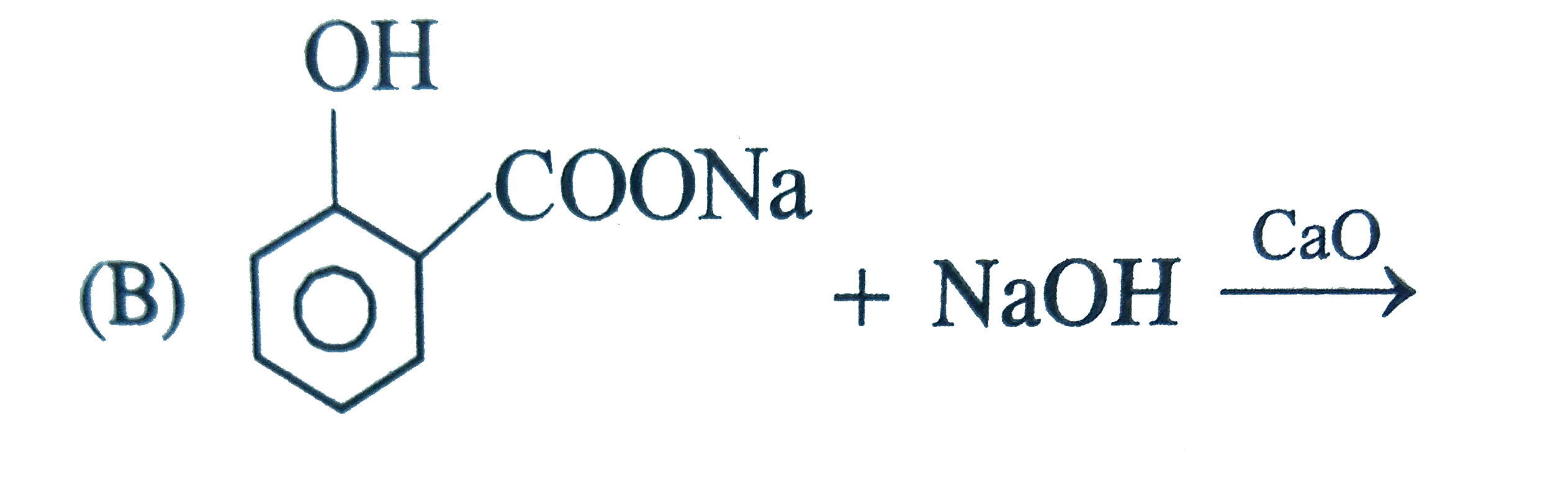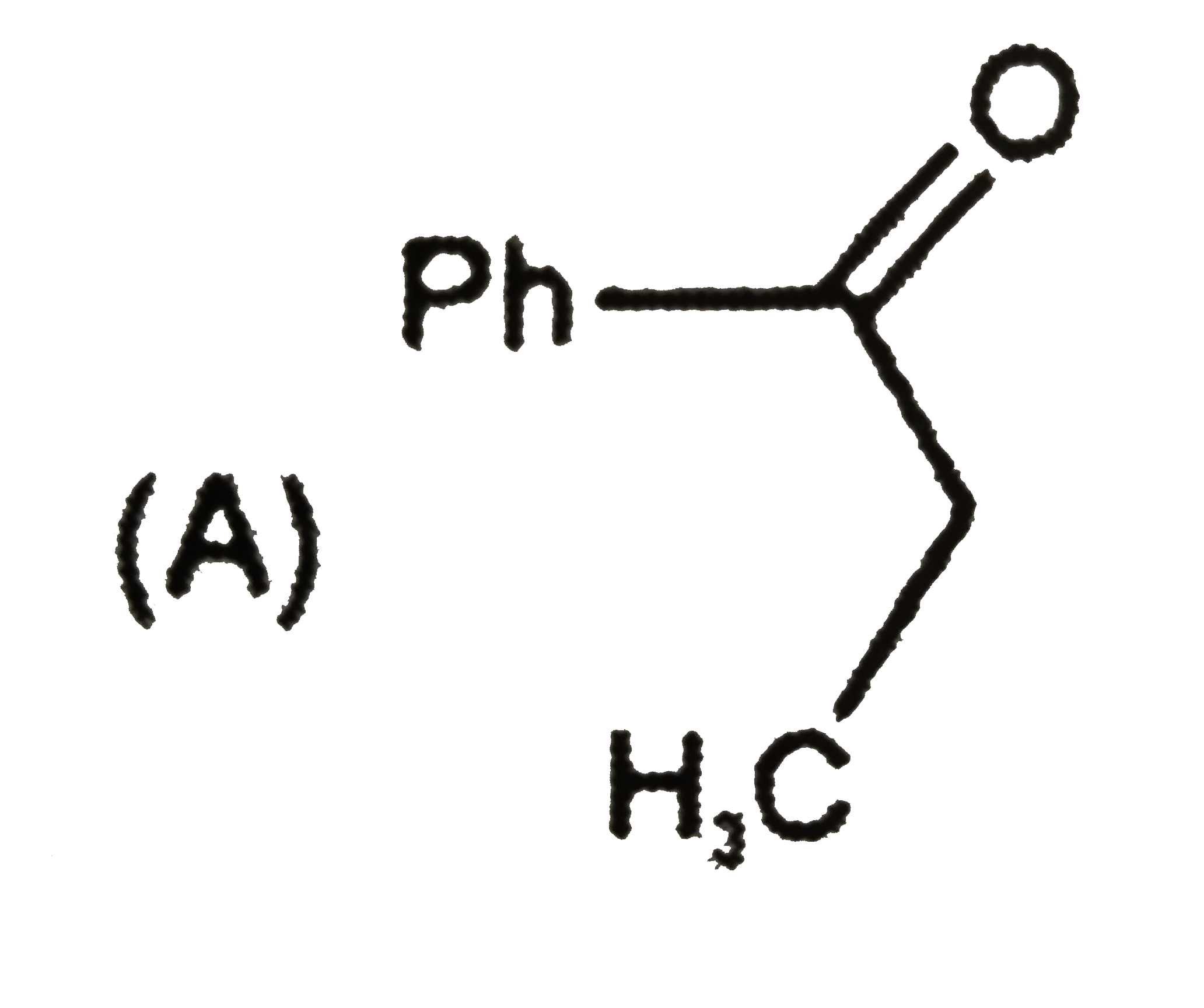Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Phenol on exposure to air produces a red coloured product due to the formation of |
|
Answer» p-benzoquinone 
|
|
| 2. |
Phenol is treated with bromine water and shaken well to get white precipitate is: |
|
Answer» 1-Bromophenol |
|
| 3. |
Phenol is more readily soluble in |
|
Answer» dil. HCl |
|
| 4. |
Phenol is less acidic than |
|
Answer» Acetic acid |
|
| 5. |
Phenol is heated with C Cl_(4) and alkaline KOH when salicylic acid is produced. The reaction is known is |
|
Answer» Friedel-Craft REACTION |
|
| 6. |
Phenol is heated with a solution of mixture KBr and KBrO_(3). The major product obtained the above reaction is : |
|
Answer» 3-Bromophenol 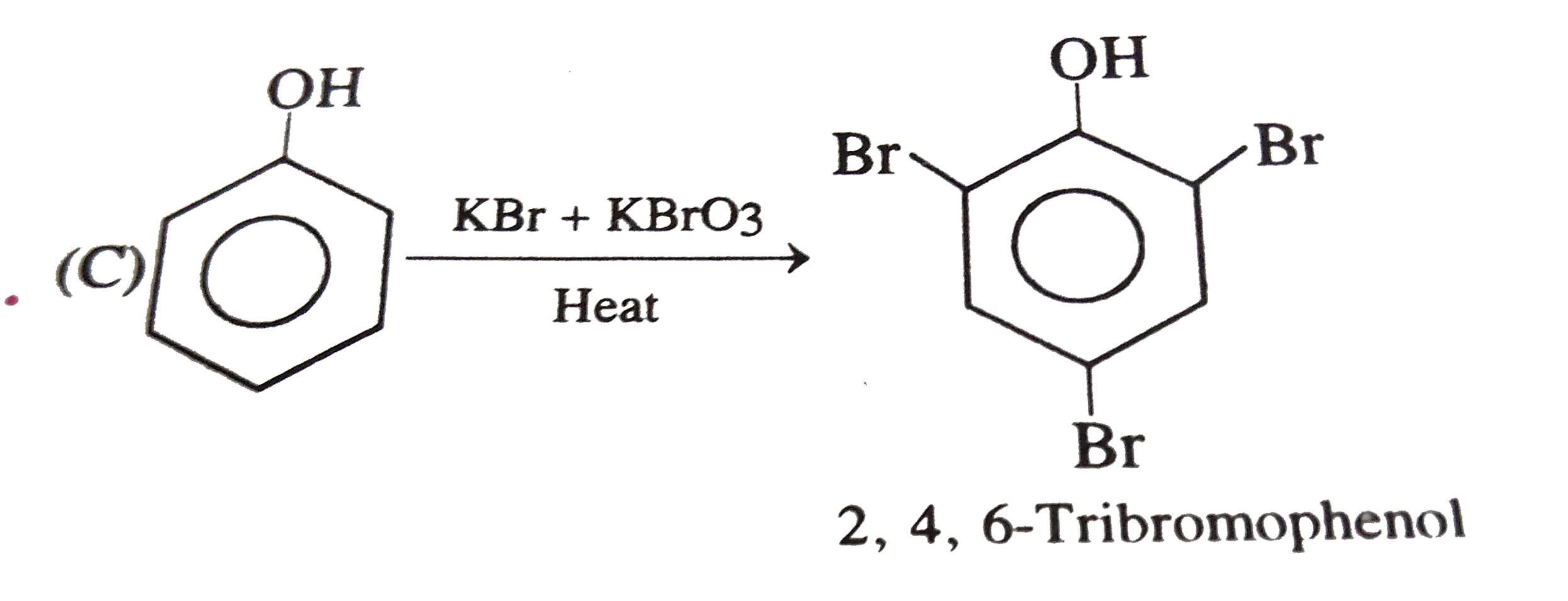
|
|
| 7. |
Phenol gives.......colour with neutral FeCl_(3) solution |
|
Answer» VIOLET |
|
| 9. |
Phenol dimerises in benzene having Van't Hoff factor 0.54. What is the degree of association? |
|
Answer» SOLUTION :`I= 0.54 ` `ALPHA =((1-i)N)/( (n-1)) =( (1 -0.54) 2) /((2-1)) = 0.46xx 2` ` alpha= 0.92` |
|
| 10. |
Phenol condenses with formaldehyde in the presence of acid or base to form |
|
Answer» bakelite |
|
| 11. |
Phenol dismerises in benzene having Van,t Hoff factor 0.54 What is the degree of association? |
|
Answer» `0. 46` ` alpha = ( (1-i)N)/((n -1))= ((1- 0.54)2)/((2-1))= 0.46 xx 2 =0.92` |
|
| 12. |
Phenol cannot be obtained by one of the following reactions |
|
Answer» `C_(6)H_(5)N_(2)^(+)Cl^(-)+H_(2)O overset(dil. H_(2)SO_(4))rarr` |
|
| 13. |
Phenol can be distinguished from ethanol by the following reagents except |
|
Answer» SODIUM |
|
| 14. |
Phenol are more acidic than alcohols because |
|
Answer» phenoxide ion is stabilised by resonance |
|
| 15. |
Phenol and benzoic acid may be distinguished by their reaction with |
|
Answer» AQUEOUS `NAOH` (C) Phenol gives characteristic colout (violet) with neutral aqueous `FeCl_(3)` while benzoic acid gives buff coloured precipitates |
|
| 16. |
pH=7.40, K_(1) "of" H_(2)CO_(3)=4.5xx10^(-7). What will be the ratio of [HCO_(3)^(-)] to [H_(2)CO_(3)] ? |
|
Answer» SOLUTION :`H_(2)CO_(3) hArr H^(+) + HCO_(3)^(-)` `K_(1) = ([H^(+)][HCO_(3)^(-)])/([H_(2)CO_(3)])` `:. ([HCO_(3)^(-)])/([H_(2)CO_(3)])=(K_(1))/([H^(+)])` pH = 7.40 means - LOG `[H^(+)] = 7.4` or `log [H^(+)] = - 7.4 bar(8) . 6 or [H^(+)] = 3.981 xx 10^(-8)` `:. ([HCO_(3)^(-)])/([H_(2)CO_(3)]) = (4.5xx10^(-7))/(3.981xx10^(-8))=11.3` |
|
| 17. |
PH_(3) has lower boiling point than NH_(3). Why ? |
Answer» Solution :The electronegativity of N(3.0) is much higher than that of P(2.1). Therefore, `NH_(3)` undergoes extensive intermolecular H-bonding and hence it exsists as an ASSOCIATED molecule. To BREAK these H-bonds, a LARGE amount of energy is NEEDED. On the other hand, `PH_(3)` does not undergo H-bonding and thus exists as discrete molecules. Therefore, the boiling point of `PH_(3)` is much LOWER than that of `NH_(3)`. 
|
|
| 18. |
PH_(3) forms bubbles when passed slowly in water but NH_(3) dissolves. Explain why ? |
| Answer» Solution :DUE ot HIGH electronegativity (3.0) and SMALL size of `N,NH_(3)` forms H-bonds with water and H-bonds with water and hence it is water soluble. On the other hand, due to its LOWER electronegativity (2.1) of P and its bigger size than `N,PH_(3)` does not form H-bonds with `H_(2)O`. As a result, it does not dissolve in `H_(2)O` and hence escapes as bubbles. | |
| 19. |
PH_(3) and BF_(3) form an adduct readily because they form |
|
Answer» A COORDINATE bond |
|
| 20. |
pH value of which of the followingis NOT equal to one ? |
|
Answer» `0.1 M CH_(3) CO OH` |
|
| 21. |
Ph-underset(Cl)underset(|)overset(Cl)overset(|)C-CH_3 overset(3NaNH_2)to (A) , What is product (A) ? |
|
Answer» `Ph-CH=CH_2 ` 
|
|
| 22. |
Ph-oversetoverset(CH_(3))(|)CH-undersetunderset(Br)(|)oversetoverset(CH_(3))(|)C-CH_(3) underset((S_(N)1))overset(EtoH)to(A) Major -product (A) is |
|
Answer» `PH-oversetoverset(CH_(3))(|)CH-undersetunderset(OEt)(|)oversetoverset(CH_(3))(|)C-CH_(3)` |
|
| 23. |
pH of the water coming out of cation exchange resin |
|
Answer» 7 |
|
| 24. |
pH of saturated aquoeus solution of Ba(OH)_(2) is 12. Identify the correct statement. |
|
Answer» The SOLUBILITY of `Ba(OH)_(2)` in a buffer SOLUTION of `pH = 13` is `5 xx 10^(-5)` moles/L |
|
| 26. |
pH of NH_4Cl solution is = 5.28 calculate degree of hydrolysis of its 0.02 M solution. |
| Answer» SOLUTION :`h=2.624xx10^(-4)` | |
| 28. |
pH of black coffee is 5.0 "at " 25^(@) C. Is black coffee acidic or basic ? |
|
Answer» |
|
| 29. |
pH of Ba(OH)_(2) Ssolution is 12. Its solubility product is : |
|
Answer» `10^(-6) M^(3) ` ` Ksp = 4S^(3) =5 xx 10 ^(-7) ` |
|
| 30. |
pH of aqueous solution of 0.1 M , NH_4Cl is found to be 5. The equilibrium constant for the neutralization of NH_4 OHby HCl is 10 ^(y) . The value of .y. is |
|
Answer» ` K_ N =(1)/(K_n)=(K_b)/(K_w)=(1)/(K_a)=(1)/(10^(-9) ) =10 ^(9) ` |
|
| 31. |
pH of a weakacid in 0.1 M solution is 4.3. Which statements are correct ? |
|
Answer» its 0.01 M Solution has `PH gt 4.3` ` 4.3 =(1)/(2)[P^(ka) -log 10 ^(-1)] rArrP^(ka) =7.6` ` pH =(1)/(2)[7.6-log 10 ^(-2)]rArr pH=4.8` As DIL `uparrow ,alpha " ALSO" uparrow ` |
|
| 32. |
pH of a solution of CuSO_(4) is.......... Than7and that of solution of Na_(2)CO_(3) is ..........than 7 . |
|
Answer» |
|
| 33. |
pH of a solution of a strong acid is 5.0. What would be the pH of the solution obtained after diluting the given solution a 100 times ? |
|
Answer» Solution :Given that, pH = 5 `[H^+]=10^(-5) "mol L"^(-1)` On diluting the solution 100 TIMES `[H^+]=10^(-5)/100= 10^(-7) "mol L"^(-1)` On calculating the pH using the equation `pH = -log [H^+]`, value of pH comes out to be 7. It is not POSSIBLE. This indicates that solution is very dilute. Hence, Total `H^+` ion concentration = `H^+` ions from ACID + `H^+` ion from water `[H^+]=10^(-7)+10^(-7)=2XX10^(-7)` M pH=-log `[2xx10^(-7)]` pH = 7 - 0.3010 = 6.699 |
|
| 34. |
P^(H)of a solution of the mixture of 0.1 HCl and 0.1N CH_3 COOHis (K_a =2xx10 ^(-5)) |
|
Answer» `1` |
|
| 35. |
pH of a solution of a strong acid is 5.0 . What will be the pH of the solution obtained after diluting the given solution 100 times ? |
|
Answer» Solution :PH = 5 means `[H^(+)]=10^(-5)M`. On diluting 100 times , `[H^(+)]=(10^(-5))/(100) = 10^(-7) M`. Thisshould give pH = 7 but it cannot be so because solution is acidic and pH should be less than 7 Thereason is that `[H^(+)]` from `H_(2)O` cannot be neglected. THUS, total `[H^(+)]=10^(-7)M` (from HCl) + `10^(-7)` M (from `H_(2)O ` ) `= 2xx10^(-7)M` `:. pH = - log (2xx10^(-7))=7-0.3010 = 6.699`. |
|
| 36. |
P^(H)of a solution is independent of |
|
Answer» Temperature |
|
| 37. |
Ph of a saturated solution of Ba(OH)_(2) " is "12. The value of solubility product is : |
|
Answer» `3.3 xx 10^(-7)` |
|
| 38. |
pH of a mixture of HA and A^(-)buffer is 5. K_b of A^(-)=10 ^(-10 ) .Hence [HA]/ [A^(-) ] will be |
|
Answer» 1 `pKa +PKB =14 rArr pKa =1 4 -10 =4` ` 5=4 +log ""([S])/([A]) rArr ([A^(-)])/([H_A]) =10` |
|
| 39. |
pH of a buffer solution decreases by 0.02 units when 0.12 g of acetic acid is added to 250 mL of a buffer solution of acetic acid and potassium acetate at 27^(circ)C. The buffer capacity of the solution is |
|
Answer» 0.1 |
|
| 40. |
pH of a 1.0xx10^(-8) M solution of HCl is |
|
Answer» 7.02 |
|
| 41. |
pH of 0.08 mol "dm"^(-3) HOCl solution is 2.85. Calculate its ionisation constant. |
|
Answer» Solution :pH of HOCl = 2.85 But, -pH = log `[H^+]` `THEREFORE -2.85=log [H^+]` `rArr 315 = log [H^+]` `rArr [H^+]= 1.413xx10^(-3)` For weak MONOBASIC ACID `[H^+]= SQRT(K_axxC)` `K_a=[H^+]^2/C=(1.413xx10^(-3))^2/0.08` `=24.957xx10^(-5) = 2.4957xx10^(-5)` |
|
| 42. |
pH of 0.08 mol dm^(-3) HOCl solution is 2.85. Calculate its ionisation constant. |
|
Answer» Solution :pH of Hocl = 2.85, i.e, - LOG `[H^(+)]= 2.85 ` or log `[H^(+)]=-2.85= bar(3).15` or `[H^(+)]`= antilog ` bar(3) .15 = 1.413 xx 10^(-3)M` For weak monobasic ACID, `HA hArr H^(+) + A^(-)` `K_(a) = ([H^(+)]^(2))/([HA])=((1.413xx10^(-3))^(2))/(0.08) = 2.4957xx10^(-5)`. |
|
| 43. |
pH, ionization constant K_(a) and concentration c of the solution of the salt of a weak acid and strong base (like CH_(3)CO Ona) are related as ............ |
|
Answer» |
|
| 44. |
Ph-CH=CH_2overset(IC l)to P, Identify major product 'P' is : |
|
Answer» `Ph-undersetunderset(I)(|)CH-CH_2-CL` |
|
| 45. |
Ph-CH=CH-CH_3 overset(HOCl)to X,X is : |
|
Answer» `Ph-undersetunderset(CL)(|)CH-oversetoverset(OH)(|)CH-CH_3` |
|
| 46. |
Ph-CH_(2)-overset(CH_(3))overset(|)(C)H-CH_(3)underset(("monochlorination"))overset(Cl_(2)//hv)to which statements is/are correct about protochemical chlorination of the above compound |
|
Answer» The MAJOR PRODUCT will have chiral carbon atom but is optically inacive. |
|
| 47. |
Ph-CH_2 - underset(D)underset(|)CH-CH_3 overset(Br_2// hv)to Product of the above reaction will be : |
|
Answer» DIASTEREOMERS |
|
| 48. |
Ph-CH_2-CH=CH_2overset(dil.H_2SO_4)to X, 'X' is : |
|
Answer» `Ph-CH_2-CH_2-CH_2-OH` |
|
| 49. |
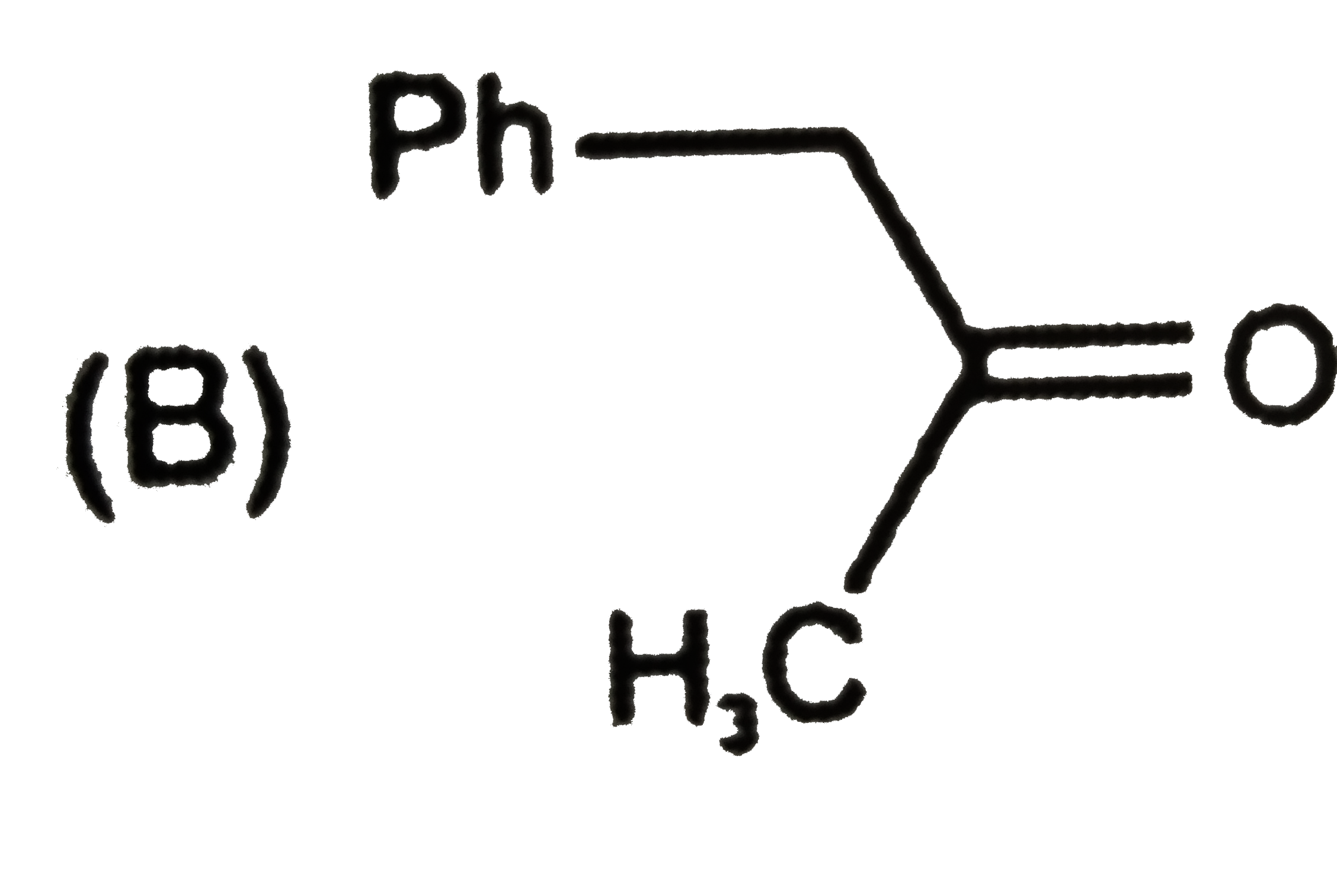
Ph-C -=CH underset(MeOH)overset(MeO)to major product of the reaction is |
|
Answer»

|
|
| 50. |
Ph-C-=C-CH_3overset(Hg^(2+)//H^(+))toA,A is : |
|
Answer»
|
|