Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following statements is/are correct? |
|
Answer» Gallium has higher ionisation enthalpy than ALUMINIUM |
|
| 2. |
Write the ozonolysis products of mesitylene. |
Answer» SOLUTION :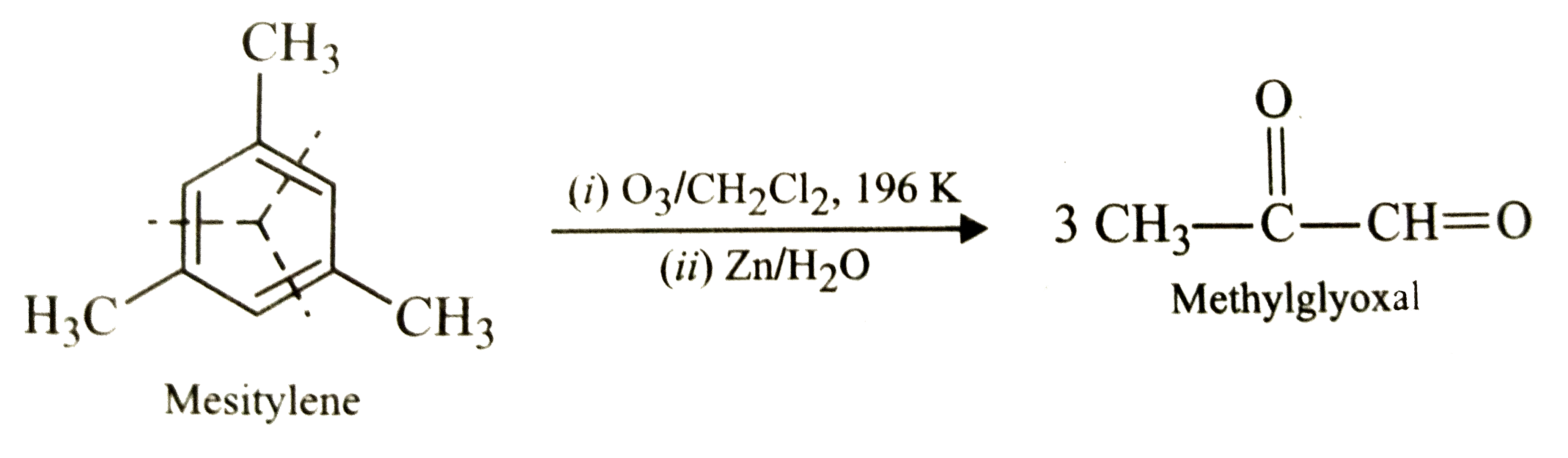
|
|
| 3. |
What are hydrogroscopic substance ? Give example |
|
Answer» Solution :Some substance are HIGHLY SOLUBLE in water as they ABSORB water from vapour of atmosphere and become soluble. Such substance are CALLED hydroscopic. e.g., Calcium flouride `(CaF_2)` is highly soluble in water. So, that it absorb also the water vapour from atmosphere and become soluble and therefore it is hydroscopic. |
|
| 4. |
The volume occupied by 0.2 mole of methane at N.T.P. is |
|
Answer» `4.48 DM^(3)` 0.2 mole of methane at N.T.P. occupies `22.4 xx0.2` =4.48 `dm^3` |
|
| 5. |
What volume of 0.1M KMnO_4is needed to oxidise 5mg of ferrous oxalate in acidc medium (MW of ferrous oxalate is 144) approximately 0.20 mL |
|
Answer» Solution : Milli equivalents of `KMnO_(4) = V xx 0.1 xx 5` milli equivalents of `FeC_(2)O_(4) = ("Wt in g")/("Ew") xx 1000` n-factor for `FeC_(2)O_(4)=3` milli equivalents of `KMnO_(4) = 5/(144//3) = (5xx3)/(144)` `:.` milliequivalents of `KMnO_4` = milli equivalents of `FeC_(2)O_4` `:. V xx 0.1 xx 5 =(5xx3)/144 :. V = 0.20 mL` |
|
| 6. |
The triad of nuclei that is isotonic is..... |
|
Answer» `._(6)C^(14), ._(7)N^(15), ._(9)F^(17)` |
|
| 7. |
Which of the following species can function both as oxidising as well as reducing agent ? |
|
Answer» `CI^(-)` |
|
| 8. |
What volume shall be occupied by 6.022 xx 10^23 molecules of any gas at NTP?: |
| Answer» SOLUTION :`6022 XX 10^23` molecules are contained in 1 mole, and 1 mole, of any gas at NTP occupies a VOLUME of 22.4 LITRES. | |
| 9. |
What is the distance of separation between 3rd and 4th orbit ofH-atom ? |
|
Answer» |
|
| 10. |
Which of the following will decolorise alkaline KMnO_(4) solution ? |
|
Answer» `C_(3)H_(4)` |
|
| 11. |
When the numerical value of the reaction quotient (Q) is greater than the equilibrium constant (K), in which direaction does the reaction proceed to reach equilibrium ? |
| Answer» SOLUTION :When `Q gt K_C`, the reaction will PROCEED in the REVERSE direction, i.e., formation of reactants. | |
| 12. |
Which element's position was left empty in periodic table by Mendeleev and what name did he gave ? |
|
Answer» SOLUTION :EMPTY SPACE for element `"" `Given NAME by him Gelium-Eka aluminium Germanium-Eka silicon |
|
| 13. |
Which of the following hybrid orbitals possess two types of angles? (1) sp^(3), (2) sp^(2), (3) sp, (4) sp^(3)d, (5) sp^(3)d^(2), (6) sp^(3)d^(3) |
| Answer» SOLUTION :`SP^(3)d and sp^(3)d^(3)`. | |
| 14. |
When a sample of a gas is compressed at constant temperature from 15 atm to 60 atm, its volume changes from 76 cm^3 to 20.5 cm^3. Which of the following are possible. |
|
Answer» The gas SHOWS non-ideal behaviour `P_2V_2 = 60 xx 20.5 = 1230` = non ideal behavious If it was ideal, `V_2 = 19` `= V_r > V_i = Z > 1 = ` repulsions dominate. |
|
| 15. |
Which among the halogens would you expect to have the least electronegativity and why? |
| Answer» SOLUTION :On moving down the group , electronegativity decreases because atomic size increases . Iodine is the LARGEST , THEREFORE it is LEAST electronegative. | |
| 16. |
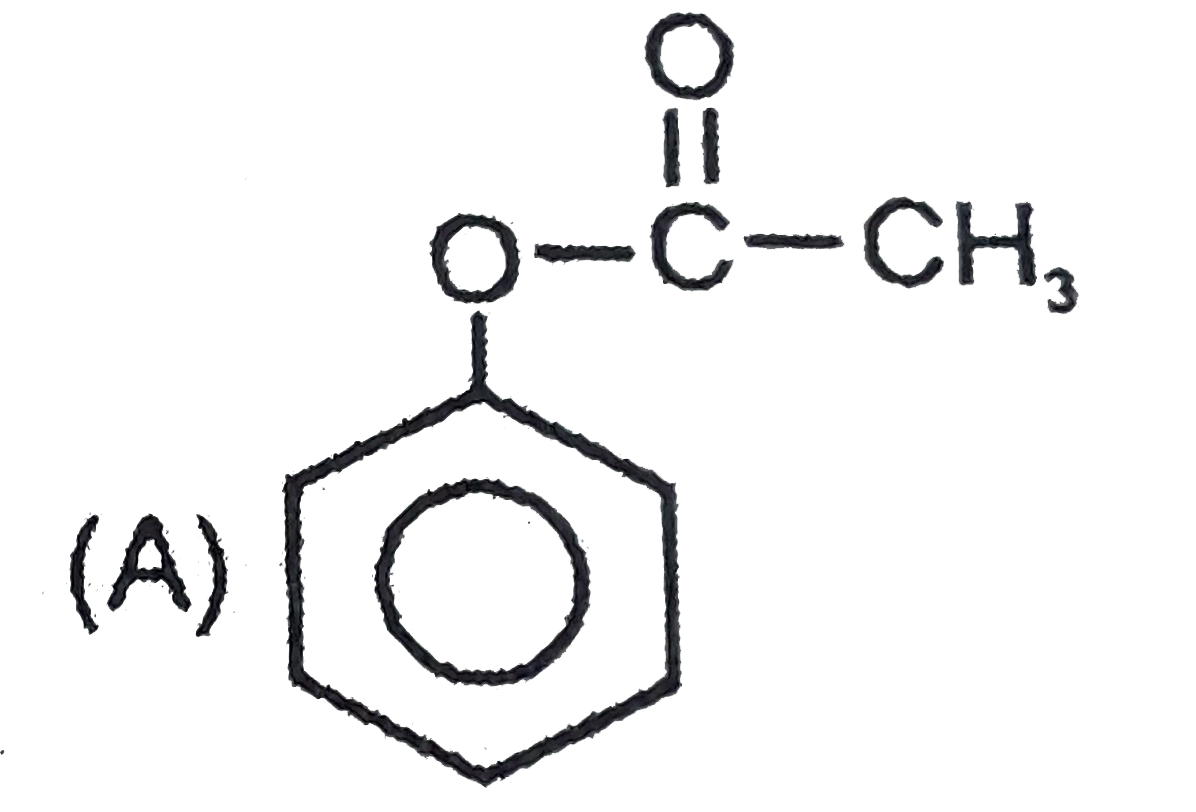
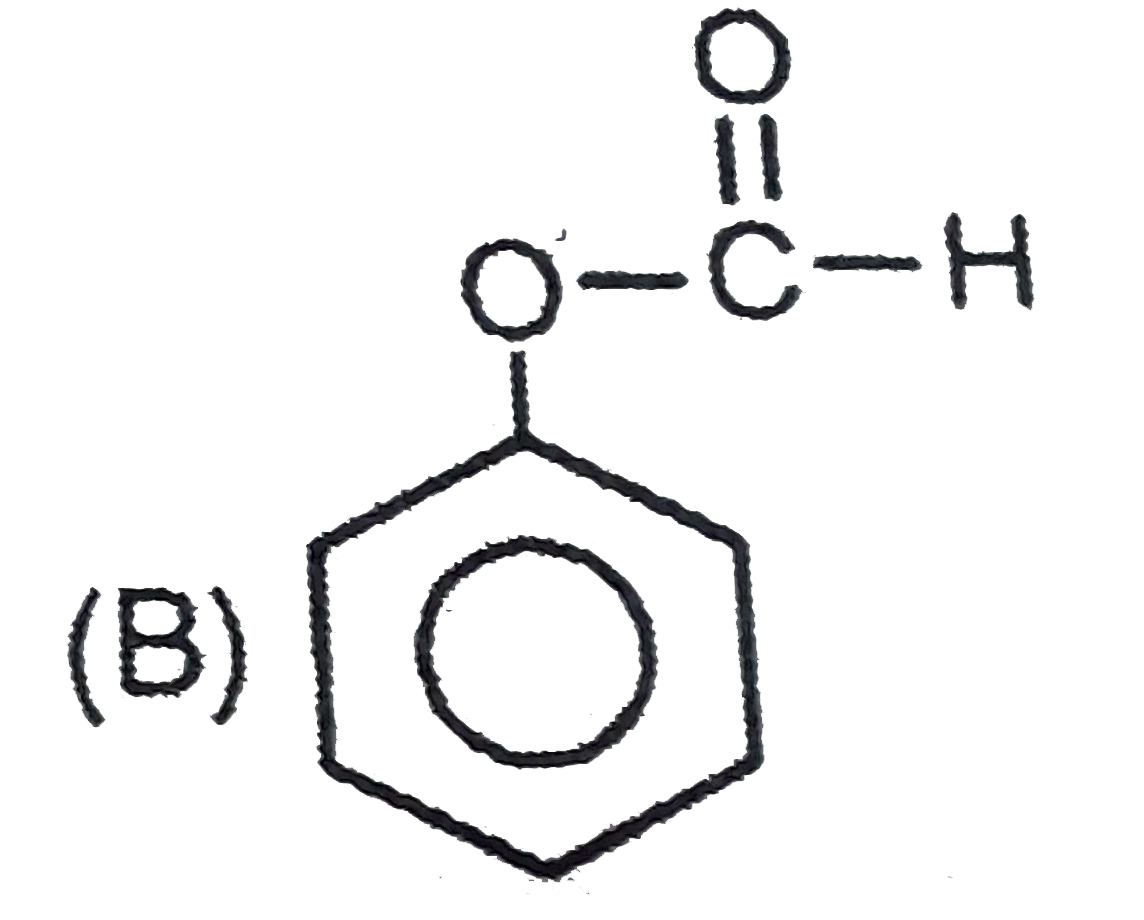
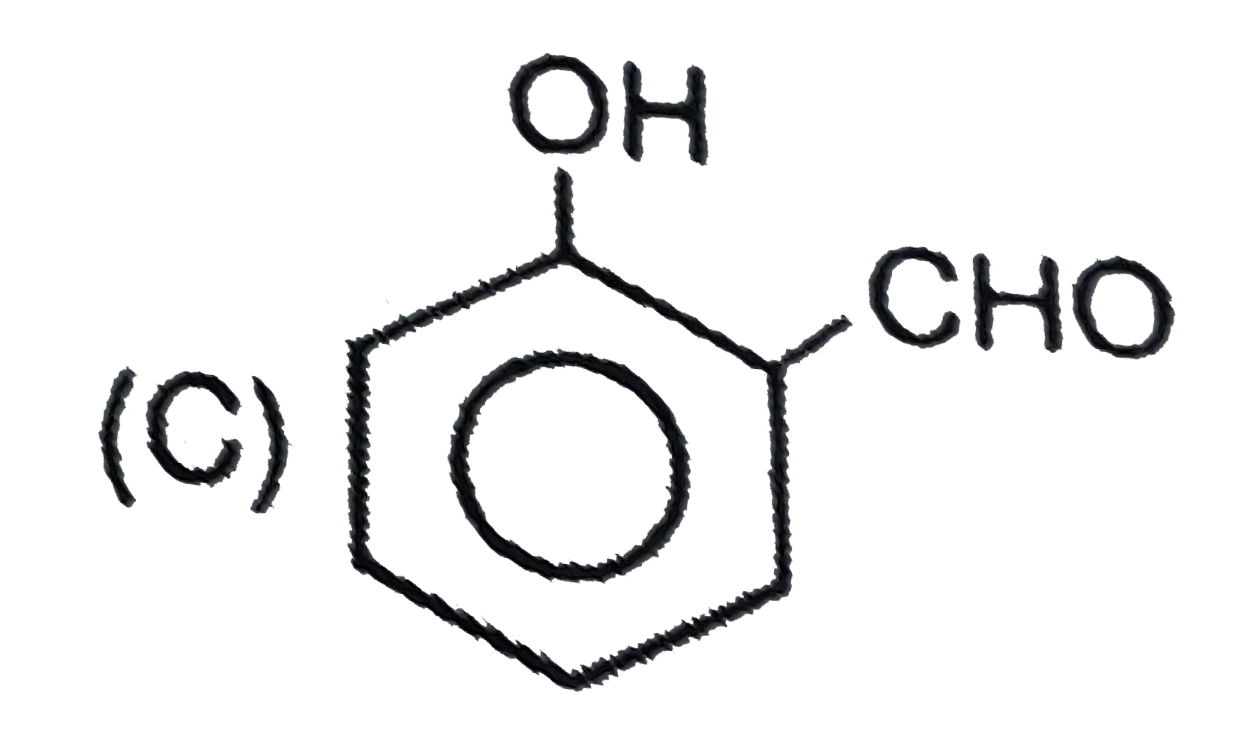
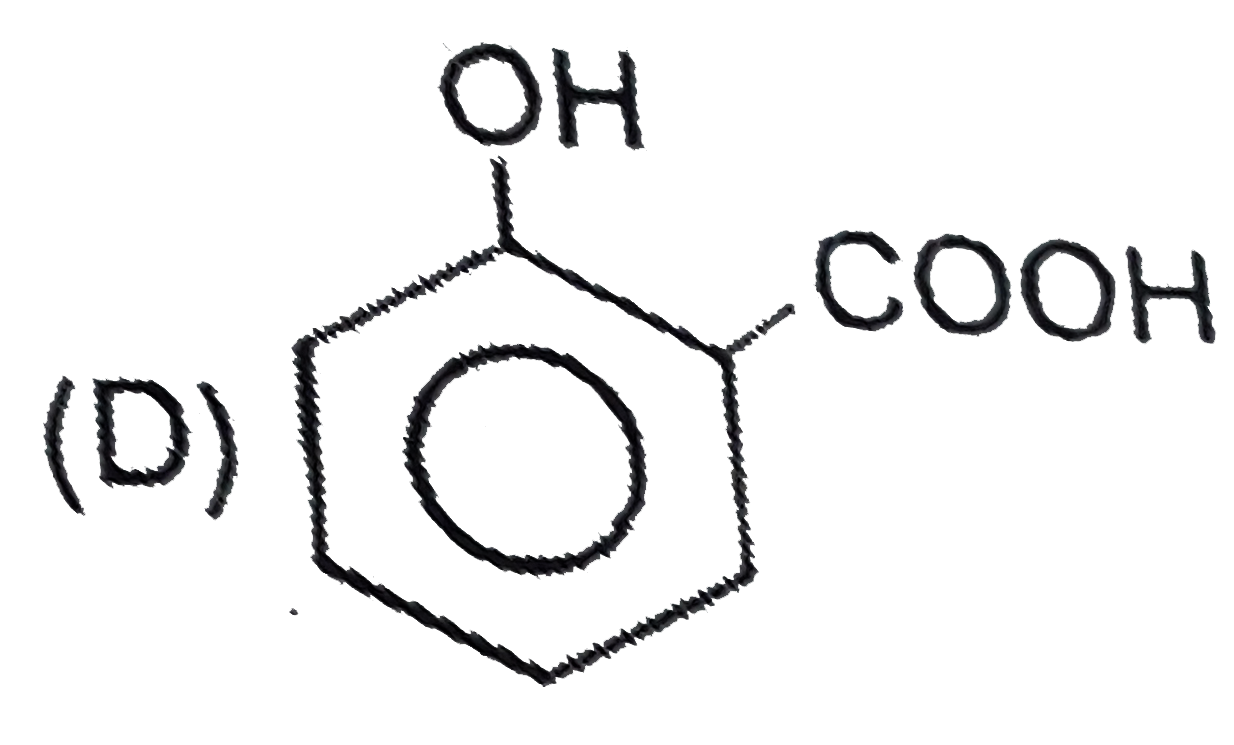
Which of the following compound is 'P' ? |
|
Answer»
|
|
| 17. |
Write short notes on (i) + E effect and (ii) -E effect |
Answer» Solution :When the ` pi` electron is transferred TOWARDS the attscking reagent it is called + E (positive electromeric ) effect .  The addition of ` H^(+)` to alkene as SHOWN above is an examples of +E effect . When the electron is transfered AWAY from the attacking reagent , it is called , - E (negative electromeric ) effect . 
|
|
| 18. |
The volume expansivity of a gas under constant pressure is 0.0037. Calculate its volume at its volume at -100^(@)C if its volume at 100^(@)C is 685 cm^(3). |
|
Answer» Solution :Volume expansivity of a gasmeans INCREASE or decrease in volume per DEGREE rise or fail in temperature of its volume at `0^(@)C`. `V_(t)=V_(0)+0.0037xxV_(0)xxt=V_(0)(1+0.0037 t)` At `100^(@)C"" V_(100^(@)C)=V_(0)(1+0.0037xx100)=685"cm"^(3)"" ("Given")"or" V_(0)=(685)/(1.37)=500" cm"^(3)` At `-100^(@)C,"" V_(-100^(@)C)=500[1+0.0037xx(-100)]=315" cm"^(3)` |
|
| 19. |
Which of the following is incorect? |
|
Answer» HydrogengtDeuteriumgtTritium, (%relative ABUNDANCE) |
|
| 20. |
The solubility of Mg(OH)_(2) is 8.352xx10^(-3) g/litre at 290^(@) C. Find out its K_(sp) at this temperature. |
|
Answer» Solution :`Mg(OH)_(2)` ionizes completely in the solution as : `Mg(OH)_(2)rarrMg^(2+)+2OH^(-)` `:. [Mg^(2+)]=[Mg(OH)_(2)] and [OH^(-)] = 2xx [ Mg (OH)_(2)]` But Molar mass of Mg`(OH)_(2) = 58 G "mol"^(-1)` `:. [Mg(OH)_(2)]=("Strength in g / litre")/("Molar mass") = (8.352xx10^(-3))/(58) = 1.44 xx 10^(-4)` moles/litre `:. [Mg^(2+)]=1.44xx10^(-4)` moles/litre and `[OH^(-)] = 2xx1.44xx10^(-4)=2.88xx10^(-4)` moles/litre `:. K_(sp) ` for `Mg(OH)_(2)=[Mg^(2+)][OH^(-)]^(2)=(1.44xx10^(-4))xx(2.88xx10^(-4))^(2)=1.194xx10^(-11)` |
|
| 21. |
Which of the following have zero electron gain enthalpy? |
|
Answer» HALOGENS |
|
| 22. |
What happens when Al_4C_3 reacts with D_2O? |
|
Answer» |
|
| 23. |
Which of the following represent a set of nucleophiles? |
|
Answer» `BF_(3), H_(2)O, NH^(2-)` |
|
| 24. |
TlI_(3) is a black coloured sparingly soluble ionic compound. In its aqueous solution, it will give |
|
Answer» `Tl^(+)` gives `I_(3)^(-)` IONS |
|
| 25. |
Water plays a key role in the biosphere. It is due to certain properties of H_(2)O as compared to other liquids. These are except |
|
Answer» higher SPECIFIC heat |
|
| 26. |
Which of the following is INCORRECT regardinginstantaneous dipole moment ? |
|
Answer» It is a non zero dipole moment of a non-polar molecule and is valid for SMALL TIME interval. |
|
| 27. |
Upon heating one litre of N/2 HCl solution, 2.67 gm of hydrogen chloride is lost and the volume of solution shrinks to 750 ml. The normality of resulting solution is |
|
Answer» 18.25 N REMAINING wt.of `HCl=18.25-2.67=15.58g` `N=(w)/(GEW)XX(1000)/(V"(in ML)")N=(15.58)/(36.5)xx(1000)/(750)=0.569N` |
|
| 28. |
Which element are known as reactive elements ? |
| Answer» SOLUTION :ELEMENTS of s-block and p-block are KNOWN as REACTIVE elements. | |
| 29. |
Which isotope of hydrogen is radioactive? |
|
Answer» PROTIUM |
|
| 30. |
The relative proportion of the atoms of tritium and protium is : |
|
Answer» `10:10^17` |
|
| 31. |
The shape of the molecule containing 3 bond pairs and one lone pair around the central atom is …………. |
| Answer» SOLUTION :TRIGONAL PYRAMIDAL | |
| 32. |
What are polar molecules ? Give examples. |
|
Answer» SOLUTION :(i) Diatomic molecules such as`H_(2), O_(2), F_(2)` have ZERO dipole moment and are CALLED non polar molecules. Molecules such as HF, HCI, CO, NO have non zero dipole moment values and are called polar molecules. |
|
| 33. |
What happens when (i) magnesium is burnt in air (ii) quick lime is heated with silica (iii) chlorine reacts with slaked lime (iv) calcium nitrate is heated ? |
|
Answer» Solution :(i) `2 Mg (s) + O_(2) (g) overset(Delta)(to) 2 MgO(s)and 2 Mg(s) +N_(2) (g) overset(Delta)(to) UNDERSET("Mag. Nitride")(Mg_(3) N_(2)(s))` (ii) `underset("Quick lime")(CaO(s)) + underset("Silica")(SiO_(2)) (s) overset(Delta)(to) underset("Calcium silicate") (CaSiO_(3) (s))` (iii) It reacts with `Cl_(2)` to form calcium hypochlorite , `Ca(OCL)_(2)-a` constituent of bleaching powder `underset("Slaked lime")(2 Ca(OH)_(2)) + 2 Cl_(2) to ubrace(CaCl_(2) + Ca(OCl)_2)_("Bleaching power") + 2H_(2)O` (iv) `2 Ca(NO_(3))_(2) (s) overset(Delta) (to) 2 CaO(s) + 4 NO_(2) (g) + O_(2) (g)` |
|
| 34. |
What is meant by free energy of a system ? What will be the direction of the chemical reaction when (i) Delta G = 0 (ii) Delta G gt 0(iii) Delta G lt 0 ? |
|
Answer» SOLUTION :Free energy is defined as the energy which can be converted into useful work. (i) `Delta G = 0`, the reaction will be in equilibrium (II) `Delta G gt 0`, the reaction will not take place. (III) `Delta G LT 0`, the reaction will be spontaneous. |
|
| 35. |
What weight of N_2 is present in a 22.4 lit flask at 273^@C and 380mm? |
| Answer» Answer :C | |
| 36. |
When 1 mole Zn powder is reacted with 1 mole sulphuric acid in bomb calourimeter (closed isolated system),........... |
|
Answer» `Delta U lt 0 , W =0` Reaction is erothermic, therefore `Delta U lt 0`. Volume of gas remains CONSTANT therefore work `w=0`. |
|
| 37. |
Which out of LI, Na, K, Be, Mg, Ca has lowest ionization enthalpy and why? |
| Answer» Solution :K has lowest ionisation ENERGY due to larger atomic SIZE among these elements. Ther force of attraction between VALENCE electron and nucleus is less, therefore, it can lose ELECTOR easily. | |
| 38. |
What is meant by Plasma state? Give an example. |
| Answer» SOLUTION :Gaseous STATE of matter at very high TEMPERATURE containing gaseous IONS and free electron is referred to as the Plasma state, e.g. Lightning | |
| 39. |
Which element having more ionisation enthalpy? (i) Ne or Ar (ii) Cl or F(iii) F or O (iv) N or O(v) Na or K(vi) Cl or S (vii) Kr or Xe(viii) P or S |
|
Answer» Solution :(i) NE(ii) F(iii) F (IV) N (V) NA(vi) Cl(vii) KR(viii) P |
|
| 40. |
Whichdevelopmentplayed a rolein theformationof Bohr.s modelof atom ? |
|
Answer» SOLUTION :Twodevelopments played amajorrole in the formulationof Bohr.smodelof atomResultsobservedfromthestudies of interactions ofradiationswith matterhaveprovidedimmenseinformationregardingthe structureof atomsand MOLECULES . NeilsBohrutllisedthesereusltsto giveatomicmodel (i) Dualcharacterof theelectromagneticradiationwhichmeansthatradiationspossessbothwavelikeand particlelikeproperties. (ii)EXPERIMENTAL resultsregardingatomicspectrawhichcan beexplainedonly byassumingquantizedelectronic energylevelsin ATOMS |
|
| 41. |
Which of the following compounds will not exist as resonance hybrid ? Give reason for your answer . CH_(3) - OH |
| Answer» SOLUTION :` CH_(3) - OH ` : Does not exist as resonance hybrid due to ABSENCE of ` pi`-electrons . | |
| 42. |
Which of the following statement is not correct:- |
|
Answer» The EXTENT of adsorption depends on the NATRUE of the ADSORBENT and adsorbate |
|
| 43. |
Threshold limit value of three pollutants X, Y and Z are respectively 9 ppm, 20 ppm and 5 ppm. Which one is the most toxic ? |
| Answer» Solution :LESSER the TLV value more is the toxicity of POLLUTANT. THUS Z is the most TOXIC. | |
| 44. |
Which of the following compounds will not exist as resonance hybrid ? Give reason for your answer . CH_(3) - CH = CH - CH_(2) NH_(2) |
| Answer» Solution : `CH_(3) - CH = CH - CH_(2) NH_(2)`: Does not exist as RESONANCE hybrid, because the LONE pair on N-atomis not conjugated with ` pi`-electrons of the DOUBLE bond . | |
| 45. |
Which of the following forms cationic micelles above certain concentration ? |
|
Answer» Cetyltrimethylammonium bromide |
|
| 46. |
Write the preparation of methane from methyl chloride and acetic acid. |
|
Answer» SOLUTION :(i) `UNDERSET(+H_(2),-HCl)overset("Reduction Zn", H^(+))rarr` (ii) `CH_(3)COOH underset(-H_(2)O)overset(NaOH)rarr CH_(3)COO^(-)Na^(+)` (iii) `underset("Sodium ethanoate")(CH_(3)COO^(-)Na^(+)) underset(Delta, -Na_(2)CO_(3))overset(+"Na(OH), CaO, SODALIME")rarr CH_(4)` |
|
| 47. |
Which of the following molecules has the shortest carbon single bond length ? |
|
Answer» `CH_(2) = CH - oversetoverset(O)("||")C -OH` |
|
| 48. |
The solubility of Pb(OH)_(2) in water is 6.7xx10^(-6)M. Calculate the solubility of Pb(OH)_(2) in a buffer solution of pH = 8 |
|
Answer» Solution :`Pb(OH)_(2) hArr Pb^(2) + 2 OH^(-)` `:. K_(sp)=[Pb^(2+)][OH^(-)]^(2)=s xx (2s)^(2)=4 s^(3) = 4xx(6.7xx10^(-6))^(3) = 1.20 xx 10^(-15)` In a solution with pH = 8, `[H^(+)]=10^(-8) and [OH]^(-) = 10^(-6)` `:. 1.2xx10^(-15)=[Pb^(2+)]xx(10^(-6))^(2) or [Pb^(2+)]=(1.2xx10^(-15))/((10^(-6))^(2))=1.2xx10^(-3)M` |
|
| 49. |
Type pf hybridization of complex (E): |
|
Answer» `sp^(3)d^(2)` `I rarr PbCrO_(4) (AcOH` INSOLUBLE) `HNO_(2) +I^(-) +H^(+) rarr NO` (Gas H) |
|
| 50. |
What is meant by non (homo) polar covalent bond ? |
| Answer» SOLUTION :`107^(@) and 104^(@)` RESPECTIVELY. | |