Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What isatomicnumbernucleonsand massnumber? Giverelationbetweenthem. |
|
Answer» Solution :ATOMICNUMBER (Z ) :Thenumberof protonspresentsin thenucleus is EQUALTO atomicnumber(Z ) . Which areequalnumberofelectronsin aneutralatom . Nucleons :protonsand neutronspresentin the nucleus arecollectivelyknownas nucleons MassNumber : The totalnumberof nucleonsin termedas massnumber(A )of THEATOM. massnumber (A ) = (Numberof ) + ( Numberof ) (protons (z )+ NEUTRONS (N)) |
|
| 2. |
Which of the following does not exhibit inert pair effect ? |
|
Answer» B |
|
| 3. |
What is wavelength of wave associated with electron whose velocity (v) is 10^(8)cm sec^(-1) ? |
|
Answer» 7.27 nm for n = 4 = 42 = 16 |
|
| 4. |
The structure of ice is ....... type. |
|
Answer» Linear |
|
| 5. |
What does the resulting solution contain when ammonia gas reacts with water ? |
|
Answer» `NH_2, H_3O^+` |
|
| 6. |
What is the SI unit for amount of substance ? |
|
Answer» MOLE |
|
| 7. |
Which property does aqueous solution of monoxide or peroxides carries? |
|
Answer» Acidic |
|
| 8. |
Which of the following molecules are expected to exhibit intennolccular H-bonding |
|
Answer» ACETIC ACID |
|
| 10. |
What weight of NaCI would be decomposed by 4.900 g of H_2SO_4, if 6 g of NaHSO_4 and 1.825 g of HCI are produced in the reaction ? |
|
Answer» |
|
| 13. |
What are the main constituents of LPG ? |
| Answer» SOLUTION :The main constituents of LPG are butane and isobutane. Both these isomers can be easily liquefied and hence can be CONVENIENTLY transported in iron CYLINDERS . | |
| 14. |
Write following conversions: a) nitrobenzene to acetanilide b) acetanilide to p-nitroaniline |
Answer» SOLUTION :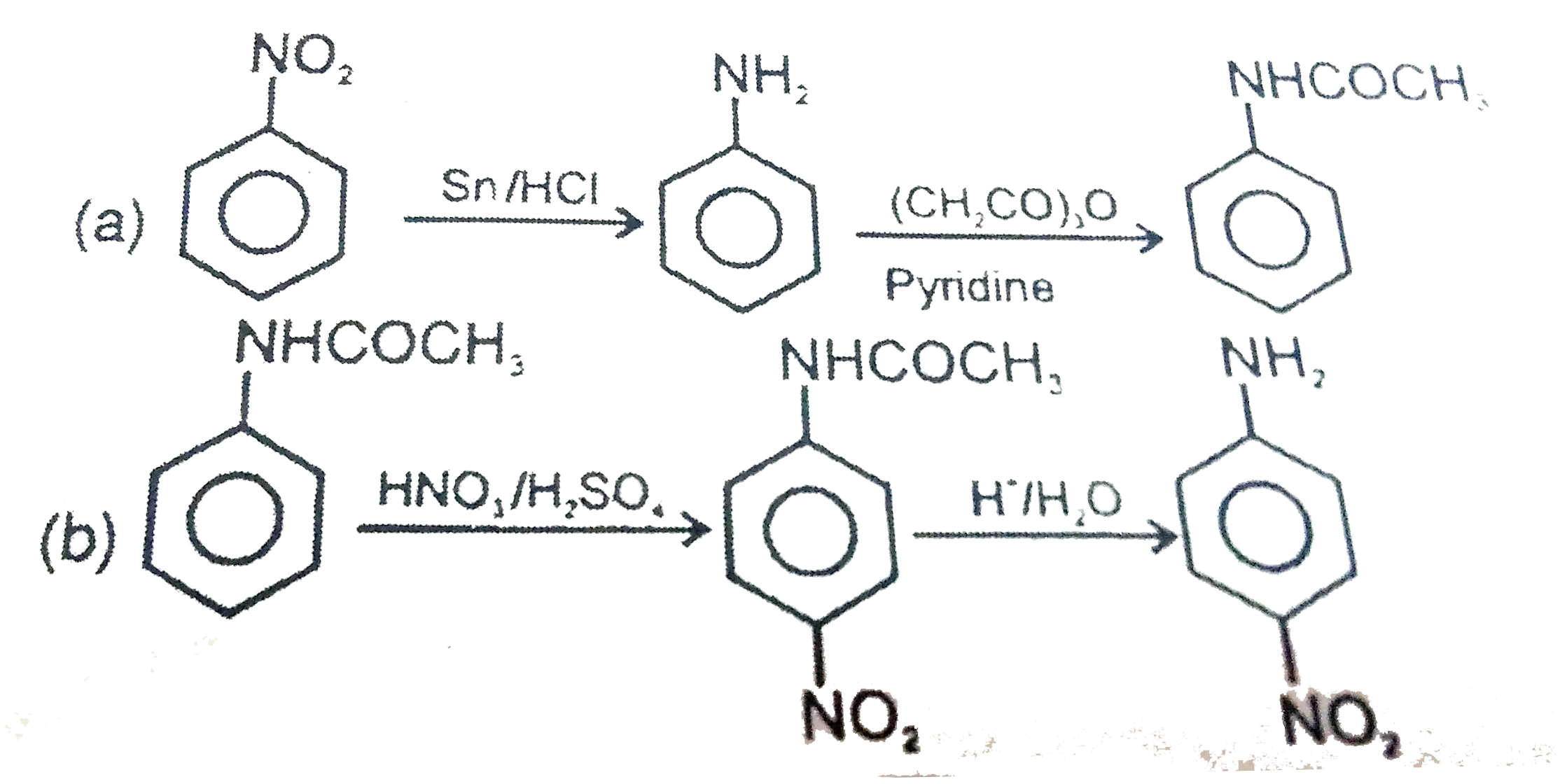
|
|
| 15. |
Which explosive reaction is used in fuel cells to generate electricity? |
|
Answer» REACTION of HYDROGEN with CARBON |
|
| 16. |
Which of the following molecules does not show any resonating structures? |
|
Answer» `NH_(3)` |
|
| 17. |
What will be the main product obtained during hydrobromination of Pent-1-ene in presence of Benzoyl peroxide ? |
|
Answer» 1-bromo PENTANE |
|
| 18. |
Weak base K_b=8.96xx10^(-7) than what is the pK_b ? |
| Answer» SOLUTION :`pK_b=-log K_b=-log (8.96xx10^(-7))`=6.04 | |
| 19. |
The root mean square speed of an ideal gas is given by : u_("rms") = sqrt((3RT)/M)Thus we conclude that u_("rms")speed of the ideal gas molecules is proportional to square root of the temperature and inversely proportional to the square root of the molar mass. The translational kinetic energy per mole can also be given as 1/2Mu_(rms)^(2) . The mean free path (lambda) is the average of distances travelled by molecules in between two successive collisions whereas collision frequency (C.F.) is expressed as number of collisions taking place in unit time. The two terms lambda and C.F. are related by : C.F = (u_("rms")/lambda) A jar contains He and H, in the molar ratio 1 : 5. The ratio of mean translational kinetic energy at the same temperature is |
|
Answer» `1:5` |
|
| 20. |
The root mean square speed of an ideal gas is given by : u_("rms") = sqrt((3RT)/M)Thus we conclude that u_("rms")speed of the ideal gas molecules is proportional to square root of the temperature and inversely proportional to the square root of the molar mass. The translational kinetic energy per mole can also be given as 1/2Mu_(rms)^(2) . The mean free path (lambda) is the average of distances travelled by molecules in between two successive collisions whereas collision frequency (C.F.) is expressed as number of collisions taking place in unit time. The two terms lambda and C.F. are related by : C.F = (u_("rms")/lambda) Which of the following relation is correct for an ideal gas regarding its pressure (P) and translational kinetic energy per unit volume (E) ? |
|
Answer» <P>`P = 2/3E ` `implies PV = 1/3 xx 2KE , (KE)/(V) = E implies P = 2/3 E` |
|
| 21. |
The root mean square speed of an ideal gas is given by : u_("rms") = sqrt((3RT)/M)Thus we conclude that u_("rms")speed of the ideal gas molecules is proportional to square root of the temperature and inversely proportional to the square root of the molar mass. The translational kinetic energy per mole can also be given as 1/2Mu_(rms)^(2) . The mean free path (lambda) is the average of distances travelled by molecules in between two successive collisions whereas collision frequency (C.F.) is expressed as number of collisions taking place in unit time. The two terms lambda and C.F. are related by : C.F = (u_("rms")/lambda) If n represents number of moles, n0 is number of molecules per unit volume, k is Boltzmann constant, R is molar gas constant, T is absolute temperature and NA is Avogadro's number then which of the following relations is wrong ? |
|
Answer» `P = m_0 kT` `implies P = (n_0)/(N_A) xx RT = n_0 RT`. (or) `PV = nRT = nKN_A xx T` . |
|
| 22. |
Which of the following alkyl group has the maximum +I effect ? |
|
Answer» `(CH_(3))_(2)CH-` |
|
| 23. |
Vapour density of a gas is 11.2. Volume occupied by 2.4 g of this at STP will be |
|
Answer» 2.4 L `=2xx 11.2 = 22.4` Number of MOLES of gas = (2.4)/(22.4)` `THEREFORE` 1 mole occupies = 22.4 L volume `therefore (2.4)/(22.4)` mole will occupy `=22.4 xx (2.4)/(22.4)` |
|
| 24. |
What Is the maximum weight of ammonia obtained by the reaction of 3kg of hydrogen and 7kg of nitrogen? |
|
Answer» |
|
| 25. |
Why benezeneundergoes substitutions reaction rather than addition reactionunder normal conditions |
|
Answer» SOLUTION :(i) Eachcarbonatomin benzenepossesan unhybrisised p-orbitalcontainingone electron.Over ALLTHE sixcarbonatomsand aresaidto bedelocalised (II) dueto delocalisationstrong`pi-`BONDIS formedwhichmakes themoleculestable.Henceunlikealkenesand alkynesben zeneundergonesubstitution reactionratherthanaddition reactionundernormalconditions |
|
| 26. |
What are the uses of zeolites ? |
| Answer» | |
| 27. |
Which of the following is an example of redox reaction ? |
|
Answer» `XeF_(6)+H_(2)OrarrXeO_(2)F_(2)+4Hf` `(b) overset(+6)XeF_(6)+2H_(2)OrarrXeO_(2)F_(2)+4HF^(-)` (c )  (d)`XeF_(2)+PF_(5)rarr[xeF]^(+)PF_(6)^(-)` In option (a) ,(b)and (d) O.N of the atoms remains the same but in option (c ) O.N of Xe increasses from +4 in `XeF_(4)` to +6 in `XeF_(6)` while the O.N of P DECREASES from +1 in `O_(2)F_(2)` to 0 in `O_(2)F_(2)`in `O_(2)` thus it is a REDOX reaction |
|
| 28. |
Write the favourable factors for the formation of ionic bond. |
|
Answer» Solution :(i) LOW IONISATION enthalpy of METAL ATOMS. (ii) High electron GAIN enthalpy of non-metal atoms. (iii) High lattice enthalpy of compound formed. |
|
| 29. |
Whatis therelationin shapeof 3d_(xy) and 3dx_(2) ? |
| Answer» Solution :bothhas ameshapeof 4 LOBES .In `d_(xy)`and`d_(xy)`bothare `45^(@)` and `d_(xy)` is inxy planebut`d_(x-y)` is onx andy axes. | |
| 30. |
Whichof the followingmetal ispresent in greencolouringpigmentchlorophyllof plants ? |
| Answer» ANSWER :B | |
| 31. |
When silicate units are linked together , they form which type of structures ? |
| Answer» Solution :When SILICATE UNITS are linked TOGETHER , they FORM chain, ring , sheet or three-dimensional structures. | |
| 32. |
Which of the following is known as pseudoalum? |
|
Answer» `KMN(SO_(4))_(2).12H_(2)O` |
|
| 33. |
When 0.1 mole of an acid is added to 2 lit of a buffer solution, the P^(H)of the buffer decreases by 0.5 The buffer capacity of the solution is |
|
Answer» `0.6` 0.05 _________ 1 lt ` PHI =(0.05)/( 0.5)=0.1 ` |
|
| 34. |
The values of K_(p1) and K_(p2) for the two equilibrium reactions X hArr +Z and A hArr 2B are in the ratio 9, 1,. If degree of dissociation of X and A beequal , calculate the ratio of the total pressure of the equilibriummixture in the two cases. |
|
Answer» Solution :Let the degree of disscoiation in the two cases be `alpha` (i) Calculattion of `K_(p1)` `{:("For equilibrium", X,hArr, Y, +,Z),("initial MOLES" : ,1,,0,,0),("moles at eqm. point",(1:alpha),,alpha,,alpha):}` TOTAL no. of ,moles `=1 -alpha +alpha+alpha=(1+alpha)` `K_(p1) = (P_(y)xxP_(z))/(P_(x)) = (((alpha)/(1+alpha)P_(1))xx((alpha)/(1+alpha)P_(1)))/(((1-alpha)/(1+alpha)P_(1)))` `=(P_(1)^(2)alpha^(2))/((1+alpha)^(2))xx((1+alpha)/((1-alpha)P_(1)))=(P_(1)alpha)^(2)/((1+alpha)(1-alpha))` (II) Calculation of `K_(P2)`. `{:("For equilibrium",A, hArr, 2B),("initial moles",1,,0),("Moles at eqm. point",(1-alpha),,2alpha):}` Total no. of moes `=1 alpha +2alpha =(1+alpha)` `K_(p2) = (pB)^(2)/(pA)=((2alpha)/(1+alpha)P_2)^(2)/(((1-alpha)/(1+alpha)P_(2)))` `P_(2)^(2) xx (4alpha^(2))/(1+alpha)^(2) xx ((1+alpha))/((1-alpha)P_(2))= (P_(2)xx4alpha^(2))/((1+alpha)(1-alpha))` Dividing eqn. (i) with eqn. (ii), `(K_p1)/(K_p2) = ((P_(1)alpha)^(2))/((1-alpha^(2)))xx ((1-alpha^(2)))/(P_(2)xx4alpha^(2)) = P_(1)/(4_(p_(2))` `" Now " ""(K_(p1))/(K_(p2))=9/1 ("given") :. P_(1)/(4P_(2))=9/1 or P_(1)/P_(2) = (36)/(1)` |
|
| 35. |
Which is a green house gas ? |
|
Answer» `CO_(2)` |
|
| 36. |
Yellow ppt. of compound (I) is insoluble in: |
|
Answer» `NaOH` `I rarr PbCrO_(4) (AcOH` INSOLUBLE) `HNO_(2) +I^(-) +H^(+) rarr NO` (Gas H) |
|
| 37. |
Which of the following is more basic in gaseous phase |
|
Answer» `CH_(3)NH_(2)` |
|
| 38. |
Two liquids X and Y on mixing gives a warm solution. The solution is ….... |
|
Answer» ideal |
|
| 39. |
Whatis isomerism mention the typeof isomerism |
|
Answer» SOLUTION :THEPHENOMENON in WHICHTHE samemolecularformulamay exhibitdifferentstructurarrangementis calledisomerism (i) Structuralisomerism (II)STEROISOMERISM |
|
| 40. |
Urea and hydrazine react to form ammonia gas along with compound X which reacts with aldehydes and ketone to form specific crystalline derivatives. X is |
|
Answer» PHENYL hydrazine 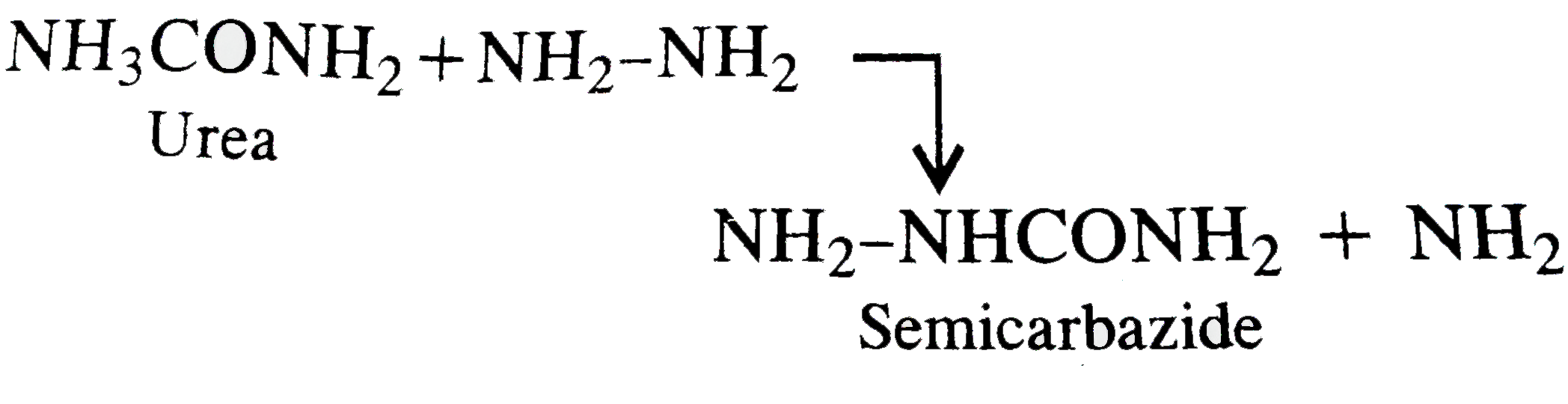
|
|
| 41. |
Which oxide is more basic, MgO or BaO? Why? |
| Answer» Solution :BaO, because basic CHARACTER of oxides increases down the group. On going down the group, the size of the cation increases and hence its acidic nature becomes less. As a result those with smaller sizes generally become amphoteric or more acidic. MgO is a basic oxide too but BaO with a larger sized `BA^(+2)` ION has greater basic nature. This can also be proven by looking at the strength of the HYDROXIDE bases. Where `Ba(OH)_2` is a strong base, `Mg(OH)_2` is a weak base and poorly SOLUBLE in water also. | |
| 43. |
The thermit rection used to weld iron rails in building a rail track, involves the following reaction Fe_(2)O_(3)+2AlrarrAl_(2)O_(3)+2Fe Calculate the mass of iron metal that can be prepared from 150 g of aluminium and 250 g of iron (iii) oxide. |
|
Answer» `underset(underset(=160g)(2xx56+3xx16))(Fe_(2)O_(3))+underset(underset(=54g)(2xx27))(2Al)rarr2Al_(2)O_(3)+underset(underset(=112g)(2xx56))(2Fe)` From the available data, it is evident that 150 g of AL metal need `Fe_(2)O_(3)=((160g))/((54g))XX(150g)=444.4g` But `Fe_(2)O_(3)` actually available = 250 g This means `Fe_(2)O_(3)` is the LIMITING reactant. The mass of Fe metal which can be PREPARED from 250 g of `Fe_(2)O_(3)=((112g))/((160g))xx(250g)=175g`. |
|
| 44. |
Which awards were given to Newland and Seaborg for their contribution or work? |
|
Answer» Solution :Newlands : Was later AWARDED Davy medal in 1887 by the Royal Society, London. Glenn T. Seaborg : He awarded NOBLE prize in chemistry. To give him HONOUR element Seaborgium (SG) was NAMED. |
|
| 45. |
When BF_(3) reacts with LiH, the gaseous product produced is |
|
Answer» HF |
|
| 46. |
Which one of the following does not show resonance? |
|
Answer» Carbon DIOXIDE |
|
| 47. |
What is the volume ratio of the product gases in the decomposition of phosphorus pentachloride? |
|
Answer» SOLUTION :`PCl_(5)toPCl_(3)(g)+Cl_(2)(g)` VOLUME ratio of `PCl_(3)` and `C_(2)` is 1:1 |
|
| 49. |
Two containers A and B have the same volume. Container 'A' contains 5 mole oxygen gas. Container 'B' contains 3 moles He gas and 2 mole CH_4 gas. Both the containers are separately kept in vaccum at the same temperature. Both the containers have very small orifices of the same area through which the gases leak out. The rate of diffusion of O_2 is how many times the rate of diffusion of He and CH_4 mixture |
|
Answer» `sqrt((8.8)/(32))` `(r_(O_2))/(r_("mix")) = sqrt((M_("mix.))/(32)) = sqrt((8.8)/(32))`. |
|