Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following reactions production of dihydrogen form synthesis gas |
|
Answer» `CH_4 + H_2O UNDERSET(Ni)OVERSET(1270K) to CO + 3H_2` s |
|
| 2. |
What typeof substances would make better permanent magnets, ferromagnetic or ferrimagnetic.Justify your answer. |
| Answer» Solution : ferromagnetic SUBSTANCES MAKE BETTER permanent magents. This is because the metal ions of a ferromagnetic substance are grouped into SMALL regions called 'dmains'. Each domain acts as a tiny magnet. These DOMAINS are randomly oriented. when the substance is placedin a magnetic field, all the domings get oriented in the direaction of magneticfield and a strong magnetic field is produced . This ording of domains presists even when the external magnetic fieldsi removed. Hence, the ferromagnetic substance becomes a permanment magnet. | |
| 3. |
Which of the following does not form double salts ? |
|
Answer» `Li_(2)SO_(4)` |
|
| 5. |
Which one of the following reactions depicts the reducing action of hydrogen peroxide? |
|
Answer» `C_(6)H_(6)+H_(2)O_(2)rarrC_(6)H_(5)OH+H_(2)O` |
|
| 6. |
Which change occurs when lead monoxide is converted into lead nitrate ? |
|
Answer» Oxidation Neither oxidation nor reduction |
|
| 7. |
Which elements among the Lanthanides has the smallest atomic radius ? |
|
Answer» CERIUM |
|
| 8. |
The decomposition temperature of BaCO_(3) (A), MgCO_(3) (B) and CaCO_(3) (C ) follow the order |
|
Answer» `Z GT X gt Y` |
|
| 9. |
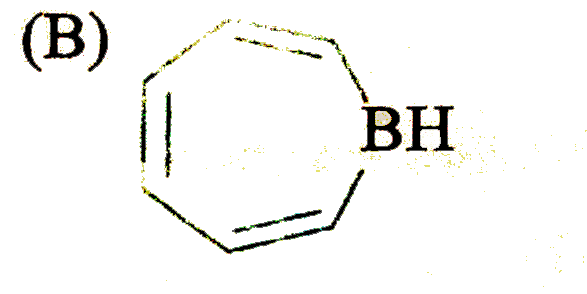
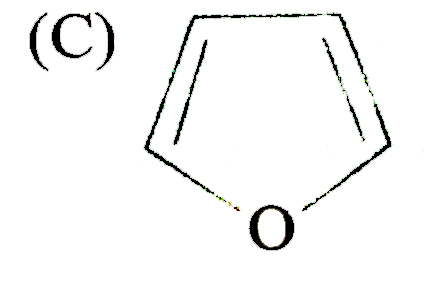
Which of the following heterocyclic compound/s might be aromatic? |
|
Answer»
|
|
| 10. |
Which of the following processes does not involve a catalyst |
|
Answer» THERMITE process |
|
| 11. |
The variation of volume V, with temperature T, keeping pressure constant is called the coefficient of thermal expansion is alpha=(1)/(V)((deltaV)/(deltaT))_(P). For an ideal gas alpha is equal to |
|
Answer» T |
|
| 12. |
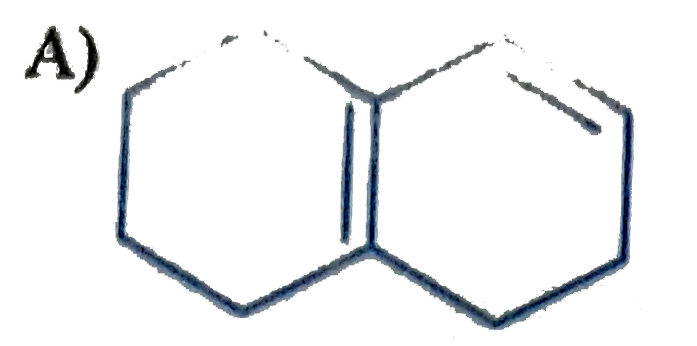
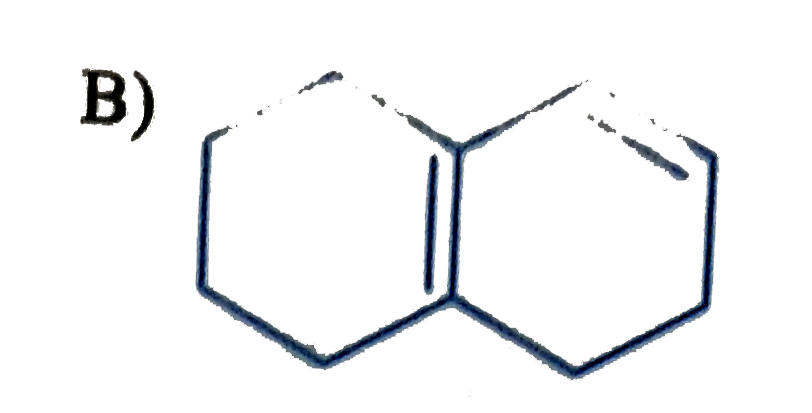
Which of the following is havin highest heat of hydrogenation? |
|
Answer»
|
|
| 13. |
Which of the following simplest alkanes with fewest number of C atom contains 1^(@) , 2^(@) , 3^(@)and 4^@C atoms? |
|
Answer» 2,2,3-Trimethyl PENTANE 2,2,4-Trimethyl pentane 2,3,3-Trimethyl pentane |
|
| 14. |
Which among the following statement is false? |
|
Answer» Increase of pressure increase the amount of adsorption |
|
| 15. |
What are the distinctive behaviour of beryllium? |
|
Answer» Solution :Beryllium is small in SIZE (ii) It has high POLARIZING power. (iii) Its ELECTRONEGATIVITY is relatively high. (iv) It has high ionization ENTHALPY (v) In valence shell, vacant d-orbitals are absent in beryllium |
|
| 16. |
What are transfermium elements? |
| Answer» SOLUTION :The ELEMENTS BEYOND FERMIUM are CALLED as transfermium elements. | |
| 17. |
Ethanol flows faster than honey.What is the effect of temperature on this? |
| Answer» SOLUTION :VISCOSITY of a liquid DECREASES with rise in temperature. They are inversely proportional. | |
| 18. |
True statement among the following about silicone |
|
Answer» Silicon is BONDED to ANOTHER silicon |
|
| 19. |
Which of the following is true. |
|
Answer» Acetylene is more reactive than ethylene to an ELECTROPHILIC attack |
|
| 20. |
What name is give to the compounds formed by more electropositive elements with carbon ? |
| Answer» SOLUTION :IONIC COMPOUNDS . | |
| 21. |
Which of the following species is not electrophile in nature? |
|
Answer» `CL^+` |
|
| 22. |
Use the bond energies in the table to estimate DeltaHfor this reaction . H_(2)C=CH_(2)+Cl_(2)toClH_(2)C-CH_(2)Cl {:overset("Bond energies")((C-C,347KJmol^(-1)),(C=C,612KJmol^(-1)),(c-Cl,331KJmol^(-1)),(C-H,414KJmol^(-1)),(Cl-Cl,243KJmol^(-1))):} |
|
Answer» `DeltaH=-684KJ` |
|
| 23. |
Which of the following alcohols on dehydration gives most stable carbocation? |
|
Answer» `CH_(3)-UNDERSET(CH_(3))underset(|)(C)H-CH_(2)OH` |
|
| 24. |
The value of K_c for the reaction 2A hArr B+ C is 2 xx 10^(-3). At a given time, the composition of reaction mixture is [A] = [B] = [C] = 3 xx 10^(-4) M. In which direction the reaction will proceed ? |
|
Answer» Solution :`{:("Reaction :",2A HARR, B+, C),("Concentration at t TIME :",3xx10^(-4), 3xx10^(-4), 3xx10^(-4)):}` At t time the reaction quotient, `Q_c=([B][C])/[A]^2=((3xx10^(-4))(3xx10^(-4)))/(3xx10^(-4))^2-1.0` Thus, `(Q_c=1.0) gt (K_c=2xx10^(-3))` Here, value of `Q_c gt K_c`, So the reaction will proceed in the DIRECTION of reactant (reverse reaction). The reactant converted into reactant till`Q=2xx10^(-3)`. |
|
| 26. |
What is the minimum volume of water required to dissolve 1 g of calcium sulphate at 298 K ? (For calcium sulphate , K_(sp)=9.1xx10^(-6) ) |
|
Answer» Solution :Suppose solubility in water of `CaSO_4` is S mol `L^(-1)` `{:(CaSO_(4(s)) hArr, Ca_((aq))^(2+)+ , SO_(4(aq))^(2-)),(S, S M, S M):}` `therefore K_(SP)(CaSO_4)=[Ca^(2+)][SO_4^(2-)]=(S)(S)=S^2` `therefore S=sqrt(K_(sp))` `=sqrt(9.1xx10^(-6))=3.0167xx10^(-3) "mol L"^(-1)` `therefore "mol L"^(-1) = "mol"/L = "weight in gm"/"molecular X VOLUME in litre"` `therefore 3.0167xx10^(-3) = "1 G"/("136 g mol"^(-1) xx "volume")` molecular mass of `CaSO_4` =40+32+64=136 g `mol^(-1)` `therefore` Volume =`1/(3.0167xx136xx10^(-3))=1000/(3.0167xx136)` =2.4374 L =2437.4 mL 1 g `CaSO_4` soluble in minimum 2437.4 mL solution. |
|
| 27. |
Which of the following isomeric heptanes can yield seven different monochlorinated products upon free radical hlorination ? |
|
Answer» 3-Methylhexane |
|
| 28. |
What is the solubility of speringly soluble salt ? |
|
Answer» More than 0.1 |
|
| 29. |
Under similar conditions, 390cm^# of gas A of molar mass M_A and 130cm^3 of gas B of molar mass M_B diffuesed out through a porous container i nthe time ratio of 2:1. The ratio of M_A to M_B is |
| Answer» ANSWER :C | |
| 30. |
Which of the following is not true regarding the nature of halides of boron? |
|
Answer» BORON trihalides are covalent |
|
| 31. |
What is entropy ? Explain its spontaneity. |
|
Answer» Solution :Entropy is the MEASUREMENT of randomness. Entropy of solid is less whereas in gaseous STATE entropy in maximum. Whenever heat is ADDED to the system, it increases molecular motions causing increased randomness in the system. Heat also depends on the temperature at which heat is added to the system. A system at higher temperature has greater randomness in it than one at lower temperature. Thus, temperature is the measure of average CHAOTIC .motion of particles in the system. Heat added to a system at lower temperature causes greater randomness than when the same quantity of heat is added to it at higher temperature. This suggests that the entropy change is inversely proportional to the temperature. `DeltaS= (q_("rev"))/( T)` `q_("rev")=` The heat gain or loss inversely by the system The total entropy change for the system and surroundings of a spontaneous process is given by `DeltaS_("total") = DeltaS_("sys") + DeltaS_("surr") gt 0` For spontaneous process `DeltaS= +ve` Entropy is a state property, we can calculate the change in entropy of a reversible process by : `DeltaS_("sys")= (q_("sys.,rev") )/( T)` We FIND that both for reversible and irreversible expansion for an ideal gas, under isothermal conditions, `DeltaU=0`, but `DeltaS_("total")=DeltaS_("sys")+ DeltaS_("surr")` is not zero for irreversible process. |
|
| 32. |
What is SI unit of viscosity coefficient (eta) ? |
|
Answer» Pascal We known that, `f= eta A(dv)/(dx)` f = force `eta =` viscosity coefficient `(dv)/(dx)=` VELOCITY gradient Now, PUT SI units of f = N, dx = m, A `m^(2)` and V `= ms^(-1)` in above equation. We get, `eta = (N xx m)/(m^(2)xxms^(-1))` `= Nm^(-2)s` Hence, unit of `eta = Nsm^(-2)` |
|
| 33. |
What is the molecular formula of the product formed when benzene is reacted with ethyl chloride in presence of anhydrous aluminium chloride? |
|
Answer» `C_8H_10` |
|
| 34. |
Two ratio-isotopes P and Q atomic weight 100 and 200 respectively are mixed in equal amount by weight. After 20 days their weight ratio is found to be 1:4 if t_(1//2) for P is 10 day calculate t_(1//2) for Q. |
|
Answer» |
|
| 37. |
Which of the following is not be correct representation of resonance structure of p-nitrophenoixde ion : |
|
Answer»
|
|
| 38. |
Which of the following species contains equal number of sigma - and pi- bonds? |
|
Answer» `HCO_(3)^(-)` number of `pi` bonds `=4` 
|
|
| 39. |
Which of the following constitutes irreversible colloidal system in water as dispersion medium? |
| Answer» Solution :All are IRREVERSIBLE colloidal systems. | |
| 40. |
Which of the following can tautomerizw? |
|
Answer»
|
|
| 41. |
What is the chemical name of calgon's? |
|
Answer» <P> |
|
| 42. |
Which law of chemical combination is not explained by the Dalton's atomic theory ? |
| Answer» SOLUTION :GAY Lussac's LAW of chemical combination is not explained by Dalton's atomoc THEORY. | |
| 43. |
Which of the following is not metal displacement |
|
Answer» `Zn+CuSO_(4) to ZnSO_(4)+CU` |
|
| 44. |
What is the difference in the cystal structures of NaHCO_(3) and KHCO_(3) ? |
Answer» Solution :The crystal structures of `NaHCO_(3) and KHCO_(3)` both show hydrozen bonding, but different. In `NaHCO_(3)`, the `HCO_(3)^(-)`ions are LINKED into an infinite CHAIN, whilst in `KHCO_(3)` a dimeric ANION is formed. 
|
|
| 45. |
Which reactions are useful to prepare Hydrogen in the laboratory |
|
Answer» Decomposition |
|
| 46. |
Which of the following shows ideal gas behaviour |
|
Answer»
|
|
| 47. |
Which among the following is coloured and paramagnetic |
|
Answer» `Li_(2)O` |
|
| 48. |
What is the difference in the approach between the Mendeleev's periodic law and the modern periodic law? |
|
Answer» |
|
| 49. |
The toal number of Na+ ions present per unit cell of NaCl is |
|
Answer» 1 |
|
| 50. |
What mass of Agl will dissolve in 1.0L of 1.0M NH_3 ? Neglect change in conc. NH_3[ Given , K_(sp) (Agl) =1.5 xx 10^(-16),K_f [Ag(NH_3) _2^(+)] =1.6 xx 10^(7)], (At .Wt . Ag =108 , 1 =127) |
|
Answer» `4.9 xx 10^(-5) g` ` K_(SP). K_f =(X^(2))/( (Z-2x)^(2) ) ` ` 1.6 xx 10^(7)xx 1.5 xx10^(-16) =(x^(2))/((1-2x)^(2))` but ` 1-2x ~~ 1, x =4.9xx 10 ^(5)" mol " //lit ` Mass of ` Agl =4.9 xx 10 ^(-5)xx 235 =0.011 gm` |
|