Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following compounds are readily soluble in water ? |
|
Answer» `BeSO_(4)` Therefore, ` BeSO_(4)` and `MgSO_(4)`are easily SOLUBLE. |
|
| 2. |
What is oxidation number of Pt in [Pt(C_(2)H_(4))Cl_(4)]^(-) ? |
|
Answer» SOLUTION :`Pt+0+4(-1)=-1` `thereforePt-4=-1` `thereforePt=+3` |
|
| 3. |
StateKelvin-Planck statementof secondlawof thermodynamics. |
| Answer» SOLUTION :Kelvin PLANK statement : It is impossible to CONSTRUCT a machine that absorbs heat from a hot source and converts it completely into work by a cyclic process WITHOUT transferring a part of heat to a cold sink. | |
| 4. |
Write the IUPAC names of the following compounds (i) {:(CH_(3)-CH-CH-CH_(3)),("||"),(""C_(2)H_(5) " C"_(2)H_(5)):} (ii) (CH_(3))_(3)"CC"_(2)H_(5) |
Answer» SOLUTION :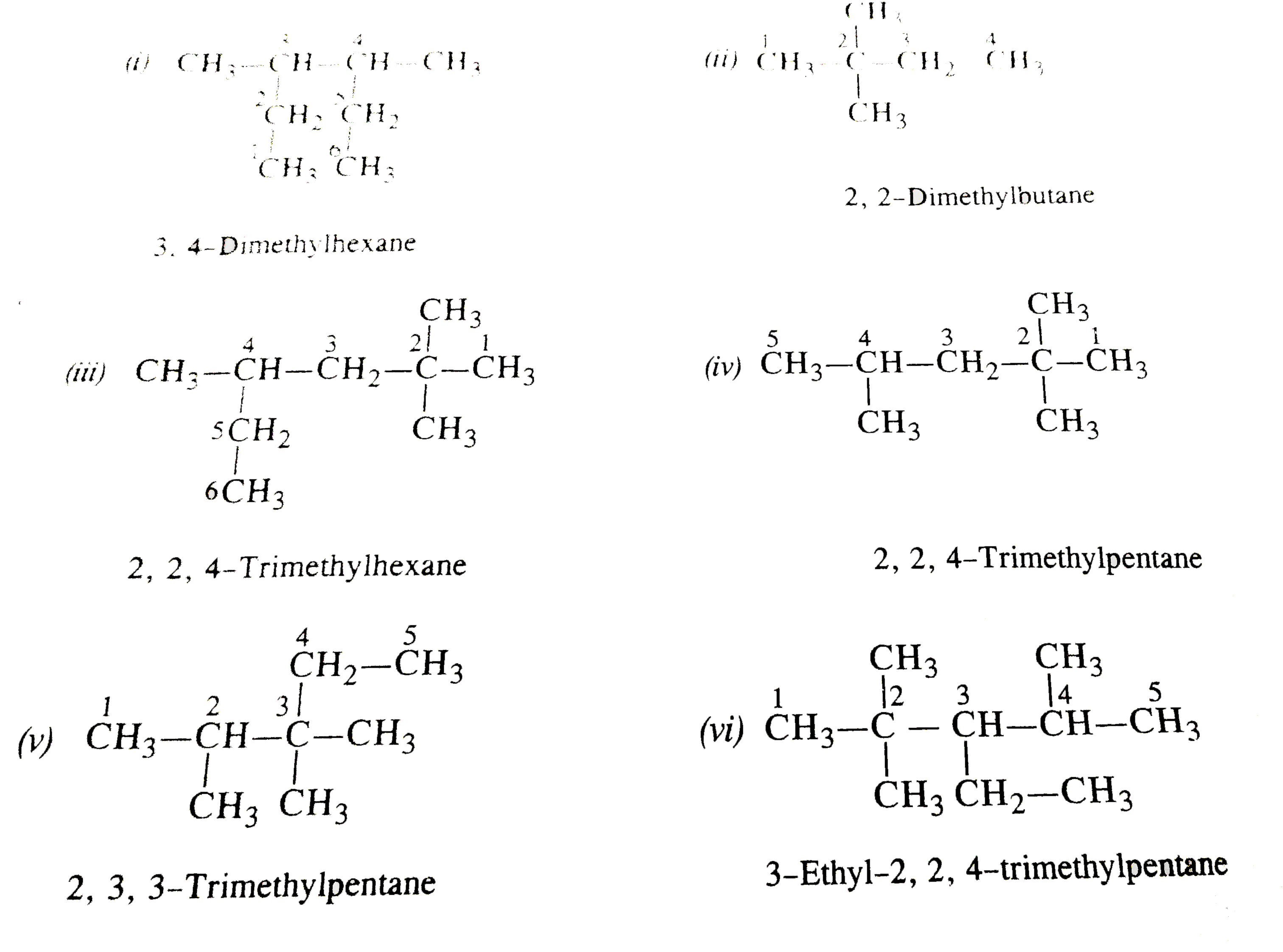 
|
|
| 5. |
whatchangesoccursin energyof Bohr'sorbit ? |
|
Answer» INCREASE awayfromnucleus |
|
| 6. |
What will happen if the equilibrium system is kept in ice bath ? underset"brown"(2NO_(2(g))) hArr underset"colourless"(N_2O_(4(g))), DeltaH=-ve |
|
Answer» COLOUR INTENSITY INCREASES. |
|
| 7. |
Which of the following has the highest molarity ? (i) 4 g of NaOH per 100 cm^3 of water (ii) 4 g of HCI per 100 cm^3 of water (iii) 4 g of H_2SO_4 per 100 cm^3 water |
|
Answer» `w=(MM.V)/1000` (w = weight of solute in grams, M. = gram molecular MASS, V = VOLUME of solution in cm ). `therefore` The molarity of NaOH solution is given by `4=(M xx 39.998 xx 100)/1000` or M=1.000 Similarly for HCI solution, `4=(M xx 36.458 xx 100)/1000` or `M=1.0971` and for `H_(2)SO_(4)` solution, `4=(M xx 98.076 xx 100)/1000` or M = 0.40785 Hence, the HCI solution has the highest molarity. |
|
| 8. |
Which of the reaction does not give quantitative yield of the expected product ? |
|
Answer»
 Maximum YIELD of `93%` is obtained in this reaction. |
|
| 9. |
What are the hybridization and shapes of the following molecules? (i) CH_(3)F (ii) HC-=N |
|
Answer» (i) `sp^(2)`, TRIGONAL PLANAR, (ii) `sp^(3)`, TETRAHEDRAL |
|
| 10. |
Which of the following aromatic compounds undergoes Friedel-Crafts alkylation with methyl chloride and aluminum chloride? |
|
Answer» BENZOIC acid |
|
| 11. |
Which of thefollowingstatements are correct? |
|
Answer» Helium hashighest firstionisationenthalpyin the periodictable. Statement( d) isbecausein any periodnoblegas has thelargestatomicradius . Please notethat inthe NCERT exemplar the answerto thisstatementis given tobe correctwhichis WRONG . |
|
| 12. |
What is average speed (barc or u_(av)) ? |
|
Answer» Solution :It is the arithmatic mean of the SPEED of different MOLECULES of the gas at a given TEMPERATURE if `n_(1)` molecules have speed `v_(1),n_(2)` molecules have speed `v_(2)n_(2)` molecules have speed `V_(3)` and so on. `barC (or U_(av))= sqrt((8RT)/(pi M))` |
|
| 13. |
Which of the following correctly depicts the fact that identical chemical equilibrium can be attained through reversible reaction H_2+I_2=2HI from either direction? |
|
Answer» |
|
| 14. |
Which is correct about tailing of Hg? |
|
Answer» It is DUE to `Hg_2O` `2Hg+O_3rarrHg_(2)O+O_2` `Hg_(2)O+H_2O_2rarrHg_(2)+H_(2)O+O_2` |
|
| 15. |
Which of the following options is correct w.r.t. ideal gas |
|
Answer» <P>`((delH)/(delT))_(P)-((delH)/(delT))_(V)=R` |
|
| 16. |
Which of the following solutions have the same concentration ? |
|
Answer» 20 g of NAOH in 200 mL of solution `= (20xx1000)/(40xx200)=2.5M` (B) `M=(0.5xx1000)/(200)=2.5M` (C) `M=(40xx1000)/(10xx100) = 10M` (D) `M = (20xx1000)/(56xx200) = 1.785 M` HENCE, 20 g NaOH in 200 mL of solution and 0.5 mol of KCl in 200 mL have the same concentration. |
|
| 17. |
Which of the following is the best reducing agent and why? Li,Cu, Br_2, F_2, H_2, K. |
| Answer» SOLUTION :Lithium (Li) is best REDUCING AGENT because it has the lowest STANDARD reduction potential, i.e., `Li^(+)` is the most stable AMONGST these. | |
| 18. |
What are enantiomers? |
|
Answer» Solution :(i) An optically active substance may exist in two or more ISOMERIC forms which have same physical and chemical properties but DIFFER in terms of DIRECTION of rotation of plane POLARISED light, such OPTICAL isomers which rotate the plane polarised light with equal angle but in opposite directions are known as enantiomers and the phenomenon is known as enantiomerism. (ii) Isomers which are non-super imposable mirror images of each other are called enantiomers. |
|
| 19. |
Which of the following oxides can act as amphoteric ? |
|
Answer» `SiO_2` |
|
| 20. |
Which of the following pairs are positive isomers ? |
|
Answer» I and II |
|
| 21. |
Which of the following aromatic compound will undergo nitration most easily? |
|
Answer»
 will UNDERGO NITRATION most EASILY will UNDERGO NITRATION most EASILY
|
|
| 22. |
Which oxidizing agent required to convert sulphur dioxide into sulphur trioxide ? |
|
Answer» DUST |
|
| 23. |
Which of the following oxide is basic ? |
|
Answer» `Na_(2)O` `:. Na_(2)O gt Al_(2)O_(3) gt CO_(2) gt P_(4)O_(10)` |
|
| 24. |
Which of the folllowing attain the linear structrue : |
|
Answer» `Be Cl_(2)` `""^(-)NCO(""^(-)N= C = O )`is linear because it is isoelectronic with `CO_(2)`. `NO_(2)`is angular with angle `132^(@)`and each O-N BOND length of `1.20Å` (intermediate between single and double bond) . |
|
| 25. |
Which gas is released on reaction of alkali metal hydride with water ? |
|
Answer» Oxygen |
|
| 26. |
When the screening effect increases, ionisation energy |
|
Answer» DECREASES |
|
| 27. |
Which ordering of compounds is according to the decreasing order of the oxidationstate of nitrogen ? |
|
Answer» `HNO_3,NO,NH_4,Cl,N_2` |
|
| 28. |
What is orbital? Explainpointand boundarysurfacediagramof orbitals |
|
Answer» Solution :Orbital : The areaaround the nucleuswhereorbital . In eachorbitalsuch AREA is maximumis calledprobabilityfindelectronis maximum approx`90%`Forthis `w^(2)`is smallbut itgivedefintevaluefordefinitedistancefromnucleus. it isnotthat enclosea region of 100%probability.so wedrawthe figurefor 90%PROBABILITYOF findingelectron. Pointand surfacediagramof orbital: in pointdiagrampointsshownforprobability .Bycombiningpointsshapeof orbitalis given. inboundarysurfaceor countersurfaceis drawn foranorbitalin WHICHTHE valueandprobabilitydensity`w^(2)`constant. e.g. the boundarysurfacediagramof 1s,2s ,`2p_(x ) 2p_(y)2p_(Z)` orbitalare GIVE . |
|
| 29. |
Which of the following statements aboutCO_(3)^(2-) ion are correct ? |
|
Answer» The C-O bond order is 1.33  Bond order `=(4)/(3) =1.33` Formal charge `=(2)/(3) =0.67` All bonds are equivalent due to resonance . Hence , (c) is wrong . Hybridization of central ATM is`sp^(2)` . Hence (d) is wrong . |
|
| 30. |
Which of the following has a net dipole moment ? |
|
Answer» `NO_(3)^(-)` |
|
| 31. |
trans-Pent-2-ene is polar with trans-but-2-ene is non polar. Explain. |
Answer» Solution :In trans-but-2-ene, the diple momens of the C-`CH_3` bonds are equal and opposite and HENCE they EXACTLY cancel out each other. Thus, trans-but-2-ene is non-polar.  However, in trans-pent-2-ene, the +I-effect of `CH_3CH_2`-group is higher than of `CH_3` group, therefore, the DIPOLE moment of `C-CH_3` and `C-CH_2CH_3` bonds are unequal . Although these two DIPOLES oppose each other , yet they do not exactly cancel out each other and hence trans-pent-2-ene has a small but finite dipole moment and thus is polar. |
|
| 32. |
What happens when ferrimagnetic Fe_3O_4 is heated to 850 K and why ? |
| Answer» SOLUTION :FERRIMAGNETIC `Fe_3O_4` on heating to 850 K BECOMES paramagnetic . This is due to greater alignment of domains (spins ) in ONE direction on heating | |
| 33. |
What is the formula of a compound in which element P forms ccp lattice and atoms of Q occupy 2/3rd of tetrahedral voids ? |
|
Answer» `therefore` Ratio `Al^(3+) : O^(2-)="2n"/3:n =2:3` , i.e., formula is `Al_2O_3` |
|
| 34. |
What is value of compressibility factor (Z) ? |
|
Answer» `(PV)/(nRT)` |
|
| 35. |
The solubility of BaSO_(4) in water is 2.42xx10^(-3)g L^(-1) at 298 K. The value of its solubility product (K_(sp)) will be (Given molar mass of BaSO_(4)=233 g L^(-1)) |
|
Answer» `1.08xx10^(-10) "mol"^(2)L^(-2)` `=(2.42xx10^(-3))/(233) "mol" L^(-1)` `=0.0104xx10^(-3) "mol" L^(-1)` `=1.04xx10^(-5) "mol" L^(-1)` `{:(BaSO_(4) ,hArr,Ba^(2+),+,SO_(4)^(2-)),(,,S,,S):}` `K_(sp)=[Ba^(2+)][SO_(4)^(2-)]=SxxS=S^(2)` `=(1.04xx10^(-5) "mol" L^(-1))^(2)` `=1.08xx10^(-10) "mol"^(2)L^(-2)` |
|
| 36. |
Which one of the elements with the following outer orbital configurations exhibits the largest number of oxidation states? |
|
Answer» `3d^3, 4s^2` |
|
| 37. |
What is thebasicdifference inapproach betweenMendeleev'sPeriodicLaw andthe Modern PeriodicLaw ? |
| Answer» Solution :MendeleevPeriodic Lawstatesthat thephysicaland chemicalpropertiesof the ELEMENTS are aperiodicfunction of theiratomicweigths while MODERN PeriodicLawstatesthat physicaland chemicalPropertiesof ELEMENTSARE aperiodicfunctionof theiratomicnumbers . thusthe basicdiffereceinapproachbetweenMendeleev's PeriodicLaw andModern PeriodicLaw is thechangein BASIS ofclassificationof elementsfromatomicweight to atomicnumber. | |
| 38. |
What are isoelectronic ions? Give an example. |
|
Answer» Solution :There are some ions of DIFFERENT ELEMENTS having the same number of ELECTRONS are called isoelectronic ions. EXAMPLE : `Na^+ , Mg^(2+) , Al^(3+) , F^(-) , O^(2-) and N^(3-)` |
|
| 39. |
What are the ozonolysis products of ortho xylene? |
Answer» SOLUTION : `{:(CH_3-C=O , , ,CHO ,CH_3-C=O),(""|,""+,""," "|," "+""|),(CH_3-C=O,,,""CHO,""CHO):}` 1,2-Dimethylbenzene (o-Xylene) on ozonolysis GIVES glyoxal, methyl glyoxal and dimethyl glyoxal in 3:2:1 molar RATIO. It proves the existance of TWO resonance structures for benzene and its derivatives |
|
| 40. |
Which of the following angle corresponds to sp^(2)hydridisation ? |
|
Answer» `90^(@)` |
|
| 41. |
What two changes on the equilibrium, N_(2) (g) + 3 H_(2) (g)hArr 2 NH_(3) (g) , Delta H= -92.4" kJ." can keepitsstate undisturbed ? |
| Answer» SOLUTION :Increase of TEMPERATURE ALONG with suitable increase of PRESSURE or increase of pressure along with suitable increase of temperature. | |
| 42. |
Two particles A and B are in motion. If the wavelength associated with particle A is 5 xx 10^(-8)m, calculate the wavelength associated with particle B if its monentum is half of A |
|
Answer» Solution :By de Broglie equation, `lamda_(A) = (H)/(p_(A)) and lamda = (h)/(p_(B))` `:. (lamda_(A))/(lamda_(B)) = (p_(B))/(p_(A))` But `p_(B) = (1)/(2) p_(A)` (Given) `:. (lamda_(A))/(lamda_(B)) = (1//2p_(A))/(p_(A)) = (1)/(2) or lamda_(B) = 2xx lamda_(A) = 2 XX 5 xx 10^(-8) m= 10^(-7)m` |
|
| 43. |
Which of the following statement is correct about alkanes? |
|
Answer» Alkanes react with acids or BASE in normal conditions |
|
| 44. |
Which of the following hydrocarbons is the most reactive towards addition of H_(2)SO_(4)? |
|
Answer» ETHENE |
|
| 45. |
What is the co-ordination number of an atom in its own layer in ABAB type arrangement |
|
Answer» |
|
| 46. |
Which of the following statements is not correct for BF_3 ? |
|
Answer» It can form adduct |
|
| 47. |
Which of the following compound has incorrect IUPAC nomenclature? |
|
Answer» `underset("Ethyl butanoate")(CH_(3)CH_(2)CH_(2) -overset(overset(O)(||))(C )-OC_(2)H_(5))` |
|
| 49. |
Which of the following properties of atom could be explained correctly by Thomson model of an atom? |
|
Answer» Overall NEUTRALITY of atom. |
|











