Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict the metallic nature of the elements with atomic numbers 34, 35, 36 and 37. |
|
Answer» Solution :Element with atomic number 34 is selenium. It is a metalloid. Element with atomic number 35 is bromine, It is a non-METAL. Element with atomic number 36 is krypton. It is NEITHER a metal nor a non-metal Element with atomic number 37 is rubidium. It is a metal . |
|
| 2. |
Predict the major product of the acid catalysed dehydration of the following alcohols : (a)(CH_3)_2C(OH)CH_2CH_3 , (b)CH_3CH_2CH_2CH(OH)CH_3 (c )(CH_3)_2C(OH)CH(CH_3)_2 , (iv)(CH_3)_3C CH_2OH |
|
Answer» (B)`CH_3-CH_2-CH=CH-CH_3` (c)`(##RES_CHM_ORM_IV_E01_003_A01##)` (d)`CH_3-oversetoverset(CH_3)(|)C=CHCH_3` |
|
| 3. |
Predict the major product of the following reactions and explain their formation. H_3C-CH=CH_2 underset"HBr"overset ((Ph-CO-O)_2)to H_3C-CH=CH_2 overset"HBr"to |
Answer» SOLUTION :In presence of peroxides , addition of HBr to propene follows anti-Markovnikov's RULE and HENCE give 1-bromopropane as the major product . The reaction occurs through a free radical mechanism VIA a more stable free radical intermediate as shown below : 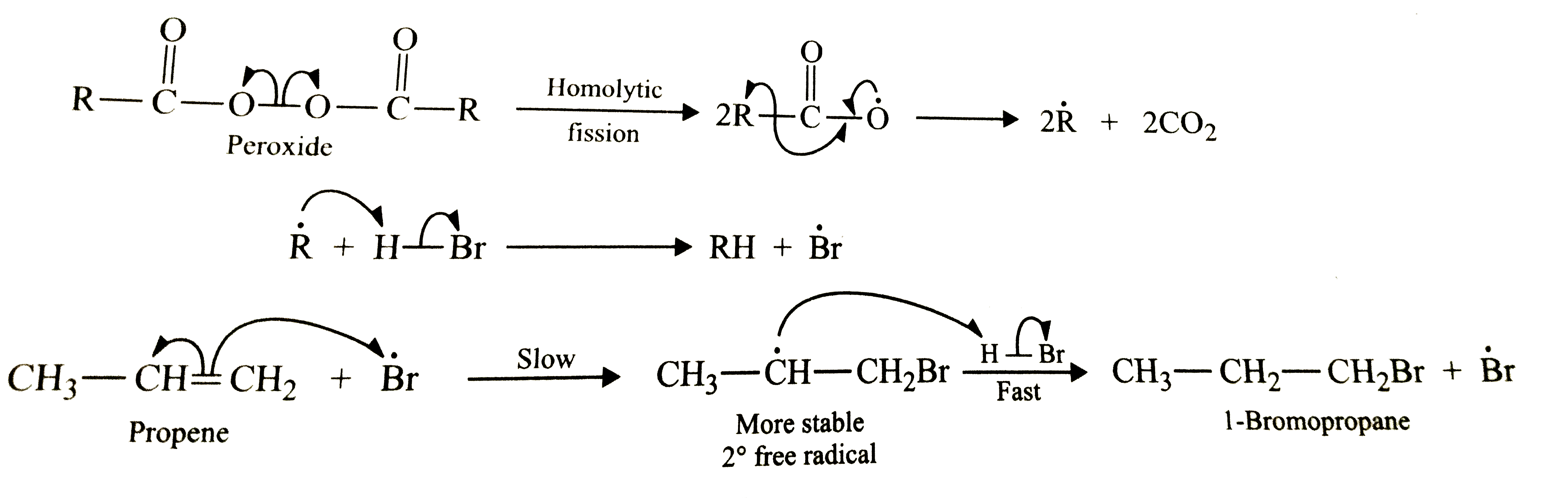 However, in absence of peroxides , addition of HBr to propene follows Markovnikov's rule and hence give 2-bromopropane as the major product. The reaction occurs through IONIC mechanism via a more stable carbocation intermediate as shown below : 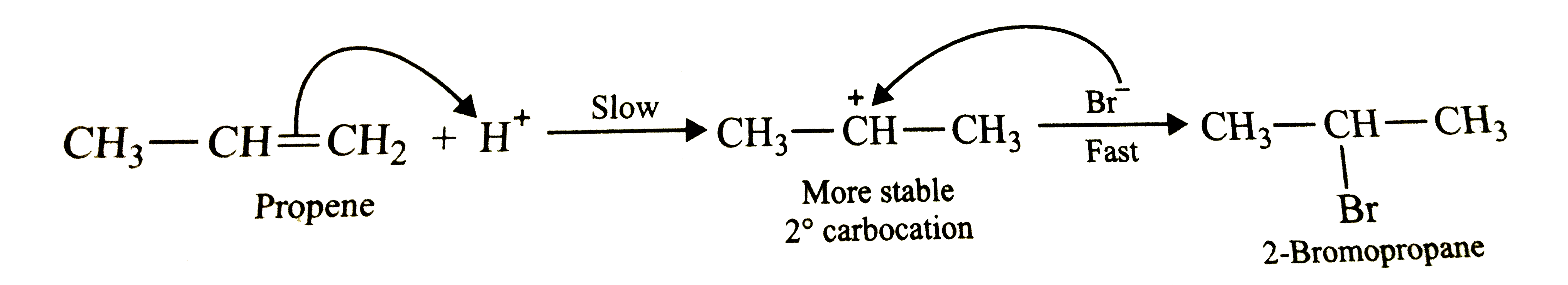
|
|
| 4. |
Predict the major product of chlorination of m-dinitrobenzene. |
Answer» SOLUTION :In m-dinitrobenzene, both NITRO GROUPS are m-directing. Further substitution occurs at third position to each `-NO_2` GROUP. The major product is l-chloro-3,5-dinitro- benzene. 
|
|
| 5. |
Predict the major product in the following reaction : R-C-=C-R underset"Lindlar's catalyst"overset(H_2)to |
Answer» SOLUTION :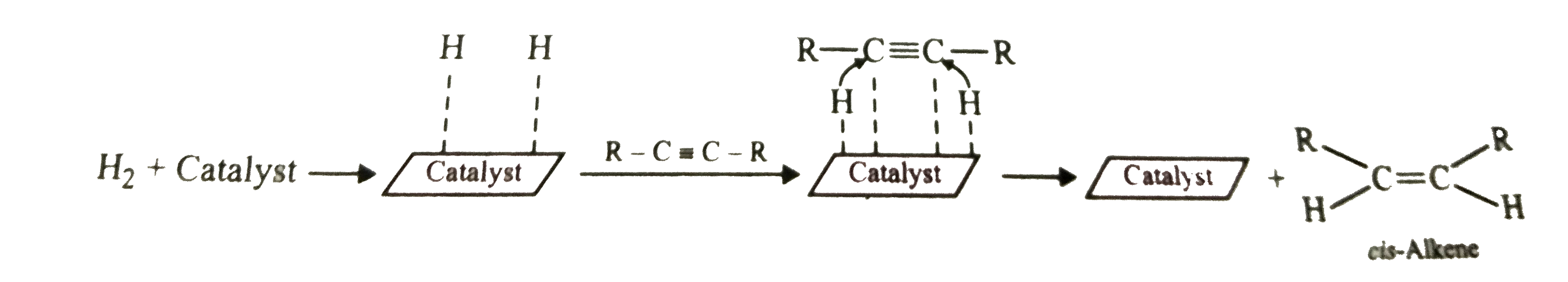
|
|
| 6. |
Predict the major product in the following reaction : C_6H_6 +(CH_3)_2CHCH_2OH overset(H_2SO_4)to |
Answer» Solution :In presence of CONC. `H_2SO_4`, isobutyl alcohol first gives `1^@` carbocation (I) which then rearranges to the more stable `3^@` carbocation (II) by 1,2-hydride shift. Carbocation (II) then REACTS with benzene to FORM tert-stable `3^@` carbocation (II) by 1,2-hydride shift. Carbocation (II) then reacts with benzene to form tert-butylbenzene. 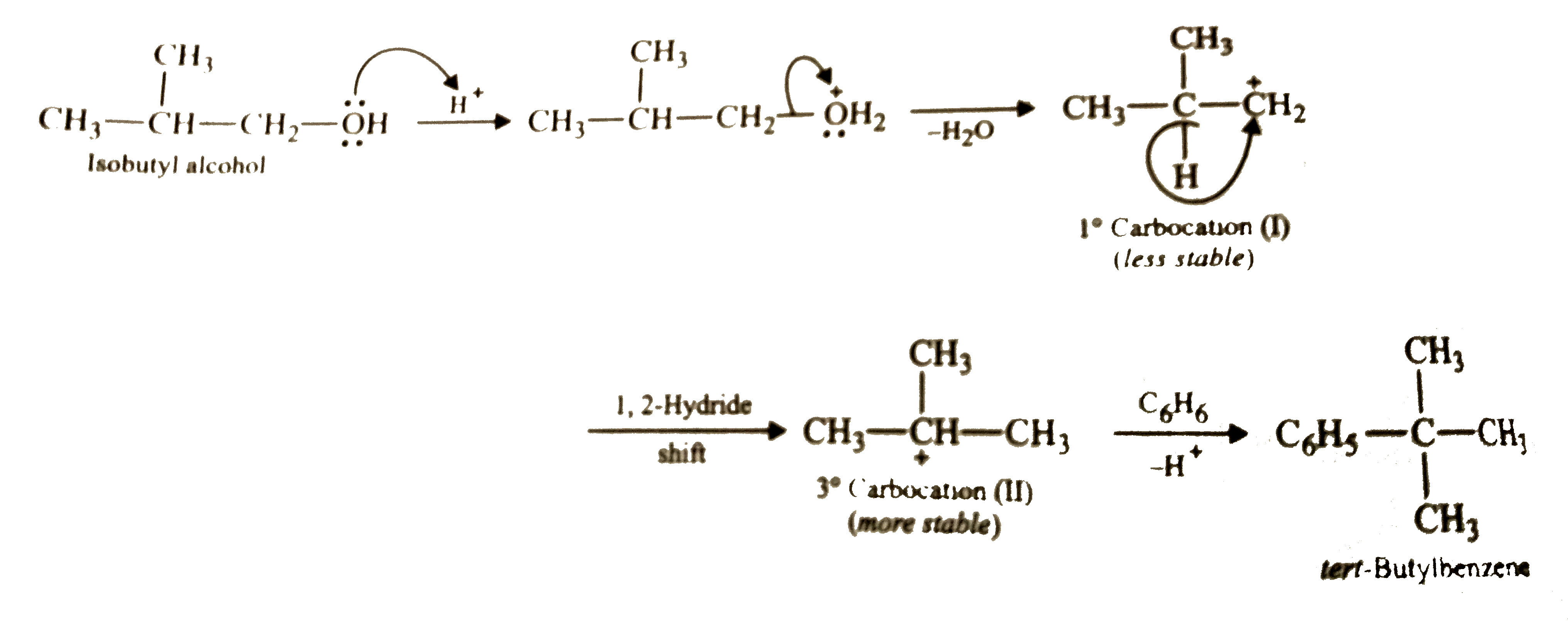
|
|
| 7. |
Predict the magnetic nature of the following heteronuclear diatomic molecules . NO . |
|
Answer» Solution :NO: No. of electrons ` = 7 + 8 = 15` Electronic configuration : `(sigma_(1s))^(2) (sigma_(1s)^(**))^(2) (sigma_(2)^(s))^(2)(sigma_(2s)^(**))^(2)(sigma_(2)p_(Z))^(2)(pi_(2)^(2)p_(x))^(2)` ` = (pi_(2)p_(y))^(2) pi_(2)^(**)p_(x)^(1)`. Bond order ` = 1/2 (N_(b) - N_(a))` ` = 1/2 (10 - 5) = 2.5` Magnetic nature : NO consist of one unpaired ELECTRON . Hence it is paramagnetic. |
|
| 8. |
Predict the major product formed when 2 - methyl-3-pentanol is dehydrated with alumina. |
|
Answer» Solution :Structure of 2-methyl-3-pentanol is `overset(5)CH_(3)-overset(4)CH_(3)-underset(OH)underset(|)overset(3)CH-underset(CH_(3))underset(|)overset(2)(CH)-overset(1)(CH_(3))` During dehydration, hydrogen from SECOND carbon is lost as it CONTAINS lesser number of HYDROGENS. So the major PRODUCT FORMED is 2-methyl-2-pentene. |
|
| 9. |
Predict the magnetic nature of the following heteronuclear diatomic molecules . CO . |
|
Answer» Solution :CO : No of ELECTRONS : ` 6 + 8= 14` Electronic configuration : `(sigma_(1s))^(2)(sigma_(1s)^(**))^(2) (sigma_(2s))^(2) (sigma_(2s)^(**))^(2) (sigma_(2)p_(z))^(2)(pi_(2)p_(x))^(2)(pi_(2)p_(y))^(2)` Bond ORDER ` = 1/2 (N_(b) - N_(a))` ` = 1/2 (10 - 4) = 3` Magnetic Nature : CO has no UNPAIRED electrons. Hence it is diamagnetic . |
|
| 10. |
Predict the highest and lowest possible oxidation state of each of the following elements: (a) Ta, 9b) Te, (c) Tc, (d)Ti, (e) Tl (f) N, (g) P, (h) F, (i) Cl, (j) Zn (k) C. |
|
Answer» |
|
| 11. |
Predict the hybridization of each carbon in the molecule of organic compound given below. Also Indicate the total number of sigma and pi bonds in this molecule. |
Answer» Solution :The HYBRIDIZATION and type of BONDS of each carbon in the MOLECULE given below
|
|
| 12. |
Predict the geometry of BF_(3), SF_(6), SO_(2)" and " NH_(3) |
| Answer» SOLUTION :`{:("Molecule","Geometry"),(BF_(3),"Triagonal Planar"),(SF_(6),"Octahedral"),(SO_(2),"Bent"),(NH_(3),"Tetrahedral"):}` | |
| 13. |
Predictthe formulae of the stablebinarycompoundsthat would be formedby thefollowingpairs ofelements (a)calcium and oxygen (b) aluminiumand carbon. |
|
Answer» |
|
| 14. |
Predict the formulas of the stable binary compounds that would be formed by given paris of elements: (1) Li and O, (2) Mg and N, (3) Al and I, (4) Si and O, (5) P and F, (6) Element with atomic number 7I and F. |
Answer» SOLUTION : . .
|
|
| 15. |
Predict the formulae of thestablebinary compoundsthat would be formedbythe followingpairs of elements (a) silicon andoxygen (b)aluminium andbromine( c )calcium and iodine (d)element 114 and fluorine (e )element 120 and oxygen. |
|
Answer» Solution :(a) Silicon belongsto GROUP 14. it has four valenceelectrons and hence its valence= 4 Oxygen belong to group 16. It has six electrons in thevalence shell andhenceits valence =8-6 =2 . Therefore theformula of silicon oxide= `SiO_(2)`. ( b)Aluminium belongs to group 13. It has three electron in the valenceshelland henceits valence =3. Brominebelongs to group 17. It hassevenelectrons in thevalence shell and hence its valence =8-7 =1 Thereforeformula of aluminium bromide= `AIBr_(3)` (c ) Calcium belongsto group 2. Ithas two -electrons in the valenceshelland henceits valence=2 .Iodine belongs to group 17. Ithas seven valence electrons and hence its valence =8-7=1. Thereforeformula of calcium iodic= `Cal_(2)` (d )In the7th period the 4TH transition seriesends at element with Z =112, thereforefilingof 7p- subshell willbegin withelement Z=113 .Henceelement with Z= 114 will have two electrons in 7p- orbitals andtwoelectrons in 7 s-orbital .In otherwordsthe element Z= 114 willlie ingroup 14 and henceits valence =4 fluorinebelongsto group 17 withsevenvalenceelectrons and henceitsvalence =8-7=1 Thereforeformulaof compoundformedby element114 andfluorineis `MF_(4)` where M representsthe element (e) The7thperiodends atelement with Z= 118. Thereforefillingof 8th shell i.e., 8 s-orbital willbegin with element Z= 119 andconsequently with Z= 120will belongto group 2 andits valencewill be=2 Oxygen belongsto group 16 withsixvalenceelectrons and henceits valence=8-6=2. Thereforeformula of compound formedby element Z= 120and oxygen will be MOwhere M respresent element. |
|
| 16. |
Predict the formulas of the stable binary compounds that would be formed by the combination of the following pairs of elements. (a) Lithium and oxygen (b) Magnesium and nitrogen (c) Aluminium and iodine (d) Silicon and oxygen (e) Phosphorus and fluorine (f) Element 71 and fluorine |
|
Answer» Solution :(a) LITHIUM belongs to group-1 with a valence of 1 while oxygen belongs to group-16 with a valence of 2. HENCE, the formula of the compound is `Li_(2)O`. (b) Magnesium belongs to group-2 with valence of 2 while nitrogen belongs to group-5 with a valence of 3. Hence, the formula of the compound `Mg_(3)N_(2)`. (C) Aluminium belongs to group-13 with a valence of 3 and IODINE belong to group-17 with valence of 1. Hence, the formula of the compound is `AlI_(3)`. (d)`SiO_(2) [:. "Silicon " 4^(TH) "valency in " 14^(th)"group valency is 4 and in " 16^(th) "group of oxygen having valency is " -2.]` (e)`PF_(5) "" [:. " In 15th group valency is 5 and flourine group 17th velency is"-1.]` (f) `71^(th)` elements means : `54 + 17 (4f^(14) 3d^(1) 6s^(2))`. Take elements E in `13^(th)` group having valency 3. Flourine `17^(th)` group having valency `-1` so formula is `EF_(3) " or " Z = 71` so, Formula : `LuF_(3)`. |
|
| 17. |
Predictthe formulae of the stable binarycompoundformedby thefollowingpairs ofelements : (a)element with Z= 116 and hydrogen (b) elementwith Z= 113 and fluorine( c) Uupand sulphur (d) Useand Uus |
|
Answer» |
|
| 18. |
Predict the formula of thestablebinarycompoundsthat wouldbe formedby theconbination ofthe followingpairsof elements ( a)Lithium and oxygen( b)Magnesium andnitrogen( c ) Aluminiumand iodine ( d ) Silicon and oxygen( e)Phosphorus andfluorine( f) Element71 and fluorine . |
|
Answer» Solution :(a) Lithium is analkalimetal ( Group 1). It has only oneelectron in thevalenceshell,thereforeitsvalenceis 1. Oxygen is agroup16 element witha valenceof 2. Therefore. Formulaof the compound formedwould be `Li_(2)O` ( lithium oxide) . ( b)Magnesium is an alkaline earthmetal ( group 2) and hencehas avalenceof 2. Nitrogen is agroup 15 ELEMENTWITH avalenceof 8-5 =3.Thus theformulaof the compoundformedwould be`Mg_(3)N_(2)` (magnesiumnitride ) ( c) ALUMINIUMIS group 13 elementwith a valenceof 3 whileiodineis ahalogen( group 17)with avalenceof 1. Thereforethe formula of thecompoundformedwould be `A1I_(3)` ( aluminium iodine ). ( d)Silicon is agroup 14 elementwith avalenceof 4 whileoxygenis a group 16 elementwith avalenceof 2 . Hencethe formulaof the compound formedis `SiO_(2)` ( silicondioxide) ( e)Phosphorus is a group 15 elementwitha valenceof 3 or 5whilefluorine isa group 17 element witha valence of 1. Hencethe formula71 is alanthanoidcalledlutetium (LU). Itscommonvalenceis 3. Fluorineis agroup 17 ( halogen )elementwith avalence of1. Thereforethe formulaof THECOMPOUND formedwould be `LuF_(3)` ( lutetiumfluoride). |
|
| 19. |
Predict the formulae of stable binary compounds that would be the formed by the combination of following pairs of elements. Mg and N |
| Answer» SOLUTION :`Mg_3N_2` | |
| 20. |
Predict the formulae of stable binary compounds that would be the formed by the combination of following pairs of elements. P and F |
| Answer» SOLUTION :`pf_3(or PF_5)` | |
| 21. |
Predict the enthalpy change, free energy change and entropychange when ammonium chloride is dissolvedin water and the solution becomes colder. |
|
Answer» |
|
| 22. |
Predict the density of Cs from the density of the following elements. K=0.86g//cm^(3)" Ca"=1.548g//cm^(3)"Sc"=2.991g//cm^(3) Rb=1.532g//cm^(3)"Sr"=2.63g//cm^(3)"Y"=4.34g//cm^(3) Cs=? "Ba"=3.51g//cm^(3)"La"=6.16g//cm^(3) |
|
Answer» Solution :The elements listed above belong to group 1, 2 and 3 respectively. Let us CALCULATE the ratios of densities of BA and Sr present in group 2 and of La and Y which belong to group 3. Ratio of densities of Ba and Sr `=(3.51)/(2.63) =1.33`Ratio of densities ofLa and 6.16 Y `=(6.16)/(4.34)=1.42` From the VALUES, we find the ratio increases by (1.42 – 1.31) = 0.11. Moving on the same lines, the expected ratio of the densities of Cs and Rb =1.31- 0.11 =1.20. Thus, `("Density of Cs")/("Density of Rb")=1.20` or Density of `Cs= 1.20 xx Density of Rb= 1.20 xx 1.532" (given) = "1.84 g/cm^(3).` The actual density of Cs is `1.87 g//cm^3` and this is QUITE close to the predicted VAULE. |
|
| 23. |
Predict the feasibility of a reaction when (i) both DeltaH and DeltaS positive (ii) both DeltaH and DeltaS negative (iii) DeltaH decreases but DeltaS increases |
|
Answer» Solution :(i)When both `DeltaH` and `DELTAS` are POSITIVE, the reaction is not FEASIBLE. (ii) When both `DeltaH` and `DeltaS` are NEGATIVE, the reaction is not feasible. (iii) When `DeltaH` DECREASES but `DeltaS` increases, the reaction is feasible. |
|
| 24. |
Predict the dipole moment off (i) a molecule of the type AB_(4) having square planar geometry, (ii) a molecule of the type, AB_(3) having trigonal bipyramidal geometry, (iii) a molecule of the type, AB_(6) having octahedral geometry, (iv) a molecule of the type, AB_(7) having pentagonal bipyramidal geometry. |
|
Answer» Solution :(i) All the four A-B bond MOMENTS act at an angle of `90^(@)` with each other. THEREFORE the net dipole moment of `AB_(4)` is zero `(mu=0)` (ii) DUE to symmetrical structure of the molecule, the equatorial bond moments cancel each other. SIMILARLY the axisl bond moments cancel each other. therefore the resultant dipole moment is zero `(mu=0)` (III) same as (ii), (iv) same as (ii). |
|
| 25. |
Predict the correctshape of BH_(4)^(-) |
|
Answer» PYRAMIDAL, `SP^(3)` |
|
| 26. |
Predict the correct shape of BH_(4)^(-) |
|
Answer» PYRAMIDAL, `sp^(3)` |
|
| 27. |
Predict the correct order among the following: |
|
Answer» Ione pair - Ione pair `GT` BOND pair - bond pair `gt ` Ione pair - bond pair |
|
| 28. |
Predict the correct order of repulsions among the following : |
|
Answer» bond PAIR-bond pair `GT ` lone pair-bond pair `gt` lone pair-bond pair ` gt ` bond pair-bond pair. |
|
| 29. |
Predict the correct intermediate and product in thefollowing reaction : H_3C-C-=CH underset(HgSO_4)overset(H_2O, H_2SO_4)to underset"(A)""Intermediate" to underset"(B)""Product" |
|
Answer» `A:H_3C-undersetunderset(OH)|C=CH_2 ""B:H_3C-undersetunderset(SO_4)|C=CH_2` `CH_3-C-=CHunderset(HgSO_4//H_2O_4)OVERSET(H-OH)to underset"Intermediate (A)"(CH_3-undersetunderset(OH)|C=CH_2) overset"Tautomerises"to underset"Aceton"(CH_3-undersetundersetO(||)C=CH_2)` |
|
| 30. |
Predict the close packed struture of an ionic compoundA^(+) B^(-)in which the radius of thecation = 148 pm and radius of anion = 195 pm. What is the corrdinationnumber of the cation ? |
|
Answer» |
|
| 31. |
Predict the close packed structure of an ionic compound A^+ B^- in which the radius of the cation = 148 pm and radius of anion=195 pm. What is the coordination number of the cation ? |
|
Answer» |
|
| 32. |
Predict the change in internal energy for an isolated system at constant volume. |
|
Answer» SOLUTION :Predict the change in internal energy for an ISOLATED SYSTEM at CONSTANT VOLUME. `:.DeltaU=q+w` `DeltaU=0+0=0` |
|
| 33. |
Predict the change, if any, in hybridization of Al atom in the following reaction AlCl_(3)+Cl^(-) rarr AlCl_(4)^(-). |
| Answer» SOLUTION :`SP^(2)` to `sp^(3)` | |
| 34. |
Predict the acidic, basic or neutral nature of the solutions of the following salts : NaCl, KBr, NaCN, NH_(4)NO_(3), NaNO_(2), KF. |
|
Answer» SOLUTION :`NaCN, NaNO_(2), KF` solutions are basic, as THEYARE salts of strong base, weak acid. NaCl, KBR solutions are neutral, as they are salts of strongacid, strong base. `NH_(4)NH_(3)` solution is acidic, as it is a salt of strong acid, weak base. |
|
| 35. |
Predict reaction of 1 N sulhuric acid with the following metals : (i) copper (ii) lead (iii) iron Given E_(Cu^(2+),Cu)^(@)=+0.34 "volt" ,E_(Pb^(2+),Pb)^(@)=-0.13 "volt" ,E_(Fe^(2+),Fe)^(@)=-0.44 volt |
|
Answer» (i)since `E_(Cu^(2+),Cu)^(@)=0.34V` is higher than `E_(H^(+),H_(2)^(@)=0.0V` therefore Cu will not react with `1NH_(2)SO_(4)` to produce `H_(2)` gas (ii)Sinc `E_(PB^(2+),pb)^(@)=-0.13V` and `E_(H^(+),H_(2)^(@)=0.0` therefore lead will react with `1N H_(2)SO_(4)` to produce `H_(2)` gas |
|
| 36. |
Predict product formed when benzene is treated with iodine chloride in presence of anhydrous aluminium chloride. |
Answer» Solution :since iodine is LESS ELECTRONEGATIVE than chlorine, therefore , `AlCl_3` reacts with IC l to form iodonium ion `(I^+)` as the electrophile which SUBSEQUENTLY reacts with benzene to form IODOBENZENE. 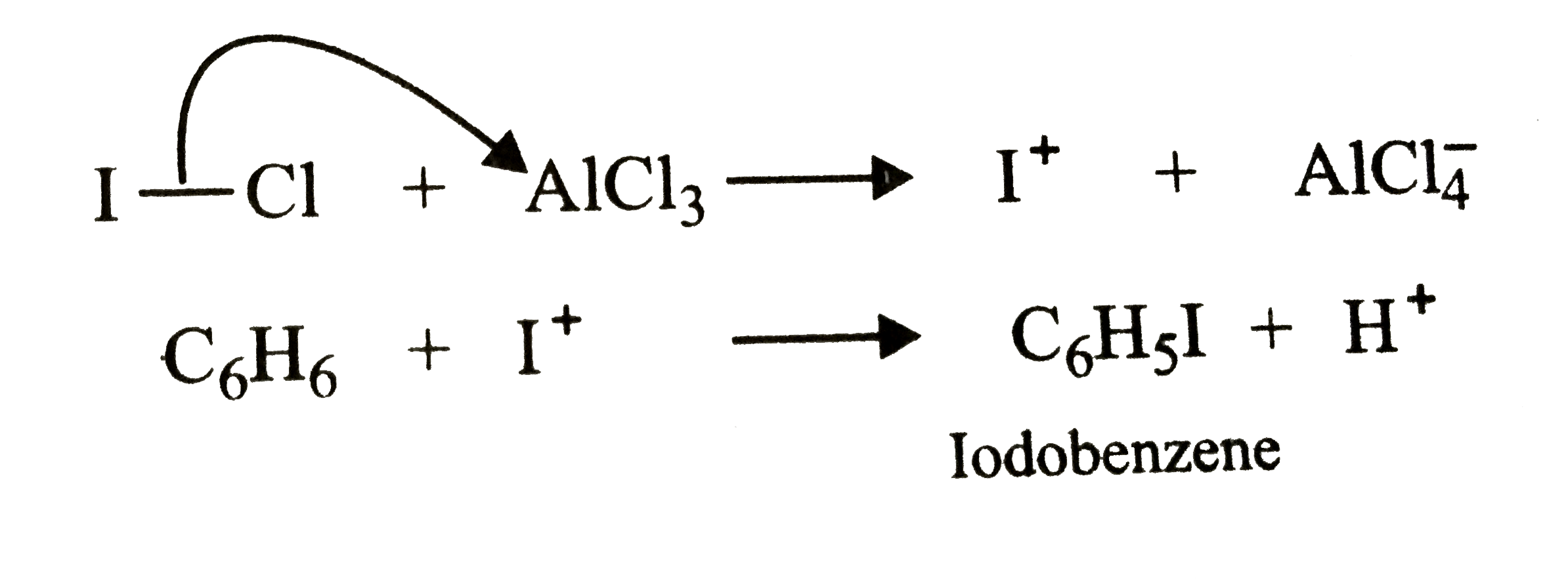
|
|
| 37. |
Predict major product of the following cannizaro' s reaction |
|
Answer» |
|
| 38. |
Predict in which of the following reactions, entropy increases/decreases Temperature of a crystalline solid is raised from 0 K to 115 K |
| Answer» SOLUTION :When TEMPERATURE is raised, DISORDER in MOLECULES increases and therefore entropy increases. | |
| 39. |
Predict in which of the following reactions, entropy increases/decreases 2NaHCO_(3) rarr Na_(2)CO_(3)(s) + CO_(2)(g) + H_(2)O(g) |
| Answer» SOLUTION :Reactant is a solid and hence has low ENTROPY. AMONG the products there are two GASES and one solid. So the products represent a condition of higher entropy. | |
| 40. |
Predict in which of the following reactions, entropy increases/decreases H_(2)(g) rarr 2H(g) |
| Answer» SOLUTION :here 2 MOLES of H atoms have higher ENTROPY than one MOLE of hydrogen molecule. | |
| 41. |
Predict in which of the following, entropy increases:(iii)H_2(g)rarr 2H(g). |
| Answer» SOLUTION :ENTROPY INCREASES | |
| 42. |
Predict in which of the following, entropy increases:(iii)2NaHCO_3(s)rarrNa_2CO_3(s)+CO_2(g)+H_2O(g). |
| Answer» SOLUTION :ENTROPY INCREASES | |
| 43. |
a) Predict whether entropy increases or decreases in the following changes: i) l_2(S)rightarrowl_2(g) ii) Temperature of a crystalline solid is raised from 0 K and 115 K. iii) Freezing of water b) Calculate the enthalpy of combustion of methane. Given that standard enthalpies of formation of CH_4,CO_2 and H_2O are -75.2, -394 and -285.6 kJ/mol respectively. |
| Answer» SOLUTION :ENTROPY INCREASES | |
| 44. |
Predict in which of the following, entropy increases // decreases . (i) A liquid crystallizes into a solid (ii) Temperaure of a crystalline solid israised from0Kto 115K (iii) 2 NaHCO_(3)(s) rarr Na_(2)CO_(3)(s) + CO_(2)(g) + H_(2)O(g) (iv)H_(2)(g) rarr 2H(g) |
|
Answer» Solution :(i) Decreases becausea SOLID has ORDERED ARRANGEMENT of constituent particles. (ii) INCREASES because at 0K,there isa perfect order of constituent particles. As temperature is increased , the particlestart vibrating about their equilibrium position. Hence, a disorder sets in. (iii)Increases becausethe reactant is solid whereas products have gaseous substances. (iv) Increases because the products havelarger (DOUBLE) number of particles. |
|
| 45. |
Predict in which of the following, entropy increases:(i) A liquid crystallizes into a solid. |
| Answer» SOLUTION :ENTROPY DECREASES | |
| 46. |
Predict in which of the following , entropy increases or decreases . (i) A liquid crystallizes into a solid (ii)Temperature of a crystallized solid is raised from 0 K to 115 K (iii)2NaHCO_(3(s)) to Na_2CO_(3(s)) + CO_(2(g)) + H_2O_((g)) (iv)H_(2(g)) to 2H_((g)) |
|
Answer» Solution :(i)After FREEZING, the molecules attain an ordered state and therefore, entropy decreases. (ii) At 0 K the constituent particles are in static form therefore, entropy is minimum. If the temperature is RAISED to 115 K particles begin to move and entropy increases (iii) Reactant, `NaHCO_3` is solid, THUS, its entropy is LESS in comparison to product which hashigh entropy (iv)Here, one molecule gives two atoms. Thus, number of particles increases and this leads to more disordered form. |
|
| 47. |
Predict in which of the following, entropy increases/decreases: (i) A liquid crystallizes into a solid. (ii) Temperature of a crystalline solid is raised from 0 K to 115 K. (iii) 2NaHCO_(3(s)) to Na_(2) CO_(3(s)) + CO_(2(g)) + H_(2) O_((g)) (iv) H_(2(g)) to 2H_((g)) |
|
Answer» Solution :(i) After freezing, the molecules attain an ordered state and therefore, entropy decreases. (ii) At 0 K, the constituent particles are static and entropy is minimum. If temperature is· raised to 115 K, these begin to MOVE and oscillate about their EQUILIBRIUM positions in the lattice and system becomes more disordered, therefore entropy increases. (iii) Reactant, `NaHCO_(3)` is a SOLID and it has LOW entropy. Among products there is one solid and two gases. Therefore, the products represent a condition of higher entropy. (IV) Here one molecule gives two atoms i.e., number of particles increases leading to more disordered state. Two moles of H atoms have higher entropy than one mole of dihydrogen molecule. |
|
| 48. |
What are the sign of the entropy change (+ or -) in the following: I : A liquid crystallises into a solid II: Temperature of a crystalline solid is raised from 0K to 115K III: 2NaHCO_(3(g)) rarr Na_(2)CO_(3(g)) + CO_(2(g)) + H_(2)O_((g)) IV : H_(2(g)) rarr 2H_((g)) |
|
Answer» Solution :i) After freezing, the molecules attain an ordered state and therefore, entropy DECREASES. ii) At 0 K, the consituent particles are STATIC and entropy is MINIMUM. If temperature is raised to 115K, these begin to move and oscillate about their equilibrium positions in the lattice and system BECOMES more disordered, therefore entropy increases. iii) Reactant, `NaHCO_3` is a solid and it has low entropy. Among PRODUCTS there are one solid and two gases. Therefore, the products represent a condition of higher entropy. iv) Here one molecule gives two atoms i.e., number of particles increases leading to more disordered state, Two moles of H atoms have higher entropy than one mole of dihydrogen molecule. |
|
| 49. |
Predict if the solutions of the following salts are neutral , acidic or basic : NaCl , KBr, NaCN, NH_4NO_3 , NaNO_2 and KF |
|
Answer» SOLUTION :Basic solution : NaCN, `NaNO_2` and KF are strong BASE and weak acid salts . So, the SOLUTIONIS basic. Acidic solution : The solution of `NH_4NO_3` is acidic because `NH_4NO_3` is a salt of strong acid `HNO_3` weak base `NH_4OH` Neutral solution :The solution of NaCl and KBr are neutral because they are strong acid ANDSTRONG base salt. |
|
| 50. |
Predict if the solution of the following salts are netural, acidic or basic: NaCl, KBr, NaCN, NH_(4)NO_(3), NaNO_(2) and KF |
| Answer» SOLUTION :`{:(NaCl,,-,,Neutral ("Salt" or S_(A)//S_(B))),(KBr,,-,,Neutral ("Salt" or S_(A)//S_(B))),(NACN,,-,,Basic ("Salt" or S_(B)//S_(A))),(NH_(4)NO_(3),,-,,Acidic ("Salt" or S_(A)//S_(B))),(NaNO_(2),,-,,Basic ("Salt" or S_(B)//S_(A))),(KF,,-,,Basic ("Salt" or S_(B)//S_(A))):}` | |


