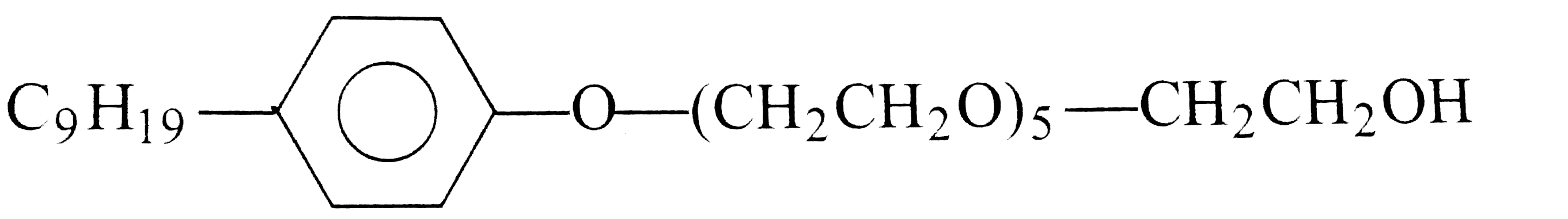Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Passage Organic compounds mainly consist of covalent bonds. The electron pair in these covalent bonds may undergo displacement either of their own or under the influence of other species. The cleavage of covalent bond between two atoms takes place in homolytic or heterolytic fashion. The homolytic fission results into free radicals while heterolytic fission results into carbocations and carbanions. These are also called reaction intermediates and are attacked by electrophiles and nucleophiles. The electrophiles seek electron rich sites while nucleophiles seek electron deficient sites. Answer the followings Questions : Which of the following series contain only nucleophiles: |
|
Answer» `AICI_3, BF_3, NH_3` |
|
| 2. |
Justify the inclusion of alkali metals in the same group of the periodic table with reference to the following. (ii) Reducing nature. |
|
Answer» Li |
|
| 3. |
PASSAGE - IV : In periodic table metals show high reducing ability and non-metals show high oxidizing ability. The properties of metalloids in between metals and non-metals. In a group from top to bottom the reducing ability increases and in a period from left to right oxidizing ability increases. F_2 is strong oxidizing agen than Cl_2 due to |
|
Answer» Bond energy of `F_2 LT Cl_2` |
|
| 4. |
PASSAGE - IV : In periodic table metals show high reducing ability and non-metals show high oxidizing ability. The properties of metalloids in between metals and non-metals. In a group from top to bottom the reducing ability increases and in a period from left to right oxidizing ability increases. In which of the following set both are metalloids |
|
Answer» As,Se |
|
| 5. |
AT673 K. in the formation of NH_(3) From N_(2) and H_(2) ,the partial pressures of N_(2) , H_(2) andand NH_(3)at equilibrium are 0.5 , 1 and 9 xx 10^(-3)atm respectively. (N_(2) + 3H_(2) hArr + Q) Report Delta K_(C) Value |
|
Answer» <P>9.3 kcal `=2.303 xx 2 xx 10^(-3) xx 673 log_(10)0.5` = 0.929 K.cal |
|
| 6. |
AT673 K. in the formation of NH_(3) From N_(2) and H_(2) ,the partial pressures of N_(2) , H_(2) andand NH_(3)at equilibrium are 0.5 , 1 and 9 xx 10^(-3)atm respectively. (N_(2) + 3H_(2) hArr + Q) Report Delta G^(@) using K_(p) value |
|
Answer» <P>22.66 kcal `=2.303 xx 2 xx 10^(-3) xx 673 log 2 xx 81 xx 10^(-6)` = 11.3 K.cal |
|
| 7. |
AT673 K. in the formation of NH_(3) From N_(2) and H_(2) ,the partial pressures of N_(2) , H_(2) andand NH_(3)at equilibrium are 0.5 , 1 and 9 xx 10^(-3)atm respectively. (N_(2) + 3H_(2) hArr + Q) Calculate K_(c)for the reaction. |
|
Answer» 0.1 `K_(P)=(PN^(2)H_(3))/(PN_(2).PH_(2)^(3))=((9 xx 10^(-3))^(2))/(0.5 xx 1)=2 xx 81 xx 10^(-6)` `K_(P)=K_(c)(RT)^(Delta n), K_(c)=(2 xx 81 xx 10^(-6))/((0.0821 xx 673)^(-2))=0.5` |
|
| 8. |
As per Lechatlier's Principle any stress applied on the equilibrium state is minimised by shifting of equilibrium. PCl_(5) hArr PCl_(3) + Cl_(2) "," SO_(2) Cl_(2) hArr SO_(2) + Cl_(2) Both equilibria exist together in a flask. If some SO_(2) is introduced into the flask ______ |
|
Answer» `SO_2CI_2` concentration decreases |
|
| 9. |
As per Lechatlier's Principle any stress applied on the equilibrium state is minimised by shifting of equilibrium. Increase in volume of container shifts the equilibrium in forward direction in the case of ____ |
|
Answer» `N_(2(G))+3H_(2(g)) harr 2NH_(3(g))` |
|
| 10. |
As per Lechatlier's Principle any stress applied on the equilibrium state is minimised by shifting of equilibrium. In which cases introduction of inert gas shifts the equilibrium in forward direction at constant pressure ? |
|
Answer» `N_(2(G))+3H_(2(g)) harr 2NH_(3(g))` |
|
| 11. |
PASSAGE - 3 : In modern periodic table there is gradation in the properties of elements. In a period the Ionisation energy, Electron affinity, Electro negativity, Non-Metallic nature increases. In a group the atomic size, metallic nature, reducing properties and densities of elements increases. In the compunds KO_2,Cr_2,O_3,SO_2 and Cl_2O_3 Which element shows more valency with respect to oxygen |
|
Answer» K |
|
| 12. |
Passage 2 Antibiotics are the chemical substances which are produced by microorganisms like bacteria, fungi and moulds. Antibiotics can inhibit the growth or even destroy other microorganisms. Now-a-days, synthetic antibiotics are also available. The first successful antibiotic produced was penicillin. The antibiotics may be either bacteriocidal (kills the organism in the body) or bacteriostatic (inhibits the growth of organism). Ampicillin and amoxycillin are modified antibiotics. Broad spectrum antibiotics are effective against several types of harmful microorganisms. 5. Which of the following antibiotics is effective against tuberculosis ? |
|
Answer» Chloromycetin |
|
| 13. |
PASSAGE - 3 : In modern periodic table there is gradation in the properties of elements. In a period the Ionisation energy, Electron affinity, Electro negativity, Non-Metallic nature increases. In a group the atomic size, metallic nature, reducing properties and densities of elements increases. Whic pair of elements have positive electron affinity values |
|
Answer» Be,Mg |
|
| 14. |
PASSAGE - 3 : In modern periodic table there is gradation in the properties of elements. In a period the Ionisation energy, Electron affinity, Electro negativity, Non-Metallic nature increases. In a group the atomic size, metallic nature, reducing properties and densities of elements increases. The pair of elements with equal electro negativities |
|
Answer» Na,K |
|
| 15. |
PASSAGE - 2 : In periodic table there are nearly 110 elements are present majority of elements are present in solid state. There is no separate placement for isotopes of elements in periodic table. The atomic weight of an element is the average atomic weight of isotopes. Among IIA,HIA, VA,VIA, VIIA groups gaseous elements are present in |
|
Answer» IA,IIA,VA only |
|
| 16. |
PASSAGE - 2 : In periodic table there are nearly 110 elements are present majority of elements are present in solid state. There is no separate placement for isotopes of elements in periodic table. The atomic weight of an element is the average atomic weight of isotopes. The element which exhibits highest number of stable isotopes |
|
Answer» Sn |
|
| 17. |
PASSAGE - 2 : In periodic table there are nearly 110 elements are present majority of elements are present in solid state. There is no separate placement for isotopes of elements in periodic table. The atomic weight of an element is the average atomic weight of isotopes. In periodic table the liquid elements at room temperature are |
|
Answer» CD,`Cl_2` |
|
| 18. |
Passage 1 Synthetic detergents are cleansing agents which have all the propeties of soaps but they actually do no contain any soap. These can be used in both hard as well as soft water as they give foam even in hard water. All the detergents are not biodegadable and hence cause water pollution in rivers and other water ways. 5. Which of the following is an example of liquid dishwashing detergents ? |
|
Answer» `CH_(3)-(CH_(2))_(10)-CH_(2)OSO_(3)^(-)Na^(+)` |
|
| 19. |
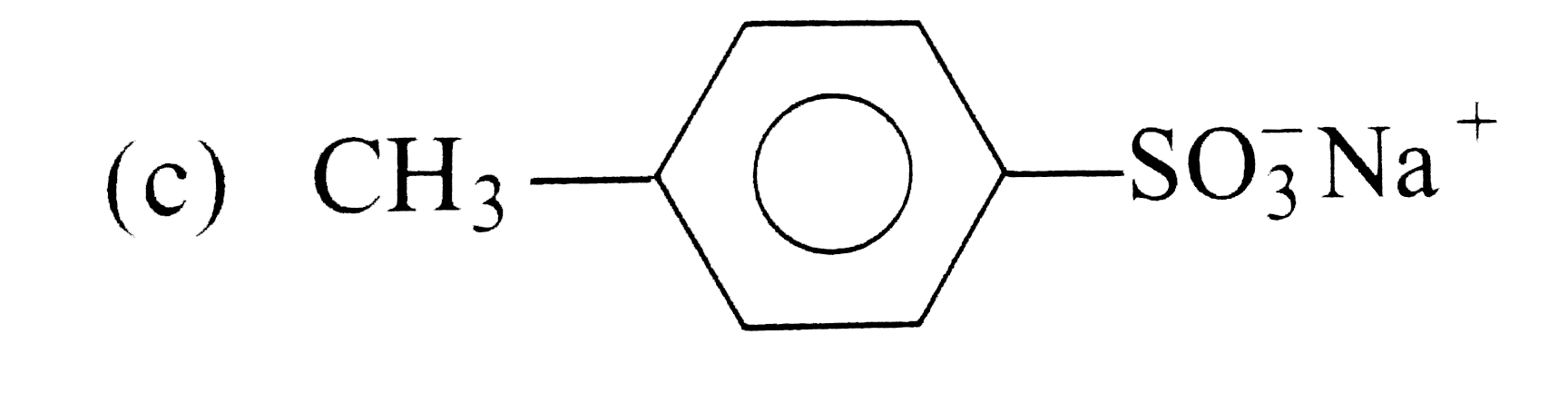
Passage 1 Synthetic detergents are cleansing agents which have all the propeties of soaps but they actually do no contain any soap. These can be used in both hard as well as soft water as they give foam even in hard water. All the detergents are not biodegadable and hence cause water pollution in rivers and other water ways. Answer the following questions : 1. Which of the following are anionic detergents ? |
|
Answer» Quarternary AMMONIUM salt of amine and acetate |
|
| 20. |
Passage 1 Synthetic detergents are cleansing agents which have all the propeties of soaps but they actually do no contain any soap. These can be used in both hard as well as soft water as they give foam even in hard water. All the detergents are not biodegadable and hence cause water pollution in rivers and other water ways. 4. Which of the following are not-ionic detergents ? |
|
Answer» Lauryl ALCOHOL ethoxylate |
|
| 21. |
Passage 1 Synthetic detergents are cleansing agents which have all the propeties of soaps but they actually do no contain any soap. These can be used in both hard as well as soft water as they give foam even in hard water. All the detergents are not biodegadable and hence cause water pollution in rivers and other water ways. 2. Which of the following are cationic detergents ? |
|
Answer» SODIUM lauryl sulphate |
|
| 22. |
PASSAGE - 1: In Mendeleeff's periodic table elements were classified on the basis of atomic weight & in modern periodic table on the atomic number. The lighter elements of 2 & 3 periods show similarities in their properties called diagonal relationship Due to digonal relation which pair are amphoteric in nature |
|
Answer» `Li_2,O,MGO` |
|
| 23. |
PASSAGE - 1: In Mendeleeff's periodic table elements were classified on the basis of atomic weight & in modern periodic table on the atomic number. The lighter elements of 2 & 3 periods show similarities in their properties called diagonal relationship The atomic number of element separated from main body of periodic table is |
|
Answer» 64 |
|
| 24. |
PASSAGE - 1: In Mendeleeff's periodic table elements were classified on the basis of atomic weight & in modern periodic table on the atomic number. The lighter elements of 2 & 3 periods show similarities in their properties called diagonal relationship 1. Which of the following element is not present in Mendeleeff's periodic table |
|
Answer» CARBON |
|
| 25. |
Particulate pollutants will result in the health hazard named as .......... |
| Answer» SOLUTION :PNEUMOCONIOSIS | |
| 26. |
Particulates are added to the atmosphere by : |
|
Answer» INDUSTRIAL processes |
|
| 27. |
Particles with 5 micron size reaches to the longs directly . |
| Answer» Solution :False statement (PARTICLE with 1 micron SIZE reaches to the LONG DIRECTLY.) | |
| 28. |
Particles of soil at the bottom of a river remain separated, but they sick together when taken out. Name the property behind this. |
| Answer» SOLUTION :SURFACE TENSION | |
| 29. |
Particle nature of electron is proved by |
|
Answer» Radioactivity |
|
| 30. |
Partial pressure of O_(2) in the reaction 1//2 P_(2) + 1//2 Q_(2) + 1//2 R_(2) hArr PQR, |
|
Answer» `K_(p)` `( :' [Ag (s)] =[Ag_(2)O (s) ] = 1 )` |
|
| 31. |
Partial pressure of (p) is in proportional to the concentration (c) - explain. |
|
Answer» Solution :For REACTIONS involving gases, however, it is usually more convenient to express the equilibrium CONSTANT in terms of partial PRESSURE. Equation of idea gas pV nRT ....(Eq.-i) `therefore p=(n/V)RT` …..(Eq. ii) If concentration =c mol `L^(-1)` OR mol/`dm^3` `therefore` p=cRT=[gas]RT ...(Eq. iii) where , R=0.0831 bar L `mol^(-1) K^(-1)` where, p = Pressure in pascal, n = Number of mol of the gas, V = Volume of gas `m^3` T = Temperature in Kelvin, `therefore n/V`= Concentration expressed in mol/`m^3` `therefore` At constant temperature, the p of the gas is proportional to its concentration p`prop`[gas], `1_"Pascal"=1 Nm^(-2)` and 1 bar = `10^5` Pa 1.013 bar =1 atm . |
|
| 32. |
Partial pressure is given as __________ |
|
Answer» `("mole fraction")/("TOTAL pressure")` |
|
| 33. |
Paramagnetism isdue to the presence of _______ electrons . |
|
Answer» PAIRED |
|
| 34. |
Paramagnetic species are- |
|
Answer» `O_(2)^(+)` |
|
| 35. |
Paramagnetic species are |
|
Answer» `O_(2)^(+)` |
|
| 36. |
Paragraph//Comprehension-2 Environment is defined as the sum total of physical and biological factors that directly influence the survival, growth ,development and reproduction of organisms.The additon of certain harmful wastes to air , water and soil by a natural source or due to certain human activitites to such a level of concentration which adversely effects the quality of environment is called environmental p[ollution and the undesirable material thus added to environment is called pollutant Photochemeical smog is the term used for the air [pollutants produced in the atmopsphere as a result of photochemical reactions .Classical smog or London smog is a combination of particulateed and gaseouir oxided of suphur present in the atomospher.CO_(2) is not considered as atmospheric polllutant but its increased concentrate leads to green house effect. Increased CO_(2) content in the atmosphee will lead to |
|
Answer» INCREASE in EARTH's temperature |
|
| 37. |
Paraffin means ………….. |
| Answer» SOLUTION :Littleactivity | |
| 39. |
Para toluidine is treated with HNO_(2) at ice cold condtions and then boiled with water. The final product obtained is: |
|
Answer» ANTHRANILIC acid |
|
| 40. |
Para and ortho H_(2) differ in |
|
Answer» ATOMIC number |
|
| 41. |
Para and ortho hydrogen differ in |
|
Answer» ATOMIC NUMBER |
|
| 42. |
Paper chromatography is also called.............chromatography in which..............constitutes thestationary phase while a....................constitutes the mobile phase. |
|
Answer» |
|
| 43. |
Paper chromatography has following mobile and stationary phases respectively |
|
Answer» liquid, solid |
|
| 44. |
PAN is one of the constituents of photochemical smog.(T/F) |
|
Answer» |
|
| 46. |
Pair of ions with similar ionic radii |
|
Answer» `LI^(+), MG^(2+)` |
|
| 47. |
Pair of ions which are having same number of unpaired electrons |
|
Answer» `Eu^(3+), Tb^(3+)` |
|
| 48. |
Pair of ions which are having only one unpair electron |
|
Answer» `CE^(3+), Yb^(3+)` |
|
| 49. |
Pair of elements with equal values of electronegativiy |
|
Answer» Be, Al |
|