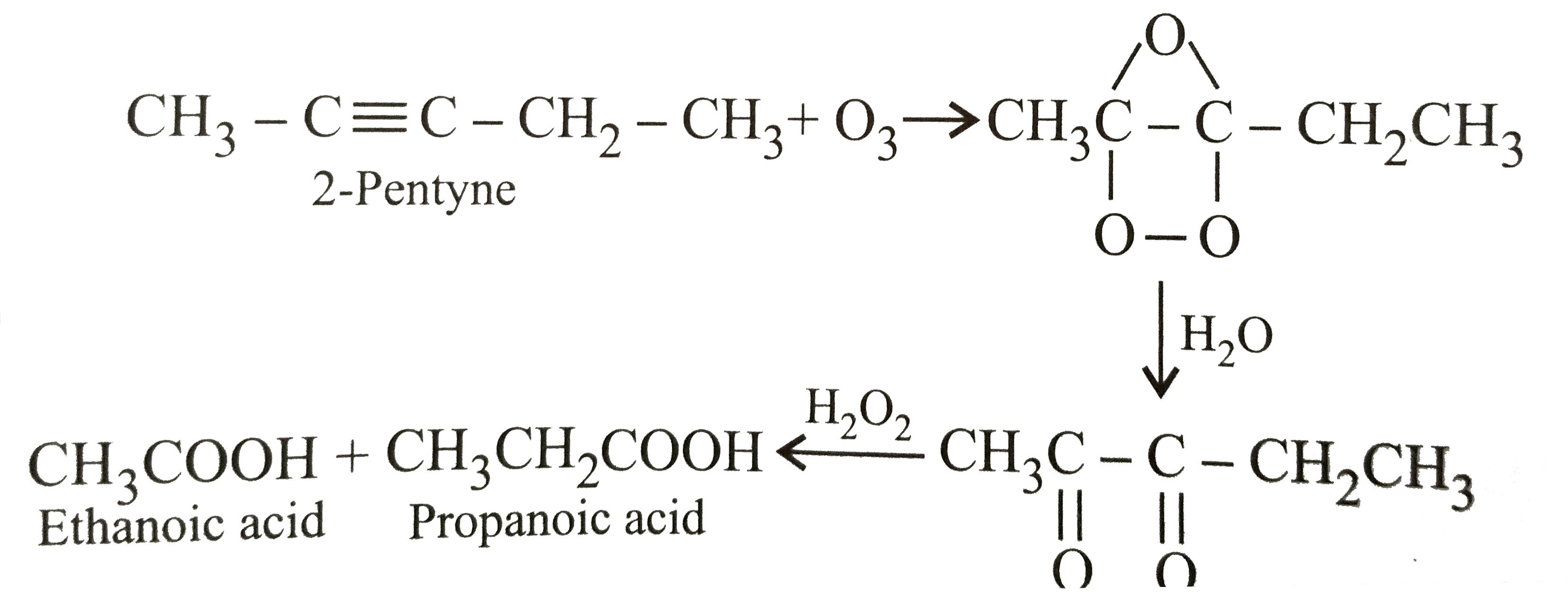Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pair of elements with the following atomic numbers have the same chemical properties |
|
Answer» 13 and 22 |
|
| 2. |
...........pair of compounds is present in acid-rain water. |
| Answer» Solution :`HNO_(3) + H_(2)SO_(4)` | |
| 3. |
...................pair of compounds is found to be highly responsible for depletion of ozone layer |
| Answer» SOLUTION :NITRIC OXIDE + CFC | |
| 4. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 Which type of stacking pattern is found insodium chloride 's crystal lattice? |
|
Answer» AB AB |
|
| 5. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 The A certain oxide of metal M crystallises in suchway that O^(2-) ions occupy hcp arrangement following AB AB....pattern. The metals ions ,however ,occupy (2)/(3)rd of the octahedral voids. The formula of the compound is: |
|
Answer» `M_(2)O_(3)` |
|
| 6. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 The spaced occupied by spheres in bcc arrangement is: |
|
Answer» 0.74 |
|
| 7. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement lt brgt "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 The closed packed lattice containing 'n' particles ,the numbers of tetrahedral and octahedral voids are: |
| Answer» ANSWER :C | |
| 8. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 The pattern of successive layers of ccp arrangement can be designated as: |
|
Answer» AB AB AB ….. |
|
| 9. |
Packing refers to the arrangement of constituent units in such a way that the forces of attraction among the constituent particles is maximum and the constituents occupy the maximum available space. In two -dimensions ,there are square close packing and hexagonal close packing .In three- dimensions ,however, there are hexagonal close packing, cubic close packing and body-centered cubic packing. (i) hcp:AB AB AB AB ..... arrangement ""coordination no. = 12 "" % occupied space = 74 (ii) ccp:ABC ABC.... arrangement lt brgt "" coordination no. = 12 ""% occupied space = 74 bcc:68% space is occupied ""coordination no.= 8 The empty space laft in three -dimensions is : |
| Answer» ANSWER :A | |
| 10. |
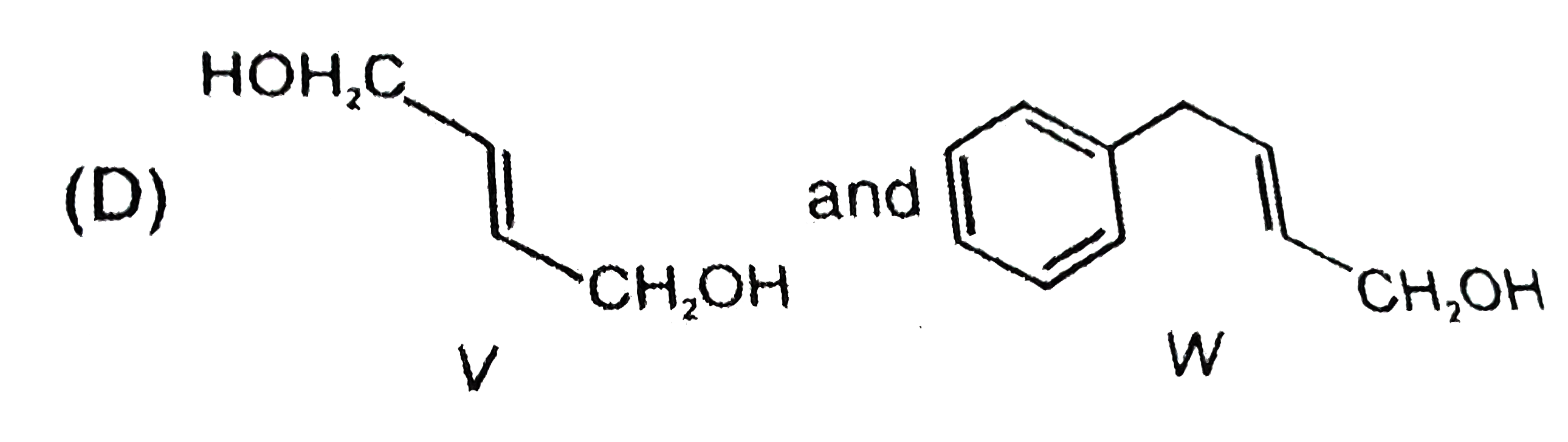
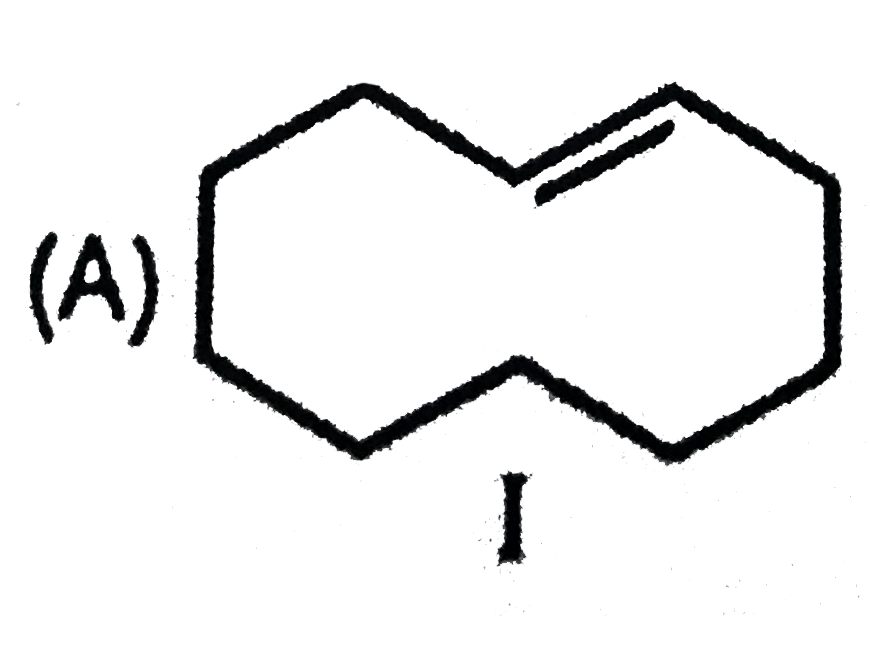
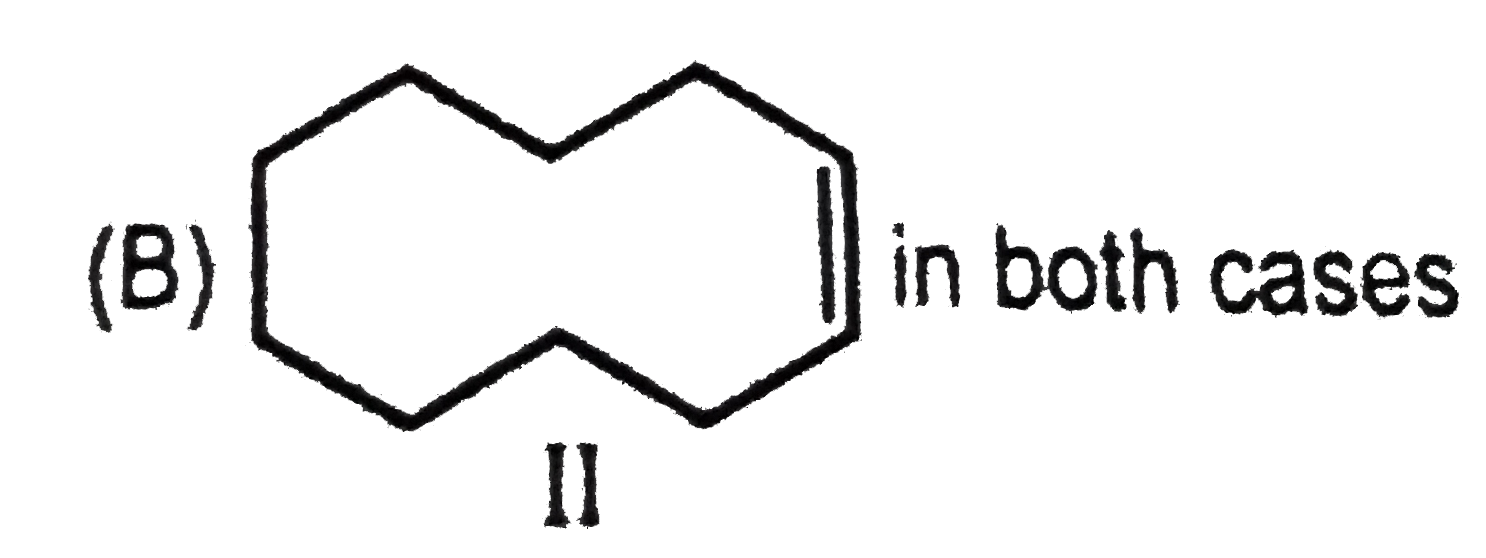
Packing fraction for which of the following nuclide is zero ? |
| Answer» Answer :D | |
| 11. |
Packing fraction in an identical solid spheres is 74%in |
| Answer» Solution :Packing fraction is same (74%) in HCP and CCP (or FCC) structure. | |
| 12. |
What is the packet of enegry called? |
|
Answer» Electron |
|
| 13. |
P_(4)O_(6) reacts with water according to equation, P_(4)O_(6)+6H_(2)Oto4H_(3)PO_(3). Calculate the volume of 0.1 M NaOH solution required to neutralise the acid formed by dissolving 1.1 g of P_(4)O_(6)" in "H_(2)O. |
|
Answer» SOLUTION :`{:(P_(4)O_(6)+6H_(2)Oto4H_(3)PO_(4)"(Hydrolysis reaction)"),(H_(3)PO_(3)+2NaOHtoNa_(2)HPO_(3)+2H_(2)O"]"xx4 "(Neutrolisation reaction)"),(overline(underset(=220g)underset(4xx31+6xx16)(P_(4)O_(6))+underset(=320 g)underset(8xx40)(8NaOH )to 4Na_(2)HPO_(3)+2H_(2)O)"(Overall reaction)"):}` Now 220 g of `P_(4)O_(6)` require NAOH for neutralization =320 g `:.` 1.1 g of `P_(4)O_(6)` will require `NaOH=(320)/(220)xx1.1=1.6g` Now 1000 mL of 0.1 MHCl contain `NaOH=40xx0.1=4g` In other words, 4 g of NaOH are present in 0.1 M NaOH `=(1000)/(4)xx1.6=400mL=0.4L` |
|
| 15. |
P_(4)O_(10) is the anhydride of |
| Answer» Solution :`4H_(3)PO_(4)toP_(4)O_(10)+6H_(2)O`. | |
| 16. |
P_4O_10 is an anhydride of which compound? |
|
Answer» `H_3PO_2` |
|
| 17. |
P_(4)+3O_(2)F_(2)toP_(4)F_(6)+3O_(2) Which is the reducing agent in the above reactions ? |
|
Answer» <P>`P_(4)andO_(2)F_(2)` both  (i) `P_(4)(0)` is convected to `P_(4)(+6)` `thereforeP_(4)` is oxidised `thereforeP_(4)` is reducing agent … (D) |
|
| 18. |
P_(1) and P_(2) are the vapour pressures of pure liquid components,1 and 2 respectively of an ideal binary solution if x_(1) represents the mole fraction of component 1, the total pressure of the solution formed by 1 and 2 will be….......... |
|
Answer» <P>`P_(1)+x_(1)(P_(2)-P_(1))` =`P_(1)x_(1)+P_(2)x_(2)``[becausex_(1)+x_(2)=1x_(1)=1-x_(2)]` =`P_(1)(1-x_(2))+P_(2)x_(2)` =`P_(1)-P_(1)x_(2)+P_(2)x_(2)=P_(1)-x_(2)(P_(1)-P_(2))` |
|
| 19. |
P_1and P_2 are the vapour pressures of pure liquid components ,1 and 2 respectively of an ideal binary solution ifx_1 represents the mole Traction of component1, and the total pressure of the solutions formed by 1 and 2 will be |
|
Answer» `p _(1) + X _(1) (P_(2) -P_(1))` `=P_(1)x_(1) + P_(2) x_(2)""[because x _(1) +x_(2) =1 x _(1) =1 - x _(2)]` ` = P _(1) (1- x _(2)) + P_(2) x _(2)` ` = P_(1) - P_(1) x _(2) + P _(2) x _(2) = P_(1) -x _(2) (P_(1) -P_(2))` |
|
| 20. |
{:("p-shell...3p orbital",-1),("" "of equal energy",0),(," "1):}}{:("3p orbital in"),("magnetic field"):} Hence each magnetic quantum number corresponds to a different quantum state in magnetic field. This explain the term magnetic quantum number. |
| Answer» | |
| 21. |
p-Phenolsulphonic acid is obtained as the major product when phenol is treated with |
|
Answer» Conc. `H_(2)SO_(4)` at 373 K |
|
| 23. |
(P) overset(Na//NH_(3)(l))rarr "Product(s)" Product (s) has/have X = degree of unsaturation and Y = number of isomeric product (s) formed. Then X + Y = ? |
Answer» 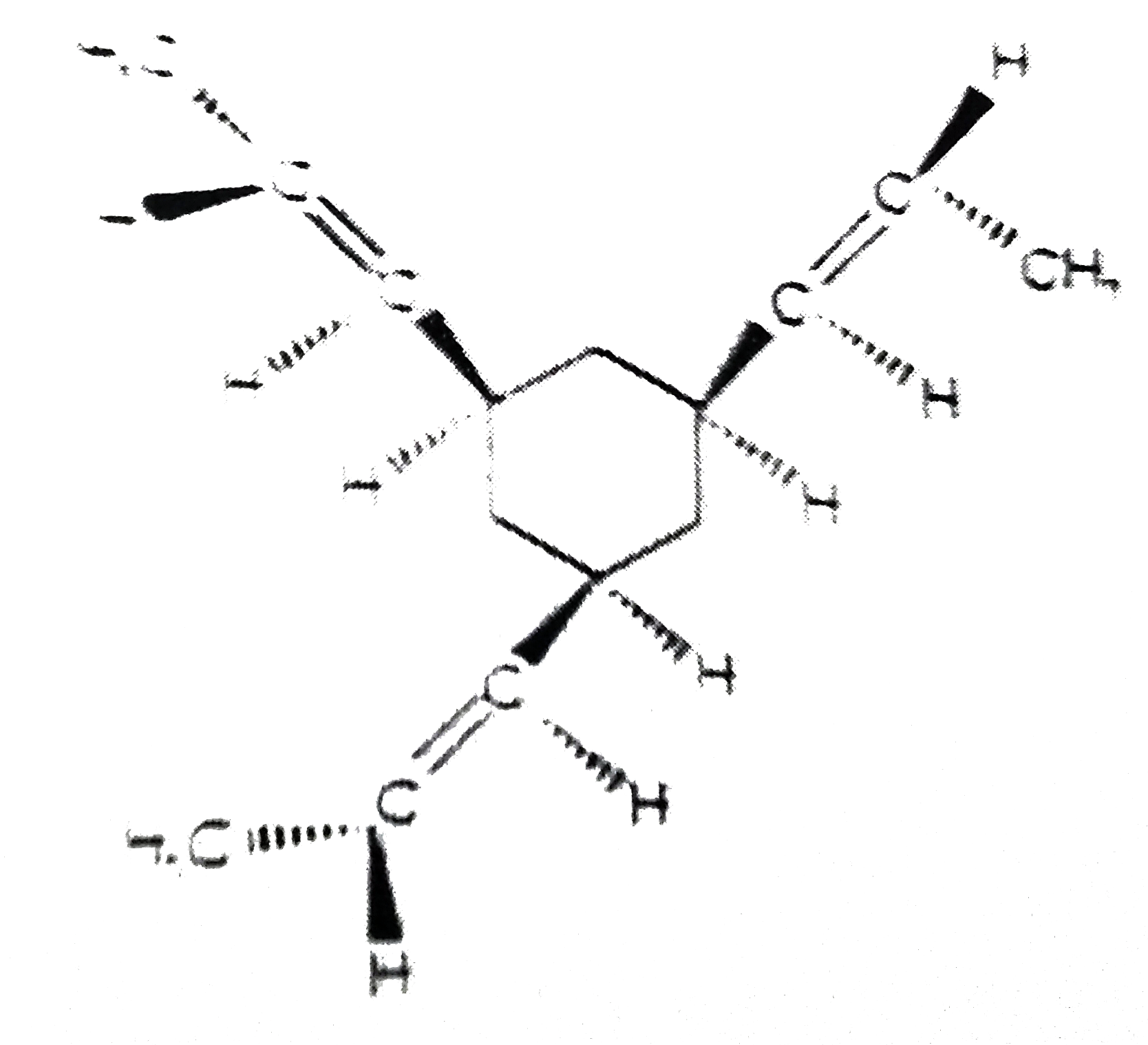
|
|
| 24. |
(P) N_2(g)+O_2(g)hArr2NO(g),""K_(1) (Q) ((1)/(2))N_2(g)+((1)/(2))O_2(g)hArrNO(g),""K_(2) (R)2NO(g)hArrN_2(g)+O_2(g),""K_(3) (S)NO(g)hArr((1)/(2))N_2(g)+((1)/(2))O_2(g),""K_(4) Correct relation between K_1,K_2,K_3and K_4 is//are: |
|
Answer» `K_1K_3=1` |
|
| 25. |
P is an alcohol which onheating withAl_(2)O_(3)_(3) forms an alkene Q. Q on ozonolysis produces R and S. when the mixture of R andS is heated with conc. NaOH, redox reaction takes place and a mixture of an acid salt and alcohol is formed.The compound R and S are |
|
Answer» `CH_(3)-OVERSET(CH_(3))overset(|)(C)=O,CH_(2)=O` `,Q= CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=CH_(2) , ""R&S=CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=O,O+CH_(2)=O` |
|
| 26. |
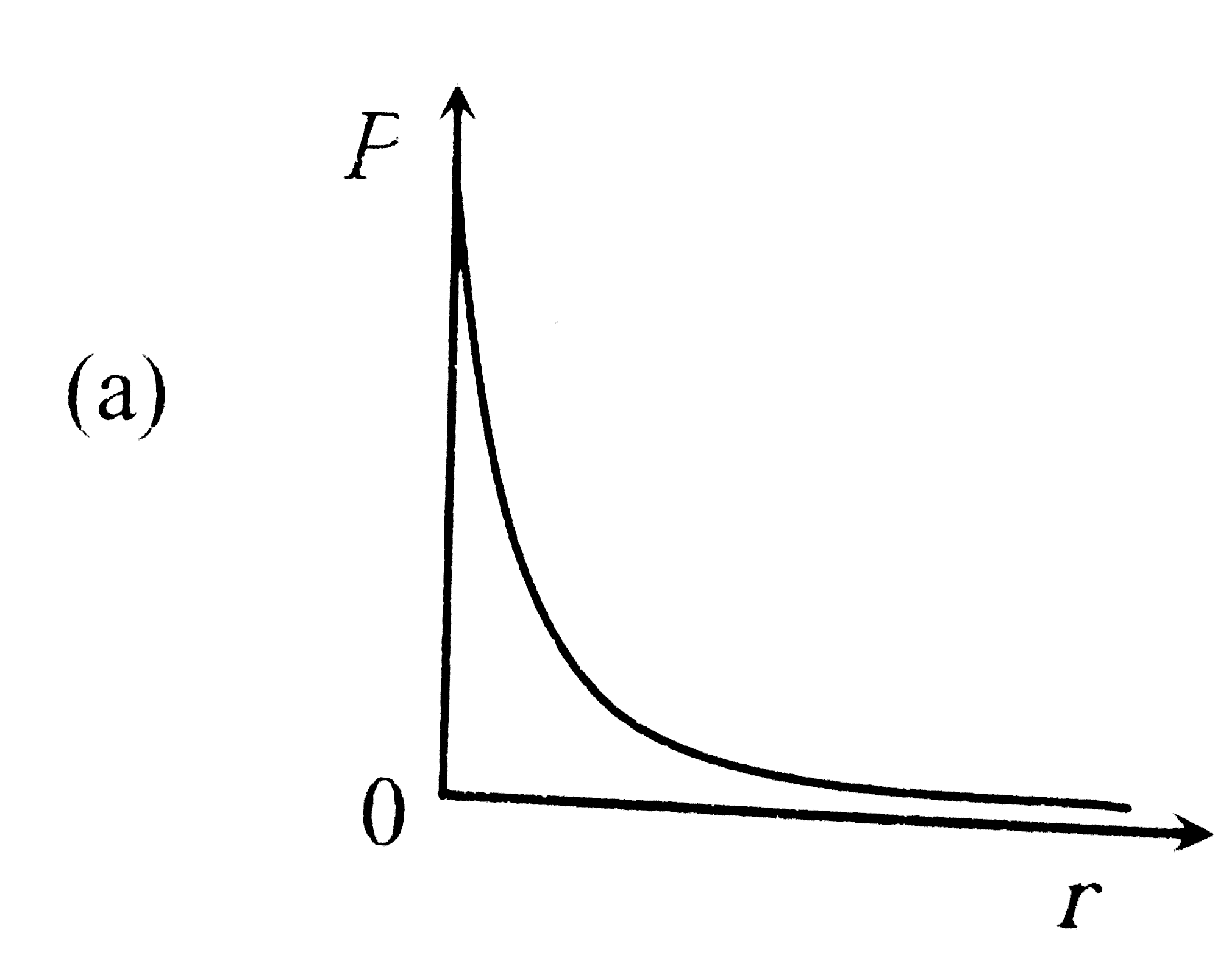
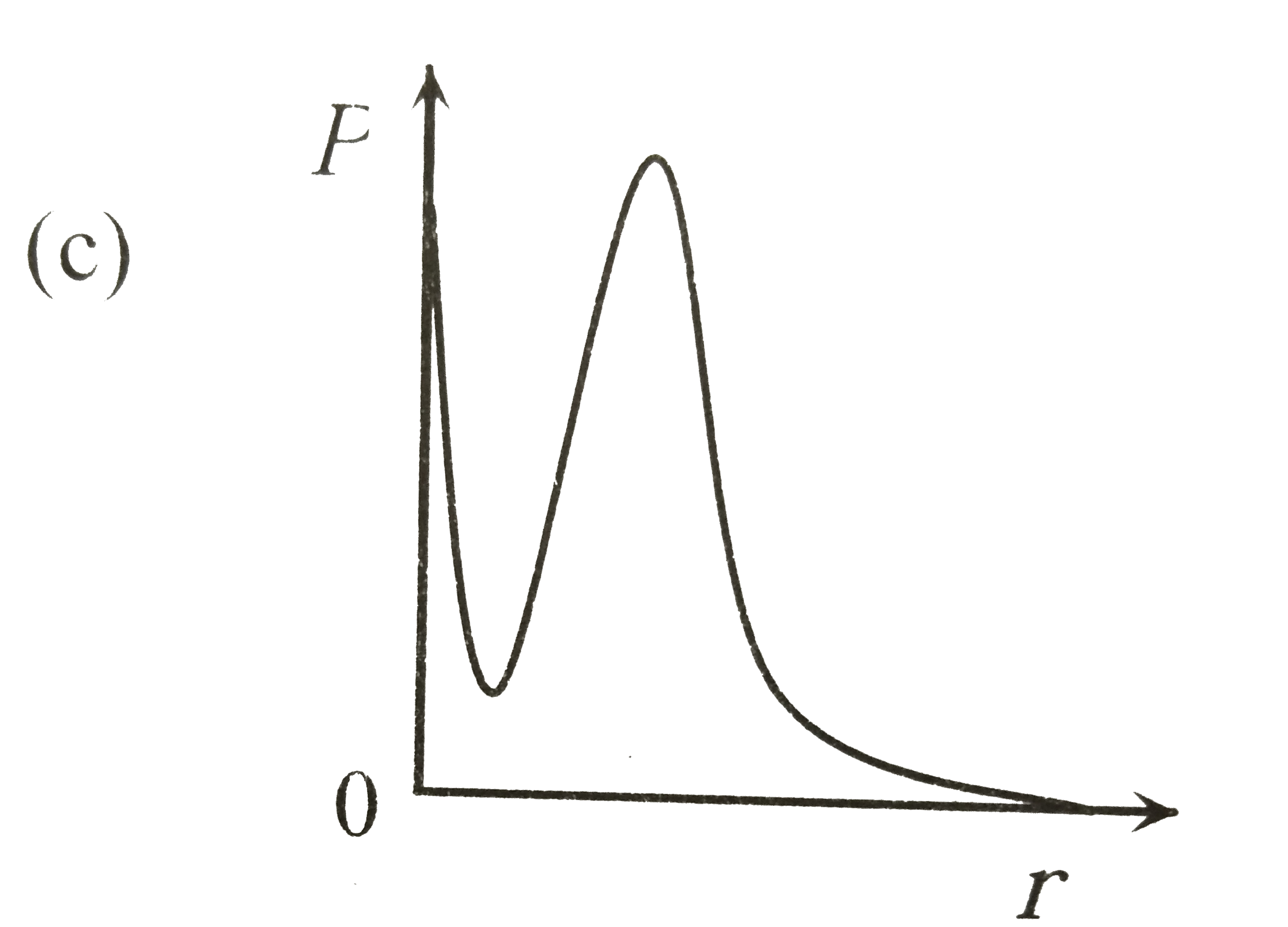
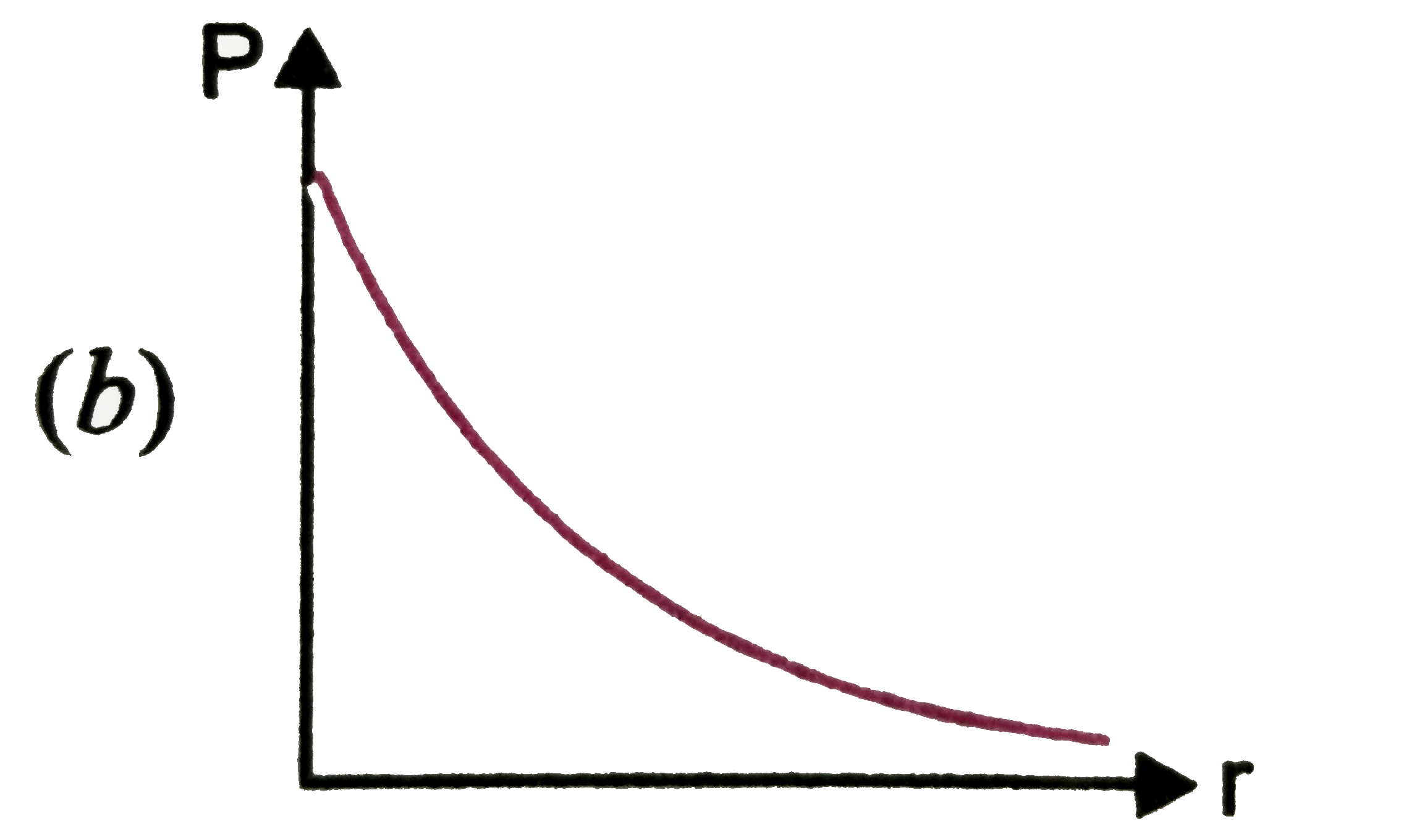
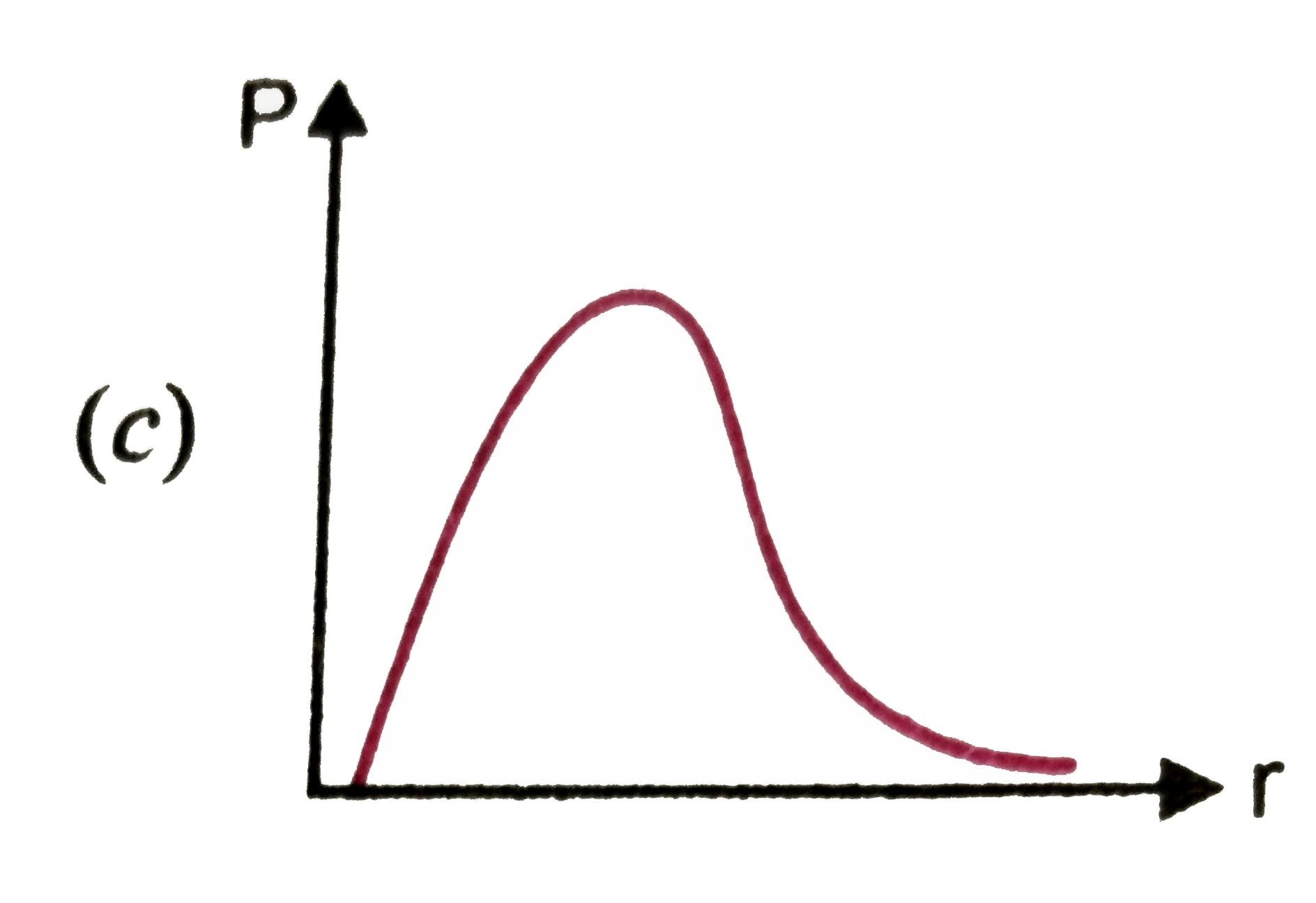
P is the probability of finding the 1 s electron of hydrogen atom in a spherical shell of infinitesimal thickness, dr , at a distance r from nucleus. The volume of this shell is 4pi r^2 dr. The qualitative sketch of thedependence of P on r is |
|
Answer»
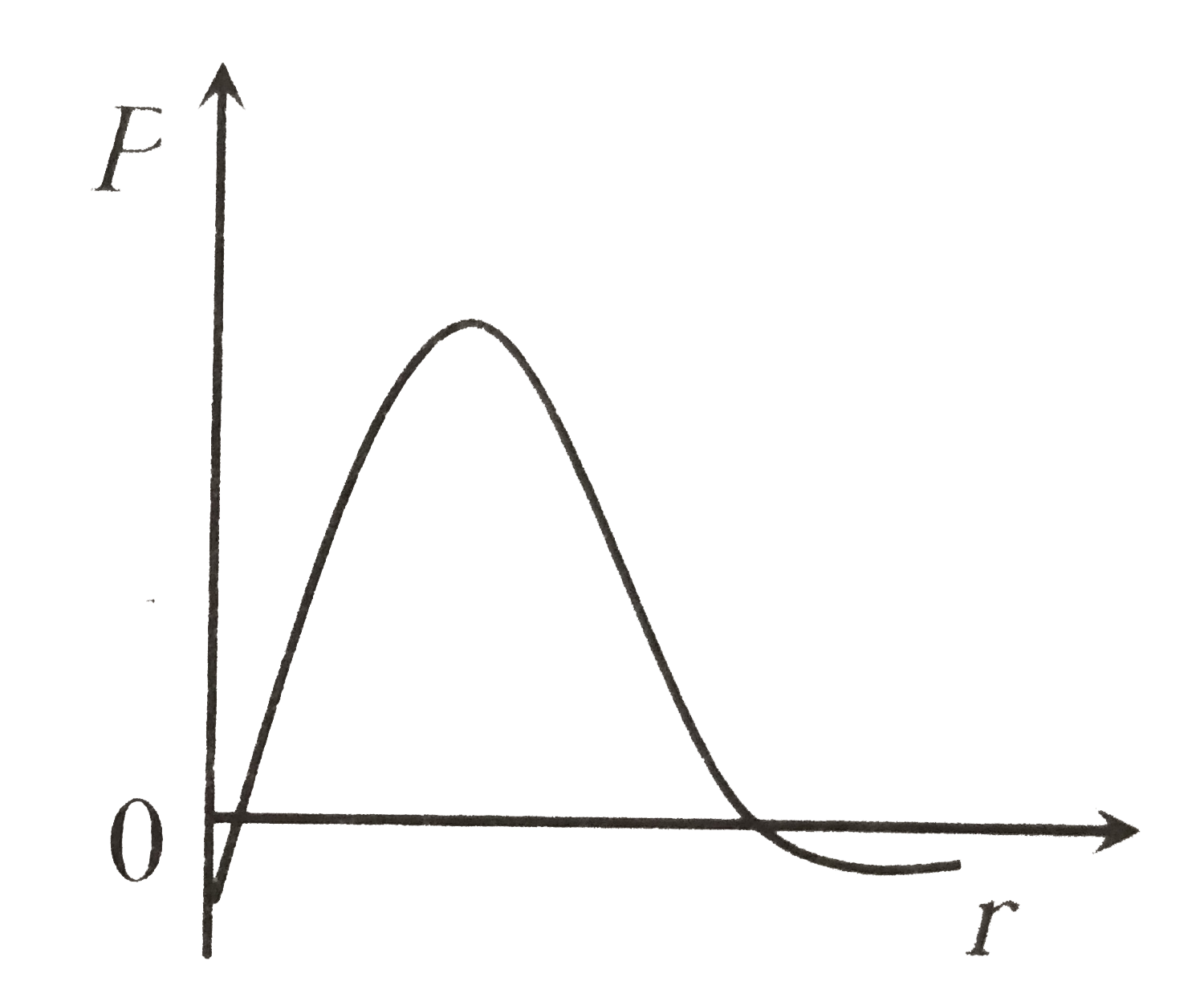
|
|
| 27. |
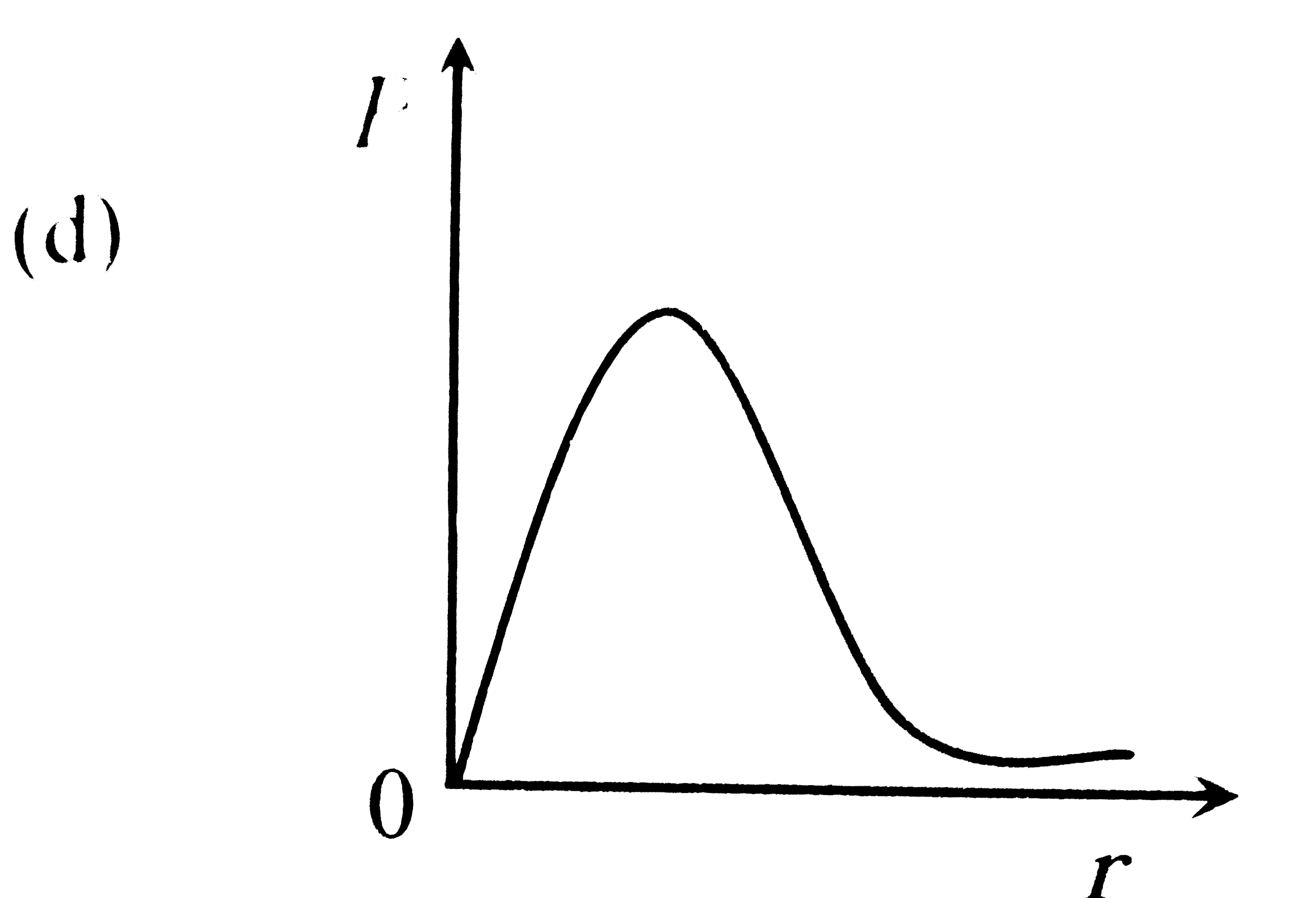
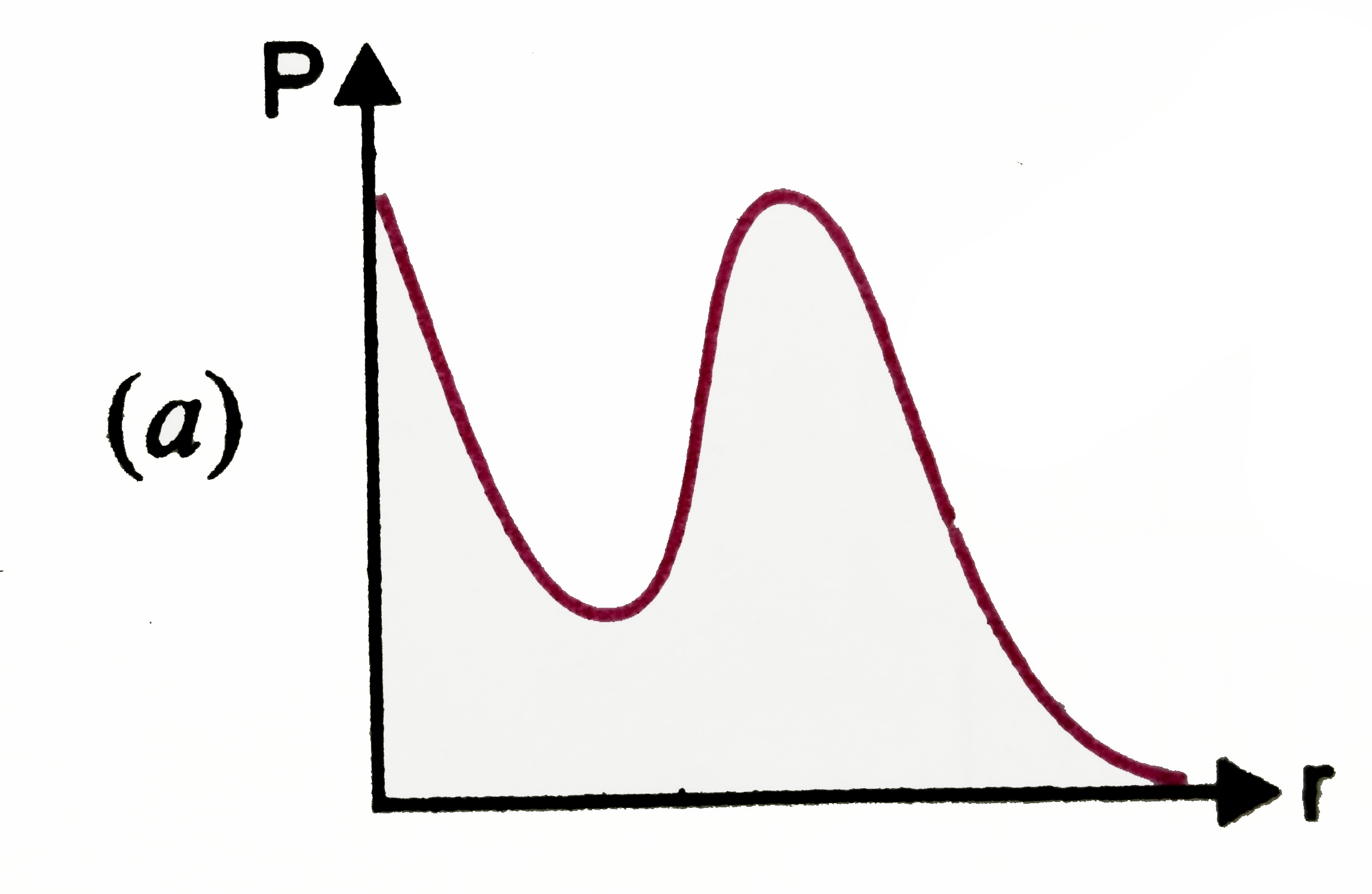
P is the probability of finding the 1s electron hydrogen atom in a spherical shell of infinitesimal thickness dr, at a distance r from the nucleus. The volume of this shell is 4pi r^(2) dr. The qualitative sketch of the dependence of P on r is |
|
Answer»
|
|
| 28. |
P is an alcohol which onheating withAl_(2)O_(3)_(3) forms an alkene Q. Q on ozonolysis produces R and S. when the mixture of R andS is heated with conc. NaOH, redox reaction takes place and a mixture of an acid salt and alcohol is formed.The compound (Q) is |
|
Answer» `CH_(3)-overset(CH_(3))overset(|)(C)=CH_(3)` `,Q= CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=CH_(2) , ""R&S=CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=O,O+CH_(2)=O` |
|
| 29. |
P is an alcohol which onheating withAl_(2)O_(3) forms an alkene Q. Q on ozonolysis produces R and S. when the mixture of R andS is heated with conc. NaOH, redox reaction takes place and a mixture of an acid salt and alcohol is formed.the alcohol (P) is obtained by |
|
Answer» `H_(3)C-underset(O)underset(||)(C)-CH_(2)-CH_(3)+CH_(3)MGBR` `,Q= CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=CH_(2) , ""R&S=CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-CH=O,O+CH_(2)=O` |
|
| 30. |
(p) ifazimythalquantum numbervalueis 3, themaximum value of spinmultiplicity is x. (Q) if valuesof priecipalquntumnumberazimuthal quantumnumber,magnetic of unparedelectronspresentin copper will be : |
|
Answer» |
|
| 31. |
P , identitify P |
|
Answer»

|
|
| 32. |
p-dichlorobenzene has higher melting point than ortho and meta dichloro benzene. Why? |
|
Answer» Solution :The MELTING point of p-dichloro benzene is higher than the melting point of the CORRESPONDING ortho and META ISOMERS. The higher melting which leads to more close packing of its moleules m the crystal lattice and consequently strong intermolecular ATTRACTIVE forces which requires more energy for melting. p-dichlorobenzene `gt` o-dichlorobenzene `gt` m-dichlorobenzene |
|
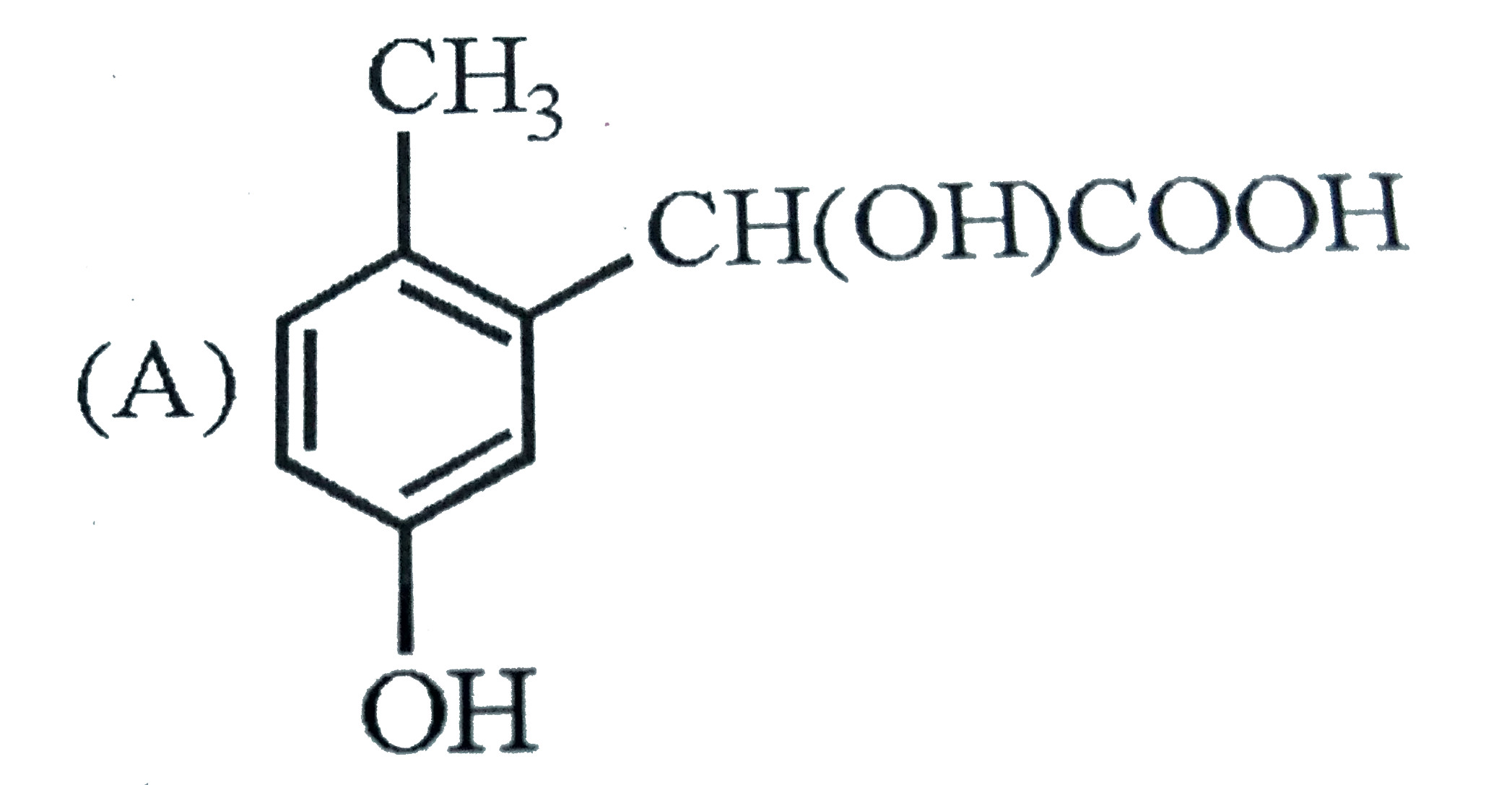
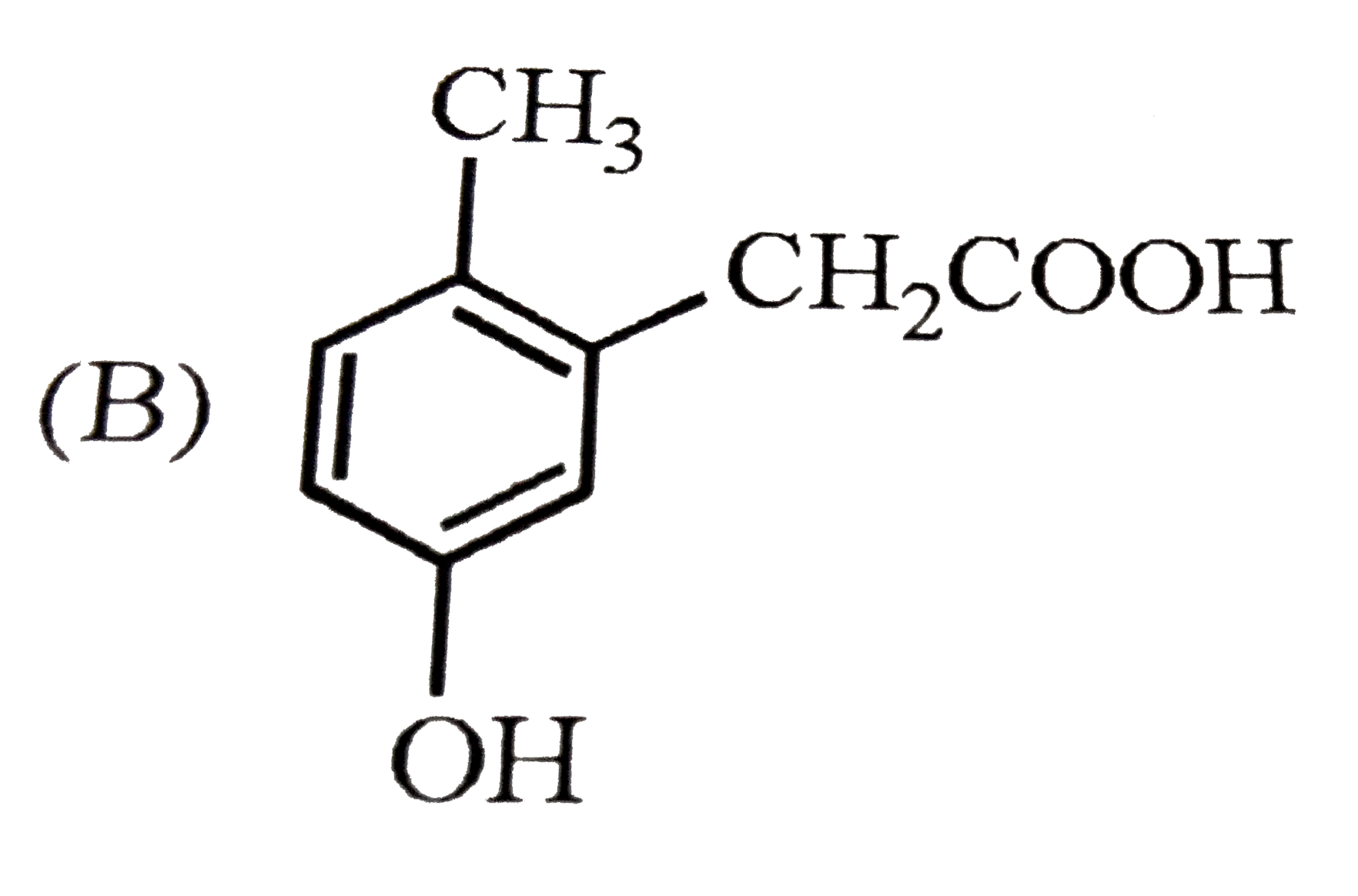
| 33. |
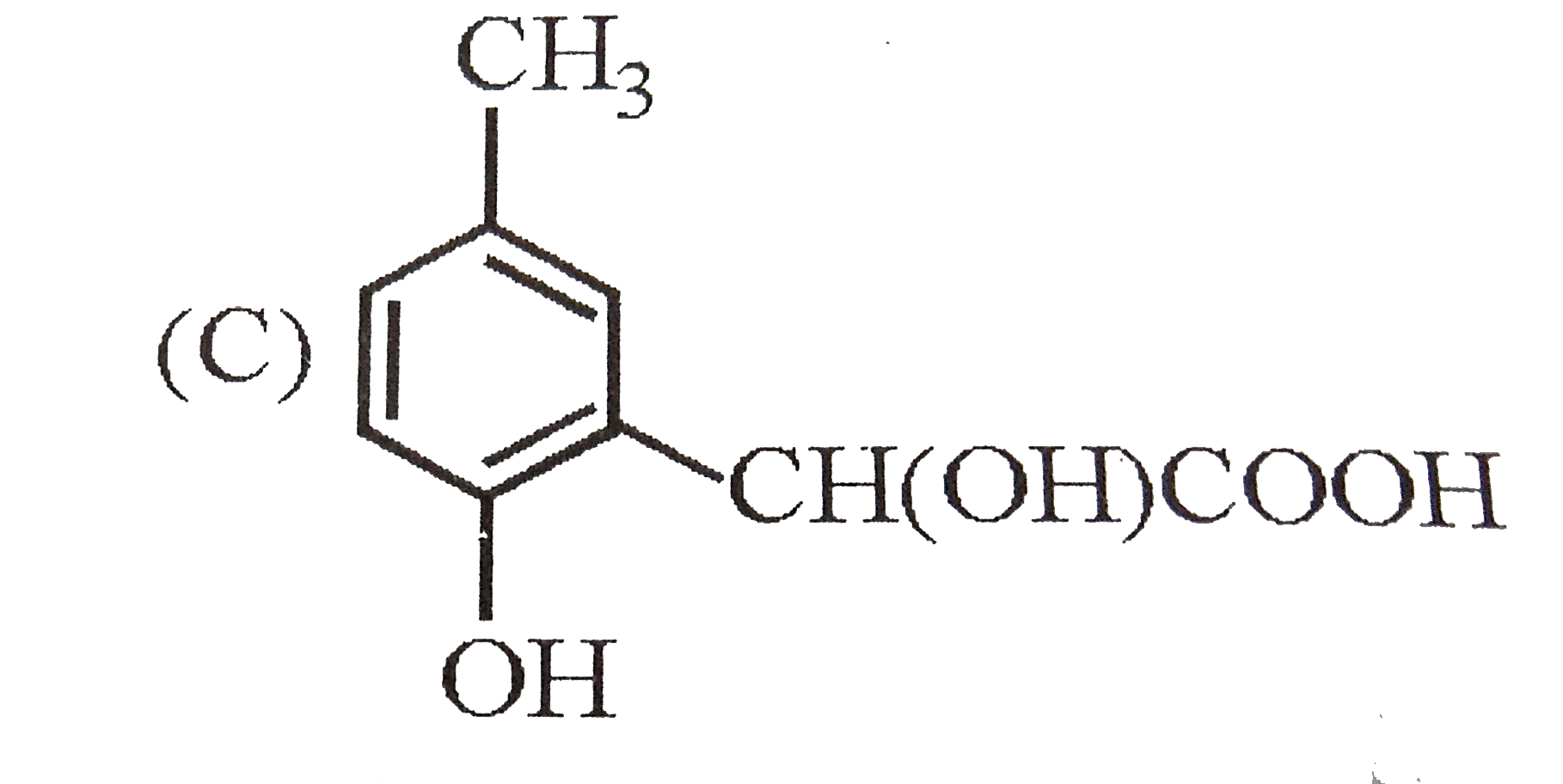
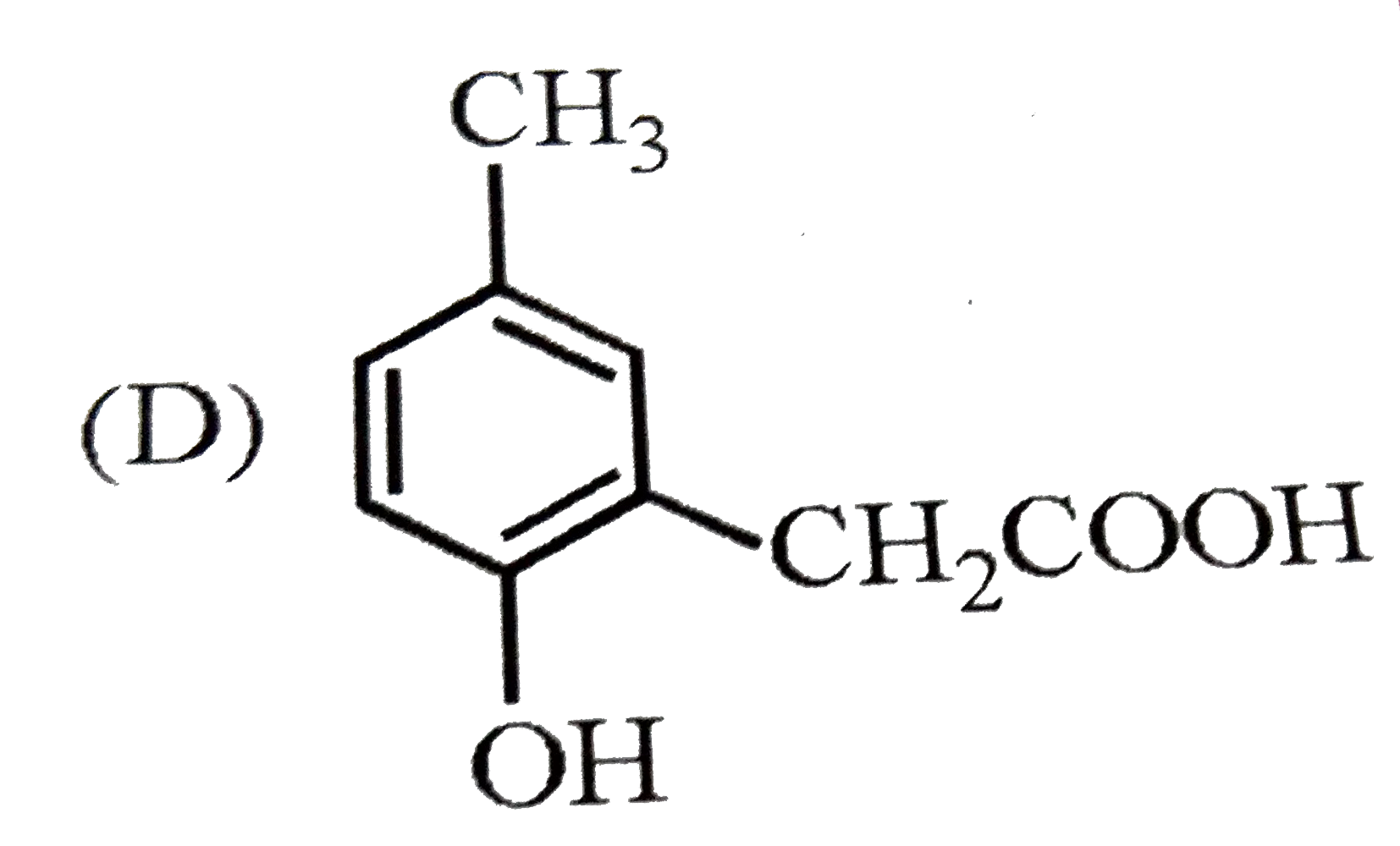
p-Cresol reacts with chloroform in alkaline medium to give the compound X which adds hydrogen cyanide to form, the compound Y. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is |
|
Answer»
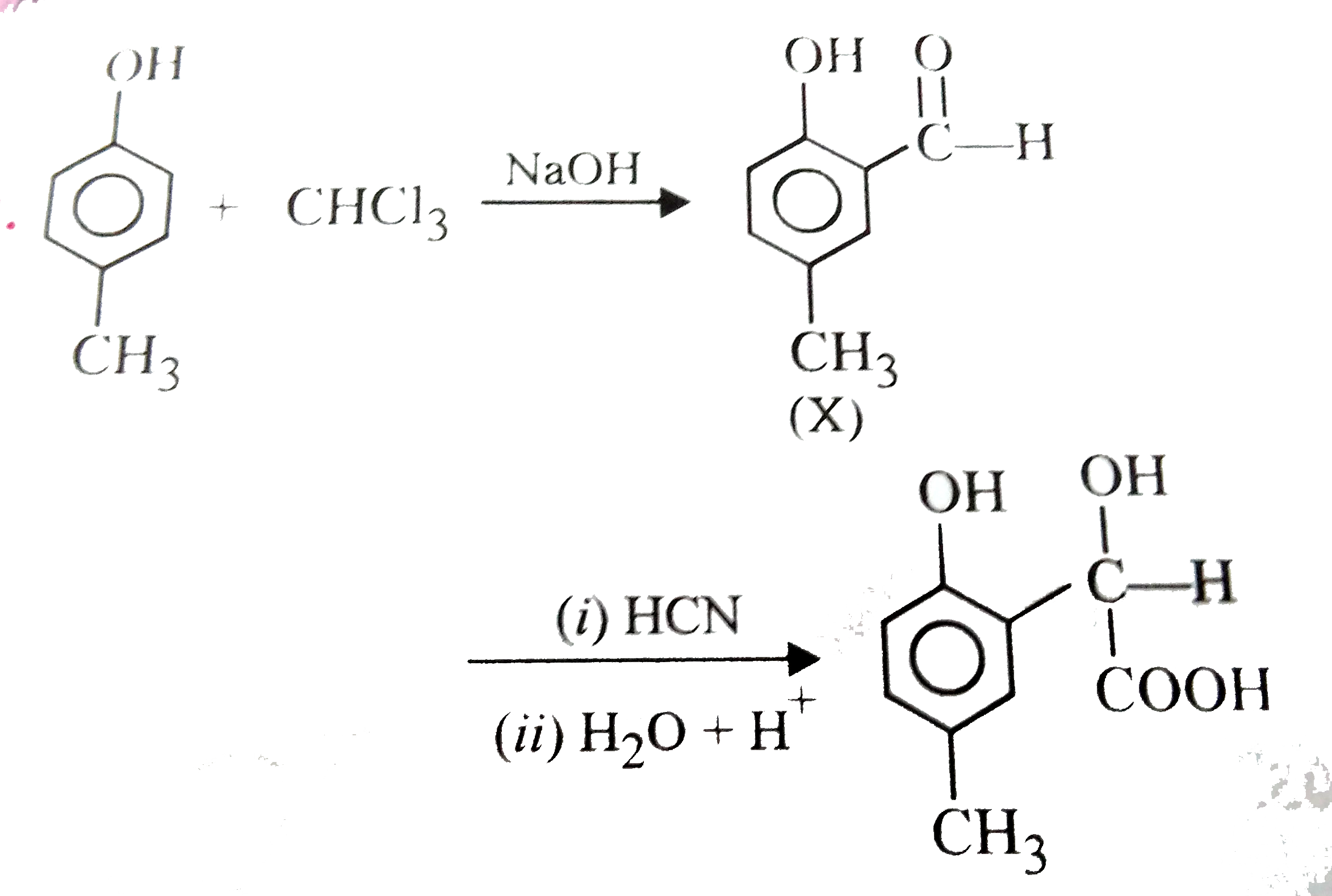 Here `-OH` group has STRONGER directive INFLUENCE than `-CH_(3)` group. |
|
| 34. |
p bond is weaker than s bond. |
| Answer» Solution :In SIGMA BAND the overlapping is maximum where as in PI BOND the overlapping is minimum. | |
| 35. |
p-block elements form acidic, basic and amphoteric oxides. Explanin each property by giving two examples, and also write the reactions of these oxides with water. |
|
Answer» i) 2nd period `B_(2)O_(3) lt CO_(2) lt N_(2)O_(3)` acidic nature in increases. ii) 3rd period `Al_(2)O_(3) lt SiO_(2) lt P_(4)O_(10) lt SO_(3)ltCl_(2)O_(7)` Acidic character increases. On MOVING down the group, acidic character decreases and basic character increases, e.g., a) Nature of oxides of 13 group elements.  b) Nature of oxides of 15 group elements. `underset("Strongly acidic")(N_(2)O_(5))` `underset("Moderately acidic")(P_(4)O_(10))` `underset("Amphoteric")(As_(4)O_(10))` `underset("Amphoteric")(Sb_(4)O_(1))``underset("Basic")(Bi_(2)O_(3))` Among the oxides of same element, higher the oxidation state of hte element, stronger is the acid, e.g., `SO_(3)` is a stronger acid than `SO_(2)`. `B_(2)O_(3)` is weakly acidic and on dissolution in water, it forms orthoboric aicd. Orthoboric aicd does not ACT as a PROTONIC acid (it does not ionise) but acts as a weak lewis acid. `underset("Boron trioxide")B_(2)O_(3) + 3H_(2)O ltimplies underset("Orthoboric acid")2H_(3)BO_(3)` `B(OH)_(3) + H- OH to [B(OH)_(4)]^(-)+H^(+)` `Al_(2)O_(3)` is amphoteric in nature. It is insoluble in water but dissolves in alkaklies and reacts with acids. `("Aluminium trioxide")Al_(2)O_(3) + 2NaOH overset(DELTA)to underset("Sodium meta aluminate")2NaAlO_(2) + H_(2)O larr Al_(2)O_(3) + 6HCl overset(Delta)to underset("Aluminium chlroide")2AlCl_(3) + 3H_(2)O` `Ti_(2)O` is an basic as NaOH due to its lower oxidation state `(+1)` `TI_(2)O + 2HCl to 2TICl + H_(2)O` `P_(4)O_(10)` on reaction with water gives orthophosphoric acid. `underset("Phosphorous pentoxide")(P_(4)O_(10)) + 6H_(2)O to underset("Orthophosphoric acid")(4H_(3)PO_(4))` `Cl_(2)O_(7)` is strongly acidic in nature and on dissolution in water, it gives perchloric acid `underset("Dichlorine heptoxide")(Cl_(2)O_(7)) + H_(2)O to underset("Perchloric acid")2HCIO_(4)` |
|
| 36. |
p-Block elements form acidic, basic and amphoteric oxides. Explain each property by giving two examples and also write the reactions of these oxides with water. |
|
Answer» Solution :In p-block, when we move from LEFT to right in a period, the acidic character of the oxides increases due to increase in electronegativity. E.g, `2^(nd)` period `:B_(2)O_(3) lt CO_(2) lt N_(2)O_(3)` acidic nature increases. `(ii) 3^(rd)` perido `Al_(2)O_(3) lt SiO_(3) lt P_(4)O_(10) lt SO_(3) lt Cl_(2) O_(7)` acidic character increases. On moving down the group, acidic character decreases and BASIC character increases. e.g., (1) Nature of oxides of 13 group elements : `underset("Weakly acidic")(B_(2)O_(3))underset("Amphoteric")ubrace(Al_(2)O_(3)GHa_(2)O_(3)) underset("Basic")(In_(2)O_(3)) underset("Strongly basic")(Tl_(2)O)` (ii) Natureof oxides of 15 group elements : `underset("Strongly acidic")(N_(2)O_(5)) underset("Moderately acidic")(P_(4)O_(10)) underset("Amphoteric")(As_(4)O_(10))underset("amphoteric")( Sb_(4)O_(10)) underset("Basic")(Bi_(2)O_(3))` Among the oxides of same element, higher the oxidation state of the element, stronger is the acid. e.g., `SO_(3)` is a stronger acid than `SO_(2)`. `B_(2)O_(3)` is weakly acidic and on dissolution in water, it forms orthoboric acid. Orthoboric acid does not act as a protonic acid (it does not ionise) but acts as a weak Lewis acid. `underset("Boron trioxide")(B_(2)O_(3))+ 3H_(2)O hArr underset("Orthoboric acid")(2H_(3)BO_(3))` `B(OH)_(3)+H-OH to [B(OH)_(4)]^(-)+H^(+)` `Al_(2)O_(3)` is amphoteric in nature. It is insoluble in water but dissolves in alkalies and reacts with acids. `underset("Aluminium trioxide")(Al_(2)O_(3))+2NAOH overset(Delta)to underset("Sodium metal aluminate")(2NaAlO_(2))+H_(2)O larr Al_(2)O_(3) +6HCI overset(Delta)to underset("Aluminum Chlorid")(2AlCl_(3))+3H_(2)O` `Tl_(2)O` is as a basic as NaOH due to its lower oxidation state (+1). `Tl_(2)O+2HCI to 2TICI+H_(2)O` `P_(4)O_(10)` on reaction with water gives orthophosphoric acid. `underset("Phosphorus pentoxide")(P_(4)O_(10)) +6H_(2)O to underset("Orthophosphoric acid")(4H_(3)PO_(4))` `Cl_(2)O_(7)` is strongly acidic in nature and on dissolution in water, it gives perchloric acid. `underset("Dichlorine heptoxide")(Cl_(2)O_(7))+H_(2) toO underset("Perchloric acid")(2HCIO_(4))` |
|
| 37. |
p-Blockelementformacidicbasicand amphotericoxides . Explaineachproperty by givingtwoexamplesand alsowritethe reactions oftheseoxides withwater. |
|
Answer» Solution :Theoxides of p- blockelementshwo acidicbasicand amphotericproperties. Thisis due tothe combinedeffectof numberof factorssome of whichare discussed below, (i)Ionizationenthalpy. Ifthe ionizationenthalpy ofan elementis highit oxidewill beacidicin natureif low , it willbe basicand ifintermediateitsoxidewill beamphotericin nature . Furtherhighertheionizationenthalpystronger will be theacid. (ii)ELECTRONEGATIVITY. Higherthe electronegativity of theelementmoreacidicis itsoxide . Forexampleelectronegativitiesof B . C and Nare -2.0 , 2.5 and3.0respectivelythereforeacidiccharacterof theiroxidesincreasesin the same order`,B_(2)O _(3) ltCO_(2)lt N_(2)O_(3)` (iii) Oxidation state(O.S) . Amongthe oxides of thesameelementhigherthe oxidationstate of theelementstrongeris theacid . Forexample `P_(4)O`(inwhichO.S. of P is + 5)is astrongeracidthant `P_(4) O_(6)` (inwhichO.S.of P is+ 3) . SIMILARLY`SO_(3)` (in whichO.S. of S i s + 6)is a strongeracidthatn `SO_(2)` (inwhichO.S.of S is+ 4) . In thelightof the abovefactorslet us nowdiscus thevariationin acid `//` baseproperties ofoxides ofp-blockelements withina groupand acrossa period. (a)Variationwithin agroup. The oxideof firstelement ofeach groupis acidicnature. Onmovingdownthe groupthe acidiccharacterfirstdecreasesthenbecomesamphotericand finallychangestobasic . For examplethe oxides ofelementsof group13 showthe followingacid - basebehaviour. `underset("Weakly acidic")(B_(2)O_(3))""underset("Amphoteric ")(A1_(2)O_(3))""underset("Amphoteric")(Ga_(2)O_(3))""underset("Basic")(In_(2)O_(3))"" underset("Strongly basic ")(T1_(2)O)` Theionizationenthalpyof B issomewhathigh. Therefore`B_(2)O_(3)` isweakly acidic. It isslightlysolubleinwater andformsorthoboricacid which doesnot ionize itselfin waterbut accepts a pair ofelectronsfrom OH group of water. Thereforeit doesnot act as a protonic acid butacts aweak lewisacid . `underset("Boron trioxide")(B_(2) O_(3) )+3 H_(2) O hArr2 H_(3) BO_(3) underset("Orthoboric acid ")(" or ") 2 B (OH)_(3)` 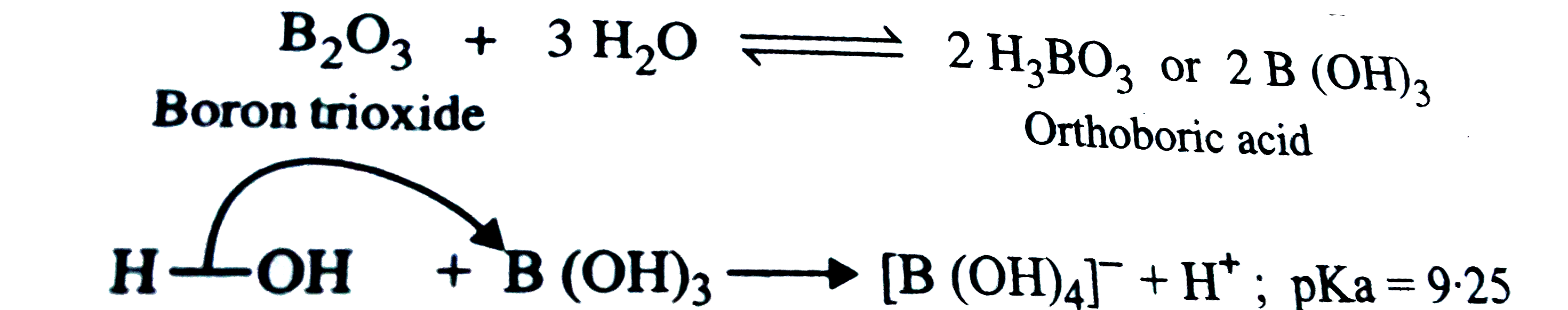 Sinceionizationenthalpyof A1 ismuchlowerthan that of B , therefore`A1_(2)O_(3)`isamphoteric . Itinsolublein waterbut dissolves inalkalies andreacts withacids. `underset("Aluminium oxide ")(A1_(2) O_(3) ) +2 Na OHoverset(Heat) (to) underset( "Sod. meta- aluminate ")(2 NaA1O_(2)) + H_(2)O` `underset("Aluminimum oxide ")(A1_(2)O_(3)) + 6HCIoverset (Heat) (to) underset("Aluminium chloride ")(2 A1C1_(3)) + 3 H_(2)O` Althoughionizationenthalpy of Ti issomewhathgiehr thant thatof A1,its oxidei.e.,`Ti_(2) O` isas basicasNaOHprobablydue toitsloweroxidationstate of + 1aas compapredto + 3or A1. `T1_(2)O_(3)+ 2 HC1 to 2 T1C1 + H_(2)O` Theoxidesof groups14,15and 16elements showsimilarytrendsbut oxidesof group 17 elements are allacids. `{:("Group" 14 "oxides ",,"Group" 15 "oxides",,"Group" 16 "oxides",,"Group" 17 "oxides"),(CO_(2) "Acidic ",,N_(2)O_(5) "Strongly acidic",,-,,-),(SiO_(2) "Less acidic",,P_(4) O_(10)"Moderately acidic",,SO_(2) "Acidic ",,C1_(2)O_(7)"Strongly acidic "),(GeO_(2) "Lesser acidic ",,As_(4)O_(10) "Amphoteric",,SeO_(2)"Weakly acidic ",,BrO_(2)"Acidic "),(SnO_(2) "Amphoteric ",,Sb_(14) O_(10) "Amphoteric ",,TeO_(2)"Amphoteric ",,I_(2)O_(5) "Acidic "),(PbO_(2) "Amphoteric ",,Bi_(2)O_(3) "Basic ",,PoO_(2) "Amphoteric " ,,-):}` ( b)Variation acrossa period. The ionzationenthalpyandelectronegativity ofelements and theirhighest oxidationstatesincreasesfrom leftto rightacrossa period . Due tocombinedeffect of thesefactors , theacidiccharacter of the oxides of elementsincreases.forexampleacrossthe thirdperiodof p-block element, theacidiccharacter of the oxidesincreasesin the order`,A1_(2) O_(3)` (amphoteric ) `lt SiO_(2)` (weakly acidic) `lt P_(4) O_(10)`(moderately acidic ) `lt SO_(3)` (strongly acidic ) `lt C1_(2) O_(7)` (verystronglyacidic) . These oxides dissovle in water toformtheircorrespondingacids : `underset("Phosphoruspentoxide ") (P_(4) O_(10))+6 H_(2) O tounderset( " Orthophosphoricacid ")(4 H_(3)PO_(4))` `underset("Dichlorine heptoxide ")(C1_(2)O_(7)) +H_(2) O tounderset( "Perchloric acid ")(2 HC1O_(4))` |
|
| 38. |
(P) and (Q) respectively are |
|
Answer»
|
|
| 39. |
P and Q are two arbitrarily chosen intensive variables then |
|
Answer» <P>(P + Q) is EXTENSIVE property |
|
| 40. |
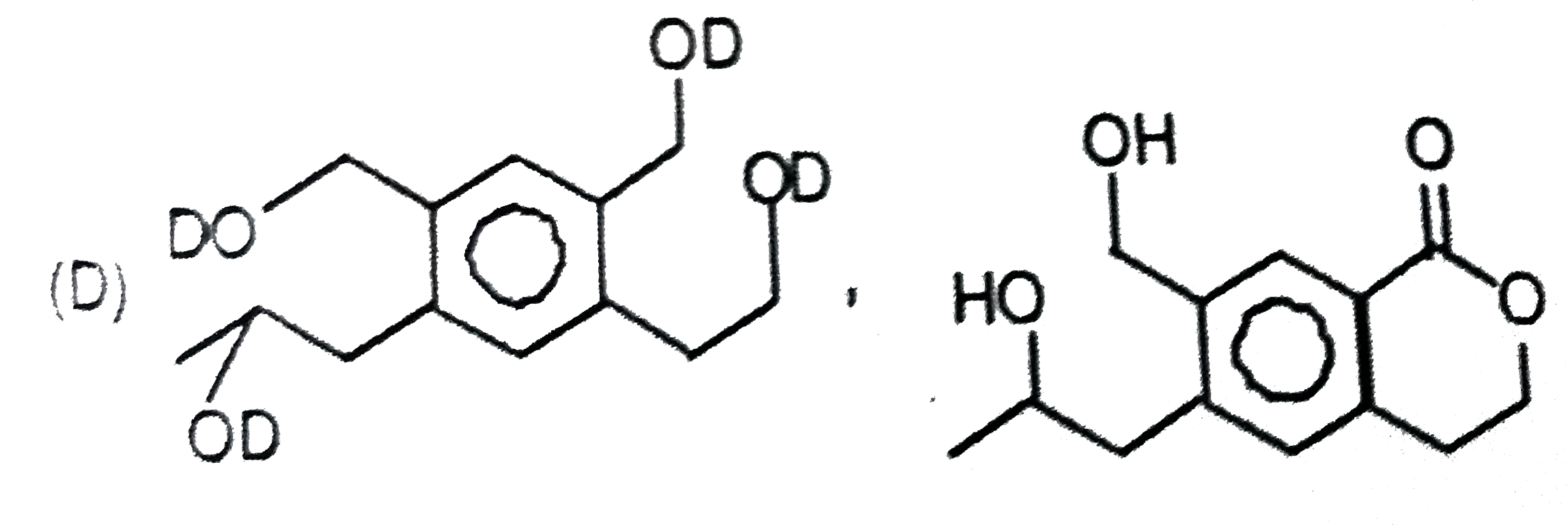
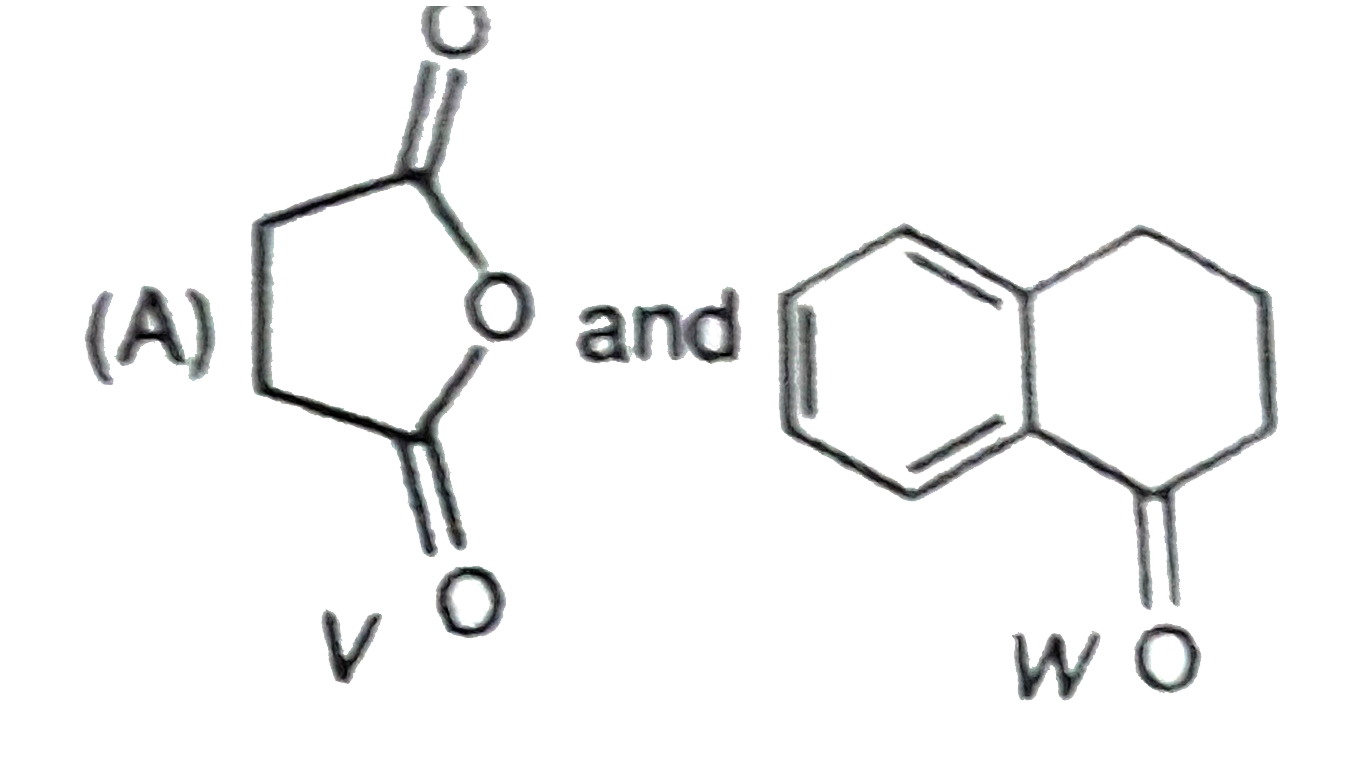
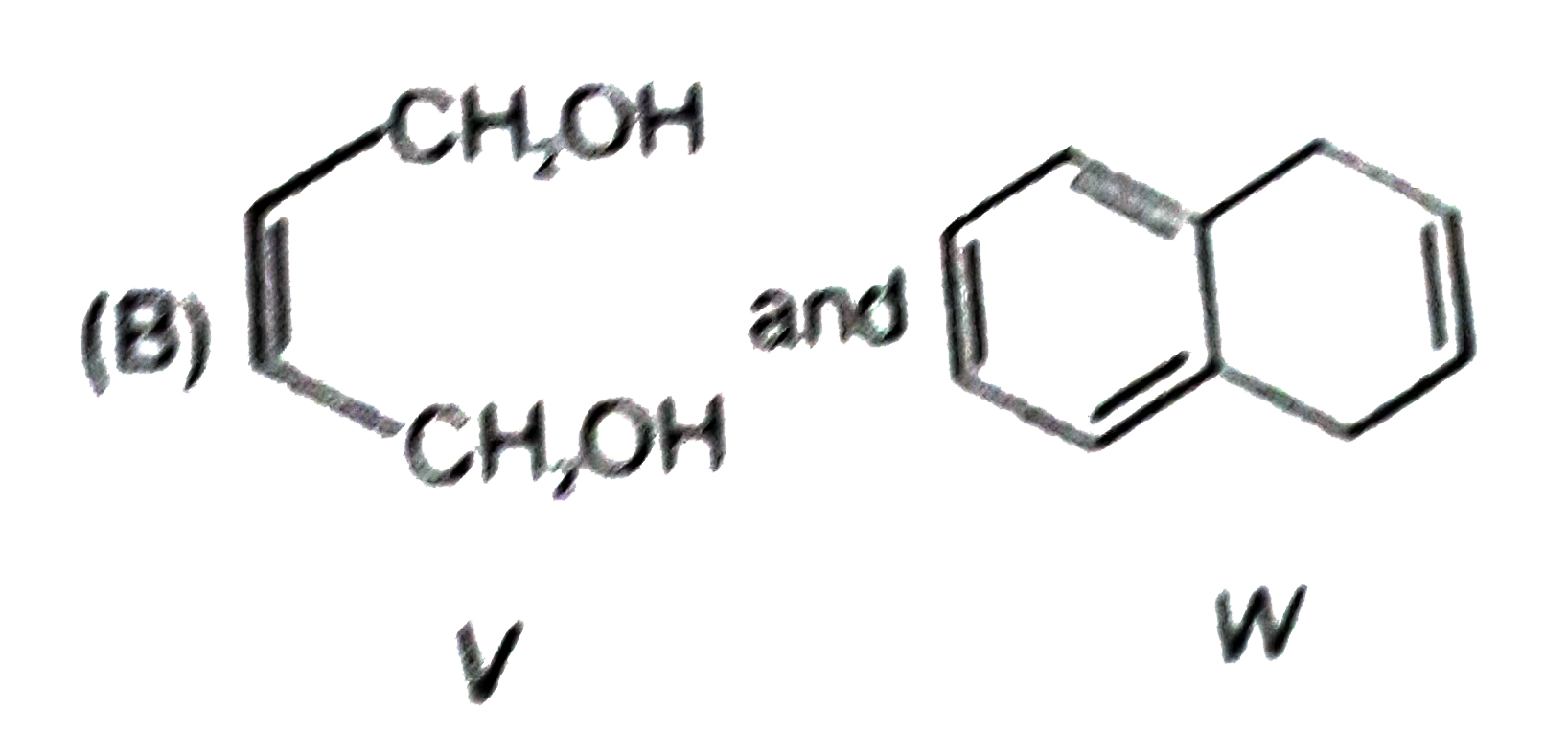
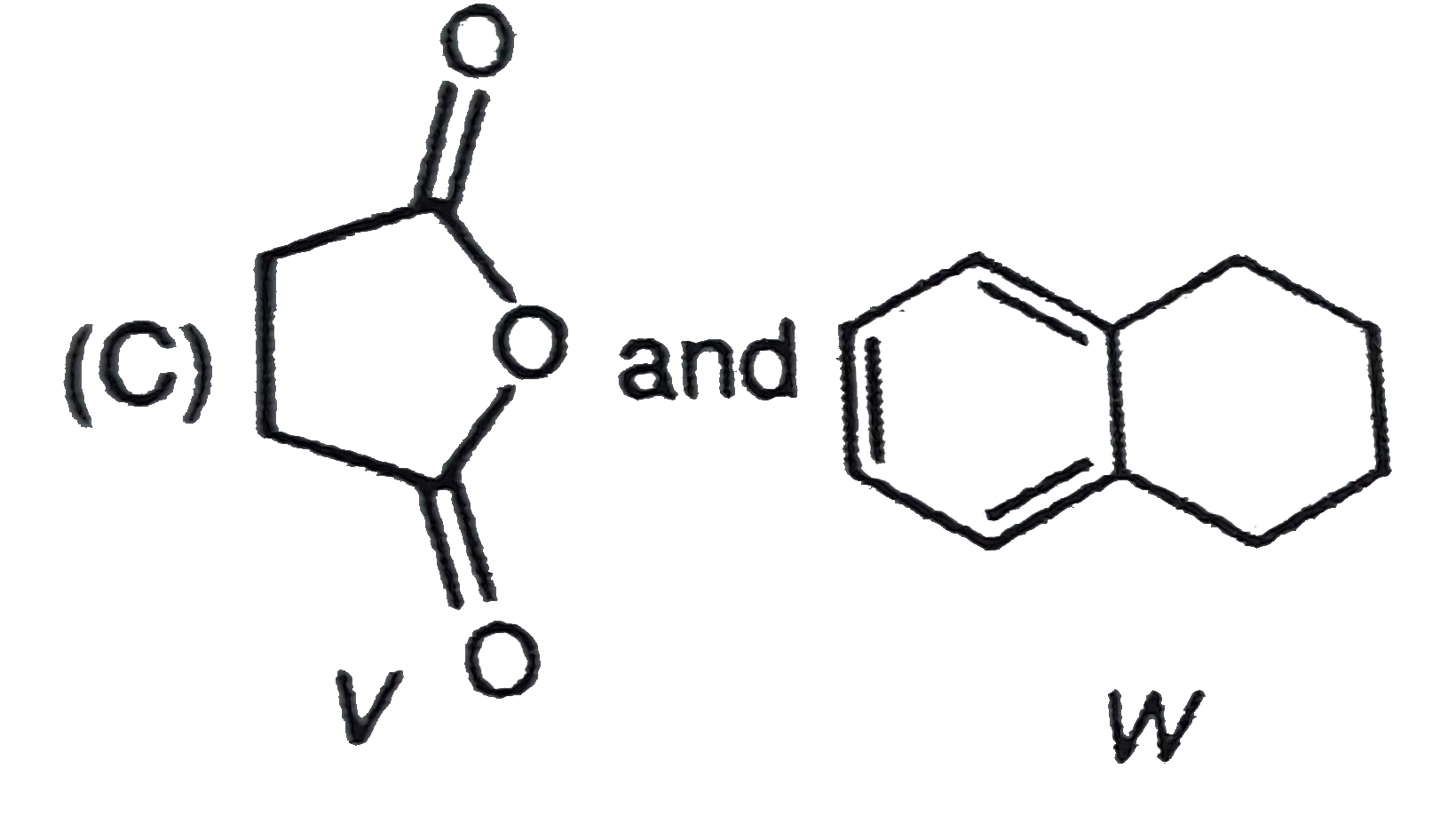
P and Q are isomers of dicarboxylic acid C_(4)H_(4)O_(4). Both decolorize Br_(2)//H_(2)O. On heating, P forms the cyclic anydride. Upon treatment with dilute alkaline KMnO_(4), p as well as Q could produce one or more than one from the S,T and U In the following reaction, sequences V and W are respectively. |
|
Answer»
|
|
| 41. |
P and Q are respectively. |
|
Answer» ACETONE and Hexane-1,2,5,6-tetraol. |
|
| 42. |
P and Q are isomers of dicarboxylic acid C_(4)H_(4)O_(4). Both decolorize Br_(2)//H_(2)O. On heating, P forms the cyclic anydride. Upon treatment with dilute alkaline KMnO_(4), p as well as Q could produce one or more than one from the S,T and U Compounds formed from P and Q are, respectively: |
|
Answer» Optically ACTIVE S and optically active pair (T,U) |
|
| 43. |
P and Q are : |
|
Answer»
|
|
| 44. |
P & Q are : |
|
Answer» POSITIONAL isomer 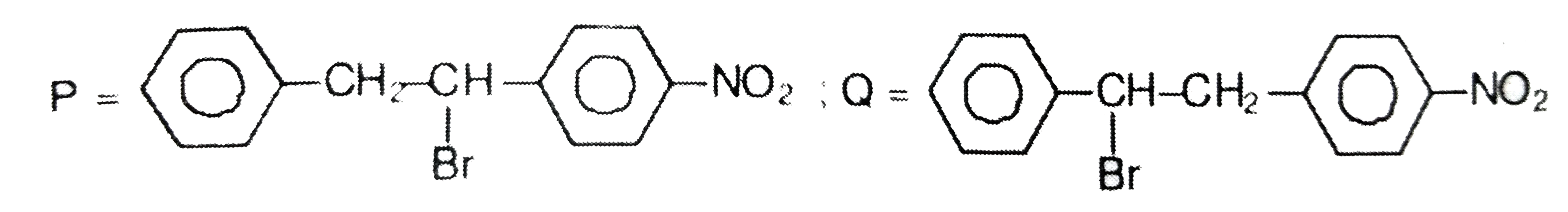
|
|
| 45. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X. the resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. The compound Y is: |
|
Answer» `MgCI_(2)` |
|
| 46. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X. the resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. The compound Z is: |
|
Answer» `Mg_(2)[Fe(CN)_(6)]` |
|
| 47. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X. the resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. The compound X is: |
|
Answer» `NaNO_(3)` |
|
| 48. |
Ozonolysis products of 3,4-dimethyl hept-3-ene are |
|
Answer» 2-BUTANONE and 3-pentanone |
|
| 49. |
Ozonolysis products of 2-ethylbut-1-ene are |
|
Answer» METHANAL and 3-pentanone |
|