Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nitrogen, sulphur and halogens are tested from the sodium fusion extract of the organic compound which is commonly known as Lassaigne's extract. For preparing Lassaigen's extract, a small quanty of the organic compound is fused with sodium metal in a fusion tube. The red tube is broken in distilled water, boiled and filtered. This filtrate is known as Lassaigne's extract or sdodium fusion extract.During testing of nitrogen in organic compound by Lassaigne's test, which of the following compounds are formed.(i)NaCN(ii) Fe_(4)[Fe(CN)_(6)]_(3)(iii) Na_(4)[Fe(CN)_(6)](iv)Fe_(3)[Fe(CN)_(6)]_(4) |
|
Answer» only (i) |
|
| 2. |
Nitrogen, sulphur and halogens are tested from the sodium fusion extract of the organic compound which is commonly known as Lassaigne's extract. For preparing Lassaigen's extract, a small quanty of the organic compound is fused with sodium metal in a fusion tube. The red tube is broken in distilled water, boiled and filtered. This filtrate is known as Lassaigne's extract or sdodium fusion extract.In the process of testing elements in Lassaigne's a test, which of the following reactions do not occur. |
|
Answer» `NA+2H_(2)O rarr 2NaOH+H_(2)` |
|
| 3. |
Nitrogen, sulphur and halogens are tested from the sodium fusion extract of the organic compound which is commonly known as Lassaigne's extract. For preparing Lassaigen's extract, a small quanty of the organic compound is fused with sodium metal in a fusion tube. The red tube is broken in distilled water, boiled and filtered. This filtrate is known as Lassaigne's extract or sdodium fusion extract.Which of the following is not applicable to Lassaigne's test |
|
Answer» The nitrogen, halogens and sulphur are converted into inorganic salts |
|
| 4. |
Nitrogen laser produces a radiation at a wavelength of 337.1 nm. If the number of photons emitted is 5.6 xx 10^(24), calculate the power of this laser. |
| Answer» SOLUTION :`E = N hv Nh (c)/(lamda) = ((5.6 xx 10^(24)) (6.626 xx 10^(-34) Js) (3.0 xx 10^(8) ms^(-1)))/((337.1 xx 10^(-9) m)) = 3.3 xx 10^(6) J` | |
| 5. |
Nitrogenlaserproducesa radiationat a wavelengthof 337.1nmifthe numberofphotonsemitted is 5.6 xx 10^(24) calculatethepowerof thislaser. |
|
Answer» Solution :Emittedlaser energy`(E ) = (hc )/(lambda)`andNumberof photon=Nlaserenergy`=E_(p)` `:. , E_(p)= (Nhc )/( lambda)` `=(5.6 xx 10^(24)6.626 xx 10^(34)js 3.0 xx 10^(8) MS^(-1))/((337.1 xx 10^(9) m))` `0.3302xx 10^(7) ` `=3.302 xx 10^(6) J` emittedlaserenergy |
|
| 6. |
Nitrogenhaspositive electrongainenthalpywhereas oxygenhas negative. Howeveroxygenhas lower ionisation enthalpythannitrogen . Explain. |
| Answer» Solution :The outermostelectronicconfigurationof nitrogen`(2s^(1)2p_(x)^(1) 2p_(y)^(1) 2p_(z)^(1))` is quitesinceit hasexactlyhalf- filled2p - ORBITALS . Thereforeit has no tendencyto ACCEPT an extra electron. Inotherwords energyhasto besupplied to add anextraelectron.thuselectron gainenthalpyof N isslightlypositive. Incontrast, outermostelecronicconfigurationof O is `2s^(2)2p_(x)^(2) 2p_(y)^(1) 2p_(z)^(1)` . Ithas highestnuclearcharge ( + 8)than N(+7)andhenceit hasa hightendencyto acceptan extraelectron. In otherwordsenergyin releasedwhenanelectronis added to O and Henceelectrongain enthalpyof O ishighly negative.Further SINCEO hasfour afterlosingone electronthereforeionization enthalpyof O islowerthanthat of N. | |
| 7. |
Nitrogen has positive electron gain enthalpy whereas oxygen has negative. However, oxygen has lower ionisation enthalpy than nitrogen. Explain. |
|
Answer» Solution :Electronic CONFIGURATION of `""_(7)N=1s^(1),2s^(2),2p_(x)^(1), 1p_(y)^(1),2p_(z)^(1)` Nitrogen has STABLE configuration because porbital is halfly filled. Adding an extra electron needs an energy into the p-orbital. Electronic configuration of `""_(8)O=1s^(2),2s^(2),2p_(x)^(2),2p_(y)^(1),2p_(z)^(1)` Oxygen has 4 ELECTRONS, in p-orbital, so process of adding an electron to that orbital is an exothermic process. Oxygen has lower ionisation enthalpy than nitrogen because if we remove one electron from 2p-orbitaloxygen attends stable configuration, i.e., `2p^(3)`. In nitrogen removal of last electron is not easy, one of the three 2p-electrons due to its stable configuration. |
|
| 8. |
Nitrogen has higher ionisation enthalpy than that of Oxygen. Give reaon. |
|
Answer» |
|
| 9. |
Nitrogen formula of Glauber's salt is |
|
Answer» `KNO_(3)` `2KNO_(3)OVERSET(Delta)rarr2KNO_(2)+O_(2)` |
|
| 10. |
Nitrogen forms several gaseous oxides. One of them has a density of 1.33 g/L measured 764 mmHg and 150^(@)C. Write the formula of the compound. |
|
Answer» NO Mw=46 |
|
| 11. |
Nitrogen forms five stable oxides with oxygen of the formula, N_(2)O, NO, N_(2)O_(3), N_(2)O_(4), N_(2)O_(5). The formation of these oxides explains fully the |
|
Answer» LAW of definite proportions |
|
| 12. |
Nitrogen exists as diatomic molecule and phosphorus as P_(4). Why ? |
| Answer» Solution :Nitrogen becaues of its small size and high electronegativity forms `ppi-ppi` multiple BONDS. Therefore, it exists as a diatomic MOLECULE having a triple bond between the two N-atoms. Phosphours, on the other hand, due to its larger size and lower electronegativity USUALLY does not form `ppi-ppi` multiple bonds with itself. Instead it prefersto form P-P single bonds and hence it exists as TETRAHEDRAL `P_(4)` molecules. | |
| 13. |
Nitrogen does not form pentahlides as it does not have …………. |
|
Answer» |
|
| 14. |
Nitrogen dioxide is not produced on heating |
|
Answer» `KNO_(3)` `2KNO_(3)overset(Delta)to2KNO_(2)+O_(2)` `2Pb(NO_(3))_(2)overset(Delta)to2PbO+4NO_(2)+O_(2)` `2Cu(NO_(3))_(2)overset(Delta)to2CuO+4NO_(2)+O_(2)` `2AgNO_(3)overset(Delta)to2Ag+2NO_(2)+O_(2)` |
|
| 15. |
Nitrogen dioxide cannot be obtained by heating |
| Answer» Answer :A | |
| 16. |
Nitrogen detection in an organic compound is carried out by Lassaigne's test. The blue colour formed is due to the formation of .............. . |
|
Answer» `Fe_(3)[Fe(CN)_(6)]_(2)` |
|
| 17. |
Nitrogen detection in an organic compound is carried out by Lassaigne's test. The blue colour formed is due to the formation of ... |
|
Answer» `Fe_(3)[FE(CN)_(6)]_(2)` |
|
| 18. |
Nitrogen containing organic compound when fused with sodium metal forms |
| Answer» Answer :B | |
| 19. |
Nitrogen containing organic compound when fused with sodium metal forms: |
|
Answer» `NaNO_(2)` |
|
| 20. |
Nitrogen atom has an atomic number of 7 and oxygen has an atomic number 8.The total number of electrons in a nitrate ion will be |
|
Answer» Solution :Numbers of electrons in nitrogen =7 and number of electron is oxygen =8 we know that formula of nitrate ion is `NO_3^-` we ALSO know that number of electron =(1 x Number of electrons in nitrogen ) + ( 3 x number of electrons in oxygen ) +1 =(1 x 7) + (3 x 8) + 1 =32 |
|
| 21. |
Nitrogen atom has 3 unpaired electrons in its ground state. It can be explained by |
|
Answer» AUFBAU PRINCIPLE |
|
| 22. |
Nitrogen and oxygen having electrons in 2p orbital which electron experiencing more repulsion ? What is effect due to repulsion ? |
|
Answer» SOLUTION :Electrons of OXYGEN experiencing more REPULSION than nitrogen. Due to more repulsion, ionisation ENTHALPY of nitrogen is more than nitrogen. |
|
| 23. |
Nitrogen and oxygen combine to form many oxides. In some oxides, 14 g of nitrogen combine with either 16 g of oxygen or 32 g of oxygen. In some other oxides, 28 g of nitrogen combine with 16, 48, or 80 g of oxygen, (i) What are the formulae of the oxides ? (ii) What law of chemical combination does the formation of these compounds illustrate ? |
Answer»  Let us TAKE a FIXED mass say 28 g of nitrogen. The weights of oxygen combining with 28 g of nitrogen in these compounds will be as follows :  The ratio of weights of oxygen combining with 28 g of nitrogen are `2 : 4 : 1 : 3 : 5` which is a simple whole number ratio. HENCE, the FORMATION of these compounds illustrate law of multiple proportions. Since, the atomic mass of nitrogen is 14 and that of oxygen is 16, the number of ATOMS of N and O in these compounds and their formulae can be obtained as follows :  Thus, the formulae of the given oxides are `NO, NO_2, N_2O, N_2O_3` and `N_2O_5`. |
|
| 24. |
Nitrogen (14.0 u) , phosphorus (31.0 u) andarsenic (74.9 u)havesimilarproperties. Do the they form aDobereiner 'striad? Comment . |
|
Answer» Solution :Sincethese elementhave similar propertiesthereforethey shouldform aDobereiner'striad. Butactuallythey donot because the atomicweight of middleelememtphosphorus(31 u)is not an arithmaticmean ofnitrogen (14.0 u)and ARSENIC(74. 9 u) . Arithmaticmean `(14 + 74. 9 ) /(2)= 44. 45 u ` |
|
| 25. |
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO_3 and conc. H_2SO_4 . In the nitrating mixture, nitric acid and acts as a |
|
Answer» base 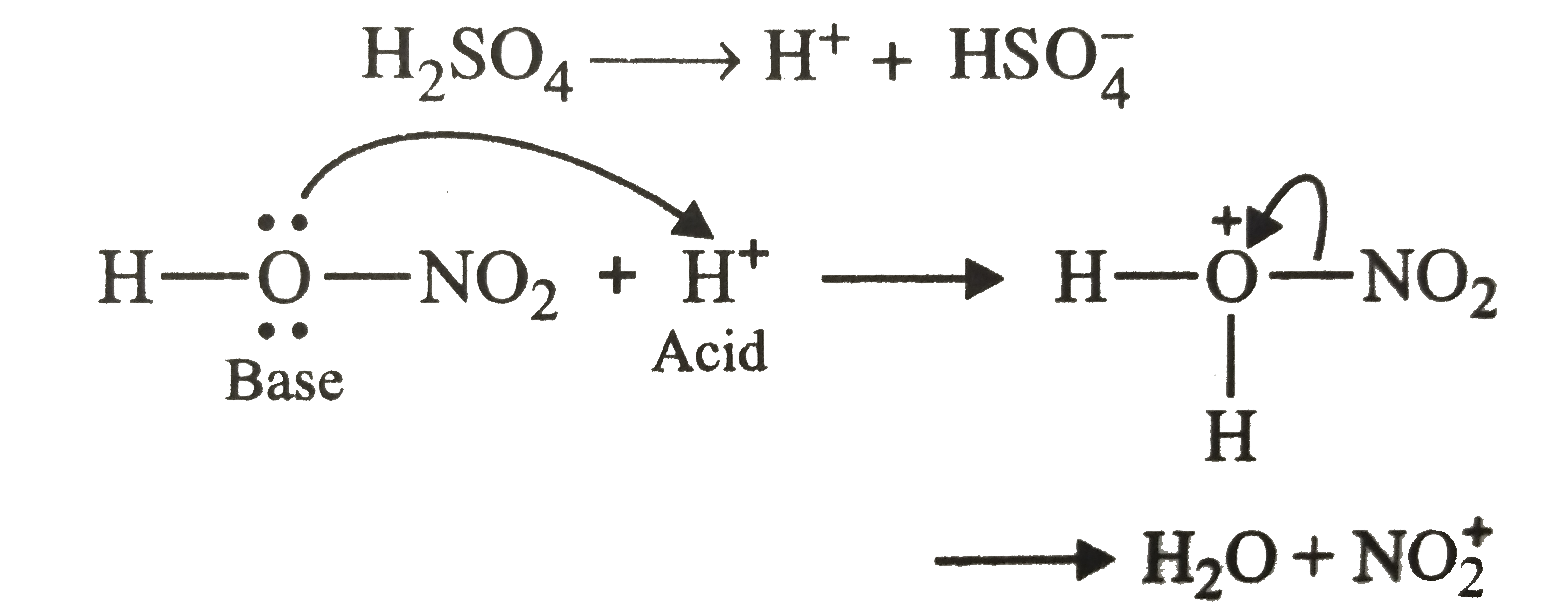 Thus, nitric acid acts as a base. |
|
| 26. |
Nitride ion is composed of |
|
Answer» 7 ELECTRONS and 7 PROTONS |
|
| 27. |
Nitric oxide reacts with bromine and gives nitrosyl bromide as per reaction given below : 2 NO (g) + Br _(2) (g) hArr 2 NOBr (g) When 0*087 mol of NO and 0*0437 mol of Br_(2)are mixed in closed container at constant temperature , 0*0518 mol of NOBr is obtained at equilibrium . Calculate equilibrium amount of nitric and bromine. |
|
Answer» SOLUTION :`0*0518 "mol of NOBR is formed from " 0*0518 "mol of NO and " 0*00259 "mol of " Br_(2) .` `:." At equilibrium, AMOUNT of NO " = 0*087 - 0*0518 = 0* 0352 "mol" ` ` " Amount of " Br_(2) = 0*0437- 0*0259 = 0* 0178 "mol" `. |
|
| 28. |
Nitric oxide reacts with Br_2 and gives nitrosyl bromide as per reaction given below: 2NO_((g)) + Br_(2(g)) hArr 2NOBr_((g)) When 0.087 mol of NO and 0.0437 mol of Br_2 are mixed in a closed container at constant temperature, 0.0518 mol of NOBr is obtained at equilibrium. Calculate equilibrium amount of NO and Br_2. |
|
Answer» Solution :According to stoichiometry of REACTION 2 MOL NO react with 1 mol `Br_2` and 2 mol NOBR formed. So, 2x mol NO react with X mol `Br_2` and from 2x mol NOBr. `{:("Reaction",2NO_((g))+ , Br_(2(g))HARR , 2NOBr_((g))),("Initial mol :",0.087 , 0.0437,Zero),("Reaction change :", "-2x mol","-x mol","+2x mol"),("Mol. at equilibrium :", (0.087-2x),0.0437-x , 2x=0.0518):}` `therefore x=0.0518/2`=0.0259 mol Thus, at equilibrium mol of NO = (0.087-2x) =0.087-0.0518 = 0.0352 mol mol of `Br_2`= (0.0437 -x) = 0.0437-0.0259 = 0.178 |
|
| 29. |
Nitric oxide (NO) is the nucleus of photochemical smog. 2NO(g) + O_(2) ("air") rarr 2NO_(2) (g) NO_(2)(g) overset("Sunlight")(rarr) NO(g) + [O] [O]+ O_(2) (g) rarr O_(3)(g) Both NO_(2) and O_(3) are strong oxidising agents and react with unburnt hydrocarbons in the polluted air to produce formaldehyde, acrolein and peroxyacetyl nitrate (PAN). These products are harmful to the plant and animal life. Catalytic converters are now installed in the automobiles to reduce the photochemical smog. Photochemical smog can also be supressed by certain compounds, which act as free radical trap. Smog is common pollutant in places having : |
|
Answer» HIGH TEMPERATURE |
|
| 30. |
Nitric oxide (NO) is the nucleus of photochemical smog. 2NO(g) + O_(2) ("air") rarr 2NO_(2) (g) NO_(2)(g) overset("Sunlight")(rarr) NO(g) + [O] [O]+ O_(2) (g) rarr O_(3)(g) Both NO_(2) and O_(3) are strong oxidising agents and react with unburnt hydrocarbons in the polluted air to produce formaldehyde, acrolein and peroxyacetyl nitrate (PAN). These products are harmful to the plant and animal life. Catalytic converters are now installed in the automobiles to reduce the photochemical smog. Photochemical smog can also be supressed by certain compounds, which act as free radical trap. Photochemical transformation of the automobile exhaust emission in UV wavelength of sunlight results into |
|
Answer» `CH_(4) and C_(6) H_(6)` |
|
| 31. |
Nitric oxide (NO) is the nucleus of photochemical smog. 2NO(g) + O_(2) ("air") rarr 2NO_(2) (g) NO_(2)(g) overset("Sunlight")(rarr) NO(g) + [O] [O]+ O_(2) (g) rarr O_(3)(g) Both NO_(2) and O_(3) are strong oxidising agents and react with unburnt hydrocarbons in the polluted air to produce formaldehyde, acrolein and peroxyacetyl nitrate (PAN). These products are harmful to the plant and animal life. Catalytic converters are now installed in the automobiles to reduce the photochemical smog. Photochemical smog can also be supressed by certain compounds, which act as free radical trap. Which are the primary constituents of photochemical smog ? |
|
Answer» `So_(2) and CO` |
|
| 32. |
Nitric oxide acts as a reducing agent in which of the following reaction ? |
|
Answer» `4NH_(3) + 5O_(2) RARR 4NO+ 6H_(2)O` |
|
| 33. |
Nitric onxide reacts with bromine to from nitrosyl bromide as follows 2NO(g)+Br_2(g)iff2NOBr(g) When 0.087 mol of NO and 0.0437 mole of Br_2 are mixed in a closed containar at constant temperature,0.0518 mol of NOBr is obtained at equilibrium. Calculate equilibrium amount of NO and Br_2. |
|
Answer» Solution :2 moles of NO gives 2 MOLE of NOBR `therefore` Number of moles NO REACTED to from 0.0518 MOL of NOBr=0.0518 Number of moles of `Br_2` reacted= `0.0518/2=0.0259` `therefore` Number of moles of NO remaining at equilibrium=0.087-0.0518=0.0352 mol `therefore` Number of moles `Br_2` at equilibrium =0.0437-0.0259=0.0178 mol |
|
| 34. |
Nitric acid is highly corroslve and a strong oxidising agent. To avoid accidents caused by possible breakage of glass containers during transporations, what other unbreakable containers can be used |
|
Answer» Solution :Nitric ACID USE can be safety transported in aluminium containers. The reasons being that AL becomes passive due to the formation of oxidelayer on itssurface. This oxide layer being IMPERVIOUS does not allow the acid to come in contact with the acid to come in contact with the metal . As a result, further action stops. `2Al + 6HNO_(3) ("conc.") rarr Al_(2)O_(3) + 6NO_(2) + 3H_(2)O` |
|
| 35. |
Nitric acid is an oxidising agent and reacts with pbo but it does not react with Pbo_(2) expain why ? |
|
Answer» SOLUTION :Since in `PbO_(2)` Pbisin the maximum oxidation STATE of +4 and in `HNO_(3)` N is in the maximum oxidation state of +5 therefore none of them can oxidation each other in other words `PbO_(2)` is passive towards `HNO_(3.)` and hence no reaction occurs in contrast PbO being BASIC undergoes an acid base reactionto form `PB(NO_(3))_(2)` and `H_(2)O PbO+2 HNO_(3) rarrPb(NO_(3))_(2)+H_(2)O` |
|
| 36. |
Nitric acid is an oxidising agent and reacts with PbO but it does not react with PbO_(2). Explain why ? |
|
Answer» Solution :PbO is a base but it reacts with `HNO_(3)` from lead nitrate. `PbO+2HNO_(3)tounderset("soluble")(Pb(NO_(3))_(2))+H_(2)O` Nitric acid does not react with `PbO_(2)` because both act as oxidising agents. In `HNO_(3)` oxidation number of N is (+5) as same `PbO_(2)` oxidation number of Pb is (+4). So, reaction is not possible. |
|
| 37. |
Nitric acid forms an oxide of nitrogen on reaction with P_(4)O_(10). Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed. |
|
Answer» SOLUTION :`P_(4)O_(10)` is a strong dehydrating agent. It reacts with `HNO_(3)` to form metaphosphric acid `(HPO_(3))` and dinitrogen pentoxide, `N_(2)O_(5)` `4HNO+P_(4)O_(10)to4HPO_(3)+2N_(2)O_(5)` For resonance STRUCTURES refer to Table |
|
| 38. |
Nitric acid can be produced from ammonia in three step process. {:(,4NH_(3)(g)+5O_(2)(g)rarr4NO_(g)+6H_(2)O(g),...(1)),(,2NO(g)+O_(2)(g)rarr2NO_(2)(g),...(2)),(,3NO_(2)(g)+H_(2)O(l)rarr2HNO_(3)(aq.)+NO(g),...(3)):} Calculate weight of NH_(3)(g) required to produce 1260kg of HNO_(3). When % teild of 1^(st), 2^(nd) and 3^(rd) reaction are respectively 69%,60% and 68% respectively. |
|
Answer» `W_(NH_(3))=1811.5 KG` |
|
| 40. |
Nitric acid can be produced from NH_(3) in three step process I) 4NH_(3(g))+5O_(2(g))rarr4NO_((g))+6H_(2)O_((g)) II) 2NO_((g))+O_(2(g))rarr2NO_(2(g)) III) 3NO_(2(g))+H_(2)O_((l))rarr2HNO_(3(aq))+NO_((g)) % yield of I, II, III reaction are respectively 50%, 60%and 80%. Then how much volume of NH_(3(g)) at STP is required to produce 2.25 gm of HNO_(3). |
|
Answer» `100rarr50` `2NO+O_(2)rarr2NO_(2),60%` `50 rarr50xx(60)/(100)=30` `3NO_(2)+H_(2)Orarr2HNO_(3)+NO,80%` `30rarr20xx(80)/(100)=16` 100 moles `NH_(3)=16` moles `HNO_(3)` `?larr (2.25)/(63)` moles `V_(NH_(3))` at STP = `(2.25xx100)/(63xx16)xx22.4=5` |
|
| 41. |
Nitric acid can be obtained from ammonia via the formation of the intermediate compounds |
|
Answer» nitric oxide and nitrogen DIOXIDE |
|
| 42. |
Nitric acid acts only as an oxidising agent while nitrous acid acts both as an oxidising as well as reducing . Explain . |
|
Answer» Solution :(i) `HNO_3` Oxidation number of N in `NHO_3` is +5. Maximum oxidation number of N is +5 because it has five electrons in the valence shell `(2s^(2)2p^3)` . Minimum oxidation number of N is -3 because it can accept 3 more electrons to get noble GAS configuration. Since O.N. of N in `HNO_3` is maximum , therefore , it can only decrease . Thus , `HNO_3` can act as an oxidising AGENT. (II) `HNO_2` Oxidation number of N = +3 Maximum O.N. = +5 Minimum O.N. of N = -3 Therefore , the O.N. of N can INCREASE by losing electrons or can decrease by accepting electrons. Thus, `HNO_2` can act both as an oxidising as oxidising as well as a reducing agent. |
|
| 43. |
Nitric acid acts only as an oxidising agent while nitrous acid acts both as oxidsing as well as a reducing agent why ? |
|
Answer» Solution :(i) `HNO_(3)`: Oxidation number of N is `HMO_(32)=+5` Maximum oxidation mnumber of N=+5 Minimum oxidation number of N =-3 Since the oxidation number of N in `HNO_(3)` is maximum (+5) therefore it can only decrease by acceptin electrons hence `HNO_(3)` ACTS only as an oxidisingagent (ii) `HNO_(2)=+3` Maximum oxidation number of N = +5 Minimum oxidation number of N=-3 THUS the oxidation number of N cna tiher increase by losin elctons of can decrease its O.N by accepting ELECTONS therefoer `HNO_(2)` acts both as an oxidsig as well as a reducing agent |
|
| 44. |
Nitration of phenol with concentrated nitric acid gives |
|
Answer» 2,4,6 trinitrophenol |
|
| 45. |
Nitration of benzene is carried out with conc. HNO_(3) in presence of conc. H_(2)SO_(4). The role of conc. H_(2)SO_(4) is to provide |
|
Answer» NUCLEOPHILE during the reaction |
|
| 46. |
Nitration of benzene by nitric acid and sulphuric acid is |
|
Answer» ELECTROPHILIC substitution |
|
| 47. |
Which one of the following is not present in the nitration mixture ? |
|
Answer» `1:1" of CONC."HNO_3 "and conc." HCI` |
|
| 48. |
Nitration and chlorination of benzene are |
|
Answer» nucleophilic and electrophilic substitution respectively `HNO_(3)+2H_(2)SO_(4) to NO_(2)^(+)+2HSO_(4)^(-)+H_(3)O^(+)` Halogenation is carried out by the help of electrophile `Cl^(+)(X^(+))` `Cl_(2)+FeCl_(3) to Cl^(+) +FeCl_(4)^(-)` Hence, both reactions are electrophilic substitution reactions. |
|
| 49. |
Nitrating agent for aromatic compound may be : |
|
Answer» `N_(2)O_(5)` |
|