Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which part of soil is formed by decomposition of animal and plant wastes ? |
|
Answer» ORGANIC PART |
|
| 2. |
The threshold frequency for a metal is 7.0 xx 10^(14) s^(-1). Calculate the kinetic energy of an electron emitted when radiation of frequency 10^(15)s^(-1) hits the metal. |
|
Answer» Solution :`KE=h(v-v_(0))=` `(6.625xx10^(-34))/(1.0xx10^(15)-7.0xx10^(14))` `=1.988xx10^(-19)J` |
|
| 3. |
Which of thefollowingtransitioninvolvesmaximumamount of energy ? |
|
Answer» `M^(-)( G) to M ( g)` |
|
| 4. |
Write the relationship between K_(sp) and S for an AB type of salt. |
|
Answer» Solution :Consider an ELECTROLYTE AB whose SOLUBILITY is S mol `dm^(-3)`. In a SATURATED solution, `AB_((s))hArrA^(+)+B^(-)` `[A^(+)]=S" mol "dm^(-3)and(B^(-))=S" mol "dm^(-3)` `K_(s)=[A^(+)][B^(-)]=S*S=S^(2)(or)K_(sp)=S^(2)`. |
|
| 5. |
Whichof thefollowinggroupshowspositiveresonanceeffect ? |
|
Answer» `-COOH` |
|
| 6. |
Write the resonance structures of CO_(3)^(2-)" and "HCO_(3)^(-). |
Answer» SOLUTION :Resonance STRUCTURES of `CO_3^(2-)` ion :  Resonances structures of `HCO_3^(-)` ion : 
|
|
| 7. |
What weight of NaOH will contain the same number of oxygen atoms as are present in 9.8 g of sulphuric acid ? (Na = 23, O = 16, S = 32, H = 1) |
|
Answer» 4 g `4xx6.02xx10^(22) = 2.408 xx10^(23)` ATOMS of oxygen Mass of NaOH containing same number of oxygen ATOM `("No. of oxygen atoms in " H_2SO_4xx "Mol. mass of NaOH" )/("Avogadro Number")` `=(4xx6.02xx10^(22)xx40)/(6.02xx10^(23)) =16 g` |
|
| 8. |
What would happen when a small quantity of H_(2)O_(2) is added toa solution of FeSO_(4) ? |
|
Answer» Colour DISAPPEARS `2FeSO_(4) + H_(2)SO_(4) + [O] to Fe_(2)(SO_(4))_(3)+H_(2)O` `H_(2)O_(2)+2FeSO_(4) + H_(2)SO_(4) to Fe^(2)(SO_(4))_(3)+2H_(2)O` |
|
| 9. |
Which one of the following is most acidic? |
|
Answer»
|
|
| 10. |
Which is the oxidation number of carbon underlined in CH_(3)underlineCOO*CH_(2)*CH_(3) ? |
Answer» SOLUTION : `CH_(3)+C+(O)+(O)+(CH_(2)-CH_(3))=0` `therefore0+C+(-2)+(-2)+1=0` `thereforeC=+3` |
|
| 11. |
The refluxing of (CH_(3))_(2) NCOCH_(3) with acid gives |
|
Answer» `2CH_(3)NH_(2)+ CH_(3)COOH` |
|
| 12. |
Which of the following reagents can be used for distinguishing the three classes of alcohols |
|
Answer» FENTON's reagent |
|
| 13. |
Derived unit is |
| Answer» SOLUTION :SI unit of DENSITY `=("SI unit of MASS")/("SI unit of volume") = KG m^(-3)` | |
| 14. |
Whensodium chloride solutionis electrolysed , thegasthatis liberatedat thecathodeis________. |
|
Answer» OXYGEN |
|
| 15. |
When lead sulphide reacts with hydrogen peroxide,The produce formed is :- |
| Answer» | |
| 16. |
The stability order of oxide, peroxide and superoxide of alkali metal is: |
|
Answer» `"NORMAL oxide" gt "SUPER oxide" gt "PEROXIDE"` |
|
| 17. |
underset((16.8gm))("Solid"(A))overset(Delta)rarr underset((4.4gm))("Acidic gas") (B)+ Neutral liuqid (C)+ Solid (D) Aqueous solution of (D) overset(BaCI_(2)sol)underset("in excess")rarr (Solid E weighing 19.7 gm) Identify A: |
|
Answer» `KHCO_(3)` `C rarr H_(2)O, D rarr Na_(2)CO_(3), E rarr BaCO_(3)` |
|
| 18. |
Which of the following hydroxide does not shows basic properties? |
| Answer» Answer :A | |
| 19. |
underset((16.8gm))("Solid"(A))overset(Delta)rarr underset((4.4gm))("Acidic gas") (B)+ Neutral liuqid (C)+ Solid (D) Aqueous solution of (D) overset(BaCI_(2)sol)underset("in excess")rarr (Solid E weighing 19.7 gm) Identify E: |
|
Answer» `BaCO_(3)` `RARRX = 10.6 gm` `BaCO_(3) ("mole") = (19.7)/(197) = 0.1` mol 0.1 mole of `D rArr (10.6)/(x gm//mol)` |
|
| 20. |
When KI is added to acidified solution of solution of sodium nitrite : |
|
Answer» `NO` GAS is liberated and `I_(2)` is set free |
|
| 21. |
What volume of NH_(2) is formed when 2.24lit of N_(2) combine with 4.48 litres of H_(2) at STP. |
|
Answer» |
|
| 22. |
Which of the following is not ore of sodium metal ? |
|
Answer» Borex |
|
| 23. |
Which of the following has number of molecules present equal to those present in 16 grams of oxygen |
|
Answer» `16gO_(3)` |
|
| 24. |
Which of the following metal oxide is antiferromagnetic in nature ? |
|
Answer» `MnO_(2)` |
|
| 26. |
Which orbitals on overlapping will form sigma and pi-bond in C=C of alkene ? Explain. |
Answer» Solution :For the FORMATION of `SIGMA`-bond the `sp^(2)-sp^(2)` ORBITALS are overlap on the same edges on two carbon atoms.  Every carbon atoms has three `sp^(2)` orbital where two other atoms are attached and with other carbon atoms from `sigma`-bonds. Two unhybridized 2p orbitals of two carbon perpendicular to planner overlap with each other and forms `PI`-bond. 
|
|
| 27. |
What is dead burnt plaster? How is it obtained from gypsum? |
| Answer» Solution :`CaSO_(4)` is CALLED dead burnt plaster. It is obtained by HEATING gypsum at HIGH temperature. | |
| 28. |
What is octet rule ? |
| Answer» SOLUTION :Element s having 8 electrons in the outermost shell are STABLE. | |
| 29. |
What isthe valueof Delta v . Delta x(uncertaintly) forelectron?Whatit indicate ? |
| Answer» Solution :For electron `Delta v .Delta x `is `10^(4) ` itmeansthatif onetriesro fieldexactlocationof theelectronthanuncertaintyof only `10^(18)` m .So`Delta x . Delta v` is `10^(4) MS^(-1)` | |
| 30. |
What is the correct IUPAC name of the following compound ? |
|
Answer» `1-`Bromo`-5-`cyclobutyl`-2-`cyclopropyl BENZENE 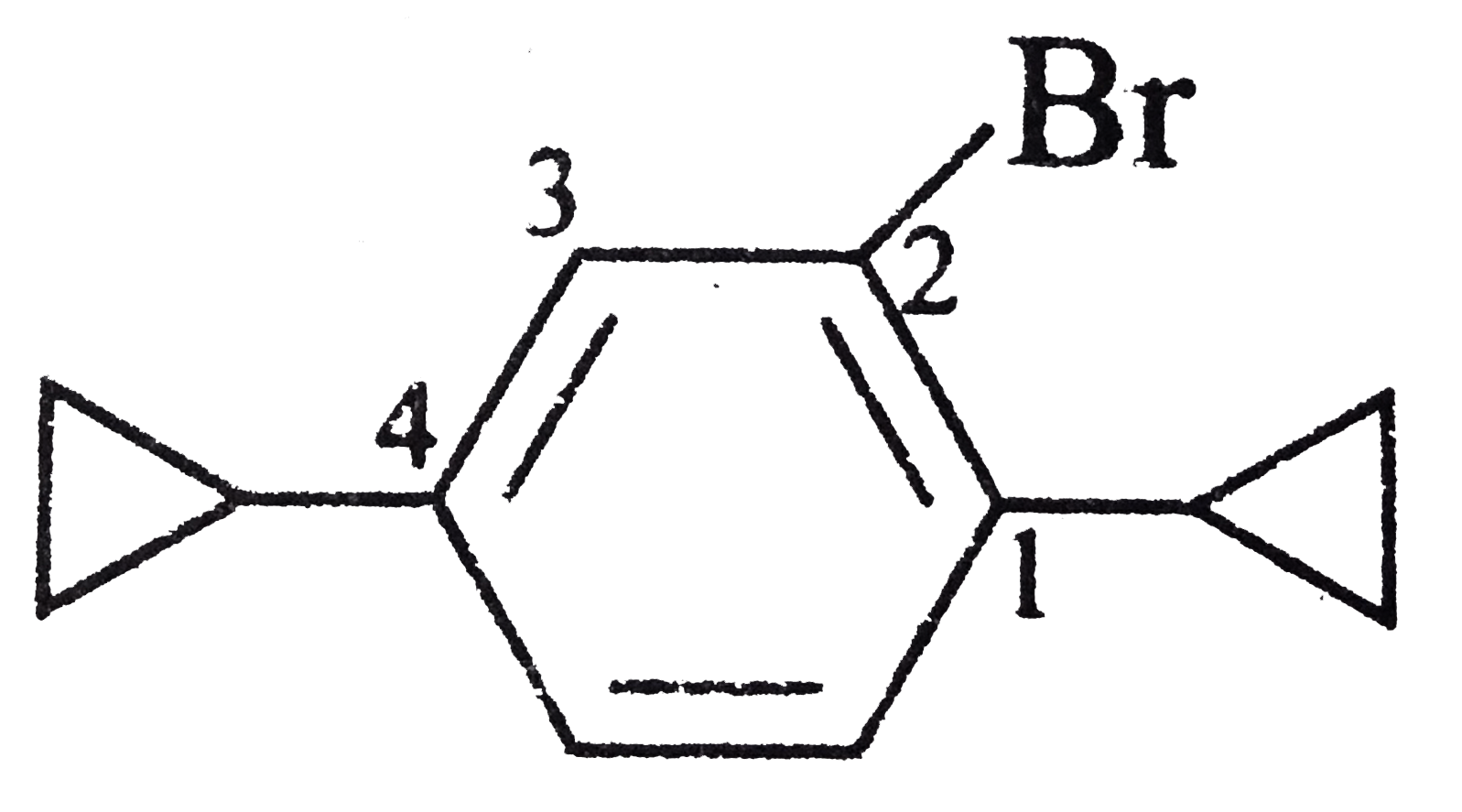 ALSO, cyclobutyl comes before cyclopropyl in alphabetical ORDER, hence, the correct `IUPAC` NAME is `2-`bromo`-4-`cyclobutyl`-1-`cyclopropyl benzene. |
|
| 31. |
What is incompressible nature of a gas molecules ? How is it shown ? |
| Answer» Solution :The van der WALLS constant for volume (b) remains constant overa WIDE RANGE of pressure and TEMPERATURE. This IMPLIES that gas molecules are incompossible. | |
| 32. |
What are electron deficient compounds ? Are BCl_(3) and SiCl_(4) electronsdeficient species ? Explain |
|
Answer» Solution :Species in which the central atom eitherdoes not have eight electrons in the valence shell or those which have 8 electronsin the valenceshell but can EXPAND their covalency beyond 4 due to the PRESENCE of d-orbitals, are called electron deficientmolecules. For EXAMPLE. (i) In `BCl_(3)`, the centralboron atom has only six electrons. Therefore, it is an electrons deficient compound. As such, it ACCEPTS a pair of electrons from `NH_(3)` to form an adduct  (ii) In `SiCl_(4)`, the central Si atom has 8 electrons but it can expandits covalency beyond 4 due to the presenceof vacant d-orbitals. Thereforein principle, `SiCl_(4)` shouldalso be an electron-deficient molecule but, in fact it does not accept two more `Cl^(-)` ions to form `[SiCl_(6)]^(2-)` becauseof the following two reasons. (i) Six large sized `Cl` atoms cannot be accommodated AROUND small `Si` atom. (ii) Due to bigger size of `Cl`, interaction betweenlone pairs of chlorine atom and the empty d-orbitals silicon atom is weak. |
|
| 33. |
xKI+yH_(2)SO_(4)rarrI_(2)+SO_(2)+KHSO_(4). In the above balanced equation, what are x and y? |
|
Answer» SOLUTION :2 moles of KI can gives one mole of `I_(2)` Therefore, x=2 `2KI+y H_(2)SO_(4) to I_(2)+SO_(2)+2KHSO_(4)` Verifying the COEFFICIENTS, y=3 |
|
| 34. |
When n-hexane is heated with anhydrous AlCl_3 and HCl gas, the major product obtained is |
|
Answer» 1-chlorohexane |
|
| 35. |
Which of the following contains same number of carbon atoms as are in 6.0g of carbon (C-12)? |
|
Answer» 6.0 g ETHANE `C_(2)H_(6)` = molar mass = 24 + 6 = 30g 30 g of ethane CONTAINS `2xx6.023xx10^(23)` Carbon atoms. (b) 8.0 g of methane (`CH_(4)` ) `CH_(4)` = molar mass = 12 + 4 = 16 g 16 g of methane contains `6.023xx 10^(23)` Carbon atoms. (c) 21.0 g of propane (`C_(3)H_(8)` ) `C_(3)H_(8)` = molar mass = 36 + 8 = 44 g 44 g of propane contains `3 xx6.023xx 10^(23)` Carbon atoms. (d) 28.0 g of Carbon monoxide (CO) CO = molar mass = 12 + 16 = 28 g 28 g of Carbon monoxide contains `6.023xx 10^(23)` Carbon atoms. 6.0 g of Carbon contains `(6.023xx10^(23))/12xx 6 = 3.0115xx10^(23)` Carbon atoms. Among the (a), (b), (c), (d)-8g g of `CH_(4)` contains `(6.022xx10^(23))/16xx8` `=3.0115xx10^(23)`carbon atoms |
|
| 36. |
The shape of the molecule is determined approximateiy by …………………….. |
| Answer» SOLUTION :BOND ANGLE | |
| 37. |
Whichof the followingwill havethe mostnegativeelectrongainenthalpyand whichthe leastnegative? P, S, Cl, F Explainyouranswer. |
Answer» Solution :Arrangingthe givenatomsintodifferent groupsandperiodsin orderof increasingatomicnumbers Aswe moreacrossa periodfromleftto rightthe electrongain enthalpybecomesmore andmorenegative while withthe groupfrom top tobottom it BECOMES lessand less NEGATIVE. Therefore , Fshouldhave themostnegativeelectrongainenthalpywhile Pshouldhave theleastnegativeelectron gain enthalpy. Butaddingan electron to LARGER 3P- orbitall of CI.HenceCl has themostnegativeelectron gainenthalpywhile PHAS thelest negativeelectron gainenthalpy . |
|
| 38. |
Which types of orbitals stabilise a molecule ? |
|
Answer» |
|
| 39. |
Which of the following point defects shown by AgBr(s) crystals ? (A)Schottky defect , (B)Frenkel defect , (C )Metal excess defect , (D)Metal deficiency defect |
|
Answer» (A) and (B) |
|
| 40. |
Which of the following acts as a self-indicator ? |
|
Answer» `K_(2)Cr_(2)O_(7)` |
|
| 41. |
Which one of the following is not the characteristic of Planck's quantum theory of radiation |
|
Answer» The ENERGY is not ABSORBED or emitted in whole NUMBER or multiple of QUANTUM |
|
| 42. |
Weakest conjugate base is obtained from |
|
Answer» `HNO_3` |
|
| 43. |
Weak Acid HA K_a is 1.0 xx 10^(-5) in 1 litre 0.100 mole Acid is solubic. At equilibrium dissociation is ……. |
|
Answer» `1.00%` `{:(,HA_((aq))+ H_2O_((L)) hArr, H_3O_((aq))^(+) + , A_((aq))^(-)),("At equilibrium",(1-x)M, xM,xM):}` `K_a=[H_3O^+]^2/((1-x)) approx [H_3O^+]^2/C^0` `therefore [H_3O^+]=SQRT(K_axxC^0)=sqrt(1.0xx10^(-5)xx0.1)` `=1.0xx10^(-3)` M % of ionization = `(1.0xx10^(-3))xx100` = 0.1 |
|
| 44. |
Which of the following on reductive ozonolysis followed by reaction with base gives |
|
Answer»

|
|
| 45. |
Which of the following is/are proper method to dispose sludge ? (A) Incineration (B) Dumping (C) Anaerobic diestion by microbes (D) Filtration |
| Answer» Answer :4 | |
| 46. |
Which of the following has highest boiling point ? |
|
Answer» Neo-pentane |
|
| 47. |
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions (a) K and S (b) Ca and O (c) Al nnd N |
Answer» Solution :K and S :  Alkali metal K lose one electron and S gain two electron and attain stable octet like Ar noble gas (Ar] containing `K^(+) and s^(2-)` ions and they are con1bine to FORM `K_(2)`S. (b) Ca and O : The electron configuration of Ca (Z = 20) and O (Z = 8) are `[Ar]4s^(2) and [He] 2s^(2) 2p^(4)`respectively Ca lose two electron and O gain two electron and form `Ca^(2+) and O^(2-)` . `underset([Ar] 4s^(2))overset(..)(Ca) rarr underset([A])(Ca^(2+)) + underset(Ca "lose " e^(-))(2e^(-))` In CaO, `Ca^(2+) and O^(2-)` POSITIVE and negative ions are combine by elector valence bond.  (c) Al and N : The configuration of Al (Z = 13) is (Ne]`3S^(2) 2p^(-1)`. This positive element lose 3 electron and attain stable octet like Ne noble gas and form `Al^(3+)` ION. `underset([Ne] 3s^(2)3p^(1))overset(.)(Al) rarr underset([Ne])(Al^(3+) + 3e^(-)` Nitrogen (Z = 7) is negative element. It accept 3 electron of Al and convert into `N^(3-)` Nytride ion like stable octet of Ne noble gas. In Al and N, the elector valence (ionic) bond between positive ion `AI^(3+)` and negative ion `N^(3-)` by electrostatic attraction. 
|
|
| 48. |
Which one of the following is an example for negative mesomeric effect? |
| Answer» ANSWER :D | |
| 49. |
Which of the following species have the same shape and same bond order ? (i) CO_(2) (ii) N_(3)^(-) (iii) O_(3) (iv) NO_(2)^(-) |
|
Answer» (i) and (II) case `O_(3) and NO_(2)^(-)` are V-shaped with bond order 1.5 in each case . |
|







