Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is combination reaction ? Give example. |
|
Answer» Solution :When two or more substances COMBINE to form a single SUBSTANCE, the REACTIONS are called COMBINATION reactions. A + B `rarr` C Example: 2 Mg + `O_(2) rarr` 2MgO |
|
| 2. |
The solubility product of BaSO_4 is 1.1 xx 10^(-10) at 298 K temp. calculate its water solubility. |
|
Answer» Solution :`BaSO_(4(s)) hArr Ba_((aq))^(2+) + SO_(4(aq))^(2-)` If `S=BaSO_4` Solubility of `BaSO_4` in mol `L^(-1)` `THEREFORE K_(SP)=S^2=1.1xx10^(-10)` `therefore S=sqrt(1.1xx10^(-10))` `=1.049xx10^(-5) "mol L"^(-1)` |
|
| 3. |
Which method of purification is represented by the following equation Ti(s) |
|
Answer» ZONE refining |
|
| 5. |
What are silicates ? |
| Answer» SOLUTION :SILICATES are MINERALS which consist of Si`O_(4)^(4-)` UNITS arranged in different ways. | |
| 6. |
What effect the branching of an alkane has on its melting point ? |
| Answer» Solution :In GENERAL , as the branching increases, the packing of the molecules in the crystal lattice becomes less CLOSE and HENCE the m.p. decreases ACCORDINGLY. However , if the branching makes the molecule symmetrical , the packing of the molecules in the crystal lattice becomes close and hence the m.p. increases. Now since isopentane is less symmetrical than n-pentane but neopentane is the most symmetrical of the three isomeric pentanes, THEREFORE , the m.p. of isopentane (113 K)is lower than of n-pentane (143 K) but the m.p. of neopentane (256 K) is much higher than that of isopentane and n-pentane . | |
| 7. |
Write the complete chemical equation for the following . Reduction of benzene |
Answer» SOLUTION :
|
|
| 8. |
The units of surface tension and viscosity of liquids are respectively "______________". |
|
Answer» `KG m^(-1) s^(-1), NM^(-1)` |
|
| 9. |
Why cannot sodium and potassium be prepared by the electrolysis of their aqueous solutions ? |
| Answer» Solution :The electrode potential , i.e., reduction potential of Na `(-2*71 V)` or `K(-2*92)` is much LOWER than that of `H_(2)O (-0*83V)` , THEREFORE , upon ELECTROLYSIS , water gets reduced in preference to `Na^(+)` or `K^(+)` ions. In other words , sodium and potassium cannot be obtained by electrolytic reduction of `Na^(+)` or `K^(+)` ions in AQUEOUS solution . | |
| 10. |
What polymerisation? Explain with suitable example. |
|
Answer» SOLUTION :A polymer is a large molecule formed by the combination of large number of small molecules (monomers). This process is known as polymerisation a few examples are: (i) `underset(("ethene"))(nCH_(2) =CH_(2)) = underset(873K)overset("red hot iron tube")(rarr) underset(("Polythene"))(-CH_(2) - CH_(2)-)` (ii) `underset(("PROPENE"))(nCH_(2) - CH) = CH_(2) underset(873K)overset("red hot iron tube")(rarr) underset(("Polypropene"))((-underset(underset((CH_(3)))(|))(CH) - CH_(2)-)_(n))` (iii) `underset(("Styrene"))(nCH_(2) = underset(underset(C_(6)H_(5))(|))(CH_(2))) underset(873K)overset("red hot iron tube")(rarr) underset(("POLYSTYRENE"))((-underset(underset(C_(6)H_(5))(|))(CH_(2)) - CH-))_(n)` |
|
| 11. |
Which of the following pair is not diagonally related? |
| Answer» ANSWER :B | |
| 12. |
Why argon does not form Ar_(2) molecule ? |
| Answer» Solution :Argon has completely filled ORBITALS. It is stable and does not form COMPOUNDS. Argon EXISTS as monoatomic gas | |
| 13. |
Which of the following is correct for lyophillic sol ? |
|
Answer» Irreversible sol. |
|
| 14. |
Write the relationship between the solubility and solubility product of AB type salt. |
|
Answer» |
|
| 15. |
The condition for skidding is |
|
Answer» Solution :(i) A carbon atom whose. tetravalency in SATISFIED by four different substituents (atoms (or) GROUPS) is called asymmetric carbon (or) CHIRAL carbon. The optical isomer should have one or more chiral carbon to show optical activity (ii) The molecule possessing chiral carbon atom and is non-superimposable its own MIRROR image is said to be chiral molecule and the property is called chirality or dissymmetry. `underset("Assymetric carbon atom")(d-underset(x)underset(|)overset(a)overset(|)C-b)` 
|
|
| 16. |
What is the ratio of average molecular KE of CO_(2) to that of SO_(2) at 27^(@)C? |
|
Answer» Solution :Assume molecules to be spheres with radius r. therefore the volume of molecule is `(4pir^(3))/(3)` The closest APPROACH between TWO molecules =`2r`. Since two molecules cannot come closer than distance 2r. The excluded volume per pair of molecules in binary collision is Volume of sphere of radius `2r=(4)/(3)pi(2r)^(3)=8xx(4pir^(3))/(3)` Excluded volume per molecules is `(1)/(2)[8xx(4pir^(3))/(3)]=4xx(4pir^(3))/(3)` `=4xx`volume of one molecule |
|
| 17. |
What are lanthanides and actinides? |
|
Answer» Solution :• In 4F series, 4f orbitals are being progressively filled with electrons, `4f^(1-14)5d^(0-1)-6S^(2)`. These elements lie in 6th period and are called rare earths or lanthanides or lanthanones. • In 3F series, 5forbitals are being progressively filled with electrons, `5F^(1-14)6D^(0-1)-7s^(2)` These elements lie in 7th period and are called actinides or actonones. |
|
| 18. |
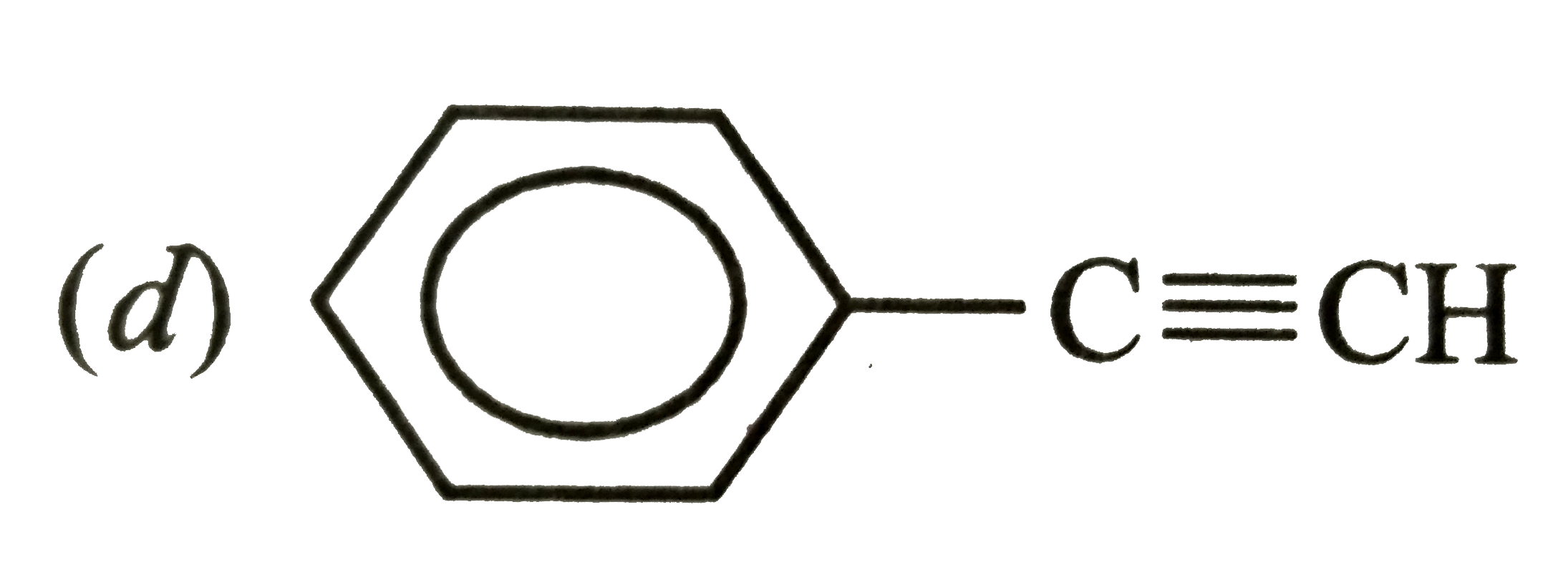
Which hybrid orbitals are used by carbon atom in the following molecules ? (a) CH_(3) - CH_(3)" (d) " CH_(3) - CHO (b) CH_(3) - CH = CH_(2) "(e) " CH_(3)COOH (c) CH_(3) - CH_(2) - OH |
Answer» SOLUTION :
|
|
| 19. |
Which one is an example of multimolecular colloid system |
|
Answer» Soap dispersed in water |
|
| 20. |
Which product is obtained by the reaction between Na_2S_2O_3 and Cl_2 gas ? |
|
Answer» `Na_2S_4O_6` |
|
| 21. |
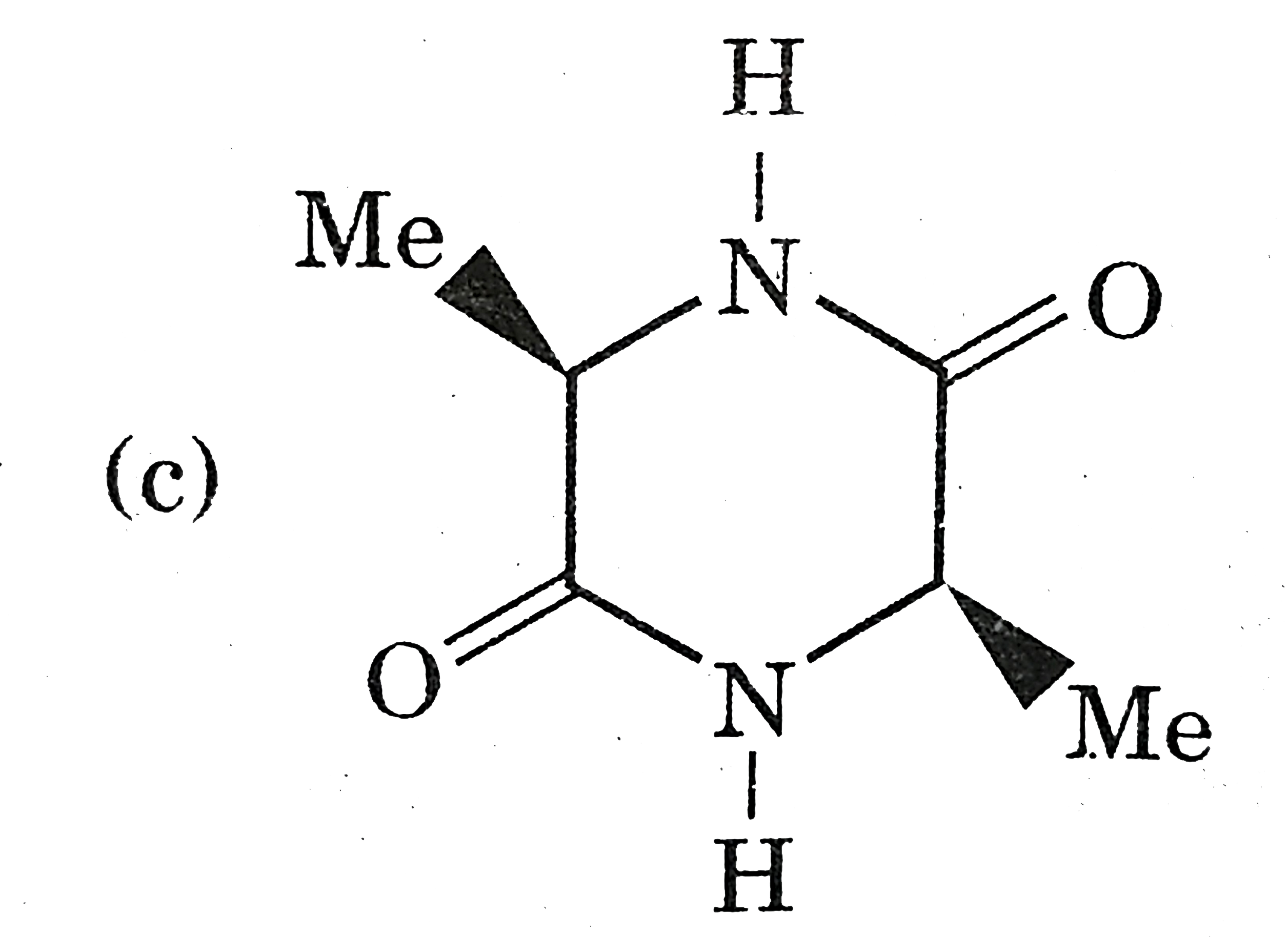
Which of the following does not have centre of symmetry? |
|
Answer»
|
|
| 22. |
Why blue colour of copper suphate solution gets dischared when zinc rod is dipped inti ? Given E_(Cu^(2+//Cu))^(@)=0.34V "and" E_(Zn//Zn^(2+))^(@)=0.76 V |
| Answer» Solution :Since `E_(Zn,Zn^(2+))^(@)=-E_(Zn^(2+),Zn)^(@)=-0.76V` is lower than `E_(Cu^(2+),Cu)^(@)`therefore Zn will lose electrons and copper will accept them in other words the FOLLOWING reaction will occur `Zn+Cu^(2+)rarrZn^(2+)+Cu` since blue `Cu^(2+)` ions are consumed and colourless `Zn^(2+)` ions are PRODUCED during the above rections therefore colour of `CuSO_(4)` solution gets DISCHARGED when zinc ROD is dipped in it | |
| 23. |
Which of the following is the smallest cation ? |
| Answer» Answer :d | |
| 24. |
which of the following is correct about species:(CH_3)_3-C^+ |
|
Answer» Its is plNNR |
|
| 25. |
Thevalence shellelectronicconfigurationofRb is __________. |
|
Answer» `3S^(1)` |
|
| 26. |
What is false about H_(2)O_(2) ? |
|
Answer» ACTS as both oxidising and reducing AGENT |
|
| 27. |
Which one of the following is the least electronegative element ? |
|
Answer» Bromine |
|
| 28. |
Which of the following is/are true for an acid -base titration? |
|
Answer» Indicators catalyse the acid BASE reactions by releasing or accepting `H^(+) ` ions. |
|
| 29. |
what is effect of heat on the following compounds? (Give equations for the reactions) (i) CaCO_(3) (ii) CaSO_(4) 2H_(2)O |
|
Answer» Solution :(i) `CaCO_(3) overset("heat")(to) CaO + CO_(2)` (decompostion / CALCINATIONS) (ii) `2CaSO_(4). 2H_(2)O underset("STRONGLY")overset("heat")(to) (CaSO_(4)) underset("Plaster of paris")(("DEAD burnt plaster"))+ 2H_(2) O + 3H_(2) O` |
|
| 30. |
Which of the following statement(s) is/are consistent with the Bohr's theory of the atom (and no others)? A) An electron can remain in a praticular orbit as long as it continuously absorbs radiation of adefinite frequency. B) The lowest energy orbits are those closest to the nucleus. C) All electrons can jump from the K shell to the M shell by emitting ratiation of a definite frequency. |
|
Answer» A,B,C |
|
| 31. |
Which of the following is a correct match? 1) Orthoboric acid - HBO_(2) 2) Metaboric acid - H_(6)B_(4)O_(7) 3) Pyroboric acid - H_(3)BO_(3) 4) Tetraboric acid - H_(2)B_(4)O_(7) |
|
Answer» 1 |
|
| 32. |
The species formed in the depietion of ozone layer by chloroflurocarbons in free radial mechanism is |
|
Answer» `CLO^(**)` |
|
| 33. |
Use of glycol as antifreezer in automobile is an important application of ………………. |
| Answer» SOLUTION :COLLIGATIVE PROPERTY | |
| 34. |
Which of the following does not shows any effect on cement strength ? |
|
Answer» CARBONDIOXIDE water |
|
| 36. |
What are silicones ? State the uses of silicones. |
|
Answer» Solution :Silicones are a GROUP of organosilicon polymers, which have `(R_2SiO)` as a repeating unit. The starting materials for the manufacture of silicones are alkyl or aryl substituted silicon chlorides, `R_nSiCl_((4-n))`, where R is alkyl or aryl group. When methyl CHLORIDE reacts with silicon in the PRESENCE of copper as a catalyst at a temperature 570K various types of methyl substituted chloramine of formula `MeSiCl_3 , Me_2SiCl_2 , Me_3SiCl` with small amount of `Me_4Si` are formed. Hydrolysis of dimethyl dichlorosilane, `(CH_3)_2SiCl_2` followed by condensation polymerization yields straight chain polymers.  Uses : They are used as SEALANT, greases, and electrical insulators and for water proofing of fabrics. Being biocompatible they are also used in surgical and cosmetic plants. |
|
| 37. |
The unit cell that best describe the CsCl crystallattice is |
|
Answer» BCC unit cell |
|
| 38. |
When a ship is sailing in ocean where temperature is 30^(@)C , a baloon is filled with 4.0 L of air. What will be the volume of the ballon when the ship reaches other ocean, where temperature is 33^(@)C. |
|
Answer» Solution :`T_(1)=30^(@)C=30+273*15=303*15 K, T_(2)=33^(@)C=33+273*15=306*15K` Apply Charle.s law , `(V_(1))/(T_(1))=(V_(2))/(T_(2))=(4*0(L)xx306.15K)/(303*15K)=4*039L` `:. V_(2)=4*039L` |
|
| 39. |
The size of molecular chlorine is about 3.5A^@. At what maximum intermolecular distance the dispersion forces operate? |
|
Answer» Solution :One-half of the size is taken as .r. T. of chlorine is 1.75`A^@` The interaction energy is PROPORTIONAL to `r^(-6)` `r^6` for molecular chlorine `= 2.9A^@` The maximum DISTANCE at which the intermolecular forces operate in chlorine is APPROXIMATELY 300pm. |
|
| 40. |
What are the reactions involved for ozone layer depletion in the stratosphere? |
|
Answer» Solution :The main reason of ozone layer depletion is belived to be the release of chlorofluorocarbon compounds (CFCs), also known as freons. These compounds are nonreaction non-flammable, non toxic organic MOLECULES. These are used in refrigerators, air conditioners in the production of plastic foam and by the electronic INDUSTRY for cleaing computer parts. CFCs are released in the atmosphere, they mix with the normal atmospheric gases and EVENTUALLY reach the stratosphere. In stratosphere, they get broken down by powerful UV radiations, releasing chlorine free radical. `CF_2 Cl_(2(g)) overset(UV)to overset(*)Cl_((g)) + overset(*)CF_2 CL_((g)) ""....(i)` The chlorine radical then react with stratospheric ozone to form chlorine MONOXIDE radicals and molecular oxygen. `overset(*)Cl_((g)) + O_(3(g)) to Cloverset(*)O_((g)) + O_(2(g)) ""......(ii) ` Reaction of chlorine monoxide radical with atomic oxygen produces more chlorine radicals. `Cloverset(*)O_((g)) + O_((g)) to overset(*)Cl_((g)) + O_(2(g)) "".....(iii)` The chlorine radicals are continuously regenerated and cause the breakdown of ozone. Thus, CFCs are transporting agents for continuously generating chlorine radicals into the stratosphere and damaging the ozone layer. |
|
| 41. |
What will happen to the wavelength associated with a moving particle if its velocity is doubled ? |
| Answer» SOLUTION :Wavelength BECOMES half of the ORIGINAL value. (because `LAMDA = (h)/(mv), " i.e., " lamda prop (1)/(v)`) | |
| 42. |
When dilute ink is treated with powdered charcoal- |
|
Answer» blue COLOUR BECOMES faint |
|
| 43. |
Which of the following on treatment with warm dil. H_2SO_4 in presence of HgSO_4 will give a methyl ketone. |
|
Answer» `Ph-C-=C-CH_3` |
|
| 44. |
What are the uses of sodium hydroxides? |
|
Answer» Solution :Sodium HYDROXIDE is USED as laboratory reagent. (ii) It is used in the purification of bauxite and petroleum refining. (iii) It is used in the textile INDUSTRIES for mercerising cotton FABRICS. (iv) It is used in the manufacture of soap, paper, artificial silk and a number of chemicals |
|
| 45. |
what is meant by nodal surface or node? |
| Answer» SOLUTION :The region WITHIN the orbital where the PROBABILITY of finding the ELECTRON is ZERO is called a node. | |
| 46. |
When we concept that one mole of a substance contains the same numbers of elementary entities or one mole of any other substance, we don’t actually need to know what that number is. Sometimes however we will need to work with the actual number of elementary entities in a mole of substance. This number is called Avogadro.s number. N_(A)=6.022137xx10^(23) molecules The unit mol^(-1) which we read as per mole signifies that a collection of N_(A) molecular level entities is equivalent to one mole at the macroscopic level. For example a mole of carbon contains 6.02xx10^(23) atoms of C. A mole of oxygen gas contains 6.02xx10^(23) molecules of O_(2). Which of the following is a reasonable value for the numbers of atoms in 1.00 g of helium? |
|
Answer» `0.25` |
|
| 47. |
What is the percentage weight of metallic element in the constitution of calcium carbonate? |
| Answer» SOLUTION :Gram molecular weight of `CaCO_(3) `=100g% wt. of Ca`=(40)/(100)xx100=40%` | |
| 48. |
When we concept that one mole of a substance contains the same numbers of elementary entities or one mole of any other substance, we don’t actually need to know what that number is. Sometimes however we will need to work with the actual number of elementary entities in a mole of substance. This number is called Avogadro.s number. N_(A)=6.022137xx10^(23) molecules The unit mol^(-1) which we read as per mole signifies that a collection of N_(A) molecular level entities is equivalent to one mole at the macroscopic level. For example a mole of carbon contains 6.02xx10^(23) atoms of C. A mole of oxygen gas contains 6.02xx10^(23) molecules of O_(2). The number of atoms present in 8 g of ozone is |
|
Answer» `N_(A)` `=(8)/(48)xxN_(0)xx3` atoms = `(N_(0))/(2)` |
|
| 50. |
When we concept that one mole of a substance contains the same numbers of elementary entities or one mole of any other substance, we don’t actually need to know what that number is. Sometimes however we will need to work with the actual number of elementary entities in a mole of substance. This number is called Avogadro.s number. N_(A)=6.022137xx10^(23) molecules The unit mol^(-1) which we read as per mole signifies that a collection of N_(A) molecular level entities is equivalent to one mole at the macroscopic level. For example a mole of carbon contains 6.02xx10^(23) atoms of C. A mole of oxygen gas contains 6.02xx10^(23) molecules of O_(2). The dot at the end of a sentence has a mass of about one micrograms. Assuming that the black stuff is carbon. No. of carbon atoms present in it is |
|
Answer» `5xx10^(16)` = `(10^(-6))/(12)` moles = `(10^(-6))/(12)xx6xx10^(23)` atoms `=5xx10^(16)` |
|