Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
The vapour density of a gas is 11.2. The volume occupied by 10g of the gas at STP is |
| Answer» ANSWER :A | |
| 3. |
whichof thefollowingis TRUEaboutcommercialproductionofcaliumoxide? |
|
Answer» Stakedlimeis usedas reactant |
|
| 4. |
Which of the following is sparingly soluble salt ? |
|
Answer» `BaSO_4` |
|
| 5. |
Which of the following reagent is helpful to diferentiate ethylene dichloride and ethylidene chloride? |
|
Answer» Zn/methanol |
|
| 6. |
Write the structures and names for all the possible isomers of benzene derivativeshaving the molecular formulae : (a) C_(8)H_(10), (b) C_(9)H_(12), ( c) C_(2)H_(2)F_(4). |
|
Answer» |
|
| 7. |
What is the volume ratio of equal masses of hydrogen,methane and oxygen present under similar conditions? |
|
Answer» |
|
| 8. |
What is the effect of heat on the following compounds? a. Magnesium chloride hexahydrate b. Gypsum c. Magnesium sulphate heptahydrate |
|
Answer» SOLUTION :a. `MgCl_(2).6H_(2)Ooverset(Delta)(to) Mg(OH)Cl+HCl+5H_(2)O` (b)`CaSO_(4).2H_(2)Ooverset(120^(@)C)(to)CaSO_(4)1/2H_(2)O+3/2H_(2)O` (plaster of PARIS) (c)`MgSO_(4).7H_(2)Ooverset(473K)(to)MgSO_(4)+7H_(2)O` |
|
| 9. |
Which transition between Bohr orbits corresponds to third line in the Balmer series of the hydrogen spectrum |
| Answer» SOLUTION :5TH ORBIT to 2ND orbit | |
| 10. |
When 100 ml of N/10 NaOH are added to 50 ml of N/5 HCI, the P^(H)of the resulting solution is |
|
Answer» `7` |
|
| 11. |
Which of the following statement is correct for Fullerene ? |
|
Answer» There are twenty rings having five carbon atoms in Fullerene. (C) Each carbon atom combines with other three carbon atoms with `sigma`-bond. The remaining electrons forms `pi`-bond and hence, carbon possess `sp^2` hybridization, so (C) is not true. (D) Fullerene is crystalline form of fullerene, so (D) is not true. (B) Fullerene possesses `C_(2n)` type of moelcular structure. |
|
| 12. |
The role of starch in iodometric titrations is that |
|
Answer» it ACTS an oxidant |
|
| 13. |
Which of the following are the common oxidising agents used in redox titrations ? |
|
Answer» `K_(2)Cr_(2)O_(7), KMnO_(4)`, Iodine |
|
| 14. |
Write the resonance structures for N_(2)O |
|
Answer» SOLUTION :Nitrous OXIDE `N_(2)O` : `UNDERSET(..)overset(..)N = N =underset(..)overset(..)O hArr :underset(..)overset(..)N = N = O: :N = N = underset(..)overset(..)O:` |
|
| 15. |
Which one of the following is an electron deficient compound? |
|
Answer» 1) `Al_(2)Cl_(6)` |
|
| 16. |
Z the position number of NO_(2) Group X,Y,Z aer P,Q,R respectively. The value of P+Q+R is |
Answer» 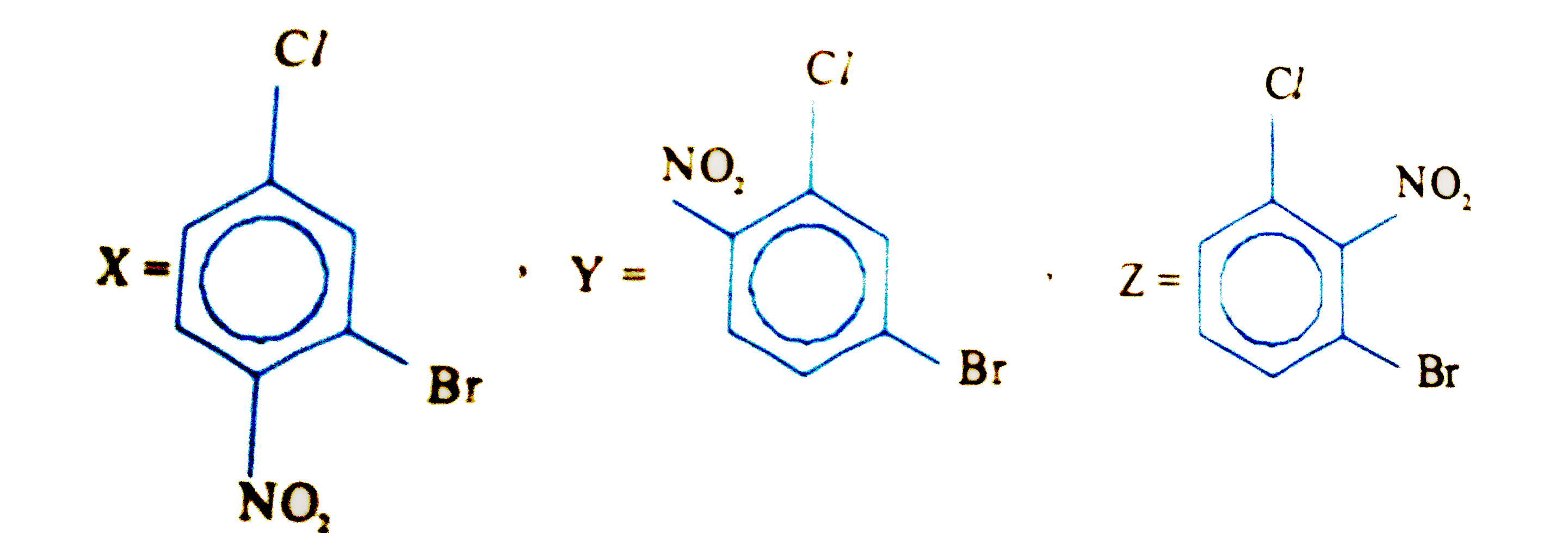
|
|
| 17. |
What happens to the ph of a solution of Acetic acid when some sodium acetate is dissolved in it? Explain. |
|
Answer» SOLUTION :`pH` of the ACID solution increases. Because of COMON ion effect dissociation of acetic acid DECREASED and henc `[H^(+)]` DECREASES. |
|
| 18. |
Which compound will yield 5-keto -2-methyl hexanal upon treatment with O_3 ? |
|
Answer»

|
|
| 19. |
Which of the following reactions is not an electrophilic addition reactions - |
|
Answer»
|
|
| 20. |
What is stone leprosy? How is it formed? |
|
Answer» SOLUTION :The attack on the marble of BUILDINGS by acid rain is called stone LEPROSY (ii) Acid rain causes EXTENSIVE damage to buildings MADE up of marble. `CaCO + H_(2)SO_(4)to CaSO_(4) + H_(2)O + CO_(2)uarr` |
|
| 21. |
Which of the following is an example of homogeneous equilibrium ? |
|
Answer» `H_(2) O (l) hArr H_(2) O (g)` |
|
| 22. |
The standard enthalpies of combustion of C_(6)H_(6)(l), C("graphite") and H_(2)(g) are respectively -3270 kJ"mol"^(-1), -394 kJ"mol"^(-1) and -286 kJ"mol"^(-1). What is the standard enthalpy of formation of C_(6)H_(6)(l) in kJ"mol"^(-1)? |
|
Answer» `-48` (II) `C(gr) + O_(2)(g) to CO_(2)(g) DeltaH = -394 kJ"mol"^(-1)` (iii) `H_(2)(g) + 1/2O_(2)(g) to H_(2)O(l) DeltaH = -286 kJ"mol"^(-1)` The required equation is (iv) `6C(gr) + 3H_(2)(g) to C_(6)H_(6)(l),DeltaH` = ? Multiply eq.(ii) by 6 and eq(ii) by 3 and adding we get : (v) `6C(gr) + 15/2O_(2)(g) + 3H_(2)(g) to 6CO_(2)(g) + 3H_(2)O(l), DeltaH = -3222 kJ` Subtract eq.(i) from eq.(v) `6C(gr) + 3H_(2)(g) to C_(6)H_(6)(l) DeltaH = + 48 kJ"mol"^(-1)`. |
|
| 23. |
The standard enthalpy of formation of FeO and Fe_(2)O_(3) is -65kcal mol^(-1) and -197kcal mol^(-1) respectively. A mixture of two oxides contains FeO and Fe_(2)O_(3) in the mole ratio 2:1. If by oxidation, it is changed into a 1:2 mole ratio mixture, how much of thermal energy will be released per mole of initial mixture ? |
|
Answer» |
|
| 24. |
Which one of the following is not paramagnetic- |
|
Answer» `O_(2)` |
|
| 25. |
What is bond order according to Lewis ? Write structure and bond order of H_(2) , O_(2) ,N_(2), CO, NO. |
Answer» Solution :In the LEWIS DESCRIPTION of covalent BOND the Bond Order is given by the number of bonds between the two ATOMS in a molecule.
|
|
| 26. |
Which of the following reactions is an example ofredox reactions ? |
|
Answer» `XeF_(4) + O_(2) F_(2) RARR XeF_(6) + O_(2)` |
|
| 27. |
Which of the following orbitals will have zero probability of finding the electron in the yz plane ? |
|
Answer» <P>`p_(X)` |
|
| 28. |
Which of the following gives Tollen 's test |
|
Answer» ACETYLENE and propyne |
|
| 29. |
Match List-I with List-II and select the correct option. |
|
Answer» P, R |
|
| 30. |
Which of the following has incorrect relation |
|
Answer»
 They are not ISOMERS MF is DIFFERENT. |
|
| 31. |
Which of the following is not a general characteristic of equilibrium involving physical process? |
|
Answer» Equilibrium is possible only in a closed system at a given temperature |
|
| 32. |
Which of the following is less stable thermally- |
|
Answer» LiF |
|
| 33. |
What are the uses of Beryllium? |
|
Answer» Solution :(i) Due its low atomic number and very low absorption for X-rays, it is used as radition windows for X-ray tubes and X-ray detectors. (ii) The sample holder in X-ray EMISSION studies USUALLY made of beryllium (III) Since beryllium is transparnet to energetic particles it is used to build the 'beam PIPE' in accelerators. (IV) Because of its low density and diamagnetic nature, it is used in various detectors. |
|
| 34. |
Which ofthe followingelementshas mostpositiveelectrongainenthalpy ?Fluorinenitrogen neon. |
|
Answer» SOLUTION :Bothnitrogenand neonhave positive electrongainenthalpies. Butneonhas muchhigherpositiveelectron gain enthalpy because of itsmuch morestable INERTGAS CONFIGURATION than the lessstableexactlyhalf-filledelectronicconfigurationof nitrogen. Alternatively in NEON , the newelectronhas to beplacedin a muchhigherenergy 3S- orbital of the newshellwhilein case of N , thenewelectronis toplacedin the 2p-orbital of the sameshell. |
|
| 35. |
Which gas effuses fastest under identical conditions? |
|
Answer» `N_(2)` |
|
| 36. |
Which statement is correct? |
|
Answer» It is the product of ionic concentration of a slightlysoluble salt in its saturated solution at a CERTAIN temp. |
|
| 37. |
When SO_2is passed in a solution of potassium iodate, the oxidation state of iodine changes from |
|
Answer» `+5 " to " 0` |
|
| 38. |
Which of the following is a heterogeneous catalysis? |
|
Answer» `2C_(2)H_(5)OH OVERSET("Conc."H_(2)SO_(4))rarrC_(2)H_(5)OC_(2)H_(5)+H_(2)O` |
|
| 39. |
Write suitable regent to distinguish the following compounds. |
|
Answer» |
|
| 40. |
Which of the following concentration terms is temperture independent (i) Molarity (ii) Molallity (iii) Normality (iv) Mole frection |
| Answer» ANSWER :D | |
| 41. |
What is the role of CaO in sodalime ? |
| Answer» SOLUTION : It HELPS in the FUSION of REACTION MIXTURE. | |
| 42. |
Which concept can justify that CaO + SO_(3) rarr CaSO_(4)is an acid-base reaction ? |
| Answer» SOLUTION :LEWIS CONCEPT. | |
| 44. |
The vapour pressure of any liquid is………………proportional to the magnitude of the intermolecular forces and is…………….proportional to the temperature employed. |
| Answer» SOLUTION :INVERSELY, DIRECTLY | |
| 45. |
There is a collection of crystalline substances in a hexagonal close packing. If the density of matter is2.6 g//cm^(3), what would be the average density of matter in colleciton ? What fractionofthe space is actually unoccupied ? |
|
Answer» Solution :In hexagonal CLOSE packing, packingefficiency is 74 % Density of matter= Packing FRACTION ` xx ` TOTAL density = ` 74/100 xx 2.6 = 1.924 " g/cm"^(3)` % EMPTY space ( space unoccupied )= 100 - 74 = 26% . |
|
| 46. |
Which of the following phenomena does not involve surface tension ? |
|
Answer» Mercury drops acquire SPHERICAL shape . |
|
| 47. |
Which one is biodegradable substance ? |
|
Answer» Paper |
|
| 48. |
Which one of the following contains most acidic hydrogen atom. |
|
Answer»
|
|
| 49. |
What gas leaked to bring havoc in Bhopal tragedy ? |
| Answer» SOLUTION :METHYLISOCYANATE (MIC). | |