Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
Which branch is not included in the branches of chemistry ? |
|
Answer» Organic CHEMISRTY |
|
| 3. |
Which of the following statements is/are correct ?(1) Two organic compounds with the samse general formula must belong to the same homologous series.(2) Two organic compound with one of the functional groups the same must belong to the same homologous series.(3) Two organic compounds with the molecular mass differing by 14 must belong to the same homologous series. |
|
Answer» 1 only |
|
| 4. |
What weight of magnesia is obtained by complete combustion of two grams of metal? |
|
Answer» SOLUTION :The BALANCED EQUATION for the combustion of magnesium METAL is `2Mg+O_(2) to 2MgO` 2 moles of Mg =2 moles of MGO 1 mole of Mg =1 mole of MgO 24 grams of Mg =40 grams of MgO The weight of magneisa (MgO) produced by burning 2 grams of metal `=(40)/(24) xx 2=3.333gr` |
|
| 5. |
Which of the following alkaline earth metal sulphates is least soluble in water ? |
|
Answer» `BaSO_(4)` |
|
| 6. |
When electromagnetic radiation of wavelength 300 nm falls on the surface of sodium, electrons are emitted with a kinetic energy of 1.68 xx 10^(5)J mol^(-1). What is the minimum energy needed to remove an electron from sodium ? What is the maximum wavelength that will cause a photoelectron to be emitted? |
|
Answer» Solution :Energy of a photon of radiation of wavelength 300 nm will be `E = hv = h (c)/(lamda) = ((6.626 xx 10^(-34) Js) (3.0 xx 10^(8) ms^(-1)))/((300 xx 10^(-9) m)) = 6.626 xx 10^(-19) J` `:.` Energy of 1 mole of PHOTONS `= (6.626 xx 10^(-19)J) xx (6.022 xx 10^(23) mol^(-1)) = 3.99 xx 10^(5) J mol^(-1)` As `E = E_(0) +` K.E. of photoelectrons emitted. `:.` Minimum energy `(E_(0))` required to remove 1 mole of electrons from sodium `= E -` K.E. `= (3.99 - 1.68) 10^(5) J mol^(-1) = 2.31 xx 10^(5) J mol^(-1)` `:.` Minimum energy required to remove one electron `= (2.31 xx 10^(5) J mol^(-1))/(6.022 xx 10^(23) mol^(-1)) = 3.84 xx 10^(-19) J` The wavelength corresponding to this energy can be calculated using the formula, `E = hv = h (c)/(lamda)` `:. lamda = (hc)/(E) = ((6.626 xx 10^(-34) Js) (3.0 xx 0^(8) ms^(-1)))/(3.84 xx 10^(-19)J) = 5.17 xx 10^(-7) m = 517 xx 10^(-9) m = 517 nm` which CORRESPONDS to the green light. As `lamda prop (1)/(E)`, hence when E is minimum, `lamda` is maximum |
|
| 7. |
which of the most thermally stable carbonate among MgCO_3,CaCO_3,SrCO3and BaCO3? |
| Answer» SOLUTION :`BaCO_3`(DUE to GREATER ELECTROPOSITIVITY of BA) | |
| 8. |
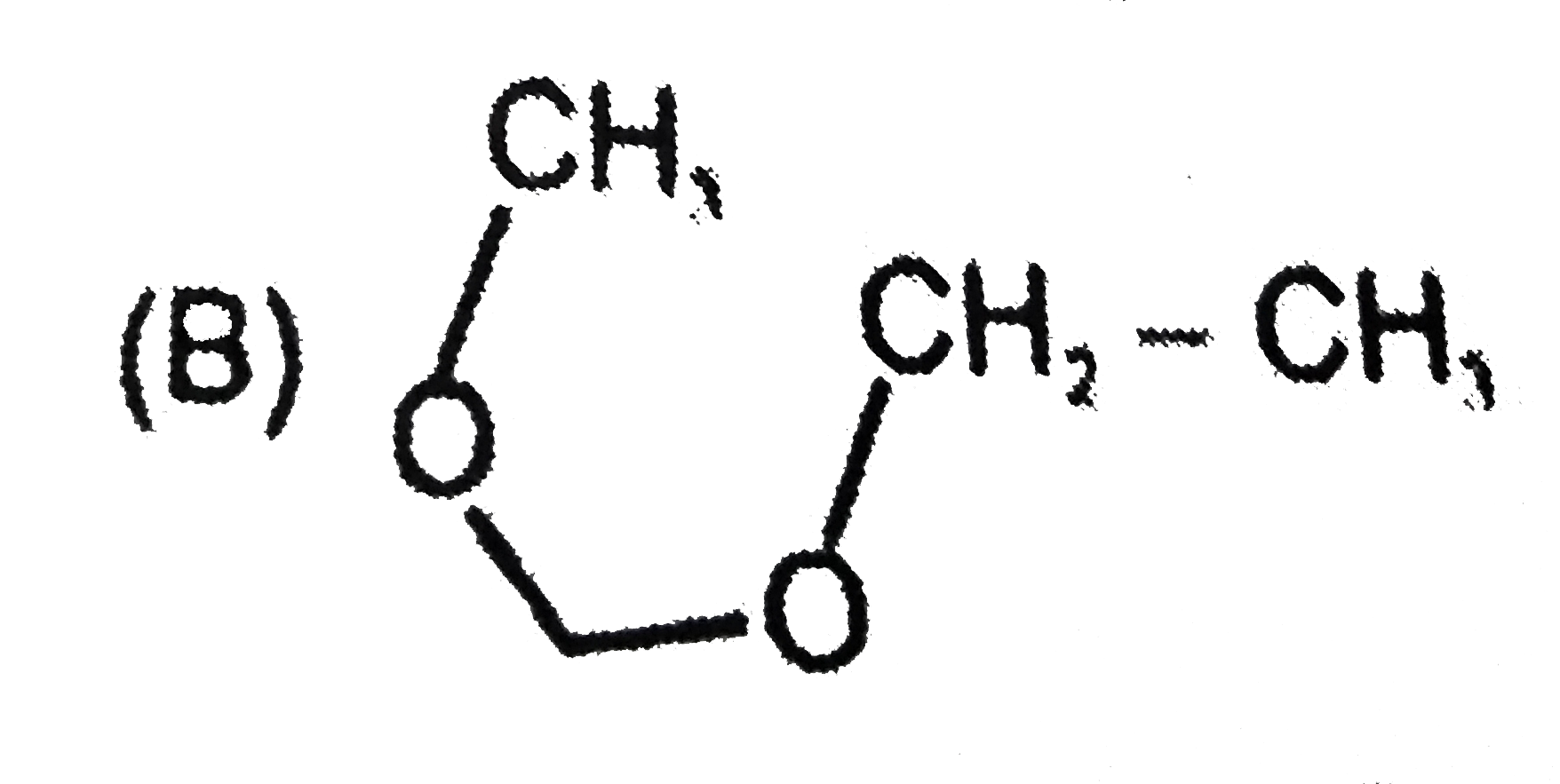
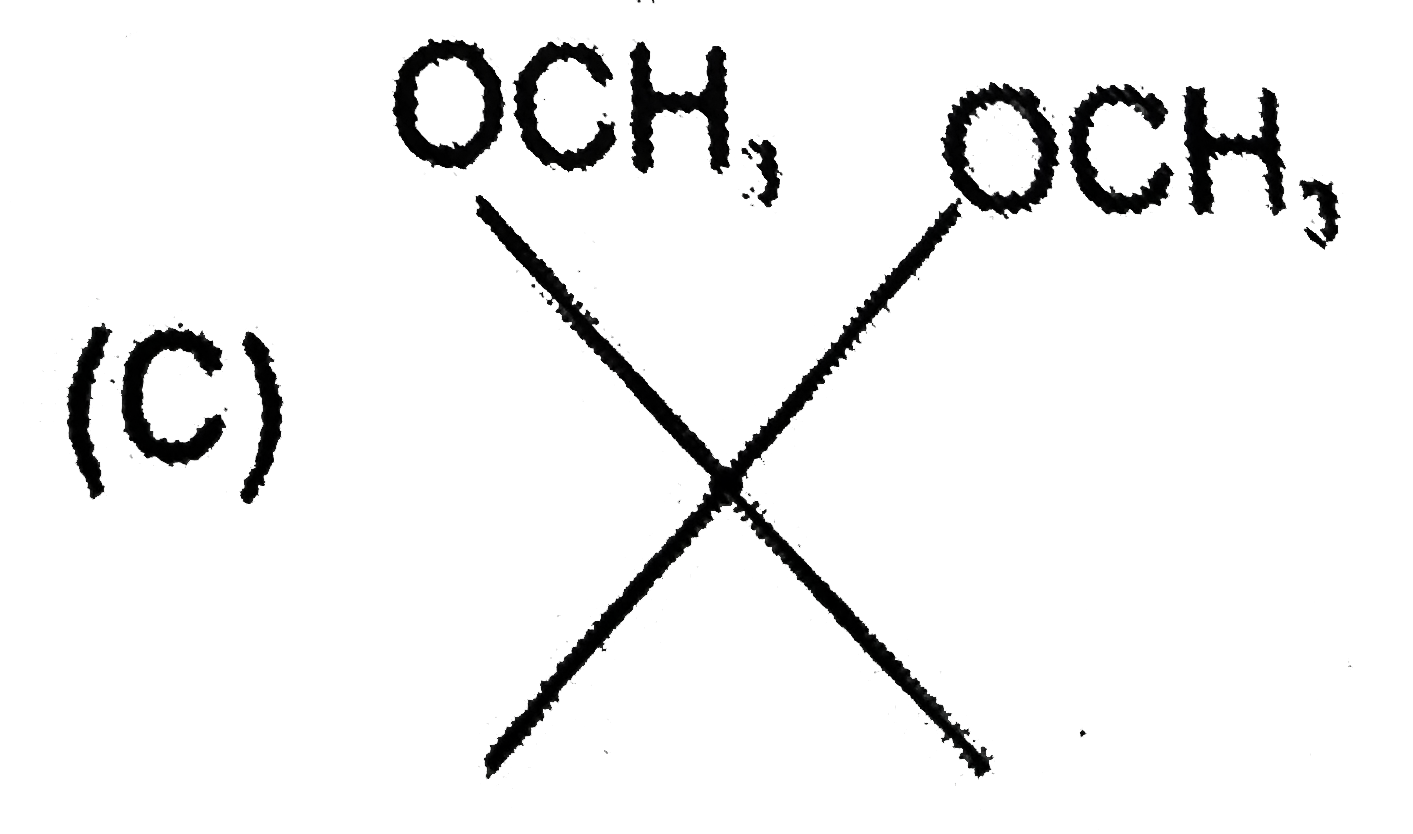
Which of the following compounds do not exhibit steroisomerism |
|
Answer»

|
|
| 9. |
The unimolecular nucleophilic substitution is represented by : |
|
Answer» (II),(III),(Q) |
|
| 10. |
Which one of the following is correct about stability of the given ions |
|
Answer» `PB^(2+) gt Pb^(4+)` |
|
| 11. |
What is the normality of 1 M aq solution of H_(3)PO_(4) ? |
|
Answer» 1 N |
|
| 12. |
Which one of these is not true for benzene ? |
|
Answer» it FORMS onlye one TYPE of monosubstituted product 
|
|
| 13. |
Which is more polar -CO_(2) or N_(2) O? Givereason . |
|
Answer» Solution :`N_(2) O ` is more polar than `CI_(2)` . This is because `CO_(2)`is linear and symmetrical . Its net DIPOLE moment is zero `(Ooverset(larr)=Coverset(rarr)(=O)) . N_(2) O`is linear but unsymmetrical .It is considered as a reasonance hybrid of the FOLLOWING two STRUCTRURE : `:OVERSET(-)overset(. .)N= overset(+)N= overset(. .)O: harr :N-=overset(+)N-overset(-)overset(. .)underset(. .)O:` It has a net dipole moment of 0.116 D. |
|
| 14. |
Using the Periodic Table, predict the formulas of compounds which might be formed by the following pairs of elements, (a) silicon and bromine (b) aluminium and sulphur. |
|
Answer» Solution :(a) Silicon and Bromine : Silicon is group 14 element with a valence of 4, bromine belongs to the halogen family with a valence of 1. Hence the formula of the COMPOUND formed would be `SiBr_(4)`. (b) Aluminium and Sulphur : Aluminium belongs to group 13 with a valence of 3, sulphur belongs to group 16 elements with a valence of 2. Hence, the formula of the compound formed would be `Al_(2)S_(3)`. |
|
| 15. |
Which of the following gives CH_(3)-OH and CH_(3)-overset(O)overset(||)(C)-CH_(3) on hydrolysis with H_(3)O^(+). |
|
Answer»
|
|
| 16. |
Which of the following statement is not true about the hexagonal close packing ? |
|
Answer» The coordination number is 12. |
|
| 17. |
Which mixture of gases at room temperature does not obey Dalton's law of partial pressure? |
|
Answer» `NO_(2)andO_(2)` |
|
| 18. |
Which effect is observed only below a certain temperature? |
|
Answer» Joule-Thomson effect |
|
| 19. |
The reagent(s) that can selectively precipitate S^(2-) from a mixture of S^(2-) and SO_(4)^(2-) in aqueous solution is (are): |
|
Answer» `CuCI_(2)` |
|
| 20. |
Yield of Ammonia will be more in Haber's process under _____ conditions (L = Low, H = high T = Temp, p = Pressure ) |
|
Answer» LT , LP |
|
| 21. |
Which one is easy, safe and reliable method to make water germ free ? |
|
Answer» by passing `Cl_2` GAS |
|
| 22. |
Which are heterogenous equilibrium ?state the unit of K_c. (i)H_2O_((l)) hArr H_2O_((g)) (ii)Ca(OH)_(2(s)) + aq hArr Ca_((aq))^(2+) + 2OH_((aq))^(-) (iii)CaCO_(3(s)) hArr CaO_((s)) + CO_(2(g)) (iv)CH_3COOC_2H_(5(l)) + H_2O_((l)) hArr CH_3COOH_((l)) + C_2H_5OH_((l)) (v)Fe_((aq))^(3+) + SCN_((aq))^(-) hArr [Fe(SCN)]_((aq))^(2+) |
|
Answer» Solution :(i)`K_c=[H_2O]="MOL L"^(-1)` (ii)`K_c=[Ca^(2+)][OH^-]^2 ("mol L"^(-1))^3` (iii) `K_c=[CO_2] "mol L"^(-1)` (iv)`K_c=([(CH_3COOH)][(C_2H_5OH)])/([CH_3COOC_2H_5][H_2O])` no UNIT (V)`K_c=([Fe(SCN)^(2+)])/([Fe^(3+)][SCN^(2+)])` Thus unit of `K_c` is `("mol" L^(-1))^(-1)` or L `mol^(-1)` |
|
| 23. |
Which one of the following electrophile used for nitration of benzene? |
|
Answer» `BR^(OPLUS)` |
|
| 24. |
Which of the following ions is the most resonance stabilised? |
|
Answer» Ethoxide |
|
| 25. |
Valence electron in theelementA are 3and that inelementB are6. Mostprobablecompound formedfrom A and Bis |
|
Answer» `A_(2) B` |
|
| 26. |
What type of difference is observed in hybridization of carbon of benzene during nitration and formaiton of 6 complex ? |
| Answer» SOLUTION :Intermediate `SIGMA`-complex is formed in which one carbon POSSESS `sp^(3)` hybridization and rest all carbon possess `sp^(2)` hybridization. | |
| 27. |
Which of the following does not exhibit tautomerism ? |
|
Answer»
|
|
| 28. |
Which of the following acts as Lewis base? |
|
Answer» `SCN^(-)` |
|
| 29. |
Which one of the following reagents distinguish ethylene from acetylene? |
|
Answer» A. Aqueous alkaline `KMnO_4` |
|
| 30. |
What is the oxidation number of carbon in diamond ? |
|
Answer» `+2` |
|
| 31. |
Which fungi is known as insecticide ? |
|
Answer» TRICHODERMA |
|
| 32. |
The values of DeltaH and DeltaS for the reaction, C("graphite") + CO_(2)(g) to 2CO(g) are 170 kJ and 170 J/K respectively. This reaction will be spontaneous at |
|
Answer» 910 K Here, `DeltaG`will be negative when `T DELTAS gt DeltaH` `DeltaH = 170 kJ = 170000`J `DeltaS - 170 J K^(-1)"mol"^(-1)` `TDeltaS gt DeltaH` only when ` T = (DeltaH)/(DeltaS) = (170000)/(170) = 1000K` i.e greater than 1000 K or 1110 K. |
|
| 33. |
What is catenation? Describe briefly the catenation property of carbon. |
| Answer» Solution :The tendency of an atom to form a chain of BONDS with the ATOMS of the same element is CALLED CATENATION. The high STRENGTH of C—C bond is responsible for its catenation property. | |
| 34. |
Which one of the following elements does not form triiodide on reacting with iodine? |
| Answer» ANSWER :B | |
| 35. |
Which of the following are the values of electron affinities in kJ/mol for the formation of O^(-) and O^(2-) from O ? |
|
Answer» `-142, -702` |
|
| 36. |
What is the moe fraction of solute in 10% aqueous caustic soda solution? |
|
Answer» |
|
| 37. |
Which of the following processes is most appropriate for the separation of o-nitrophenol and p-nitrophenol from their mixture |
|
Answer» FRACTIONAL crystallisation |
|
| 38. |
The valencies of nitrogen and boron in barazole re |
|
Answer» 3,3 |
|
| 39. |
Which of the following molecules represents the order of hybridisation sp^(2), sp^(2), sp, sp from left to right atoms ? |
|
Answer» `CH_(3)-CH=CH-CH_(3)` |
|
| 40. |
Which of the following compounds reacts with NaHCO_(3)" giving "CO_(2) |
|
Answer»
|
|
| 41. |
Write a note on williamson synthesis. |
|
Answer» Solution :Williamson's synthesis : HELO ALKANES when BOILED with SODIUM alkoxide gives the CORRESPONDING other. `underset("ethenc")underset("Bromo")(CH_(3)-CH_(2))Br+underset("sodium")underset("ethoxide")(CH_(3)CH_(2)ONa)toCH_(3)-CH_(2)-underset("Diethyl ether")(CH_(2)-CH_(3)+)NaBr` |
|
| 42. |
Two substances with the same reduced pressure and the same reduced temperature will have the same reduced volume. This is called |
|
Answer» Gay-Lussac's LAW |
|
| 43. |
When 2-bromobutane reacts with alcoholic KOH, the products formed are ………………… . |
| Answer» SOLUTION :1-butene & 2-butene | |
| 44. |
The stabillity of +5 oxidation state dereases down the group 15 of the periodic table. Explain this observation giving appropriate reasons. |
| Answer» Solution :All the elements of GROUP 15 have five electrons in the valence shell, two in the s-subshell and three in the p- subshell. Therefore, among others, they can show positive oxidation states of +3 (due to pacticipation of only p-electrons) and +5 (due to the participation of both s- and p-electrons of the valence shell). Now N does not have d- or -f -electrons but all other elements have either d-or both d-and f-electrons in the inner shells. Since d-and f-electrons shield the s-electrons of the valence shell more effectively than the p-electrons, therefore on moving down the group tendency of s-electrons to participate in bond FORMATION decreases while that of p electrons increases. As a resulty among hevier elements of group 15, the electron pair in s-orbtial of the valence shell shows reluctance to participate in bond formation. In other words, among heavier elements, only p-electrons participate in bond formation and hence show an oxidation state of +3. This is called inert pair effect. Further, as the NUMBER of d-and f-electrons increases down the group, the inert pair efferct becomes more and more pronounced. In other words, down the group from As to Bi, the stability of +3 oxidation state increases while that of +5 oxidation state decreases. | |
| 45. |
What are unsaturated hydrocarbons |
| Answer» SOLUTION :Hydrocarbonshavinglocalised CARBON- carbon multinglebondsare calledcalledunsaturatehydrocarbons . Examplealkenesalkynes. | |
| 46. |
The synthesis of higher alkyne from lower alkyne is achieved by adding a bromo alkane in to amixture of Sodium amide and an alkyne. Which of the following combination(s) of bromoalkane and alkyne, can lead to the formation of 3-Octyne. |
|
Answer» 1-bromo pentane and propyne |
|
| 47. |
Which is the only isotope that does not contain a neutron? |
|
Answer» Protium |
|
| 48. |
Which of the following statement/s is/are incorrect about H_(2)O_(2) ? |
|
Answer» It trus BLUE litmus RED |
|
| 49. |
What is reversible reaction ? Give examples. |
|
Answer» Solution :A reaction in which not only the reactants react to form the PRODUCTS under certain condition but also the products react to form reactants under the same condition is CALLED a reversible reaction. It is represented by puting (`hArr` ) double arrow. e.G. `H_(2(g)) + I_(2(g)) overset(445^@ C)hArr 2HI_((g))` |
|
| 50. |
Which of the following equations depict the oxidising nature of H_2O_2? |
|
Answer» `2MnO_4^(-) + 6H^(+) + 5H_2O_2 to 2Mn^(2+) + 8H_2O + 5O_2` 
|
|