Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the correct stability order of oxy anions given below ? |
|
Answer» `(II) GT (I) gt (IV) gt (III)` |
|
| 2. |
Whichunits are used to measure fraction of mass ? |
|
Answer»
|
|
| 3. |
Which statement are correct ? |
|
Answer» The ENERGIES of the virous sub-levels in the same shell of H-atom are in order `s gt p gt d gt f`. |
|
| 4. |
Which of the following has coordinate covalent bond? |
|
Answer» `H_(2) O ` |
|
| 5. |
Write the cell reaction for each of the following cells. Pt,H_(2)(g)|H^(+)(aq)|Ag^(+)(aq)|Ag(s) |
| Answer» Solution :`H_(2)(G)+2AG^(+)(AQ)to2H^(+)(aq)+2Ag(s)` | |
| 6. |
Which of the following compounds will show cis-trans isomerism ? (i) (CH_(3))_(2)C=CH-C_(2)H_(5) (ii) CH_(2)=CBr_(2) (iii) C_(6)H_(5)CH=CHC_(6)H_(5) (iv) CH_(3)CH=C ClCH_(3) |
Answer» Solution :(i) `(CH_(3))_(2)C=CH-C_(2)H_(5)` : This structure do not show isomerism because ONE carbon having two same `CH_(3)` groups. This structure do not show isomerism because ONE carbon having two same `CH_(3)` groups. (ii) `CH_(2) = CBr_(2)` :  This COMPOUND do not show isomerism because two same FUNCTIONAL group is present on one carbon. This COMPOUND do not show isomerism because two same FUNCTIONAL group is present on one carbon. (III) `C_(6)H_(5)CH = CHC_(6)H_(5)` : This compound shows isomerism because carbon is having different groups.  (IV) `CH_(3)CH=C ClCH_(3)` : This compound shows isomerism as both carbon is having different groups. 
|
|
| 7. |
When the first element of the periodic table is treated with dioxygen, it gives a compound whose solid state floats on its liquid state. This compound has an ability to act as an acid as well as a base. What products will, be formed when this compound undergoes autoionisation ? |
|
Answer» Solution :The first element of the periodic table is hydrogen and its molecular FORM is dihydrogen `(H_2)`. When dihydrogen reacts with dioxygen water is formed. Water is a liquid at room temperature. When liquid water freezes, it expands to form ice. In other words DENSITY of ice is lower than that of liquid water and hence ice floats over water. Water is amphoteric in nature. It acts as an acid in presence of STRONG bases and as a base in presence of strong acids. `underset("Base"_1)(H_2O_((l)))+ underset("Acid"_2)(H_2S_((aq)))to underset("Acid"_1)(H_3O_((aq))^(+))+ underset("Base"_2)(HS_((aq))^(-))` `underset("Acid"_1)(H_2O_((l)))+ underset("Base"_2)(NH_(3(aq))) to underset("Acid"_2)(NH_(4(aq))^(+)) + underset("Base"_1)(OH_((aq))^(-))` Due to amphoteric character , water undergoes self ionisation as FOLLOWING : `underset("Acid"_1)(H_2O_((l)))+ underset("Base"_2)(H_2O_((aq)))hArr underset"(Conjugate acid)"underset("Acid"_2)(H_3O_((aq))^+)+ underset"(Conjugate acid)"underset("Base"_1)(OH_((aq))^(-))` This self ionisation of water is called auto protolysis. |
|
| 8. |
What is thedifferencebetweenphotonandquantum ? |
| Answer» SOLUTION :Photonis onlyfor lightbutqunatumis apacketof ENERGY`(E= 2Sigma )` and itcanbe fromanysource | |
| 9. |
What happens when bromoethane is treated with moist silver oxide? |
|
Answer» SOLUTION :When bromoethane is treated with MOIST silver oxide. ethanol will be formed as PRODUCT: `underset("Bromoethane") (CH_(3) - CH_(2) Br) + underset("silver oxide") underset("Moist") (AgOH) overset("Boil") CH_(3) underset("Ethanol") (- CH_(2) OH) + AgBr` |
|
| 10. |
Which one of the following reactions does not correspond to the preparation of ..Synthetic gasoline.. during the Fscher-Tropsch process ? |
|
Answer» `CO+3H_(2)toCH_(4)+H_(2)O` |
|
| 11. |
Which of the following compounds gives blood red colouration when its Lassaigne's extract is treated with alkali and ferric chloride? |
|
Answer» THIOUREA |
|
| 12. |
Which of the following hydrogen bonds is the strongest ? |
|
Answer» O - H ---F Because of HIGHEST electronegativity of F, HYDROGEN bonding in F - H - - - F is STRONGEST |
|
| 13. |
The viscosity order of water , n - hexane and glycerolis |
|
Answer» n - hexane `GT` water `gt` glycerol |
|
| 14. |
Write the descending order of electron affinity values of chalcogens. |
|
Answer» Solution :Decreasing ORDER of electron affinity values of chalcogens: S > Se > TE > O Electron affinity of oxygen is less because oxygen hassmall atomic size and the added electron EXPERIENCES greater repulsion on oxygen atom. |
|
| 15. |
The Schrodinger wave equation for hydrogen atom is Ps ("radial") =1/(16sqrt4) ((Z)/(a_0))^(3//2) [ (sigma -1) (sigma^2 - 8 sigma + 12 )]e^(-sigma//2) where a_0 and z are the constant in which answer can be expressed and sigma =(2pi)/(a_0) minimum and maximum position of radial nodes from nucleus are |
|
Answer» `(a_0)/(Z), (3a_0)/(Z)` `IMPLIES(sigma -1) = 0 impliessigma =1` or `r_1 =(a_0)/(2z) ` or `(sigma^2 - 8 sigma + 12) =0 ` and `(sigma-6 ) (sigma-2)=0` `sigma=6, r= (3a_0)/(z) , sigma = 2 , r = (a_0)/(z) , r_2 = (3a_0)/(z)` |
|
| 16. |
The volume of gas is reducedto half from its original volume. The specific heat will "…................". |
|
Answer» REDUCE to half |
|
| 17. |
Which one of the following contains ionic , covalent and coordinate bonds ? |
|
Answer» `NaOH` coodinate BONDS. |
|
| 18. |
Which of the following has maximum bond angle ?H_(2)O, CO_(2), NH_(3), CH_(4). |
| Answer» Solution :`CO_(2), 180^(@)` (due to LINEAR STRUCTURE) | |
| 19. |
Which compond of sodium is used for washing purposes in laundry ? |
| Answer» SOLUTION :`Na_2CO_3` | |
| 20. |
You have the mixture of three liquids (A), (B) and (C). There is a large difference in the boiling points of (A) and rest of the two liquids i.e. (B) and (C). Boiling point of liquids (B) and (C) are quite close. Liquid [A] boils at a higher temperature than [B] and [C] and boiling point of (B) is lower than that of (C). How will you separate the components of the mixture. draw a diagram showing set up of the apparatus for the process. |
|
Answer» Solution :(i) SUBJECT the mixture of three liquid to simple distillation. Both liquids (B) and (C) will distil since these are low boiling, leaving behind liquid (A) in the distillation flask. This can be recovered from the flask. (II) Now subject the mixture of liquids (B) and (C) obtained as distillate to farctional distillation. Liquid (B) will GET distilled since it has less boiling point. Lquid (C) will be LEFT in the distillation flask. In this manner, the separation can be DONE. The separation can be represented by a flow sheet diagram.  For the apparatus of simple distillation, consult section 13.45 For the apparatus of fractional distillation, consult section 12.36. |
|
| 21. |
Which of the following anions is present in thechain structure of silicated ? |
|
Answer» `SiO_(4)^(4-)` |
|
| 22. |
There are equal volumes of N_2 and CO_2 gases at 25^@C and 1 atm. Then indicate the correct statements among the following |
|
Answer» The RMS velocity remains the same for `N_2` and `CO_2` gases average `KE = 3/2 KT , d alpha M` Total KE = `3/2 NRT`. |
|
| 23. |
To which of the following species, the Bohr theory is not applicable ? |
| Answer» Answer :A::C | |
| 24. |
Which type of mix solution Ba(OH)_2 of 20 ml 0.01 M H_2SO_4 and 20 ml 0.01 M ? |
| Answer» Answer :A | |
| 25. |
Which are the conjugate base of strong acid HClO_4, H_2SO_4, HNO_3 and H_3PO_4 ? It is strong or weak ? |
| Answer» SOLUTION :`ClO_4^(-) , HSO_4^(-), NO_3^(-)`and `H_2PO_4^(-)` all are CONJUGATE BASE of STRONG ACID | |
| 26. |
Which calcium compound is not aapreciably more soluble in 0.1 M hydrochloric acid than it is in pure water? |
|
Answer» Limestone, `CaCO_(3)` |
|
| 27. |
Whodevelopedfundamentalequationofquantummechanics ? |
| Answer» SOLUTION :The fundamentalequantumof quantummechanicswas DEVELOPED by ErwinSchrodingerphysisin 1933. | |
| 28. |
Which of the following oxides shows electrical properties like metal ? |
|
Answer» `SiO_2` |
|
| 29. |
The correct match is |
|
Answer» II 
|
|
| 30. |
The van der Waals constant 'b' for oxygen is 0.0318 L mol^(-1). Calculate the diameter of the oxygen molecule. |
|
Answer» Solution :b=4 vor`v=(b)/(4)=(0.0318)/(4)=7.95xx10^(3) L MOL^(-1)=7.95 cm^(3) mol^(-1)` `:. "Volume occupied by one " O_(2) " molecule"=(7.95)/(6.02xx10^(23))=1.32xx10^(-23)cm^(3)` Cosidering the molecule to be SPHERICAL, `(4)/(3)pi r^(3)=1.32xx10^(-23)" or " r^(3)=3015xx10^(-24)` `:. 3 log r=log(3.15xx10^(-24))=-24+0.4983=-23.5017` or`log r=-7.8339=bar(8).1661` `:. "Diameter of oxygen molecule "=2xxr=2xx1.466xx10^(-8)cm=2.932xx10^(-8)cm=2.932Å` |
|
| 31. |
Why boron trihalides act as Lewis acids ? |
| Answer» Solution :The boronatom intrihaldieshas only SIX electrons in the VALENCE shell and HENCE canaccept a pair ofelectronsin the VACANT p-orbitalto completeits octet. As a RESULT,boron trihaldiesact as aLewis acids. | |
| 32. |
Write one chemical reaction for the preparation of D_(2)O_(2). |
|
Answer» Solution :By the ACTION of `D_(2)SO_(4)` in dissolved in WATER over `BaO_(2)` `BaO_(2) + D_(2)SO_(4) to BaSO_(4) + D_(2)O_(2)` |
|
| 33. |
Which product is obtained when benzene is heated with CH_(3)Cl in presence of AlCl_(3) ? Why ? |
Answer» Solution :Toluene and MIXTURE of o-and p-XYLENE is obtained when BENZENE is heated with `CH_(3)Cl` in presence of `AlCl_(3)`.  We ALWAYS write the reaction of toluene from the benzene. But the reactivity of `-CH_(3)` group oftoluene, which is ortho, para DIRECTING, on alkylation of toluene ortho xylene and para xylene is obtained. |
|
| 34. |
When the speed of electron increases, its specific charge |
|
Answer» Increases |
|
| 36. |
Two students performed the same experiment separately and each one of them recorded two readings of mass which are given below. However, the correct reading is 5.0 g. On the basis of the data, mark the correct option {:("Student",:underset("(i)(ii)")("Readings")),("A","5.014.99"),("B","5.054.95"):} |
|
Answer» RESULTS of both the students are neither accurate nor PRECISE |
|
| 38. |
When will be K_(p) lt K_C ? Give two example |
|
Answer» <P> Solution :When `Delta n_(G) = - ve K_(p) lt K_(c). (RT)^(-vc)``K_(P) lt K_(c) .` Examples `2H_(g) + O_(2)(g) hArr 2H_(2) O(g)` `Delta n_g = 2-3 = -1` `K_p = K_c. (RT)^(-1)` `thereforeK_(p) lt K_(c)` |
|
| 39. |
Which law is signified by the equation : V_t = V_0 (1+ t/273) State the law in a different manner. |
| Answer» SOLUTION :CHARLES. LAW. | |
| 41. |
The temperature at which the solid and liquid phases of a substance are at equilibrium is called ………….. |
| Answer» SOLUTION :FREEZING POINT | |
| 42. |
What happens when butanoic acid is heated with soda-lime at 630 K ? Is there any other acid which also gives the same product under similar conditions. Write its name and the reaction involved . |
|
Answer» Solution :Butanoic ACID undergoes decarboxylation to give propane when heatedwith soda-lime at 630 K . `underset"Butanoic acid"(CH_3CH_2CH_2COOH)+2NaOH underset"(Decarboxylation)"OVERSET"CaO, 630 K"to underset"Propane"(CH_3CH_2CH_3)+ Na_2CO_3+ H_2O` 2-Methylpropanoic acid is the position isomer of butanoic acid. This ALSO on decarboxylation with soda-lime give propane. 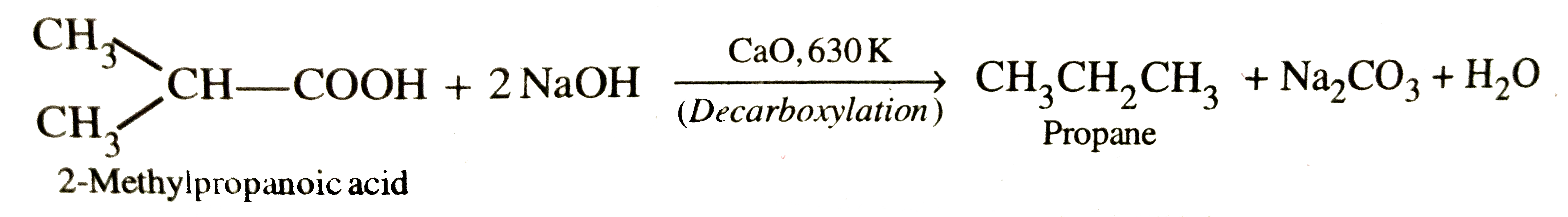
|
|
| 43. |
What is the difference between atomic mass and mass number ? |
| Answer» Solution :MASS NUMBER is a whole number because it is the sum of number of protons and number of neutrons whereas ATOMIC mass is fractional because it is the average relative mass of its atoms as compared with mass an atom of `C - 12` ISOTOPE TAKEN as 12 | |
| 44. |
Which one of the following pairs of species have the same bond order ? |
|
Answer» `NO^(+) and CN^(+)` In the given pair of species NUMBER of electron in `NO^(+)` = number of electron in `CN^(-)` = 14 electrons. So they are isoelectronic in nature. HENCE bond order of these two species will be also si.milar which is shown below. `N^(+) rarr sigma 1S^(2) sigma^(**) 1s^(2) sigma 2s^(2) sigma^(**) 2s^(2) sigma 2p_(z)^(2) pi 2p_(x)^(2) pi 2p_(y)^(2)` B.O =1/2 `[N_(b) - N_(a)] ` = 1/2 [ 10 - 4] or B.O = 3 `CN^(-) rarr sigma 1s^(2) sigma^(**) 1s^(2) sigma 2s^(2) sigma^(**) 2s^(2) pi 2p_(x)^(2) pi 2p_(y)^(2) sigma 2p_(z)^(2)` B.O = 1/2 [ 10 - 4] or B.O = 3 |
|
| 45. |
What is wurtz reaction |
| Answer» SOLUTION :WHENA solutionof haloalkane in dryetheris treatedwithsodiummetalhigheralkanesareproduce.thisreactionis knowas WurtzreactionForexample<BR> `CH_(3) - Br +2Na+ Br -CH_(3)overset(" dryether") (to)CH_(3)- CH_(3) + 2NaBr` | |
| 46. |
Which of the following gases will have least volume if 10 g of each gas is taken at same temperature and pressure? |
|
Answer» `CO_(2)` Molecular MASSES : `CO_(2)=44,N_(2)=28,CH_(4)=16`,and `HCI =36.5` `CO_(2)` will have lesast volume. |
|
| 47. |
You are given normal boiling points and standard enthalpies of vaporization, Calendly. the entropy of vaporization of liquids listed below. |
|
Answer» Solution :For ETHANOL : GIVEN : `T_b=78.4^@C=(78.4+273)` =351.4 K `DeltaH_V` (ethanol)=`+42.4 "KJ MOL"^(-1)` `DeltaS_V=(DeltaH_V)/T_b` `DeltaS_V=(+42.4 "kJ mol"^(-1))/"351.4 K"` `DeltaS_V=(+42.400 "J mol"^(-1))/"351.4 K"` `DeltaS_V=+120.66 J K^(-1) "mol"^(-1)` For Toluene : Given : `T_b=110.6^@C`=(110.6+273) =383.6 K `DeltaH_V`(toluene )=+35.2 kJ `"mol"^(-1)` `DeltaS_V=(DeltaH_V)/T_b` `DeltaS_V=(+35.2 "kJ mol"^(-1))/(383.6 K)` `DeltaS_V=(+35200 J mol^(-1))/(383.6 K)` `DeltaS_V=+91.76 J K^(-1) mol^(-1)` |
|
| 48. |
Which of the following Statement are Correct ? (1) DeltaH=DeltaU+Deltan_(g)RT for chemical and phase change (2) DeltaH=DeltaU"for the reation" (3) C(s)+O_(2)(g)toCO_(2)(g) (4) Heat of formation for C_(6)H_(6)can be calculated by bond enthalpies only |
|
Answer» 2,3 |
|
| 49. |
What is the action of alkali metals with hydrogen? |
|
Answer» Solution :ALKALI metals react with hydrogen to form ionic hydries. `2M + H_(2) overset(Delta)(to) 2M^(+) H^(-)` (Metal hydride) Ex: `2Li + H_(2) overset(1.073 K)(to) 2Li^(+) H^(-)` (Lithium hydride) `2Na + H_(2) overset(673K)(to) 2Na^(+) H^(-)` (sodium hydride). |
|
| 50. |
What is the percentage hydrolysis of NaCN in N/80 solution when the dissociation constant for HCN is 1.3xx10^(-9) and K_(w)=1.0xx10^(-14) |
|
Answer» 2.48 |
|