3141xx10^(-2)` bar L `K^(-)mol^(-1)` `R=8.314 J K^(-1)mol^(-1)` At STP conditions `0^(@)C` (273.15 K) and 1 atm. pressure value of R is as under. `R = 8.20578xx10^(-2)` L atm `K^(-1)mol^(-1)` = 0.082 L atm `K^(-1) mol^(-1)` |
|
| 5. |
When few drops of glycerol is put on powdered KMnO_(4) |
|
Answer» it becomes colourless |
|
| 7. |
When some of Si^(+4)in frame work silicates are replaced by Al^(+3)and an additional metal ion, it results in the formation of |
|
Answer» ZEOLITES |
|
| 8. |
Why are boron halides and diborane referred to as 'electron deficient compounds' ? |
| Answer» SOLUTION :In these compounds, boron ATOM has an incomplete OCTET. | |
| 9. |
What is the equivalent weight of oxalate ion C_(2)O_(4)^(-2) as reducant? |
|
Answer» SOLUTION :`Cr_(2)O_(4)^(-2) to 2CO_(2)+2E^(-)` Equivalent weight of `C_(2)O_(4)^(-2)ion`= `=("Formula weight of "C_(2)O_(4)^(-2))/("No. of electrons involved in the reaction")=(88)/(2)=44` Equivalent weight of `Na_(2)C_(2)O_(4).2H_(2)O=("Mol.Wt. of"Na_(2)C_(2)O_(4).2H_(2)O)/(2)=(175)/(2)=85` |
|
| 10. |
What is Vander Waal's force ? |
| Answer» Solution :It is a weak INTER ATOMIC or inter molecular FORCE of attractions existing in substances without FORMATION of any CHEMICAL bond. | |
| 11. |
Which of the following molecules represents the order of hybridisation sp^(2), sp^(2), sp, sp from left to right atoms? |
|
Answer» `CH_(3)- CH= CH-CH_(3)` |
|
| 12. |
What are silicones? |
|
Answer» |
|
| 13. |
Which of the following molecules have (-O-O-) bund i.e., peruxide bond? |
|
Answer» `H_(2)S_(2)O_(8)` |
|
| 14. |
Which of the following reactions can produce R - CO - Ar? |
|
Answer» `ArCOCl + H - AR overset(AlCl_(3))rarr` |
|
| 15. |
Whyalknenes are more reactive than alkanes |
|
Answer» SOLUTION :(i)ALKENES are movereactivethan alkanesdue to thepressureof a DOUBLEBOND (ii)The`alpha`- BONDIS stronggut the`pi-` bondis weakthe typicalreactionsof alkenesinvolvetheadditionof anelectrophilcproccedthroughfree-radicalmechanism alos .OZONOLYSISAND polymerisationare someof thecharacteristicreactionof alkenes |
|
| 16. |
What is the maximum number of electrons that can be associated with the following set of quantum numbers ? n = 3, I = 1 and m = -1 |
|
Answer» 25 |
|
| 17. |
Which one of the alkali metals forms only the normal oxide, M_(2)O on heating in air ? |
| Answer» Answer :A | |
| 18. |
What is the maximum concentration of equimolar solutions of ferrous sulphate and sodium sulphide so that when mixed in equal volumes, there is no precipition of iron sulphide ? (For iron sulphide , K_(sp)=6.3xx10^(-18)). |
| Answer» SOLUTION :`5 xx 10^(-9) MOL L^(-1)` | |
| 19. |
Which of the following methods cannot be used to prepare propane? |
|
Answer» REACTION of isopropylmagnesium BROMIDE with water |
|
| 20. |
Write the equation involved in the preparation of hydrogen peroxide from 2 - ethylanthraquinol. |
Answer» SOLUTION :`H_(2)O_(2)` can be PREPARED by autoxidation of ALKYL anthraquinol. 
|
|
| 21. |
Which electrophile does carry out the nitration of benzene? |
| Answer» SOLUTION :`NO_2^+` | |
| 22. |
When would the wavelength associated with an electron become equal to that with proton ? (Mass of electron = 9.10 xx 10^(-31) kg, Mass of proton =1.6725 xx 10^(-27)kg) |
|
Answer» Solution :Wave LENGTHS of electron and proton are `lambda_(c)=(h)/(m_(e)v_(e)).lambda_(p)=(h)/(m_(p)v_(p))" But "lambda_(e)=lambda_(p)` THEREFORE, `(h)/(m_(e)v_(e))=(h)/(m_(p)v_(p)) or m_(e)v_(e)=m_(p)v_(p)` `V_(e)=(m_(p)v_(p))/(m_(e))=(1.6727xx10^(-27)kg)/(9.10xx10^(-31)kg)xxv_(p)=1837v_(p)` `"Velocity of electron "=1837xx" velocity of proton."` |
|
| 24. |
What isquantummechanics? |
| Answer» SOLUTION :Thebranchof SCIENCETHAT takeintoaccuntthe DUALBEHAVIOUR of MATTERIS calledquantummechanics. | |
| 25. |
What mass of K_(2)Cr_(2) O_(7) is required to produce 5.0 litre CO_(2) at 75^(@)C and 1.07 atm pressure from excess of oxlaic acid? Also report the volume of 0.1N NaOH required to neutralise the CO_(2) evolved. |
|
Answer» |
|
| 26. |
What is Grignard reagent? How is it prepared from ethyl bromide? |
|
Answer» Solution :When a.solution of haloalkane in either is treated with magnesium, we will get alkyl magnesium halide known as GRIGNARD reagent, ethyl magnesium bromide is PREPARED from ethyl bromide as: `CH_(3) underset("Ethyl bromide") (- CH_(2) Br) + Mg OVERSET("dry ETHER") (to) underset("Grignard reagent") underset("(Or)") underset("Ethyl magnesium bromide") (CH_(3) CH_(2) Mg Br)` |
|
| 27. |
The substances, which do NOT act as catalyst but when added to reaction increase the activity of the catalyst are called ________. |
|
Answer» inhibitors |
|
| 28. |
The set of gases causing green house effect is |
|
Answer» `CO_(2),CO,SO_(2),N_2` |
|
| 29. |
Which of the following compounds may give white precipitate or turbidity with dilute HCI? |
|
Answer» `Pb(NO_(3))_(2)` |
|
| 30. |
Which of the following compounds contains only sp^(3)-hybridized carbons ? |
|
Answer» Cycloalkanes |
|
| 31. |
Water destroysgrignard reagents why? |
|
Answer» SOLUTION :`CH_(3)MGX= HOH toCH_(4)+ Mg (OH) X` Waterwouldprotonatethe grignardreagent and destroythe grignardregentbecausethegrignardcarbonatomis highlynucleophilic.thiswouldforma hydrocarbon.Thereforetogrignardsolutiononlyetheris thebestsolventand WATEROR alcoholare notusedforthatpurpose. |
|
| 32. |
What type of crystal defect is produced when sodium chloride is doped with MgCl_2 ? |
| Answer» Solution :It is called impurity defect. A cation vacancy is produced . A substitutional SOLID solution is formed (because 2 `Na^+` IONS are replaced by one `Sr^(2+)` ion in the lattice site. ) | |
| 33. |
Which of the following have the same number of electrons in outermost shell? |
|
Answer» Elements with ATOMIC numbers 30, 48, 80 |
|
| 34. |
What isquantummechanics?giveitsprimaryinformation . |
|
Answer» Solution :Quantummeachanicsis ABRANCH of science . It takesintoaccountthisdualbehaviourof matterAnd it isbasedon theideasof wavemotion. Quantum mechanics wasdevelopedandErwinSchrodinger.Thefoundamentalby Schrodingerand it won himthe NobelPrizein PHYSICSIN 1933. ThisSchrodingmechanicsequation is quatecomplexand knowledgeof highermathematicsis neededto solve it. Totalenergy( E) : The totalenergyof the systemtakesintoaccountthe kineticenergy of all thesub- atomicparticlesatractivepotentialbetweente electronsand nucleiindividually . It isknownas wavefunctionof ATOM anddoesnotcarryanyphysicalmeaning . Itcorrespondingto allenergystate. |
|
| 35. |
underset(("Dark purple almost black"))((A)) overset(Delta)rarr underset(("Green"))((B))+underset(("Black"))((C))+(D) Find the number of correct statements: (a) Compound B is K_(2)MnO_(4) (b) Compound C is MnO_(2) (c) Compound D is O_(2) (d) Compound B is paramagnetic in nature (e) Compound D has two unpaired electrons in bonding molecular orbital |
|
Answer» |
|
| 36. |
Which of the following substances acts as an oxidising as well as a reducing agent |
|
Answer» `Na_(2)O` Oxidising property: `NaNO_(2)+2KI+2H_(2)SO_(4)toNa_(2)SO_(4)+K_(2)SO_(4)+2NO+2H_(2)O+I_(2)` Reducing Property : `H_(2)O_(2)+NaNO_(2)toNaNO_(3)+H_(2)O` |
|
| 38. |
Which of the following expression is wrong? |
|
Answer» `[H^(o+)] = [OVERSET(Theta)OH] = 10^(-7)` for a neutral solution at all TEMPERATURES. |
|
| 39. |
Which quantum number reveal information about the shape,energy,orientation and size of orbitals? |
| Answer» SOLUTION :Magneticquantum number reveal INFORMATION about the SHAPE,ORIENTATION and size of orbitals. | |
| 40. |
What is the meaning of VSEPR ? |
|
Answer» <P> Solution :V = ValenceS = SHELL E = Electron P = Pair R = Repulsion i.e., VSEPR MEANS (valence shell electron pair repulsion theory) |
|
| 41. |
Which one in each of the following pairs is expected exhibit hydrogen bonding together? a.CH_(3)-CH_(2)-OH and CH_(3)-O-CH_(3) b. CH_(3)NH_(2) and CH_(3)SH c. CH_(3)OCH and (CH_(3))_(3)N |
|
Answer» Solution :a. `CH_(3)-CH_(2)OH` and H connected to O atoms. b. S does not form HYDROGEN BONDS. c.Both do not form hydrogen bonds because H atom is absent on O and on N. |
|
| 42. |
Which of the following hydrocarbons givesame product on hydrogenation. |
|
Answer» 2-methyl hex-1-ene & 3-Methyl hex-3 -ene |
|
| 43. |
What is the important of Periodic table ? OR What is Periodic table according to Glenn T-Seaborg? |
|
Answer» Solution :The periodic table is arguably the most important concept in chemistry, both in principle and in practice. It is the everyday support for students, it SUGGESTS new avenues of research to professionals and it PROVIDES a succinct organization of the whole of chemistry. It is a remarkable demonstration of the fact that the chemical elements are not a random cluster of ENTITIES but instead display trends and lie together in families. An awareness of the periodic table is essential to anyone who WISHES to DISENTANGLE the world and see how it is built up from the fundamental building blocks of the chemistry, the chemical elements. |
|
| 44. |
What are pesticides and herbicides ? Explain giving examples. |
|
Answer» Solution :Pesticides are a combination of two or more SUBTANCES. It is useful to DESTROY other insects like locusts and pests. e.g., ALDRIN and Dieldrin anesthesizers are substance used to destroy weeds. e.g., Sodium CHLORATE : `NaClO_3` Sodium Arsenate : `NO_3 ASO_3` |
|
| 45. |
Which elements are normally not detected in an organic compound ? |
| Answer» SOLUTION :CARBON & HYDROGEN. | |
| 46. |
Write the halogenation of alkene. |
Answer» Solution :(i) Common reaction : here there is breaking of `pi`-bond, Halogen is attached to the CARBON with `SIGMA`-bond. <BR>  (ii) Type of reaction : Alkene reaction with `X_(2)` (X=Cl or Br) vicinal dihalide is formed. This reaction is called electrophilic addition reaction. In this reaction unsaturated hydrocarbon is CONVERTED to saturated dihalide. Halonium ion is formed between the reaction and used as electrophilic addition reaction. (iii) Alkene on chlorine `(Cl_(2))` and bromine `(Br_(2))` give addition product and forms vicinal dihalide and vicinal dibromide. Example (a) Chlorination : `underset("Ethene")(CH_(2)=CH_(2))+underset("Dichlorine")(Cl-Cl) overset("Chlorination")RARR underset("1,2-dichloro propane")(underset(Cl)underset(|)(CH_(2))-underset(Cl)underset(|)(CH_(2)))` `CH_(3)CH_(2)=CH_(2)+Cl-Cl overset("Chlorination")rarr underset("1,2-dichloro propane")(CH_(3)-underset(Cl)underset(|)(CH_(2))-underset(Cl)underset(|)(CH_(2)))` Example - (b) unsaturation test (Bromination) : `underset("Ethene")(CH_(2)=CH_(2)) + underset("Bromine")underset("orange")underset("Red-")(Br_(2)) underset("Bromination")overset(C Cl_(4))rarr underset("1,2-dibromoethane")(underset(Br)underset(|)(CH_(2))-underset(Br)underset(|)(CH_(2)))` `underset("Propene")(CH_(2)=CH_(2)) + underset("Bromine")underset("orange")underset("Red-")(Br_(2)) underset("Bromination")overset(C Cl_(4))rarr underset("1,2-dibromoethane")(CH_(3)-underset(Br)underset(|)CH-underset(Br)underset(|)(C)H_(2))` When product forms red-orange, colour of bromene is removed and solid addition product is obtained. Bromination reaction is unsaturation test of alkene. |
|
| 47. |
Thoseelementsimpartcolour to theflame on heating init , theatom ofwhichrequire lowenergyfor the ionisation(i.e.,absorb energy inthe visibleregion ofspectrum ). Theelementsof which of thefollowinggroupswillimpart colour ot theflame ? |
|
Answer» 2 |
|
| 48. |
Which of the following statement regarding catalyst is not true:- |
|
Answer» A catalyst REMAINS unchanged in composition and quantity at the END of the reaction |
|
| 49. |
What are the major differences between metals and non-metals ? |
Answer» SOLUTION :
|
|