Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
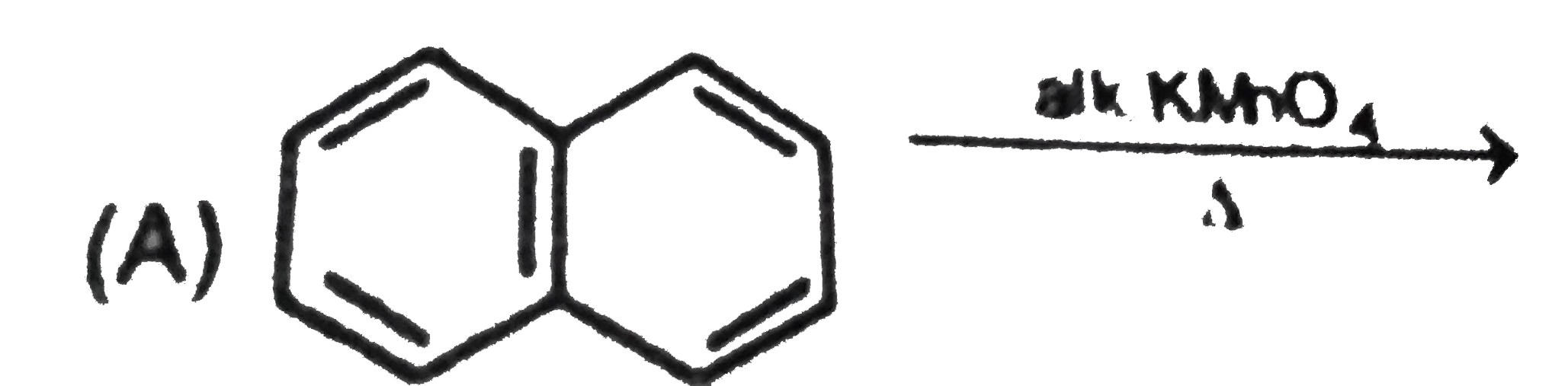
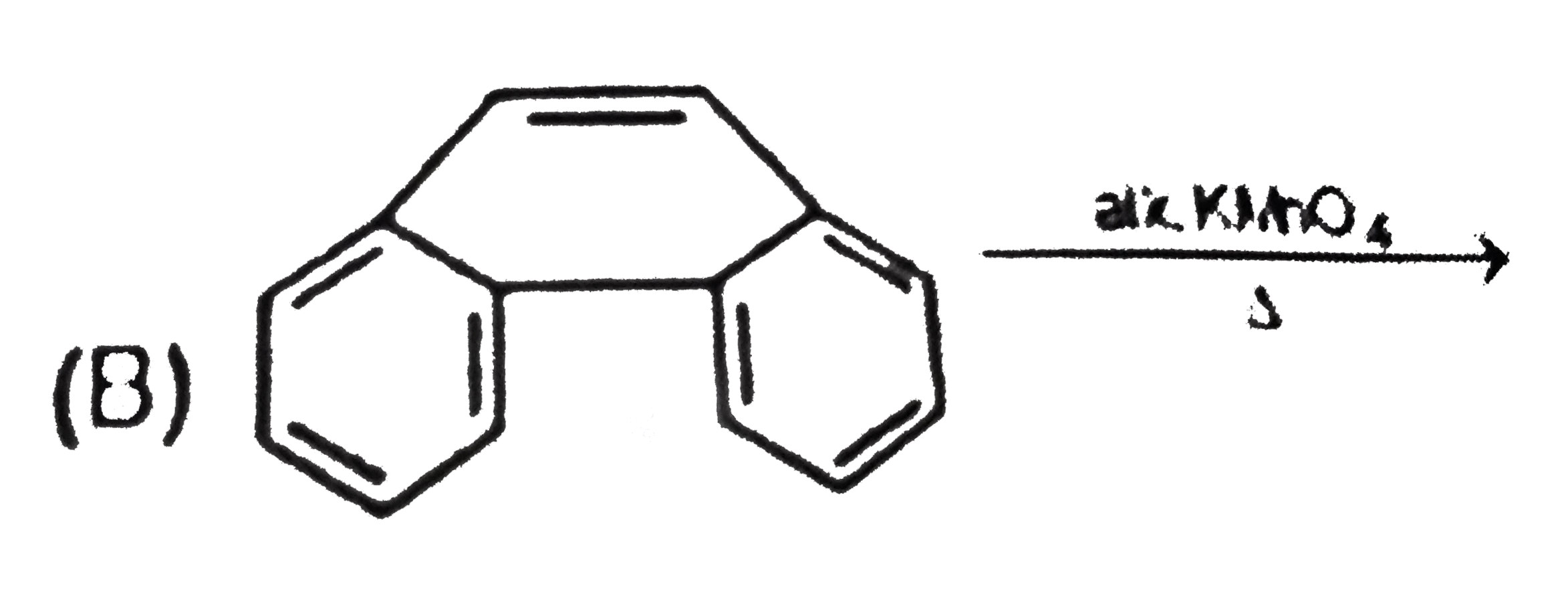
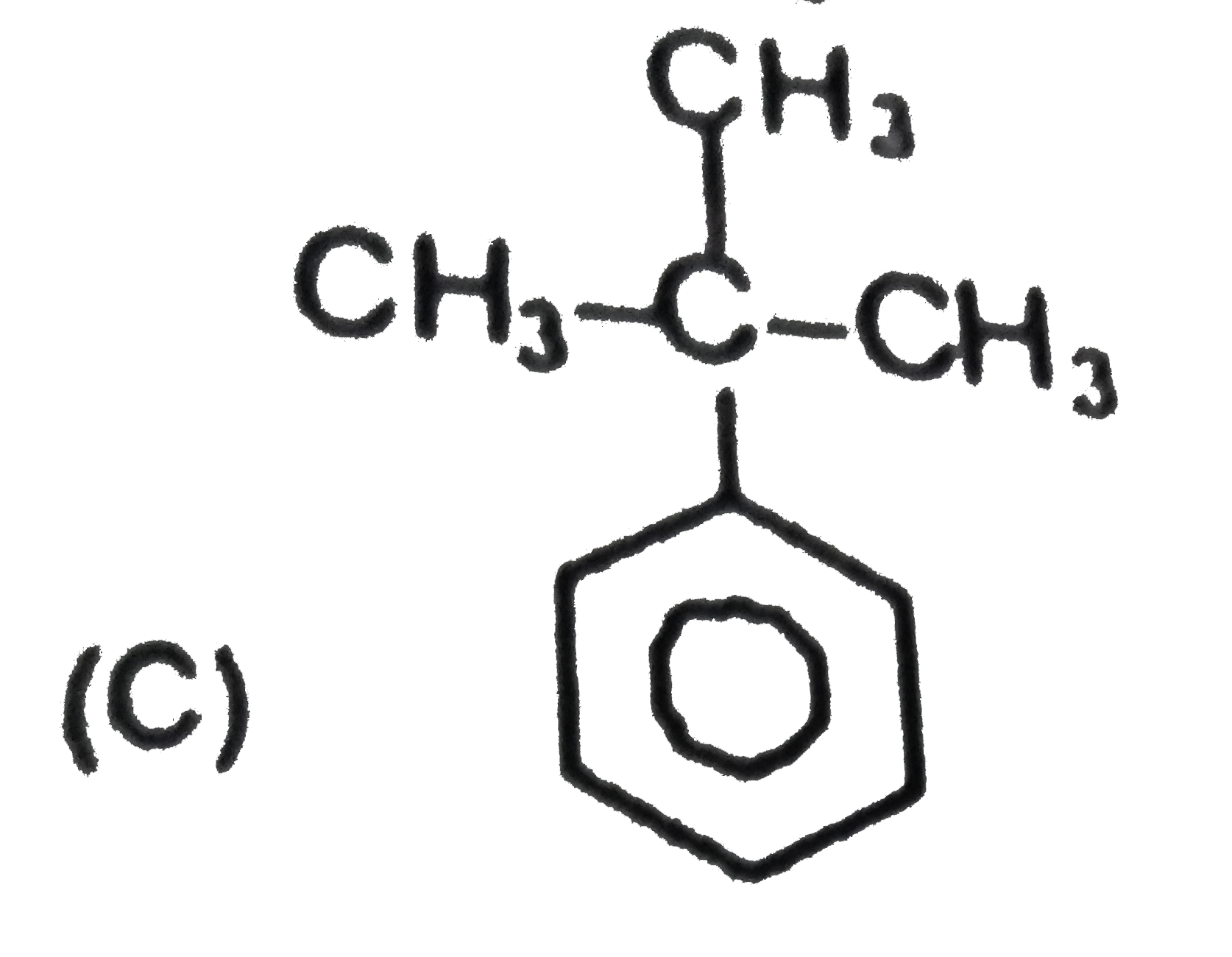
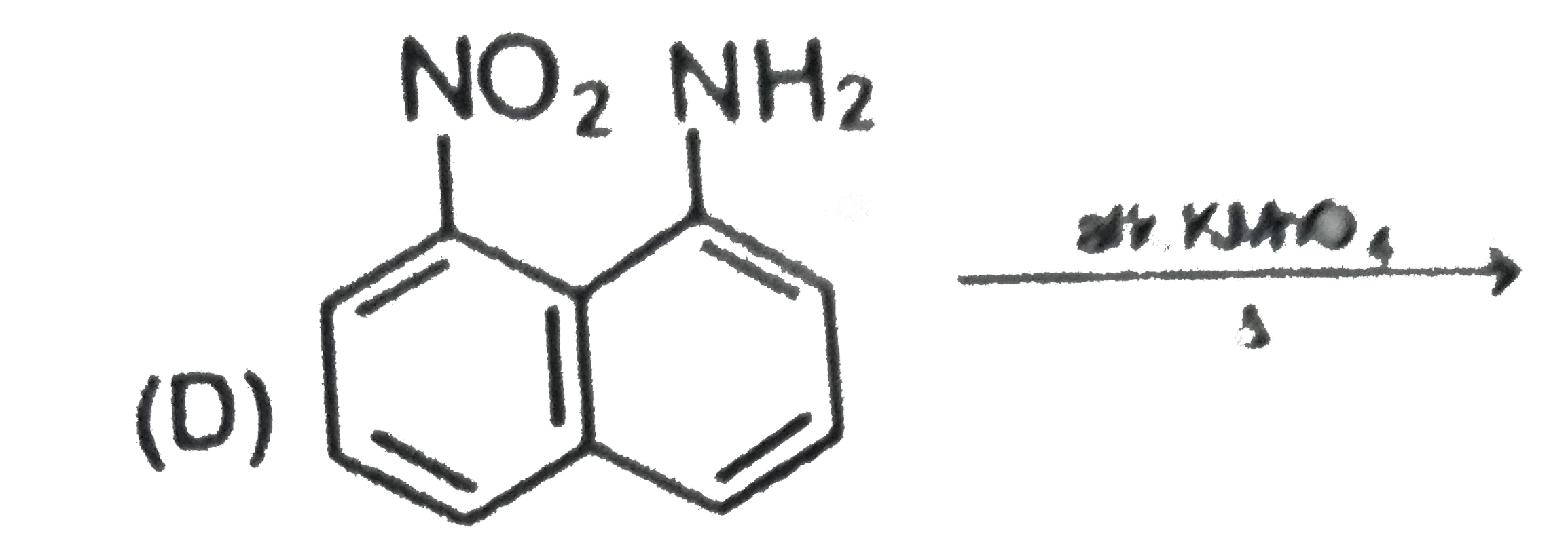
Two isomeric forms of an organic compound (A), C_11H_13OCI readily decolourise bromine water and give same compound (B) on catalytic hydrogenation. Both the isomeric forms on vigorous oxidation give (C) which on treatment with soda lime gives 2-chloro ethoxy benzene. However, (C) treatment with Ni/Al alloy in alkaline medium gives 3-ehtoxy benzoic acid. Only one of the isomers of (A) gives geometrical isomers (D) and (E). Identify (A) to (E) with proper reasoning. |
Answer» Solution :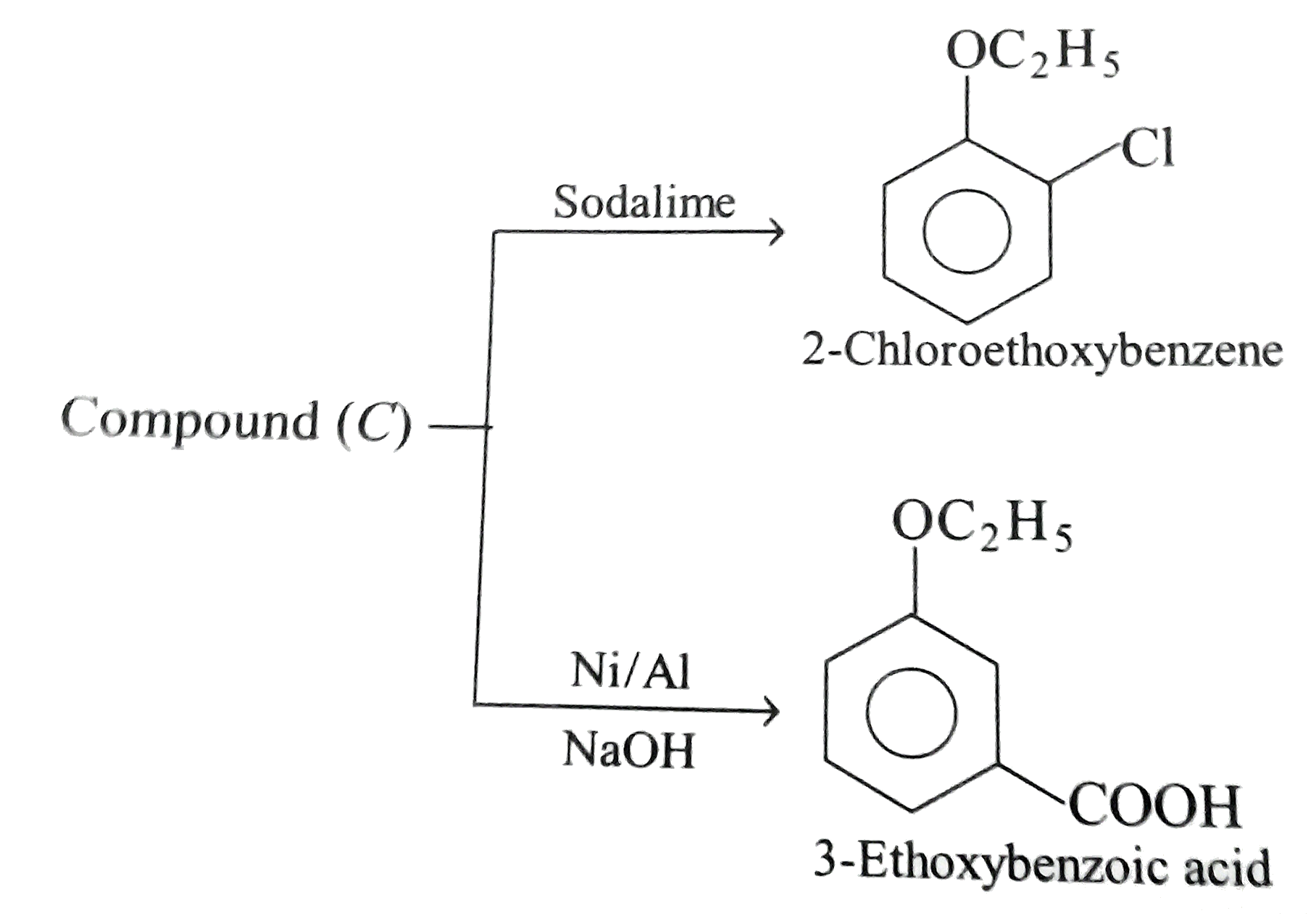 HENCE,the structure of compound (C) can be, 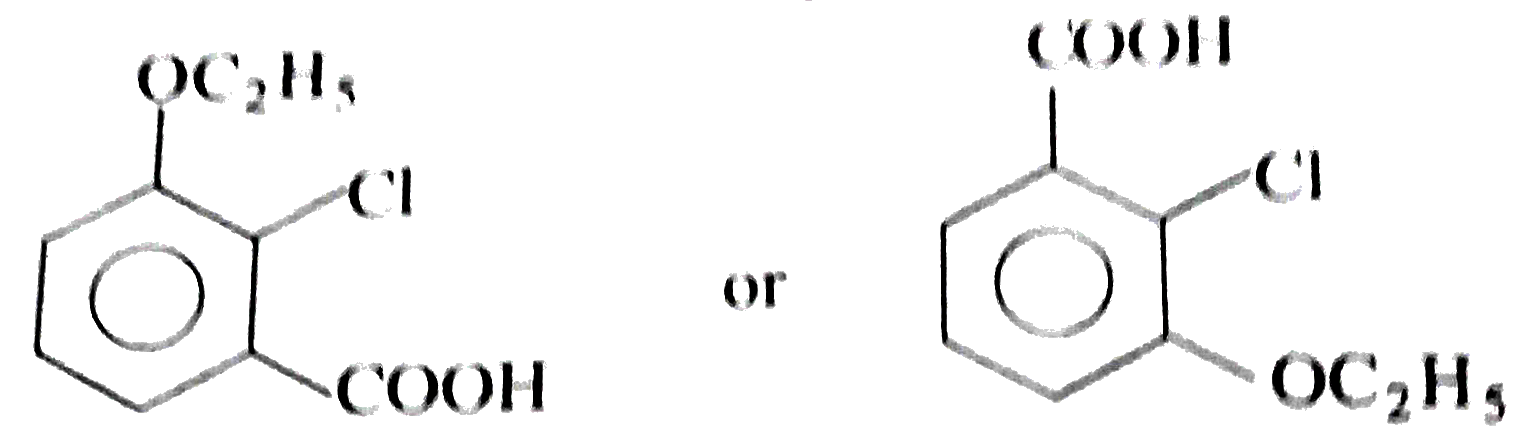 The compound (A) is unsaturated which POSSESSES `-CI` and `-OC_2H_5` groups also. The TWO isomers of (A) are 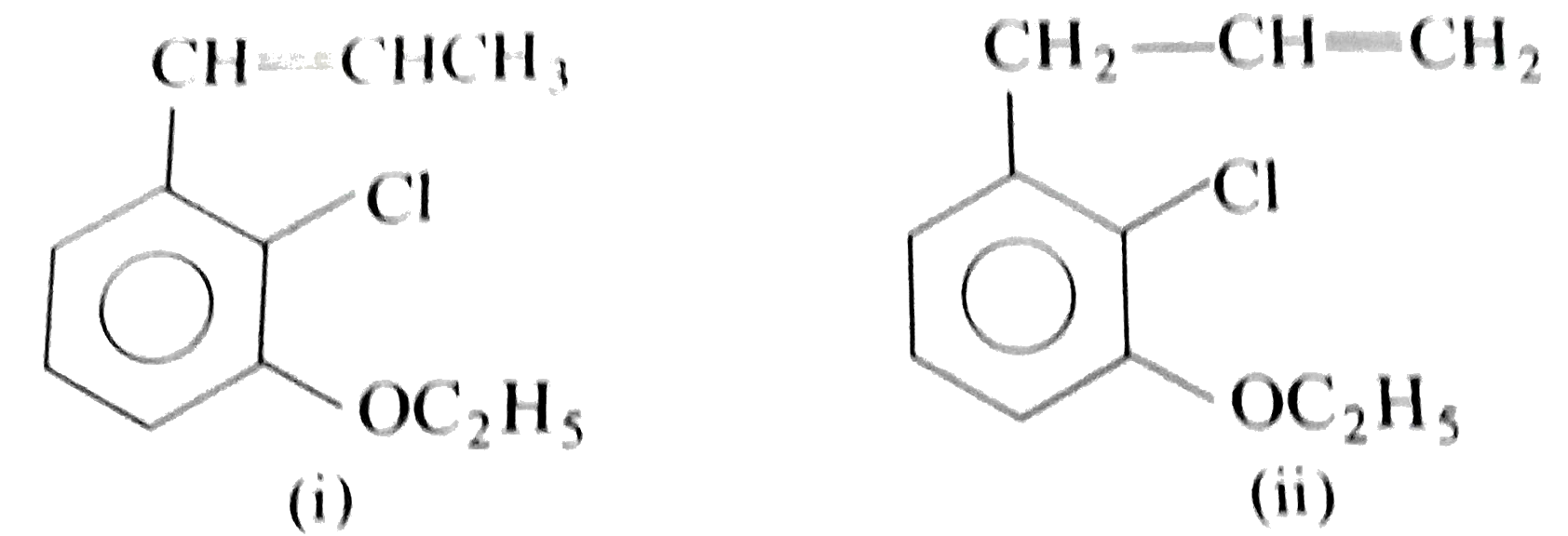 Both the isomers decolourise bromine water and on hydrogenation give the compound (B) 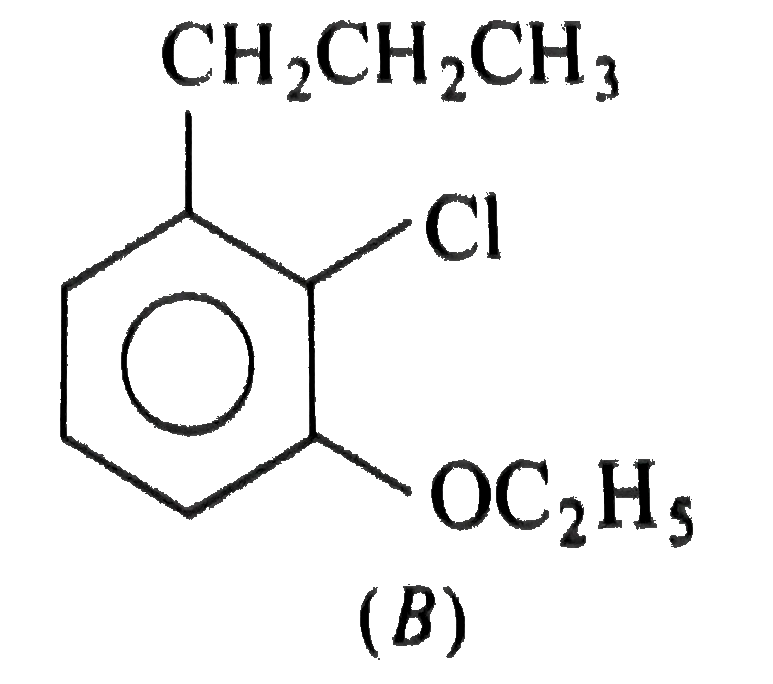  (i) isomer of (A) shows GEOMETRICAL isomerism, (D) and (E). 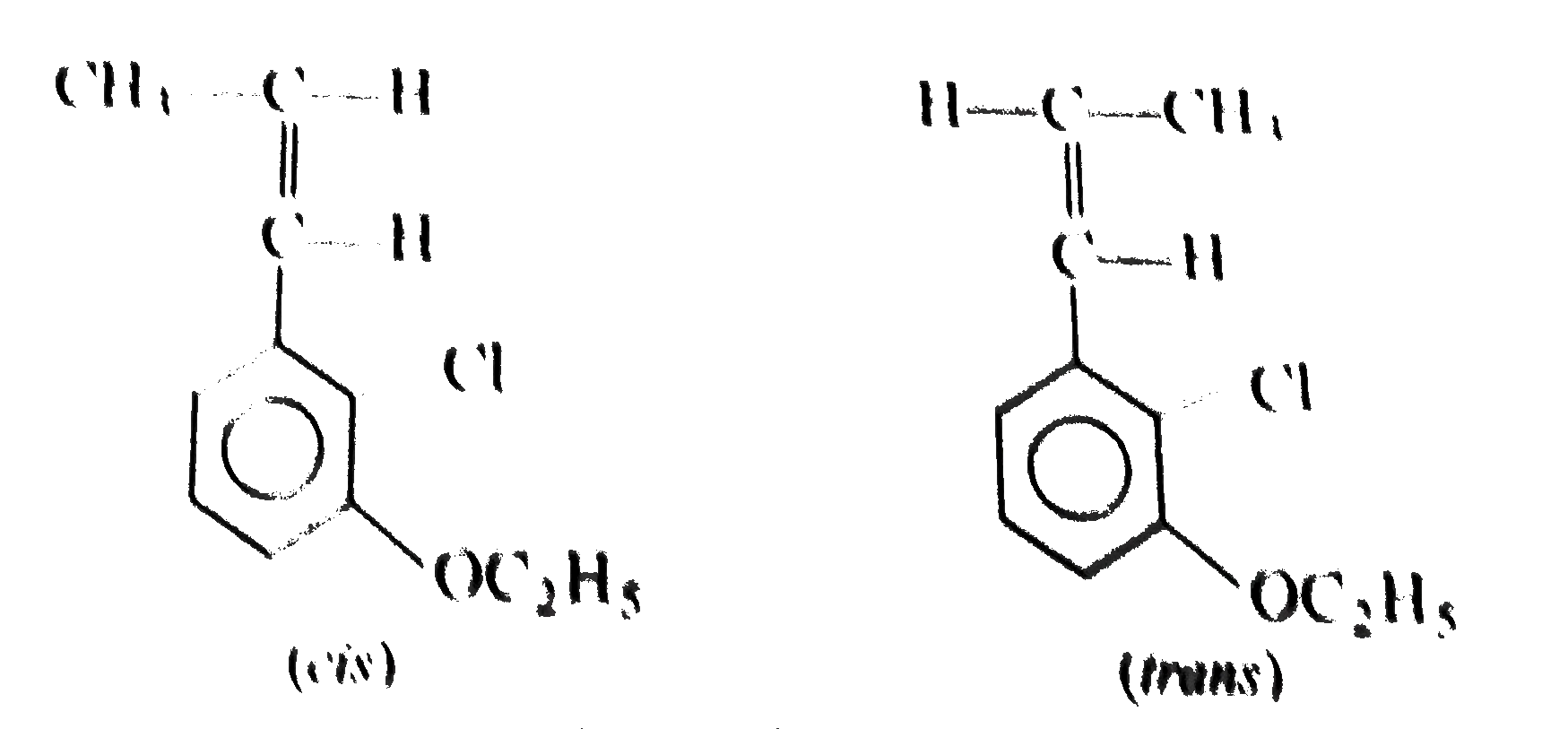
|
|
| 2. |
Which of the following will react with sodium metal ? |
|
Answer» `C_2H_6` |
|
| 3. |
The table below shows the data for three titrations to determine the concentration of a NaOH solution With standard 0.200 M HCl solution using phenolphthalein as the indicator Which explanation best accounts for the lower value of the NaOHM in Trial 3 ? |
|
Answer» Some of the neutralized solution from Trial 2 was left in the flask for Trial 3. |
|
| 4. |
The standard enthalpies of formation of CO_(2) gas and HCOOH_((l)) are -393.7 kJ mol^(-1) and -409.2 kJ mol^(-1) respectively. Which of the following are correct? |
|
Answer» `C_((s)) + O_(2(G)) RARR CO_(2(g)), DELTA H = - 393.7 kJ mol^(-1)` `C + H_(2) + O_(2) rarr HCOOH, Delta H_(2) = - 409.2` `H_(2)CO_(2) rarr HCOOH` `Delta H = Delta H_(2) - Delta H_(1) = - 15.5KJ` |
|
| 5. |
Which of the following ions does not show resonance? |
| Answer» Answer :C | |
| 6. |
What is Dipole - induced dipole forces ? Give its characteristic. |
|
Answer» Solution :Dipole - induced dipole forces : This type of attractive forces operate between the POLAR molecules having permanent dipole and the molecules lacking permanent dipole. ex. `HCl - H_(2), CO_(2)` (non Polar) in air `O_(2)-H_(2)O`. Formation of Dipole - Dipole forces : Permanent dipole of the polar molecule induces dipole on the electrically neutral molecule by deforming its electronic cloud.  When permanent polar and non polar molecule come NEAR to each other then dipole - induced dipole forces becomes reactive. Permanent dipole of the polar molecule induces dipole on the electrically neutral molecule by deforming its electronic cloud. So, polarity developed in non polar molecule.  There are attraction forces PRESENT between polar molecule (AB) and induced dipole molecule `(X_(2))`. Which is interaction forces between dipole molecule and non - polar molecule. Characteristics : Thus an induced dipole is developed in the other molecule. In this case also interaction energy is proportional to `1//r^(6)` where r is the distance between two molecules. Induced dipole moment depends upon the dipole moment present in the permanent dipole and the polarisability of the electrically neutral molecule. High polarisability increases the strength of attractive interactions. In this case also cumulative effect of dispersion forces and dipole - induced dipole interactions exists. |
|
| 7. |
Which of the following molecule contains no pi bond ? |
|
Answer» `SO_(2)` 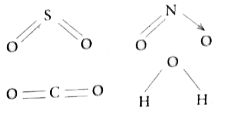 WATER `(H_(2)O)` contain only o bonds and no `pi` bonds. |
|
| 8. |
X_(2) +3Y_(2) to 2XY_(3)In this reaction 2 moles of X_(2)and 4 . 5molesof Y_(2)react to give products. Which is the limiting agent and calculate the no.of moles X_(2) , Y_(2) and XY_(3)in the reaction mixture? |
|
Answer» Solution :`X_(2) + 3Y_(2) to 2XY_(3)` `{:("No.of moles",2,4.5,?),("SC",1,3,2),("RATIO",2//1,4.5//3,=),(,2(ER),1.5(LR),=):}` ` = (2)/(1) = (4.5)/(3) = (nXY_(3))/(2)` `(4.5)/(3) = (nXY_(3))/(2) = 3 `moles No. of moles of ` 2XY_(3) `= 3 moles |
|
| 9. |
Which of the following are the correct reasons for anomalous behavior of lithium ? |
|
Answer» Exceptionally small size of its atom On account of small size and high nuclear CHARGE it shows high POLARIZING power. |
|
| 10. |
The statement that is not correct for modern classification of element is(1)The properties of elements are periodic function ofthese aromic numbers. (2)The ionisation enthalphyof the elements generally increase with increase in atomic number . (3) For transition elemens the 3d orbitals are filled after 3p-orbitals and befire 4sorbitals . (4)Fifth period contain 18 elements |
|
Answer» 1 and 2 |
|
| 11. |
When one mole of a monoatomic ideal gas at initialtemperature T K expandsadiabatically from 1 litre to 2litres , the final temperature in Kelvin would be |
|
Answer» <P>T or `(T_(2))/(T_(1))= ((V_(1))/(V_(2)))^(gamma-1)` or`(T_(2))/(T) = ((1)/(2))^((5)/(3)-1)= ( 1)/( 2^(2//3))` ( for monoatomic ideal gas , `gamma=(C_(p))/(C_(v))=((5//2)R)/((3//2)R)=(5)/(3))` `:. T_(2) =(T)/( 2^(2//3))` |
|
| 12. |
Which of the following halogens do not exhibit positive oxidation number in its compounds? |
|
Answer» Fluorine |
|
| 13. |
Whathappens to the pH ifa few drops of acid are added to CH_(3)CO ONH_(4) solution ? |
| Answer» SOLUTION :pH will REMAIN ALMOST CONSTANT (being a BUFFER solution). | |
| 14. |
Which of the following oxides of group 15 is most acidic ? |
|
Answer» `Bi_2O_3` |
|
| 15. |
When 1 mole of an ideal monatomic gas is compressed adiabatically the intenal energy change involved is 24 cals. The temperature rise is |
|
Answer» `""^(n)C_(V) Delta T= 1 xx (3)/(2) R. Delta T rArr 24 = (3)/(2) xx 2 xx Delta T` `rArr Delta T = 8` |
|
| 16. |
When a compressed real gas is made to expand adiabatically through a porous plug, how does the temperature change ? |
|
Answer» Solution :When a compressed real gas is allowed to expand adiabatically through a porous PLUG to a region of low against the molecules attractive forces, so work is DONE by the gas molecules at the expense of INTERNAL energy of the gas. THEREFORE, COOLING occurs as the gas expands. |
|
| 17. |
Which of the followingg is an isomer of ethanol? |
|
Answer» Methanol |
|
| 18. |
The reductive ozonolysis of an alkene gave butanone and ethanal. Give the structure and IUPAC name of the alkene. |
|
Answer» |
|
| 19. |
The volume occupied by any gas at S.T.P. is ________ |
|
Answer» 22.4 LITRES |
|
| 20. |
What is homogeneous equilibrium ? Give examples. |
|
Answer» Solution :Definition : In a homogeneous SYSTEM, all the reactants and PRODUCTS are in the same phase. e.g.-1: Following in the all gaseous reaction. `N_(2(g)) + 3H_(2(g)) hArr 2NH_(3(g))` ….. (homogeneous system) e.g.-2: Following equilibrium all the reactants and products are in homogeneous solution phase. (i)`Fe_((aq))^(3+) + SCN_((aq))^(-) hArr [Fe(SCN)^(2+)]_((aq))` (ii)`CH_3COOC_2H_(5_(aq)) + H_2O_((L)) hArr CH_3COOH_((aq)) + C_2H_5OH_((aq))` |
|
| 21. |
Write the relationship between enthalpy change, DeltaH and internal energy change, DeltaU , for a process occuring at constant pressure and constant temperature. |
| Answer» SOLUTION :`DeltaH = DELTAU + Deltan_(G) RT`. | |
| 22. |
When sodium is dissolved in liquid ammonia, a solution of deep blue colour is obtained. The colour of the solution is due to... |
|
Answer» ammoniated electron. `Na+(x+y)NH_(3)to underset("positive ion")underset("Ammoniated")([Na(NH_(3))_(x)]^(+))+underset("electron")underset("Ammoniated")(e(NH_(3))_(y)^(-))` |
|
| 23. |
Which statement is correct about photochemical bromination of Butane ? CH_3-CH_2-CH_2-CH_3overset(Br_2,h upsilon)to |
|
Answer» 1-Bromobutane and 2-Bromobutanes are FORMED in equal amounts. |
|
| 24. |
Which of the following is INCORRECT regarding VSEPR theory? |
|
Answer» The number of VALENCE SHELL electron pairs around the central atom determines the shape of the molecule |
|
| 25. |
The value of a cylinder containing initially 10L of an ideal gas at 25 atm and 25^(@)C is opened to the atmosphere, where the pressure is 760 torr and the temperature is 25^(@)C. Assuming that the process is isothermal the work in L-atm in the process is: |
|
Answer» 240 L ATM `n_(1) = (25 xx 10)/(RT), n_(2) = (1 xx 10)/(RT)` moles LOST `= (24 xx 10)/(RT)` Volume occupied by these moles `=(24 xx 10)/(RT) xx RT = 240L` `:. V_(2) = 10 + 240= 250L` `rArr W = -1 xx (250-10) = - 240 L`-atm |
|
| 26. |
Which gases are evolved when carbon is treated with conc. HNO_3 ? |
| Answer» SOLUTION :`CO_2 and NO_2` | |
| 27. |
Which are paramagnetic from O_(2), O_(2)^(-) and O_(2)^(2) ? |
| Answer» SOLUTION :`O_(2) and O_(2)^(-)` are PARAMAGNETIC | |
| 28. |
Total number of groups which can have higher-I effect than -F -NR_(3)^(oplus)-SR_(2)^(oplus)-NO_(2)-SO_(2)R-CN, -OR-OH-NH_(2)-Cl-COOH |
|
Answer» |
|
| 30. |
What are optical fibers ? What are their advantages over ordinary glass like that of window panes ? |
| Answer» Solution :Optical fibers are OBTAINED by drawing GLASS into fibers thinner than even human hair but still possess very high mechanical strength. The advantage of optical fibers is that unlike ordinary glass like that of WINDOW panes which stops light in less than one meter, optical fibers transmit light over kilometers without any noticeable decrease in intensity. That is why they are being used in radio BROADCAST, television programmes and telecommunication including mobile and INTERNET services. They are cheaper than copper coils and take much less space. | |
| 31. |
There are equal volume of 0.02 M CaCl_2 and 0.00004 M Na_2SO_4 solutionare mixed will a precipitation of CaSO_4 ? K_(sp)=2.4xx10^(-5) |
| Answer» Solution :Precipitation of `CaSO_4` will not OCCUR, `Q_(SP)=2XX10^(-7)` | |
| 32. |
What isthe totalnumberof orbitalsassociatedwith theprinciplequantumnumbern-3 ? |
|
Answer» Solution :Forn=3the possible VALUEOF l are 0.1 and 2Thus3s ,3pand 3dorbitals Nown=3 andl=0`m_(l)=0 `and ONE 3sorbitals `l =1 m_(1)= 1 , 0 +1 ` threevalue= there3p -orbitals Secondmethod. Numberof orbitalsin norbit`=n^(2)= (3)^(2)=9` |
|
| 33. |
Two separateexperiments were carried out involving adiabaticreversible expansion, taking monoatomic gas in one case and a diatomic gas in the second case.In each case, the initialtemperature wasT_(i) and expansion was from volume V_(1)to volume V_(2). The finaltemperaturesattained were T_(M) formonoatomic gasand T_9D) for diatomicgas. Then |
|
Answer» `T_(M) =T_(D) ltT_(i)` HENCE, for monoatomicgas, `(T_(M))/( T_(i)) =((V_(1))/(V_(2)))^(0.66)` and for diatomcgas, `(T_(D))/(T_(i))=((V_(1))/(V_(2)))^(0.40)` As`(V_(1))/(V_(2)) lt 1( :' V_(2) gt V_(1)) `. Hence,`(T_(M))/(T_(i)) LT1` i.e.,`T_(M) lt T_(i)` and`(T_(D))/(T_(i))lt 1` , i.e., `T_(D) lt T_(i)` Suppose `V_(2)= 10V_(1) `. Then `(T_(M))/(T_(D)) =((1)/(10))^(0.66-0.40) = ((1)/(10))^(0.26)` i.e. `T_(M)lt T_(D)`. Hence, `T_(M) ltT_(D) lt T_(i)` |
|
| 34. |
Whichone hashigherenergy4s and3d? why ? |
|
Answer» Solution :E 4 s `LT ` E 3d because4s = (n +1) = (4+0) =4 …. MIN value3 = (n+l)= (3+2)=5 …. Maxvalue |
|
| 35. |
Which one of the following is used in the conversion of ethyliden dichloride to Acetylene? |
| Answer» Solution :KOH+Ethanol | |
| 36. |
What volume of CO_(2) at NTP will be liberated by the action of 100 mL of 0.2 N HCl on CaCO_(3) ? |
| Answer» Solution :N//A | |
| 37. |
Which of the following has lower value of D_(2)O than for H_(2)O? |
|
Answer» MOLECULAR mass |
|
| 38. |
What factors impart characteristic properties to the transition elements? |
|
Answer» |
|
| 39. |
What is correct about the signs and magnitudes of the free energy, DeltaG^circ and the equilibrium constant, K, for a thermodynamically spontaneous reaction under standard conditons? |
|
Answer» `DeltaG^circ LT 0, K lt 0` |
|
| 40. |
Which of the following does not have a linear structure? |
|
Answer» `BeCl_(2)` |
|
| 41. |
Which one of the following is used in purification of sugar and as drying agent? |
| Answer» Answer :D | |
| 42. |
Which of the following compound(s) give yellow precipitate with BaCI_(2) solution? |
|
Answer» `Na_(2)Cr_(2)O_(7)+CH_(3)COONa` |
|
| 43. |
What is the molality of ammonia in a solution containing 0.85 g of NH^3 in 100 cm of a liquid of density 0.85 g cm^(-3)? |
|
Answer» |
|
| 44. |
Which of the following Reaction is not possible ? |
|
Answer»
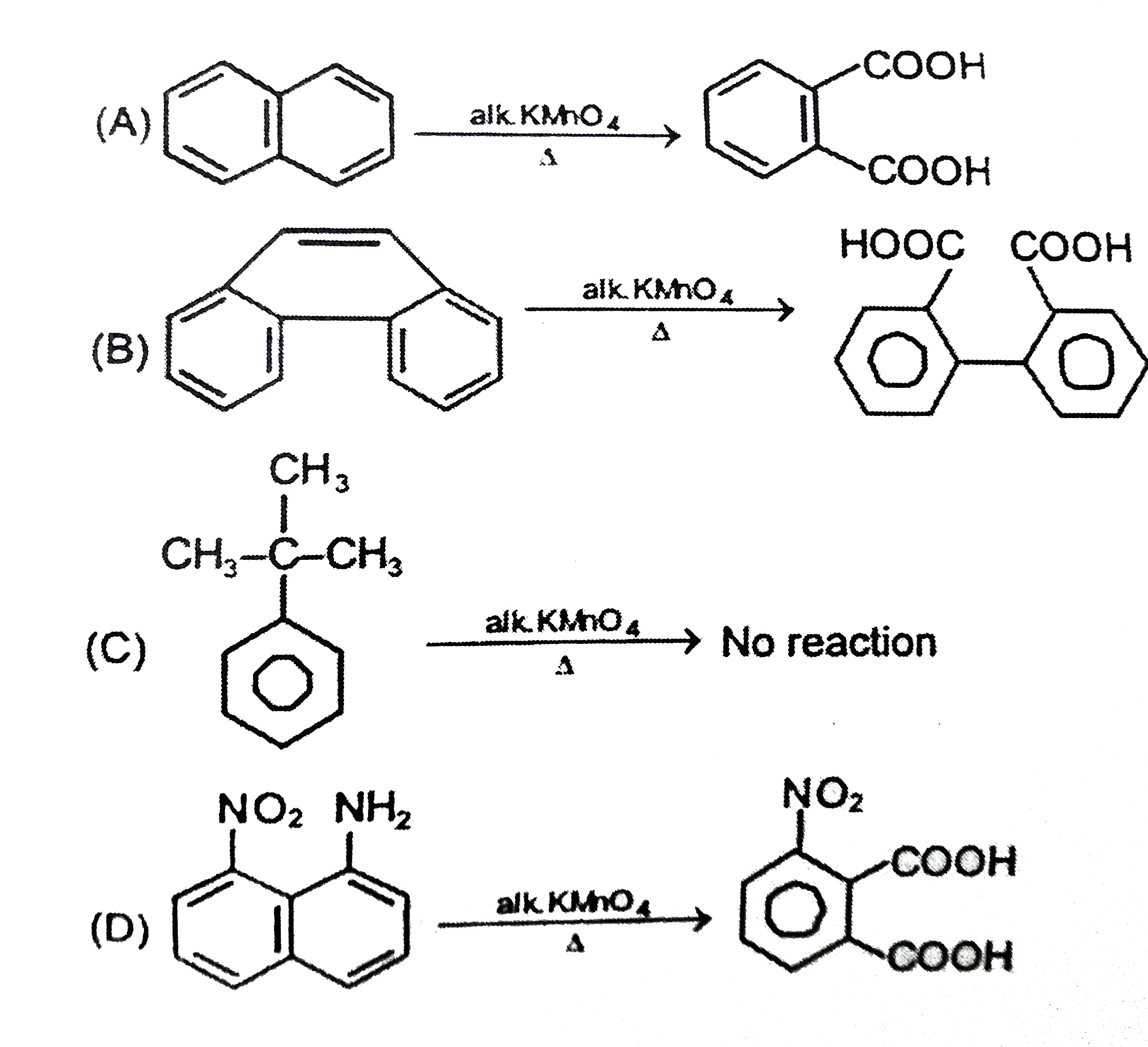
|
|
| 45. |
Which of the following is incorrect reagarding ionisation enthalpy ? |
|
Answer» `Na^(+)GT Na` |
|
| 47. |
Which of the following statement is /are correct ? (i) The presence of reacting species in a covered beaker is an example of open system. (ii) There is an exchange of energy as well as matter between system and the surroundings in a closed system. (iii) The presence of reactants in a closed vessel is an example of closed system. (iv) The presence of reactants in a thermos flask is an example of closed system. |
|
Answer» (II) &(iii) |
|
| 48. |
XY_(2) dissociates as, XY_(2(g))hArrXY_((g))+Y_((g)) Initial pressure of XY_(2) is 600mm Hg. The total pressure at equilibrium is 800mm Hg. Assuming volume of system to remain constant, the value of K_(p) is |
|
Answer» 50 |
|
| 49. |
Which of the following does not form grignard reagent on reaction with Mg in the presence of ether? |
| Answer» Solution :Vinyl chloride | |